Multiscale Entropy Analysis of Unattended Oximetric Recordings to Assist in the Screening of Paediatric Sleep Apnoea at Home
Abstract
:1. Introduction
2. Methodology
2.1. Dataset and Sleep Studies
2.2. Automated Signal Processing
2.2.1. Multiscale Entropy
2.2.2. Conventional Oximetric Indexes
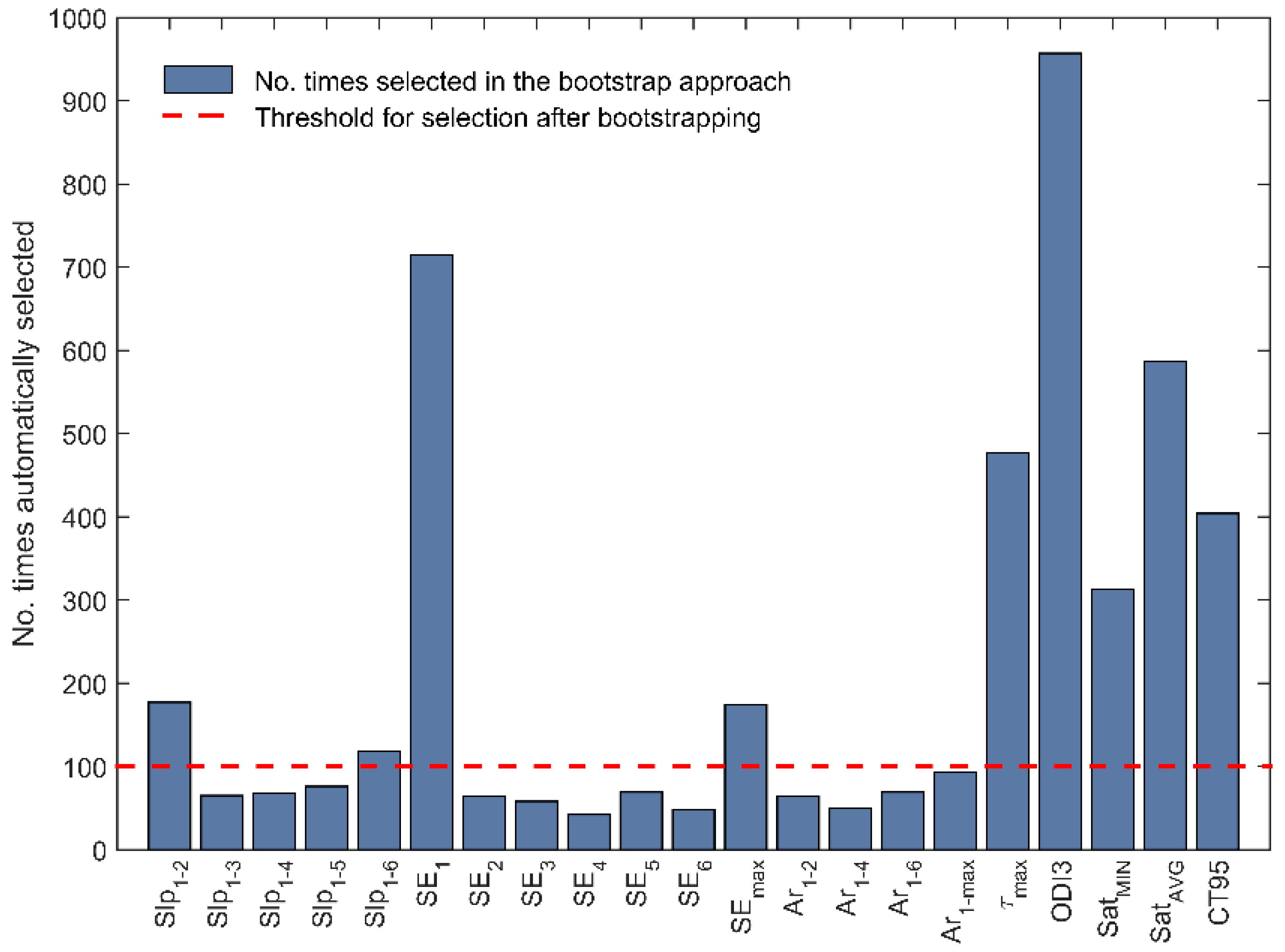
2.2.3. Feature Selection and Classification
2.2.4. Statistical Analyses
3. Results
- Slope of the MSE curve between scale τ = 1 and scales τ = 2 (Slp1-2), τ = 3 (Slp1-3), τ = 4 (Slp1-4), τ = 5 (Slp1-5) and τ = 6 (Slp1-6). It is estimated as the slope of the straight-line connecting the MSE values of the time scales under study. Higher slope accounts for a larger entropy increase between the original signal (τ = 1) and coarse-grained versions in consecutive short time scales (τ = 2 to 6), i.e., the control mechanisms regulating peripheral blood oxygen saturation on such short time scales are the most affected by recurrent apnoeic events.
- Individual SampEn values from scale τ = 1 to scale τ = 6 (SE1 to SE6). Single-scale SampEn is a measure of entropy or disorderliness and thus larger individual values are linked with more complex underlying mechanisms governing the dynamics of the oximetric signal for these time scales.
