Abstract
In some unique cases, liquids can divert from pure isotropy due to the formation of ordered molecular assemblies with acquired “negative entropy” and information storage. The energy stored in such ordered domains can be combined with an independent quantitative parameter related to the degree of order, which can then translate the dormant information to the quantitative energetic term “information capacity”. Information storage in liquids can be thus expressed in absolute energy units. Three liquid systems are analyzed in some detail. The first is a solution of a chiral substance, e.g., amino acid in water, where the degree of optical rotation provides the measure for order while the heat liberated upon racemization is the energy corresponding to the negative entropy. The second is a neat chiral fluid, e.g., 2-butanol, complying with the same parameters as those of chiral solutions. The third is electronically excited fluorescent solute, where the shift in the emission spectrum corresponds to the energy acquired by the transiently oriented solvent envelopes. Other, yet unexplored, possibilities are also suggested.
1. Introduction
In a large collection of particles, deviation from randomness can store information which upon transmission can be implemented for energy saving [1,2]. Information in such assemblies corresponds to a pattern of organization and is not an energy term on its own. In liquids or gases, microscopic reversible intermolecular condensations can take place spontaneously, for example, at the freezing or boiling points. In such cases deviation from homogeneity corresponds to a defined energetic process, i.e., an enthalpy feature, which does not include quantitative information. There are, however, a few cases in liquids where specific configurations are formed just on the basis of organization, i.e., an entropy feature, irrespective of intermolecular binding energies. Information storage in such cases is discussed in this article.
2. Negative Entropy Residing in Molecular Assemblies
The term negative entropy, or “negentropy”, is somewhat ambiguous. It was initially introduced [2,3,4] to describe a diversion from isotropy in a large collection of particles (e.g., molecules in the gaseous phase) and in some way it is opposite to entropy, the basic factor which corresponds to order dissipation in spontaneous processes. The absolute values of entropy, S, and negative entropy, SI, are essentially “floating terms” and in principle can serve only as base lines for directional processes like those associated with transmission of information [3,5]. Statistical mechanics is a leading tool in this direction which can be also applied in statistical thermodynamics of gases and liquids at their steady state [5,6]. In the most common cases, where thermodynamic values are presented as the difference between two states of equilibrium , the absolute temperature, T, and its energy coefficients k (per molecule) or R (per mole), provide absolute scales for the energy involved in such transitions. The quantitative measures for the two types of entropy then become TΔS and TΔSI.
3. Information Capacity in Fluids
The free molecular motion in liquids and gases at their steady state, is characterized by isotropic molecular distribution and orientation. The information, I, stored in such systems can be regarded as occupying the basal value I0. In cases of liquids with ordered molecular assemblies prevailing at their steady state, the stored information of I > I0 is correlated with the acquired negative entropy, –ΔSI = I − I0. The latter can be presented analogously to ΔS of classical thermodynamics, namely in units of J·mole−1·K−1. The corresponding energy stored in the negative entropy domains is then TΔSI, which has been defined as information capacity [7,8]. Quantitative evaluation of ΔSI entails an introduction of an experimental parameter which corresponds to the prevailing order. Subsequently, the information capacity, TΔSI, can be evaluated.
In fluid systems of large number of particles, order W, and information, I, are virtual terms which are proportional to each other, namely, W/W0 = I/I0. Both can be presented in terms of diversion from isotropy [1,2,3,4,5,6], as expressed in the fundamental Boltzmann Equation:
SI = RlnW = Rln I
The relation between information and entropy, presented in Equation 1 [4], can provide a basis for integration of information into classical thermodynamics of fluids, where systems under two states of equilibrium with different molecular anisotropy are evaluated energetically. Equation 1 then turns into the classical comparative scale presented in Equation 2:
where I0 is the basal information at complete isotropy. Insertion of ordered regions into the liquid, i.e., induction of anisotropy, negative entropy is acquired due to I > I0 as expressed in Equation 2 and energy amounting to –TΔSI is then accumulated:
–ΔSI = R ln W/W0 = R ln I/I0
–TΔSI = RT lnW/W0 = RTln I/I0
Alternative presentation can be based on the other edge of the scale, where order reaches its maximal value [7]:
–TΔSI = RT lnWmax/(Wmax – I) = RT ln Imax/(Imax – I)
4. Information Storage in Solvent Envelopes
Homogeneous fluids can accommodate assemblies of defined order, which in principle, store negative entropy. The most common cases are homogeneous solutions, where the solvent molecules coat the solute in an ordered configuration. The energy stored in such assemblies is provided by the liberated free energy in the initial process of solubilization. The experimental resolution of the entropic component in such assemblies can be achieved in only a few systems, most prominently in the solvent envelope which surrounds a chiral solute.