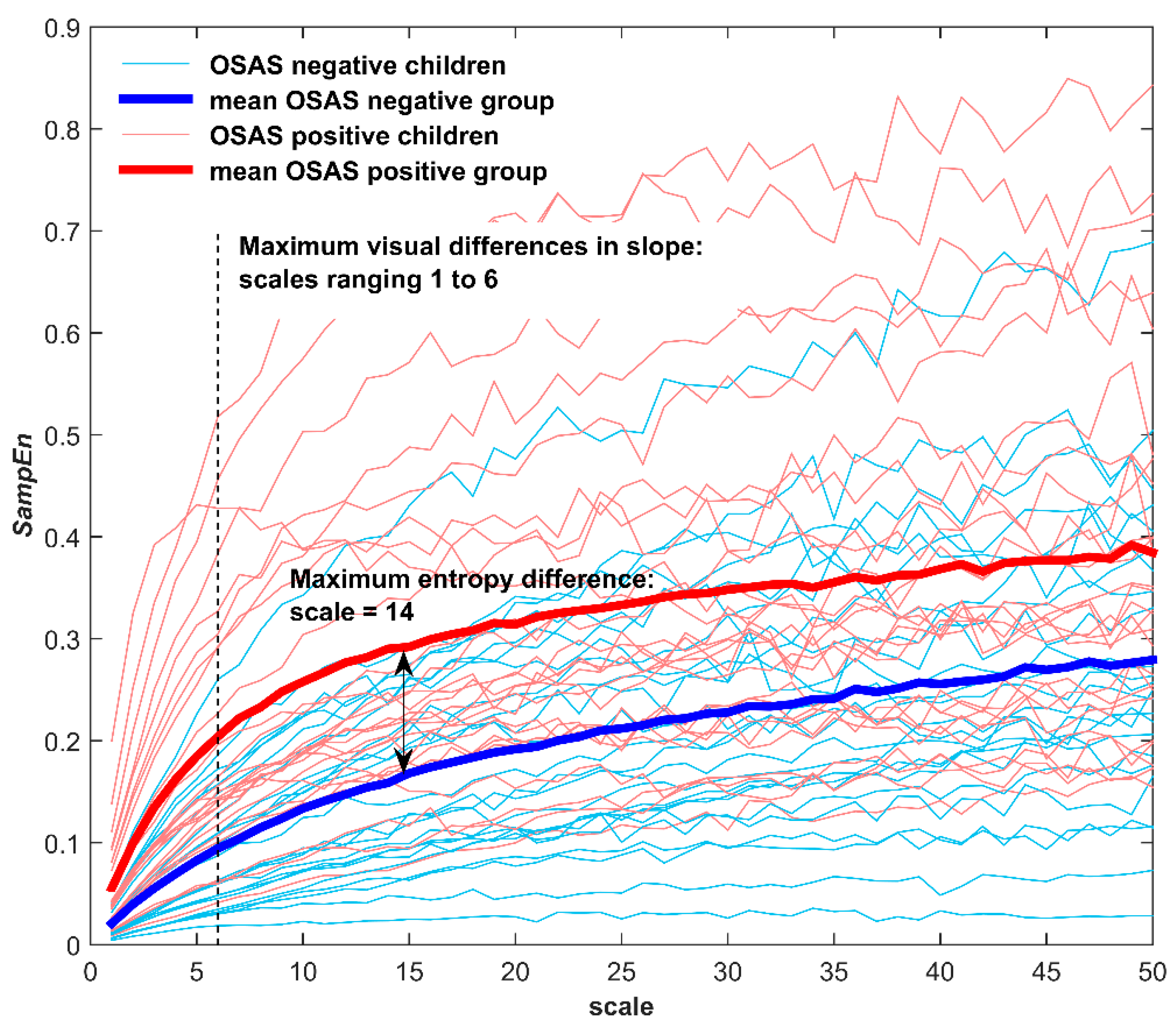
- SampEn single value in the scale reaching the maximum margin between MSE curves of the groups under study, i.e., τ = 14 (SEmax). This feature quantifies the irregularity of the oximetric recording for the time scale where the maximum difference between the classes under study (OSAS-negative vs. OSAS-positive) is expected.
- Area enclosed under the MSE curve between scale τ = 1 and scales τ = 2 (Ar1-2), τ = 4 (Ar1-4) and τ = 6 (Ar1-6). MSE curves allow us to compare the relative complexity of time series [33]. Higher area is achieved when SampEn values are higher for the majority of the time scales, suggesting that the time series is more complex.
- Area enclosed under the MSE curve between scale τ = 1 and the scale reaching the maximum margin (τ = 14) between the averaged MSE curves (Ar1-max). After time scale τ = 14, the MSE curves of OSAS-negative and OSAS-positive groups monotonically increase with a similar slope, showing almost equal behaviour. From short time scales to scale τ = 14, the MSE curves of both groups show the greatest differences regarding shape and individual entropy values. Thus, this feature gathers the contribution of the time scales showing the maximum differences in the dynamics of nocturnal oximetry between the groups under study.