The solvent envelope is constructed of several consecutive layers. The immediate solvent—solute layer is dominated by electrostatic forces, van der Waals forces and hydrogen bonds amounting to enthalpies in the range of kJ per mole. The next layers, unlike the immediate one, can be viewed as being under mild structural deviation from the isotropy in the bulk. The acquired order in these layers has arisen on the expense of the initial heat of solubilization and can be therefore viewed as storing negative entropy. The energy stored in such structures, determined so far in just a few cases [7,8,9,10,11], is of only several Js per mole, i.e., about three orders of magnitude smaller than a mole of hydrogen bonds. The local topology of the solvent assembly around the solute molecules is therefore only marginally different than that in the bulk domain. It can be assessed in either enthalpy terms, namely as a distortion in the intermolecular bonds, or in terms of acquired local order, i.e., negative entropy. At such minute energetic changes the distinction between enthalpy and entropy is blurred and these terms become intermingled [12]. In practice, however, the acquired order in such assemblies is best inferred to in terms of negative entropy [7,8].
As outlined above, the ordered solvent molecules surrounding the solute can, in principle, be expressed in terms of negative entropy. However, the subsequent translation of the order in such assemblies to stored information capacity depends on the availability of an independent experimental or theoretical order parameter. In chiral solutions this value can be presented by the optical activity, as summarized in the following.
5. Chiral Solutions
The solvent envelope surrounding a chiral solute amounts, in general, to over 100 solvent molecules which adopt a chiral configuration [11,13,14]. The chiral topology of such envelopes can be of any asymmetry like a spiral, a ring with a chiral twist etc. In Figure 1, such a solvent configuration is presented by a model of a right handed Moebius ring.
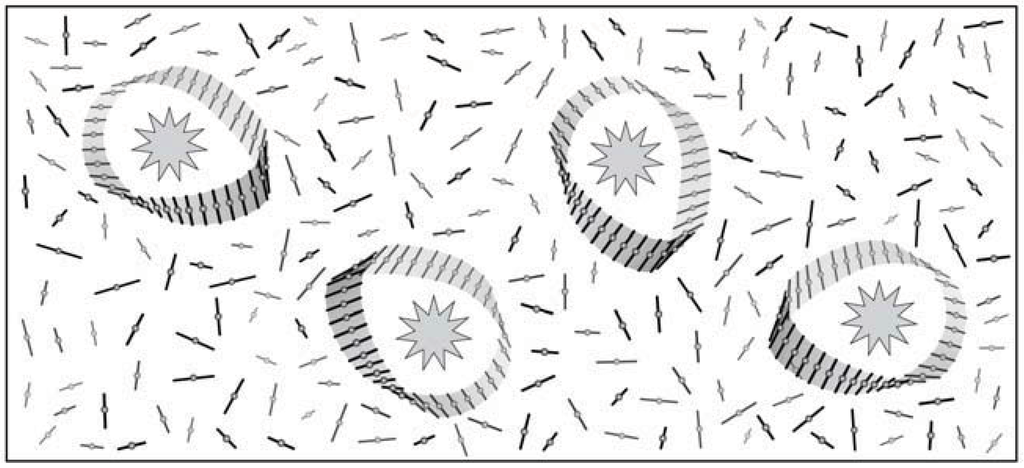
Figure 1.
Configuration of a right handed Moebius ring acquired by solvent molecules (Ι) surrounding a chiral solute (۞).
Figure 1.
Configuration of a right handed Moebius ring acquired by solvent molecules (Ι) surrounding a chiral solute (۞).
The degree of optical rotation, α, of a chiral solution provides an exclusive order parameter which, in principle, can be correlated with the acquired information, I. More specifically, the latter can range from I0 which corresponds to complete isotropy, as in racemic solutions, to Imax which corresponds to a saturated chiral solution. Accordingly:
where αmax corresponds to saturated solution. Equation 4 is then extended to:
I / Imax = α / αmax
–TΔSI = RT ln Imax/(Imax – I) = RT ln αmax / (αmax – α)
Under conditions where Imax >> I or αmax >> α, Equation 6 is reduced by a simple exponential approximation to the linear forms:
–TΔSI ≈ RT I/Imax = RT α / αmax
As such, quantification of information capacity, –TΔSI, in chiral solutions remains corresponding to the energy evolved in the loss of order in the system. Furthermore, TΔSI is expected to increase linearly with α, namely with concentration, a key point in its experimental verification [7].
Quantification of −TΔSI in chiral solutions can be achieved by heat release ensuing two independent processes; heat of dilution and heat of intermolecular racemization. However, these processes are, in general, of just a few Joules per mole, which fall near the border line of accuracy in conventional calorimetric measurements. With the availability of micocalorimetry instrumentations, in particular isothermal titration calorimetry, ITC [15,16], such processes can be now determined. These have up to now been applied to aqueous solutions of d- and l-amino acids and neat chiral liquids, which will be described below as specific examples.
The observed heat release upon dilution of concentrated chiral solutions with their solvent can be formally interpreted as the release of the excess chemical potential. In molecular terms, the solute molecules in “ideal” solutions are separated by the bulk solvent beyond the level that permits static or dynamic correspondence between neighboring envelope layers. In line with this view, at concentrations higher than the ideal solution, all solvent molecules in the bulk are at a state of mutual contact, leading to an overall ordered network of the solvent which deviates from the isotropy prevailing in its pure state. Furthermore, this putative overall degree of order increases with the solute concentration [7]. It has been thus shown, that heat release upon dilution of concentrated d- or l-alanine in water has a TΔSI component attributed to the disintegration of supramolecular chiral hydration assemblies [11].
The energy corresponding to TΔSI can be alternatively released upon intermolecular racemization by mixing solutions of opposite enantiomers [7]. As hypothesized above, the released heat, Q, upon dilution or intermolecular racemization of chiral solutions, corresponds predominantly to the dissipation of the order in the chiral domains:
where If is defined here as the "information coefficient". As a coefficient it provides a constant which converts a measurable parameter to a distinct physical value. In our case, it translates the apparent |α| value, or its change, into the stored thermal energy, in units of RT. It formally corresponds to the reciprocal of |α|max (see Equation 7) and can be derived from the slopes of the linear dependence of Q on |α|, which are obtained experimentally [7]. If values obtained for proline and alanine at different concentrations in water and their derived energy parameters, are presented in Table 1. As shown, this coefficient increases markedly with concentration. Other factors like temperature may also affect If.
Q = −TΔSI ≈ RT α / αmax = If RT |α|
The expressions for the change in entropy following the induction of order, presented in Equations 4–8, correspond to a sum-up of all domains acquired with anisotropic distribution. Its translation to information capacity, as described in Equation 8, has a priori an upper limit of RT, namely approx. 2.5 kJ per mole at room temperature. Values calculated for 1M solutions of proline and alanine, presented in Table 1, are as expected, below this limit, while the values deduced from experimental racemization, are markedly lower. One possible account for it is that upon intermolecular racemization the hydration coating of the individual molecules keeps retaining most of the chiral configuration of the initial hydration layer. An alternative explanation, which seems more plausible, is that in chiral solutions there are actually two distinct domains for information storage, the first corresponds to the chiral hydration, which is dissipated upon intermolecular racemization, and the other corresponds to the global asymmetry presented by the overall optical activity.

Table 1.
Information capacity of 1M proline and alanine in water, at 30° [7].
| Resident solute | Liberated heat upon point racemization, Q, (J·mole−1) | IfRT (J·mole−1 ·optdeg−1) | If (optdeg−1) | Information capacity, RT α/αmax, estimated (kJ·mole−1) |
|---|---|---|---|---|
| L-Proline | 19.2 | 18.0 | 7.1·10−3 | 0.3 |
| D-Proline | 26.4 | 22.6 | 9.0·10−3 | 0.3 |
| L-Alanine | 2.5 | 104.7 | 4.2·10−2 | 1.2 |
| D-Alanine | 2.6 | 108.9 | 4.3·10−2 | 1.2 |
Another particular information storage, corresponds to the somewhat unexpected chiral discrimination between solutions of opposite enantiomers [7,9,17] (compare, for example, the values presented in Table 1). It presumably originated from an intrinsic asymmetry between enantiomers which is extrapolated into their solutions [18]. Through such discrimination, one could ascribe absolute directions to right versus left in space [19], a long standing problem in space coordinates.
6. Neat Chiral Liquids
A pure liquid, undiluted, under conditions of a steady state is at a complete isotropy of molecular orientation, which can be defined as the state of minimal order. In homogeneous liquids of chiral molecules, integration of the molecular coordinates over the whole space yields an overall asymmetry, which is reflected in optical activity. In practice, however, it is also a state of minimal information, unless a selective abolishment of the chiral center can be achieved. An example for such a possibility is calorimetric evaluation of the energy associated in the hypothetical turning of liquid ethanol from the chiral form CH3CHD-OH, to its common non chiral form CH3CH2-OH.