- Time scale where the maximum SampEn value is reached (τmax). This feature is related to the level of depth of changes in the underlying complexity of the signal, i.e., it shows the time scale up to which entropy increases.
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chang, L.; Wu, J.; Cao, L. Combination of symptoms and oxygen desaturation index in predicting childhood obstructive sleep apnea. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Marcus, C.L.; Brooks, L.J.; Ward, S.D. Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 2012, 130, 714–755. [Google Scholar] [CrossRef] [PubMed]
- Capdevila, O.S.; Kheirandish-Gozal, L.; Dayyat, E.; Gozal, D. Pediatric obstructive sleep apnea: Complications, management, and long-term outcomes. Proc. Am. Thorac. Soc. 2008, 5, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Kheirandish-Gozal, L. What is “abnormal” in pediatric sleep? Respir. Care 2010, 55, 1366–1376. [Google Scholar] [PubMed]
- Tarasiuk, A.; Simon, T.; Tal, A.; Reuveni, H. Adenotonsillectomy in children with obstructive sleep apnea syndrome reduces health care utilization. Pediatrics 2004, 113, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Kadmon, G.; Shapiro, C.M.; Chung, S.A.; Gozal, D. Validation of a pediatric obstructive sleep apnea screening tool. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 1461–1464. [Google Scholar] [CrossRef] [PubMed]
- Lesser, D.J.; Haddad, G.G.; Bush, R.A.; Pian, M.S. The utility of a portable recording device for screening of obstructive sleep apnea in obese adolescents. J. Clin. Sleep Med. 2012, 8, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Katz, E.S.; Ron, B.M.; D'ambrosio, C.M. Obstructive sleep apnea in infants. Am. J. Respir. Crit. Care Med. 2012, 185, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Álvarez, M.L.; Terán-Santos, J.; Cordero-Guevara, J.A. Reliability of respiratory polygraphy for the diagnosis of sleep apnea-hypopnea syndrome in children. Arch. Bronconeumol. 2008, 44, 318–323. [Google Scholar] [CrossRef]
- Kaditis, A.G.; Alonso-Alvarez, M.L.; Boudewyns, A. Obstructive sleep disordered breathing in 2–18 year-old children: Diagnosis and management. Eur. Respir. J. 2016, 47, 69–94. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Álvarez, M.L.; Terán-Santos, J.; Ordax-Carbajo, E. Reliability of home respiratory polygraphy for the diagnosis of sleep apnea in children. Chest 2015, 147, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Brockmann, P.E.; Schaefer, C.; Poets, A.; Poets, C.F.; Urschitz, M.S. Diagnosis of obstructive sleep apnea in children: A systematic review. Sleep Med. Rev. 2013, 17, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Álvarez, M.L.; Navazo-Egüia, A.I.; Cordero-Guevara, J.A.; Ordax-Carbajo, E.; de La Mata, G.; Barba-Cermeño, J.L.; Terán-Santos, J. Respiratory polygraphy for follow-up of obstructive sleep apnea in children. Sleep Med. 2012, 13, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Brouillette, R.T.; Morielli, A.; Leimanis, A.; Waters, K.A.; Luciano, R.; Ducharme, F.M. Nocturnal pulse oximetry as an abbreviated testing modality for pediatric obstructive sleep apnea. Pediatrics 2000, 105, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Kaditis, A.; Kheirandish-Gozal, L.; Gozal, D. Pediatric OSAS: Oximetry can provide answers when polysomnography is not available. Sleep Med. Rev. 2016, 27, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Garde, A.; Dehkordi, P.; Karlen, W.; Wensley, D.; Ansermino, J.M.; Dumont, G.A. Development of a screening tool for sleep disordered breathing in children using the Phone OximeterTM. PLoS ONE 2014, 9, e112959. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.; de Chazal, P. Automated detection of sleep apnea in infants: A multi-modal approach. Comput. Biol. Med. 2015, 63, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Tobal, G.C.; Alonso-Álvarez, M.L.; Álvarez, D.; del Campo, F.; Terán-Santos, J.; Hornero, R. Diagnosis of pediatric obstructive sleep apnea: Preliminary findings using automatic analysis of airflow and oximetry recordings obtained at patients’ home. Biomed. Signal Process Control 2015, 18, 401–407. [Google Scholar] [CrossRef]
- Álvarez, D.; Alonso-Álvarez, M.L.; Gutiérrez-Tobal, G.C.; Crespo, A.; Kheirandish-Gozal, L.; Hornero, R.; Gozal, D.; Terán-Santos, J.; Del Campo, F. Automated screening of children with obstructive sleep apnea using nocturnal oximetry: An alternative to respiratory polygraphy in unattended settings. J. Clin. Sleep Med. 2017, 13, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Kirk, V.G.; Bohn, S.G.; Flemons, W.W.; Remmers, J.E. Comparison of home oximetry monitoring with laboratory polysomnography in children. Chest 2003, 124, 1702–1708. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.; Kang, C.; Su, M. Usefulness of desaturation index for the assessment of obstructive sleep apnea syndrome in children. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 1286–1290. [Google Scholar] [CrossRef] [PubMed]
- Pincus, S.M. Assessing serial irregularity and its implications for health. Ann. N. Y. Acad. Sci. 2001, 954, 245–267. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, 2039–2049. [Google Scholar]
- Cohen, M.E.; Hudson, D.L.; Deedwania, P.C. Applying continuous chaotic modeling to cardiac signals analysis. IEEE Eng. Med. Biol. 1996, 15, 97–102. [Google Scholar] [CrossRef]
- Stam, C.J. Nonlinear dynamical analysis of EEG and MEG: Review of an emerging field. Clin. Neurophysiol. 2005, 116, 2266–2301. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, D.; Hornero, R.; Abásolo, D.; del Campo, F.; Zamarrón, C. Nonlinear characteristics of blood oxygen saturation from nocturnal oximetry for obstructive sleep apnoea detection. Physiol. Meas. 2006, 27, 399–412. [Google Scholar] [CrossRef] [PubMed]
- Hornero, R.; Álvarez, D.; Abásolo, D.; del Campo, F.; Zamarrón, C. Utility of approximate entropy from overnight pulse oximetry data in the diagnosis of the obstructive sleep apnea syndrome. IEEE Trans. Biomed. Eng. 2007, 54, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, F.; Hornero, R.; Zamarrón, C.; Abasolo, D.E.; Álvarez, D. Oxygen saturation regularity analysis in the diagnosis of obstructive sleep apnea. Artif. Intell. Med. 2006, 37, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, D.; Hornero, R.; Marcos, J.V.; del Campo, F. Multivariate analysis of blood oxygen saturation recordings in obstructive sleep apnea diagnosis. IEEE Trans. Biomed. Eng. 2010, 57, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, D.; Hornero, R.; Marcos, J.V.; del Campo, F. Feature selection from nocturnal oximetry using genetic algorithms to assist in obstructive sleep apnoea diagnosis. Med. Eng. Phys. 2012, 34, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, D.; Hornero, R.; Marcos, J.V.; Wessel, N.; Penzel, T.; Glos, M.; del Campo, F. Assessment of feature selection and classification approaches to enhance information from overnight oximetry in the context of sleep apnea diagnosis. Int. J. Neural. Syst. 2013, 23, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Marcos, J.V.; Hornero, R.; Nabney, I.T.; Álvarez, D.; Gutiérrez-Tobal, G.C.; del Campo, F. Regularity analysis of nocturnal oximetry recordings to assist in the diagnosis of sleep apnoea syndrome. Med. Eng. Phys. 2016, 38, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.D.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef] [PubMed]
- Vanderlei, F.M.; Vanderlei, L.C.M.; de Abreu, L.C.; Garner, D.M. Entropic analysis of HRV in obese children. Int. Arch. Med. 2015, 8, 1–9. [Google Scholar] [CrossRef]
- Costa, M.D.; Peng, C.K.; Goldberger, A.L. Multiscale analysis of heart rate dynamics: Entropy and time irreversibility measures. Cardiovasc. Eng. 2008, 8, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.D.; Peng, C.K.; Goldberger, A.L.; Hausdorff, J.M. Multiscale entropy analysis of human gait dynamics. Phys. A 2003, 330, 53–60. [Google Scholar] [CrossRef]
- Escudero, J.; Abásolo, D.; Hornero, R.; Espino, P.; López, M. Analysis of electroencephalograms in Alzheimer's disease patients with multiscale entropy. Physiol. Meas. 2006, 27, 1091–1106. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Cho, R.Y.; Mizuno, T.; Kikuchi, M.; Murata, T.; Takahashi, K.; Wada, Y. Antipsychotics reverse abnormal EEG complexity in drug-naive schizophrenia: A multiscale entropy analysis. Neuroimage 2010, 51, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, C.S.; Placek, M.M.; Czosnyka, M.; Cabella, B.; Kasprowicz, M.; Austin, T. Complexity of brain signals is associated with outcome in preterm infants. J. Cereb. Blood Flow Metab. 2017, in press. [Google Scholar]
- Pan, W.; Su, M.; Wu, H.; Su, T.; Lin, M.; Sun, C. Multiscale entropic assessment of autonomic dysfunction in patients with obstructive sleep apnea and therapeutic impact of continuous positive airway pressure treatment. Sleep Med. 2016, 20, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Roebuck, A.; Clifford, G.D. Comparison of standard and novel signal analysis approaches to obstructive sleep apnea classification. Front. Bioeng. Biotech. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Tobal, G.C.; Álvarez, D.; Gomez-Pilar, J.; del Campo, F.; Hornero, R. Assessment of time and frequency domain entropies to detect sleep apnoea in heart rate variability recordings from men and women. Entropy 2015, 17, 123–141. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J. Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.L.; Quan, S.F. The AASM Manual for the Scoring of Sleep and Associated Events. Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression; Wiley: London, UK, 2000. [Google Scholar]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of complex physiologic time series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Tobal, G.C.; Hornero, R.; Álvarez, D.; Marcos, J.V.; del Campo, F. Linear and nonlinear analysis of airflow recordings to help in sleep apnoea–hypopnoea syndrome diagnosis. Physiol. Meas. 2012, 33, 1261–1275. [Google Scholar] [CrossRef] [PubMed]