Aliphatic secondary alcohols of four or more carbon atoms are amongst the most common chiral liquids. Calorimetric studies of mixing the apposite enantiomers of such alcohols was ensued by either heat consumption or heat release depending on the length of the aliphatic chain [20,21]. A detailed study with the chiral isomers of 2-butanol was recently carried out [10]. Introduction of neat S(+)-2-butanol into bulk neat R(−)-2-butanol, and vice versa, were associated with a relatively strong endothermic response which gradually decreased as the accumulated intermolecular racemization level increased. Formally speaking, the formation of R(−)-2-butanol envelope around the S(+)-2-butanol solute, and vice versa, are ensued by energy consumption (spontaneous endothermic reactions, like these ones, are driven by the overriding increase in entropy). This unique type of solvent envelope still retains the characteristics of negative entropy and is therefore rendered with information capacity.
7. Micellar Aggregates
Solutes which are in a dynamic equilibrium with their aggregates define a state of diversion from isotropy and as such can in principle acquire negative entropy. Micelles formed in water by amphipathic solutes are a typical example of such cases. Two counter-acting forces dominate their critical micellar concentration (CMC), the hydrophobic attraction between the long aliphatic chains and the interaction with water of the hydrophilic head groups. The micelles formed at concentrations above the CMC are surrounded with an ordered aqueous envelope which in principle acquires features of negative entropy. Yet, experimental translation of the latter to information capacity is hampered by the much greater magnitude of the enthalpies of the intermolecular interactions which override attempts to resolve it.
In the specific case of micelles formed by N-stearoyl l- or d-serine in water, a chiral surface composed of chiral spines was detected by a strong circular dichroism (CD) signal [17]. Such a distinct order could be implemented for resolution of information capacity.
8. Transient Information Acquired by Fluorescent Solutes
Fluorescent molecules in solution interact with the solvent molecules differently at their excited state. After electronic excitation, a rapid relaxation (~10−12 s) is ensued, trapping the excited fluorescent molecule in its lowest vibrational level of the first excited state for approximately 10−8 s. At this time interval, reorientation of the solvent envelope takes place. Immediately after emission of the fluorescence photon the solvent envelope relaxes back to its original configuration. This sequence of solvent reorientation upon electronic excitation is outlined in Figure 2 for the common case of a fluorescent aromatic molecule. As shown, the excited state is characterized (classically) by a transient dipole which induces relatively strong polarization in the surrounding solvent molecules. The ensuing electrostatic attraction between the excited molecule and the polarized surrounding solvent molecules is displayed by an energetic shift in the emission spectrum to longer wavelengths, i.e., lower energy. For example, in the case of tryptophan, a fluorescent amino acid, the maximum of the emission spectrum shifts from 320 nm to 355 nm when measured in a hydrophobic solvent and in water, respectively [22]. This typical 35 nm shift of the fluorescence spectrum, corresponds to 8 kcal/mole, which is invested in the strong electrostatic interaction of the excited molecules with water. A very small fraction of it, presumably in the range of several cal per mole, corresponds to TΔSI of the acquired order which precedes the full relaxation to the ground state. Thus, the instantaneous emission leaves the solvent coating around the previously excited molecule (d in Figure 2) in an ordered configuration which stores the equivalent information. This transient configuration relaxes spontaneously to the less ordered original configuration (a in Figure 2) by releasing heat amounting to TΔSI. Experimental resolution of this quantity encounters the high background of the simultaneous heat liberations (b to c in Figure 2) which are by far more intensive. It can, in principle, be approached by analyzing heat release ensuing a pulse excitation or pulse extinction of a steady state excitation.
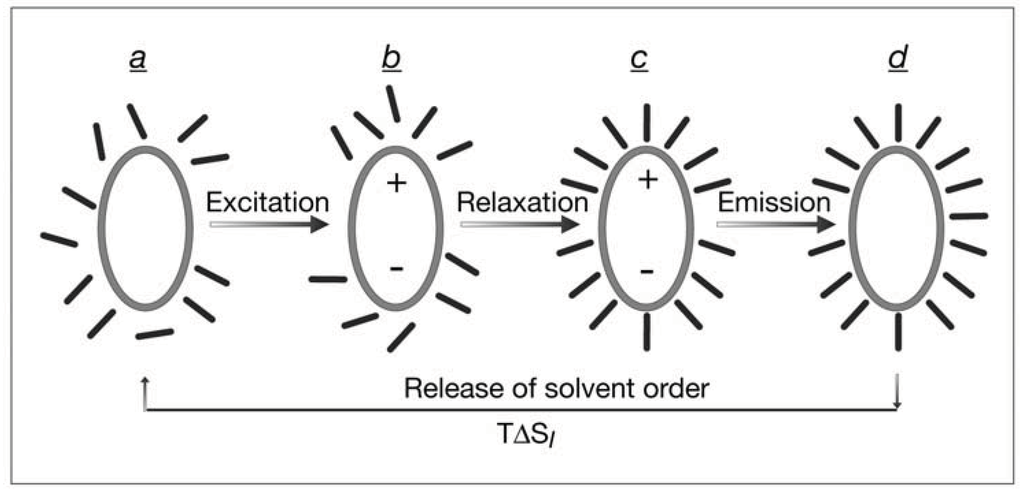
Figure 2.