- Witten, I.H.; Frank, E.; Hall, M.A. Data Mining Practical Machine Learning Tools and Techniques; Morgan Kaufmann/Elsevier: Burlington, MA, USA, 2011. [Google Scholar]
- Sahadan, D.Z.; Davey, M.J.; Horne, R.S.; Nixon, G.M. Improving detection of obstructive sleep apnoea by overnight oximetry in children using pulse rate parameters. Sleep Breath 2015, 19, 1409–1414. [Google Scholar] [CrossRef] [PubMed]
- Gil, E.; Bailón, R.; Vergara, J.M.; Laguna, P. PTT variability for discrimination of sleep apnea related decreases in the amplitude fluctuations of PPG signal in children. IEEE Trans. Biomed. Eng. 2010, 57, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Lázaro, J.; Gil, E.; Vergara, J.M.; Laguna, P. Pulse rate variability analysis for discrimination of sleep-apnea-related decreases in the amplitude fluctuations of pulse photoplethysmographic signal in children. IEEE J. Biomed. Health Inform. 2014, 18, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Li, X.; Guo, X.; Qin, J.; Li, S. A simple diagnostic scale based on the analysis and screening of clinical parameters in paediatric obstructive sleep apnoea hypopnea syndrome. J. Laryngol. Otol. 2017, 131, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Morillo, D.; López-Gordo, M.A.; León, A. Novel multiclass classification for home-based diagnosis of sleep apnea hypopnea syndrome. Expert Syst. Appl. 2014, 41, 1654–1662. [Google Scholar] [CrossRef]
- Marcos, J.V.; Hornero, R.; Álvarez, D.; del Campo, F.; Zamarrón, C.; López, M. Utility of multilayer perceptron neural network classifiers in the diagnosis of the obstructive sleep apnoea syndrome from nocturnal oximetry. Comput. Meth. Programs Biomed. 2008, 92, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Marcos, J.V.; Hornero, R.; Álvarez, D.; Nabney, I.T.; del Campo, F.; Zamarrón, C. The classification of oximetry signals using Bayesian neural networks to assist in the detection of obstructive sleep apnoea syndrome. Physiol. Meas. 2010, 31, 375–394. [Google Scholar] [CrossRef] [PubMed]
- Karandikar, K.; Le, T.Q.; Sa-ngasoongsong, A.; Wongdhamma, W.; Bukkapatnam, S.T. Detection of sleep apnea events via tracking nonlinear dynamic cardio-respiratory coupling from electrocardiogram signals. In Proceedings of the 6th International IEEE/EMBS Conference on Neural Engineering, San Diego, CA, USA, 6–8 November 2013; pp. 1358–1361. [Google Scholar]

| All Children | OSAS-Negative | OSAS-Positive | p-Value | |
|---|---|---|---|---|
| No. Subjects (%) | 50 | 24 (48.0%) | 26 (52.0%) | - |
| Age (years) | 4 [4, 6] | 4.5 [4, 6] | 4 [4, 6] | N.S. |
| No. Males (n) | 27 (54.0%) | 11 (45.8%) | 16 (61.5%) | N.S. |
| BMI (kg/m2) | 16.42 [15.00, 17.53] | 16.42 [15.61, 17.42] | 16.38 [14.57, 17.70] | N.S. |
| OAHI (events/h) | 3.56 [1.21, 17.28] | 1.18 [0.54, 1.87] | 15.88 [6.72, 23.49] | <<0.05 |
| Recording time (h) | 9.05 [8.40, 9.27] | 9.0 [8.74, 9.22] | 9.08 [8.28, 9.56] | N.S. |
| ODI3 (events/h) | 1.89 [5.19] | 0.87 [1.48] | 5.90 [7.26] | <<0.05 |
| SatMIN (%) | 90 [4] | 91 [2] | 89 [3] | <0.05 |
| SatAVG (%) | 97 [2] | 97 [1] | 97 [2] | N.S. |
| CT95 (%) | 0.82 [3.00] | 0.36 [1.10] | 1.62 [6.61] | <0.05 |
| MSE Features | OSAS-Negative | OSAS-Positive | p-Value |
|---|---|---|---|
| Slp1-2 | 0.015 [0.017] | 0.031 [0.041] | <<0.05 |
| Slp1-3 | 0.028 [0.031] | 0.057 [0.070] | <<0.05 |
| Slp1-4 | 0.038 [0.044] | 0.079 [0.094] | <<0.05 |
| Slp1-5 | 0.049 [0.059] | 0.096 [0.111] | <<0.05 |
| Slp1-6 | 0.058 [0.073] | 0.110 [0.128] | <0.05 |
| SE1 | 0.016 [0.019] | 0.039 [0.055] | <<0.05 |
| SE2 | 0.032 [0.036] | 0.071 [0.097] | <<0.05 |
| SE3 | 0.045 [0.050] | 0.097 [0.126] | <<0.05 |
| SE4 | 0.055 [0.063] | 0.118 [0.149] | <<0.05 |
| SE5 | 0.067 [0.078] | 0.135 [0.166] | <<0.05 |
| SE6 | 0.075 [0.092] | 0.147 [0.175] | <<0.05 |
| SEmax | 0.129 [0.147] | 0.229 [0.216] | <0.05 |
| Ar1-2 | 0.048 [0.055] | 0.110 [0.152] | <<0.05 |
| Ar1-4 | 0.151 [0.168] | 0.324 [0.427] | <<0.05 |
| Ar1-6 | 0.292 [0.338] | 0.599 [0.767] | <<0.05 |
| Ar1-max | 1.164 [1.382] | 2.114 [2.463] | <<0.05 |
| τmax | 48.000 [3.500] | 48.000 [5.000] | N.S. |
| Se (%) | Sp (%) | PPV (%) | NPV (%) | LR+ | LR- | Acc (%) | AUC | |
|---|---|---|---|---|---|---|---|---|
| Slp1-2 | 75.6 (47.7, 98.3) | 67.8 (36.6, 96.3) | 72.5 (50.5, 96.8) | 72.6 (48.3, 97.7) | 2.83 (1.30, 6.71) | 0.37 (0.03, 0.77) | 71.8 (56.8, 86.5) | 0.79 (0.62, 0.94) |
| Slp1-3 | 78.1 (49.4, 98.5) | 68.1 (38.4, 95.8) | 73.1 (51.2, 96.4) | 74.9 (49.7, 98.0) | 2.89 (1.34, 6.71) | 0.33 (0.02, 0.72) | 73.2 (58.8, 88.2) | 0.80 (0.63, 0.94) |
| Slp1-4 | 78.6 (50.6, 98.5) | 68.0 (37.6, 96.2) | 73.2 (51.4, 96.5) | 75.1 (49.6, 98.1) | 2.94 (1.36, 7.07) | 0.32 (0.02, 0.72) | 73.4 (57.7, 88.6) | 0.80 (0.64, 0.95) |
| Slp1-5 | 76.1 (48.7, 98.4) | 66.6 (35.7, 95.7) | 71.8 (48.7, 96.2) | 72.6 (46.9, 97.6) | 2.71 (1.26, 6.10) | 0.37 (0.03, 0.81) | 71.4 (55.2, 86.4) | 0.78 (0.61, 0.94) |
| Slp1-6 | 76.6 (45.8, 97.6) | 66.5 (35.7, 95.6) | 71.8 (48.8, 95.9) | 72.8 (47.6, 97.1) | 2.72 (1.23, 6.33) | 0.36 (0.03, 0.81) | 71.6 (55.2, 87.4) | 0.77 (0.60, 0.92) |
| SE1 | 75.2 (47.4, 98.6) | 70.2 (36.0, 96.9) | 74.0 (51.4, 97.3) | 73.0 (48.3, 98.0) | 3.10 (1.35, 7.43) | 0.36 (0.02, 0.76) | 72.7 (57.6, 88.6) | 0.81 (0.65, 0.95) |
| SE2 | 75.0 (48.4, 98.7) | 68.7 (36.6, 96.6) | 73.0 (50.9, 96.8) | 72.6 (48.3, 98.1) | 2.92 (1.34, 6.99) | 0.37 (0.02, 0.77) | 71.9 (56.8, 87.2) | 0.80 (0.64, 0.95) |
| SE3 | 76.9 (49.1, 98.3) | 68.9 (39.1, 96.3) | 73.3 (51.4, 96.7) | 74.0 (48.6, 97.8) | 2.96 (1.34, 6.92) | 0.34 (0.02, 0.74) | 72.9 (58.0, 88.2) | 0.81 (0.64, 0.94) |
| SE4 | 77.3 (50.1, 98.5) | 69.4 (36.6, 96.5) | 73.9 (51.7, 96.8) | 74.6 (48.7, 97.9) | 3.06 (1.35, 7.14) | 0.33 (0.02, 0.74) | 73.4 (57.6, 88.2) | 0.80 (0.64, 0.95) |
| SE5 | 76.4 (49.1, 98.7) | 67.9 (36.1, 96.1) | 72.8 (50.5, 96.6) | 73.3 (48.3, 98.1) | 2.88 (1.30, 6.81) | 0.35 (0.02, 0.76) | 72.2 (56.8, 87.0) | 0.79 (0.63, 0.94) |
| SE6 | 75.8 (49.9, 97.7) | 67.1 (36.2, 96.1) | 72.0 (48.6, 96.5) | 72.3 (47.9, 97.2) | 2.79 (1.25, 6.63) | 0.37 (0.03, 0.82) | 71.5 (55.2, 87.0) | 0.78 (0.61, 0.93) |
| SEmax | 74.3 (39.4, 98.7) | 61.6 (31.7, 96.0) | 68.3 (46.0, 95.7) | 70.7 (43.4, 97.8) | 2.26 (1.13, 5.49) | 0.42 (0.02, 0.95) | 68.1 (52.3, 83.6) | 0.75 (0.58, 0.91) |
| Ar1-2 | 74.8 (48.4, 98.7) | 68.8 (34.8, 96.7) | 73.1 (50.7, 96.8) | 72.5 (48.3, 98.1) | 2.94 (1.31, 7.03) | 0.37 (0.02, 0.78) | 71.8 (55.2, 87.2) | 0.80 (0.64, 0.95) |
| Ar1-4 | 76.1 (49.1, 98.6) | 68.5 (36.6, 96.5) | 73.0 (51.3, 96.8) | 73.4 (48.3, 98.0) | 2.92 (1.33, 7.07) | 0.35 (0.02, 0.76) | 72.3 (57.8, 87.2) | 0.80 (0.64, 0.95) |
| Ar1-6 | 75.1 (49.2, 98.6) | 68.7 (36.9, 96.5) | 73.0 (50.5, 96.8) | 72.5 (47.9, 98.0) | 2.94 (1.30, 7.05) | 0.37 (0.02, 0.78) | 71.9 (55.5, 87.2) | 0.80 (0.64, 0.94) |