Cycling of solvent configuration in a fluorescence process. A fluorescent solute, e.g., an aromatic molecule (a), is excited (b) and then relaxes to a polar excited state (c) which lives for about 10−8 s, when the surrounding solvent molecules reorient themselves. Upon fluorescence emission, the solvent shell transiently preserves an ordered configuration with an acquired information capacity (d) which relaxes spontaneously to the initial configuration.
Figure 2.
Cycling of solvent configuration in a fluorescence process. A fluorescent solute, e.g., an aromatic molecule (a), is excited (b) and then relaxes to a polar excited state (c) which lives for about 10−8 s, when the surrounding solvent molecules reorient themselves. Upon fluorescence emission, the solvent shell transiently preserves an ordered configuration with an acquired information capacity (d) which relaxes spontaneously to the initial configuration.
9. Chiral Conductivity
Thermodynamic processes which are restricted to two dimensions, e.g., on a membrane surface, or to one dimension along a wire, are of orders of magnitude more efficient than in the ordinary 3 dimensional realm [23]. Furthermore, in two dimensions and by far more so in one dimension, the reduction in degrees of freedom turn scalar values into directionality vectors, which may have enormous implications when acting in a three dimensional system like a biological tissue [24]. An example of such a case is presented in Figure 3, displaying two wires with opposite chiral twists, either left or right handed, acting in a three dimensional system. The additional degree of freedom which is added by the chiral elements in such wires, may impart a tunnel for transmission of specific information.
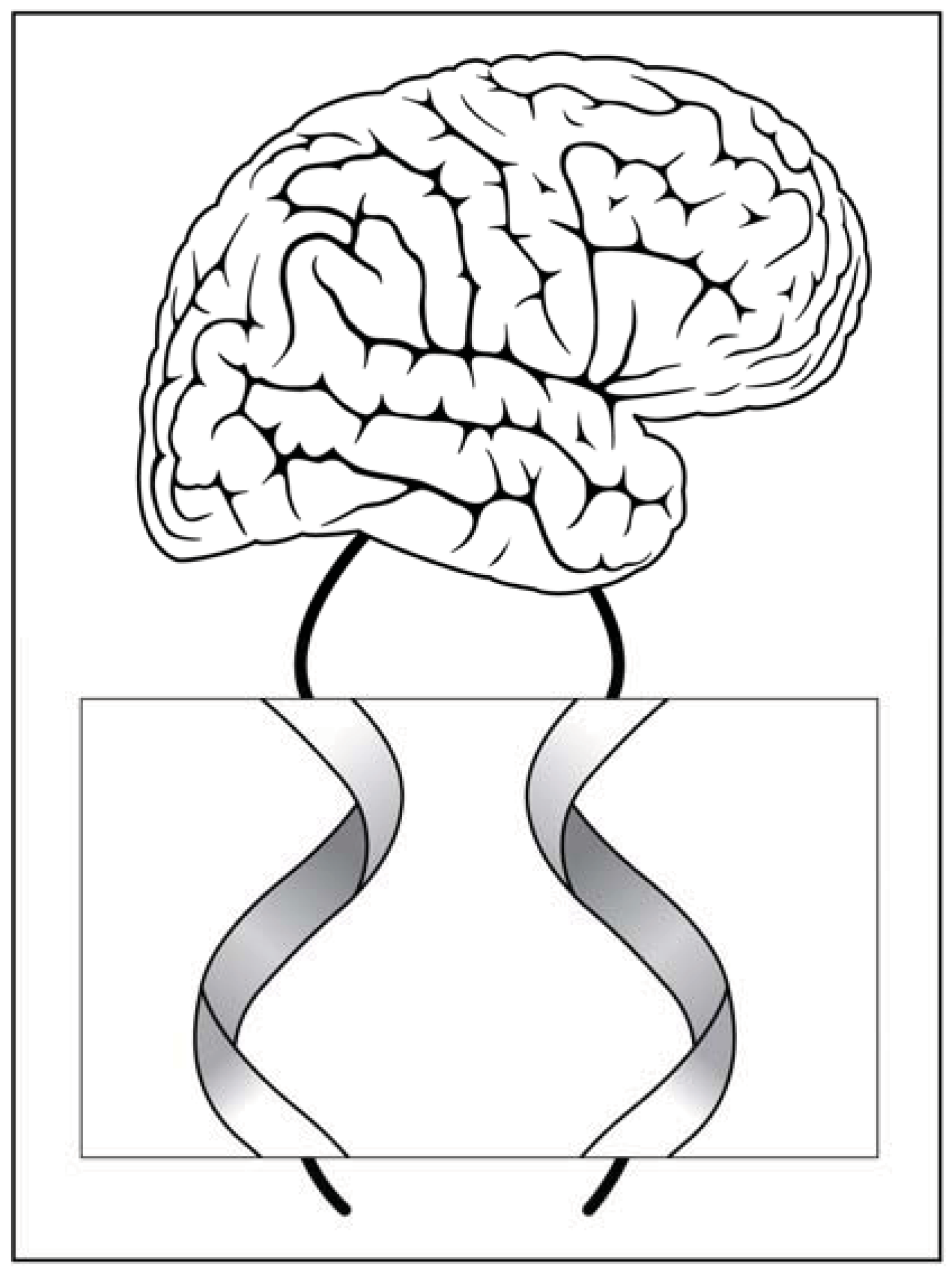
As an intriguing possibility, which is still merely a speculation, one can assume a nerve signal propagating to the brain along a fiber with a spiral structure like the ones presented in Figure 3. Remembering that the corresponding target tissues are also constructed from chiral elements, i.e., proteins, could this chiral mode of signal transmittance contribute additional putative information? Is there a distinct difference between the information provided by the right handed and left handed signals? Could these putative signals be implemented in the various enigmatic subtleties of brain function that we commonly attribute to emotional responses? Well, many other novel implications can be suggested for this unexplored virgin territory. In particular, assemblies of asymmetric objects like spiral screws or curly threads, which possess an overall asymmetry rendered with stored information [24].
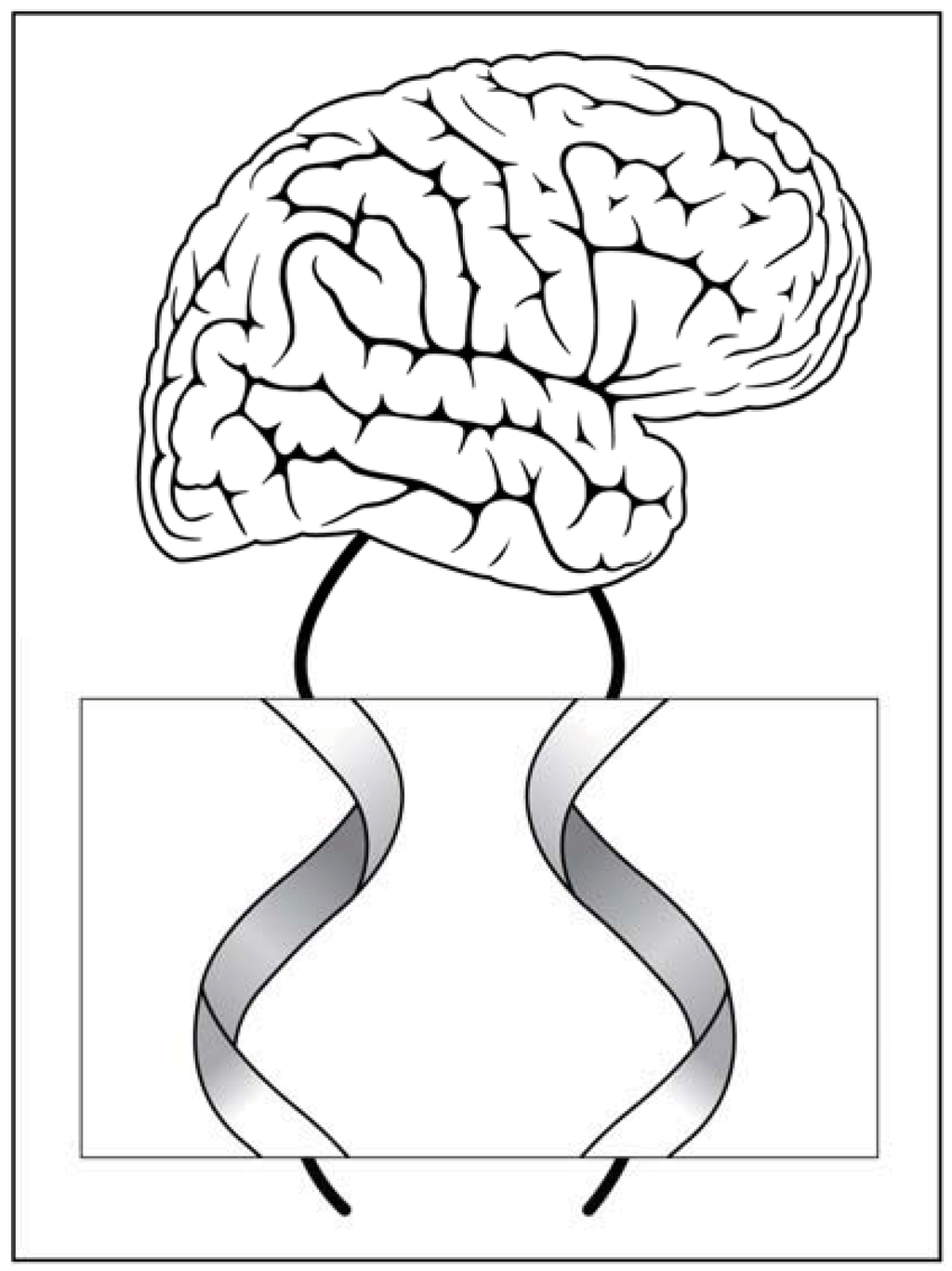
Figure 3.
Signal transmission along a right handed nerve fiber and a left handed nerve fiber which can be either wires or planar sheets (displayed in the magnified section).
Figure 3.
Signal transmission along a right handed nerve fiber and a left handed nerve fiber which can be either wires or planar sheets (displayed in the magnified section).
10. Information Processing
As outlined in this article, information in fluid domains is stored in molecular organizations which depart from isotropy, as for example in chiral solutions. Processing of such information is associated with liberation of thermal energy, corresponding to the stored information capacity. In line with classical thermodynamics, such information processing can proceed via pathways between two extremes. In the first, the processing is spontaneous, where the information storage is fully liberated as heat. In the second extreme, the processing is carried out in infinitesimal steps, where it is fully transferred as free energy to another system. An example, which is close to the latter, could be the process of enzymatic conversion of mono saccharides, like d-galactose to d-glucose. Here, the chiral configurations of the hydration layers which surround the product and the substrate, are different. Through the gradual process of conversion, their intermixing liberates the excess of information capacity which can be then absorbed as free energy, which is further implemented in the conversion process. It should be stressed, though, that the quantity of this energy is considerably smaller than the overall free energy associated in such processes. However, when an experimental information factor, like α, is monitored throughout, then the contribution of the information processing becomes apparent.
11. Conclusions
The popular statement that “information saves energy” is in general ascribed to routine activities, yet without any quantitative formulation. Level of information in model systems with patterns of order, were previously calculated, but have not reached the final goal of how to quantify the residing information in energy terms. In the special case of chiral solutions, the measurable parameters of optical rotation, heat of dilution and heat of intermolecular racemization, have been implemented, and thus yielded, for the first time, a quantitative assessment of the information capacity stored in such systems. The information capacities in other chiral systems, as well as in other non-random configurations acquired in homogeneous domains, remain at this stage just as qualitative aspects of stored information.