| Ar1-max | 75.5 (47.4, 98.3) | 67.1 (35.0, 96.2) | 72.0 (49.1, 96.3) | 72.3 (47.8, 97.3) | 2.75 (1.26, 6.47) | 0.37 (0.03, 0.82) | 71.3 (55.2, 86.4) | 0.78 (0.61, 0.93) |
| τmax | 57.0 (21.2, 98.3) | 55.0 (20.3, 89.2) | 58.3 (31.9, 84.0) | 56.6 (29.0, 96.8) | 1.47 (0.64, 3.59) | 0.81 (0.04, 1.94) | 55.8 (39.6, 71.8) | 0.60 (0.51, 0.76) |
| Se (%) | Sp (%) | PPV (%) | NPV (%) | LR+ | LR- | Acc (%) | AUC | |
|---|---|---|---|---|---|---|---|---|
| ODI3 | 71.9 (44.9, 97.6) | 77.6 (40.7, 100) | 79.7 (55.1, 100) | 72.6 (49.7, 97.3) | 3.97 (1.50, 11.20) | 0.36 (0.03, 0.70) | 74.5 (58.5, 88.9) | 0.85 (0.70, 0.97) |
| SatMIN | 54.2 (20.3, 97.8) | 64.2 (21.2, 98.2) | 64.8 (34.7, 97.3) | 58.4 (30.8, 96.1) | 1.96 (0.72, 5.62) | 0.72 (0.05, 1.44) | 58.8 (42.9, 73.6) | 0.62 (0.51, 0.79) |
| SatAVG | 69.4 (34.2, 96.7) | 66.8 (39.5, 95.9) | 69.8 (45.1, 95.6) | 68.1 (42.1, 96.0) | 2.41 (1.07, 5.40) | 0.46 (0.05, 0.94) | 68.1 (49.9, 82.9) | 0.70 (0.52, 0.88) |
| CT95 | 66.8 (34.2, 96.3) | 69.3 (35.9, 98.6) | 72.0 (48.3, 98.2) | 66.4 (42.3, 95.4) | 2.63 (1.14, 8.02) | 0.49 (0.06, 0.97) | 67.8 (52.3, 83.0) | 0.75 (0.57, 0.92) |
| Se (%) | Sp (%) | PPV (%) | NPV (%) | LR+ | LR- | Acc (%) | AUC | |
|---|---|---|---|---|---|---|---|---|
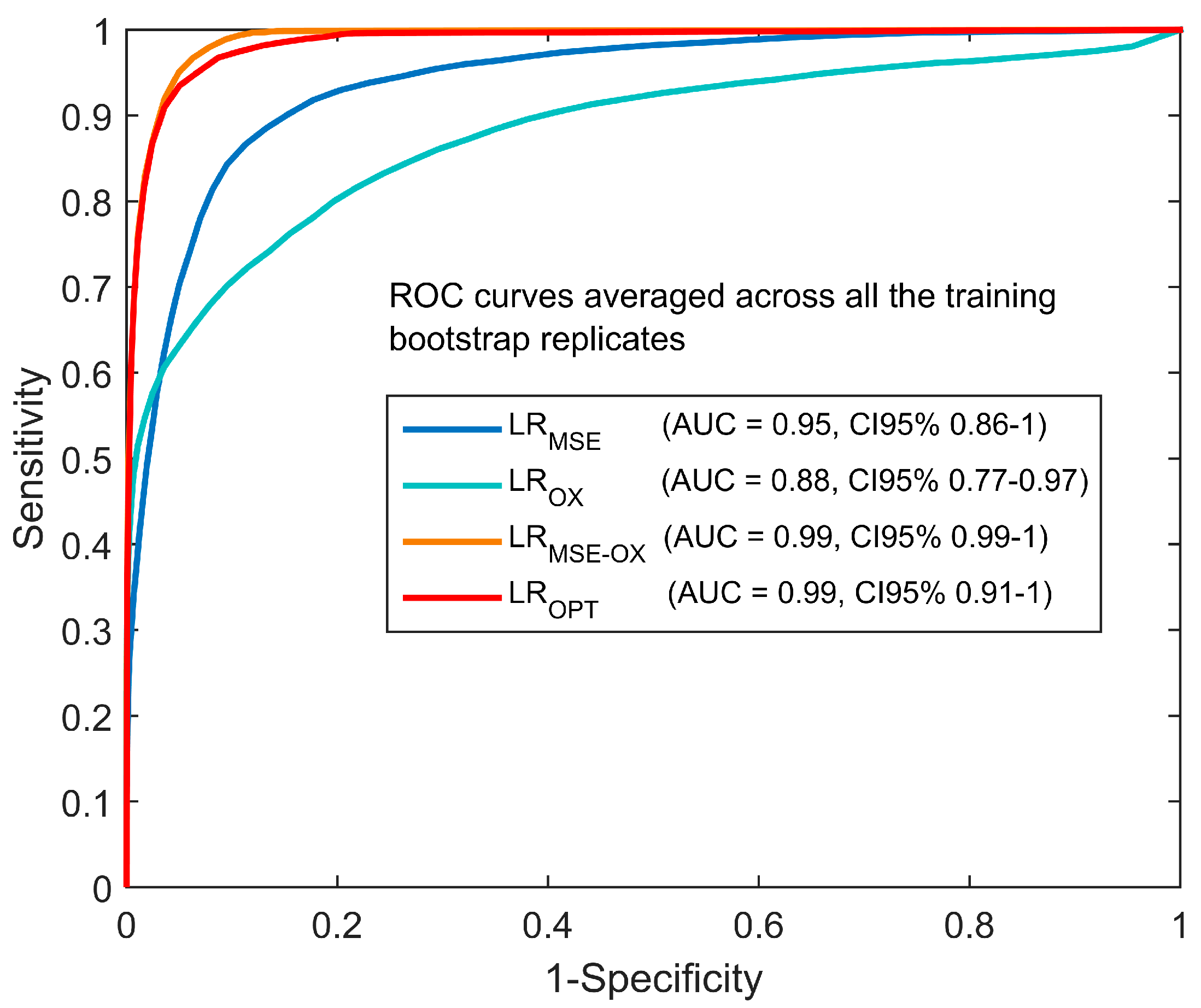
| LRMSE | 75.7 (49.0, 100) | 75.3 (43.4, 100) | 77.5 (53.6, 100) | 74.4 (48.7, 100) | 5.95 (1.78, 13.41) | 0.38 (0, 0.93) | 75.2 (57.0, 90.0) | 0.79 (0.58, 0.95) |
| LROX | 74.7 (47.3, 98.8) | 77.7 (44.4, 1) | 79.7 (55.9, 1) | 74.2 (49.9, 98.7) | 4.76 (1.62, 12.92) | 0.35 (0.01, 0.75) | 76.0 (58.8, 90.4) | 0.82 (0.64, 0.97) |
| LRMSE-OX | 79.4 (54.9, 1) | 79.3 (55.3, 1) | 80.8 (62.1, 1) | 78.4 (58.5, 1) | 6.65 (2.56, 14.39) | 0.34 (0, 0.92) | 79.0 (63.1, 92.8) | 0.80 (0.62, 0.95) |
| LROPT | 84.5 (60.1, 1) | 83.0 (54.2, 1) | 84.7 (61.6, 1) | 83.3 (58.7, 1) | 7.81 (1.58, 15.05) | 0.23 (0, 0.70) | 83.5 (63.2, 96.1) | 0.86 (0.65, 0.99) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crespo, A.; Álvarez, D.; Gutiérrez-Tobal, G.C.; Vaquerizo-Villar, F.; Barroso-García, V.; Alonso-Álvarez, M.L.; Terán-Santos, J.; Hornero, R.; Campo, F.d. Multiscale Entropy Analysis of Unattended Oximetric Recordings to Assist in the Screening of Paediatric Sleep Apnoea at Home. Entropy 2017, 19, 284. https://doi.org/10.3390/e19060284
Crespo A, Álvarez D, Gutiérrez-Tobal GC, Vaquerizo-Villar F, Barroso-García V, Alonso-Álvarez ML, Terán-Santos J, Hornero R, Campo Fd. Multiscale Entropy Analysis of Unattended Oximetric Recordings to Assist in the Screening of Paediatric Sleep Apnoea at Home. Entropy. 2017; 19(6):284. https://doi.org/10.3390/e19060284
Chicago/Turabian StyleCrespo, Andrea, Daniel Álvarez, Gonzalo C. Gutiérrez-Tobal, Fernando Vaquerizo-Villar, Verónica Barroso-García, María L. Alonso-Álvarez, Joaquín Terán-Santos, Roberto Hornero, and Félix del Campo. 2017. "Multiscale Entropy Analysis of Unattended Oximetric Recordings to Assist in the Screening of Paediatric Sleep Apnoea at Home" Entropy 19, no. 6: 284. https://doi.org/10.3390/e19060284
APA StyleCrespo, A., Álvarez, D., Gutiérrez-Tobal, G. C., Vaquerizo-Villar, F., Barroso-García, V., Alonso-Álvarez, M. L., Terán-Santos, J., Hornero, R., & Campo, F. d. (2017). Multiscale Entropy Analysis of Unattended Oximetric Recordings to Assist in the Screening of Paediatric Sleep Apnoea at Home. Entropy, 19(6), 284. https://doi.org/10.3390/e19060284