References
- Shannon, C.E.; Weaver, D.W. The Mathematical Theory of Communication; University of Illinois Press: Champaign, IL, USA, 1949. [Google Scholar]
- Brillouin, L. Science and Information Theory; Academic Press: New York, NY, USA, 1962. [Google Scholar]
- Brillouin, L. Negentropy principle of information. J. Appl. Phys. 1953, 24, 1152–1163. [Google Scholar] [CrossRef]
- Schrodinger, E. What Is Life? The Physical Aspect of Living Cell; Cambridge University Press: Cambridge, UK, 1945. [Google Scholar]
- Kondepudi, D. Non-equilibrium polymers, entropy and algorithmic information. In Complexity, Entropy and the Physics of Information; Zurek, W.H., Ed.; Addison-Wesley: Redwood City, CA, USA, 1990; pp. 199–206. [Google Scholar]
- Mahulikar, S.P.; Herwig, H. Exact thermodynamic principles for dynamic order existence and evolution in chaos. Chaos Sol. Frac. 2009, 41, 1939–1948. [Google Scholar] [CrossRef]
- Shinitzky, M.; Shvalb, A.; Elitzur, A.C.; Mastai, Y. Entrapped energy in chiral solutions: Quantification and information capacity. J. Phys. Chem. B. 2007, 111, 11004–11008. [Google Scholar] [CrossRef] [PubMed]
- Shinitzky, M. Configurational entropy in chiral solutions-Negative entropy of solvent envelopes. Entropy 2009, 11, 667–674. [Google Scholar] [CrossRef]
- Scolnik, Y.; Portnaya, I.; Cogan, U.; Tal, S.; Haimovitz, R.; Fridkin, M.; Elitzur, A.C.; Deamer, D.W.; Shinitzky, M. Subtle differences in structural transitions between poly L- and poly D-amino acids of equal length in water. Phys. Chem. Chem. Phys. 2006, 8, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Cogan, U.; Shpigelman, A.; Portnyana, I.; Rutenberg, A.; Scolnik, Y.; Shinitzky, M. Intermolecular chiral assemblies in R(−) and S(+) 2-butanol detected by microcalorimetry measurements. Chirality 2010, in press. [Google Scholar]
- Shvalb, A.; Mastai, Y.; Shinitzky, M. Chiral configuration of the hydration layers of D- and L-alanine in water implied from dilution calorimetry. Chirality 2010, 22, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.; Johnson, C.M.; Lackely, J.H.; Nollmann, M. Heat does not come in different colours: Entropy-enthalpy compensation, free energy windows, quantum confinement, pressure perturbation calorimetry, solvation and the multiple causes of heat capacity effects in biomolecular interactions. Biophys. Chem. 2001, 93, 215–230. [Google Scholar] [CrossRef]
- Clementi, E.; Cavallon, F.; Scordamaglia, R. Analytical potentials fromab initio computation for the interaction between biomolecules. 1. Water with amino acids. J. Am. Chem. Soc. 1977, 99, 5531–5545. [Google Scholar] [CrossRef] [PubMed]
- Run-Shen, C.; Xiang-Shan, N.; Xiu-Fan, S. Computer simulation of structuralorganization of water around biomolecules. In Water and Ions in Biological Systems; Pullman, A., Vasilescu, V., Packer, L., Eds.; Plenum: New York, NY, USA, 1983; pp. 137–149. [Google Scholar]
- Jelesarov, I.; Bosshard, H.R. Isothermal titration calorimetry and differential scanning calorimetry, as complementary tools to investigate the energetics of biomolecular recognition. J. Mol. Recog. 1999, 12, 3–18. [Google Scholar] [CrossRef]
- Weber, P.C.; Salemme, F.R. Applications of calorimetric methods to drug discovery and the study of protein interactions. Curr. Opin. Struc. Biol. 2003, 13, 115–121. [Google Scholar] [CrossRef]
- Shinitzky, M.; Haimovitz, R. Chiral surfaces in micelles of N-palmitoyl or N-stearoyl L-(or D-) serine. J. Am. Chem. Soc. 1993, 115, 12545–12549. [Google Scholar] [CrossRef]
- Deamer, D.A.; Dick, K.; Thiemann, W.; Shinitzky, M. Intrinsic asymmetries of amino acid enantiomers and their peptides: A possible role in the origin of biochirality. Chirality 2007, 19, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Ellizur, A.C.; Shinitzky, M. P-violation manifested at the molecular level—A simple means for an absolute definition of “left” vs. “right”. arXiv Chem. Phys. 2006. arXiv:physics/0601010v1. [Google Scholar]
- Kimura, T.; Khan, M.A.; Ishii, M.; Kamiyama, T. Enthalpy change on mixing a couple of S- and R- enantiomers of some chiral compounds at 298.15 K. Chirality 2006, 18, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Matsushita, T.; Ueda, K.; Aktar, F.; Matsuda, T.; Kamiyama, T.; Fujisawa, M. Enthalpic changes on mixing two couples of S- and R-enantiomers of heptane-2-ol, octane-2-ol, nonane-2-ol, 3-chloro-propane-1,2-diol, 2-methyl-1,4-butanediol at 298.15 K. Thermochim. Acta 2004, 414, 209–214. [Google Scholar] [CrossRef]
- Weber, G.; Teale, F.W.J. Ultraviolet fluorescence of the aromatic amino acids. Biochem J. 1957, 65, 476–482. [Google Scholar]
- Richter, P.H.; Eigen, M. Diffusion controlled reaction rates in spheroidal geometry. Application to repressor-operator association and membrane bound enzymes. Biophys. Chem. 1974, 2, 255–263. [Google Scholar] [CrossRef]
- Shinitzky, M. Symmetry, entropy and information capacity in chiral solutions. Symmetry Cult. Sci. 2010, 21, 17–26. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
