The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes
Abstract
1. Introduction
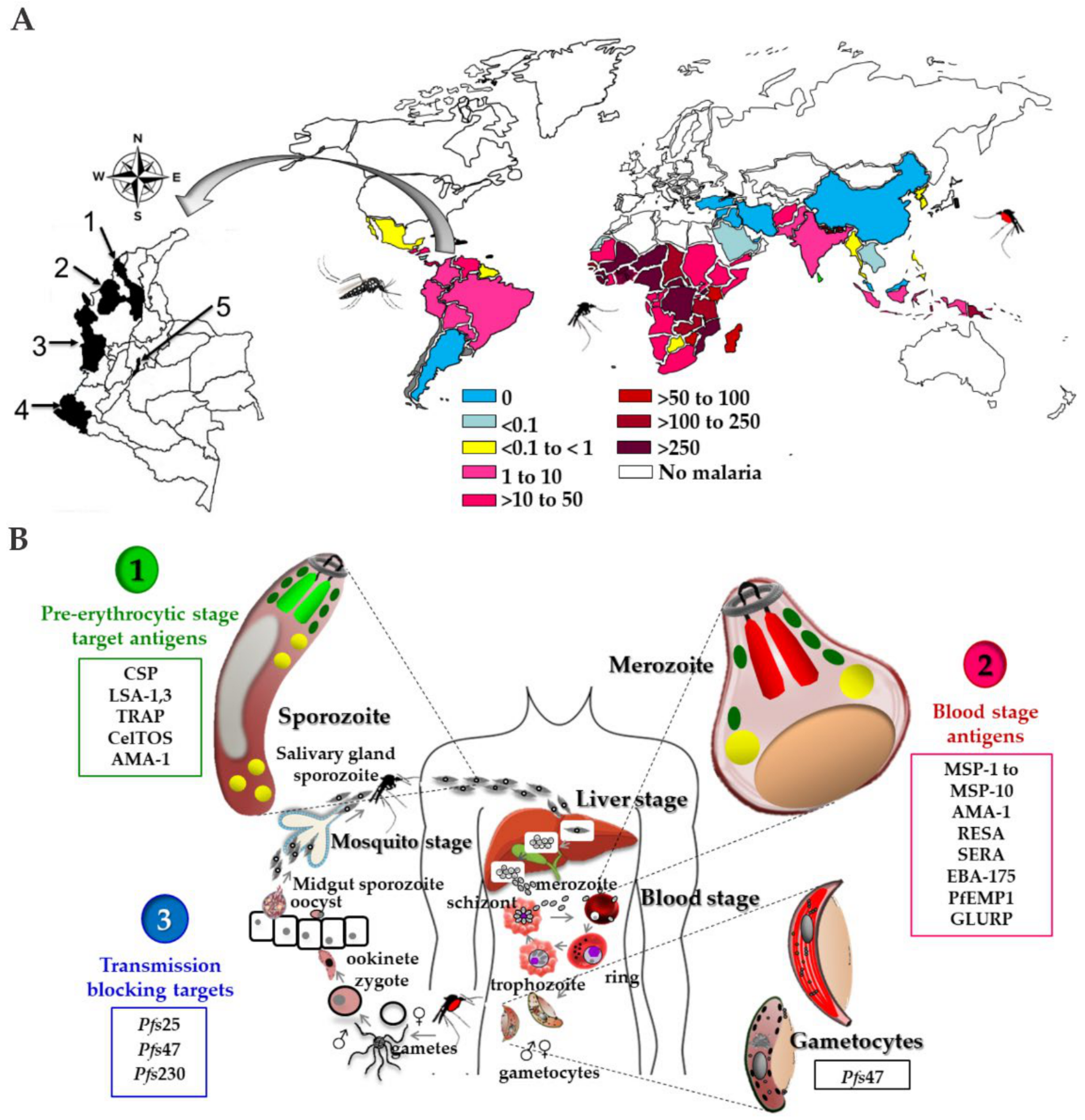
2. The Malaria Disease
2.1. Generalities
2.2. Plasmodium spp. Life Cycle
2.3. Aspects on the Biology of the Malaria Transmission Vector (Anopheles spp.)
2.4. Immune Response in Malaria
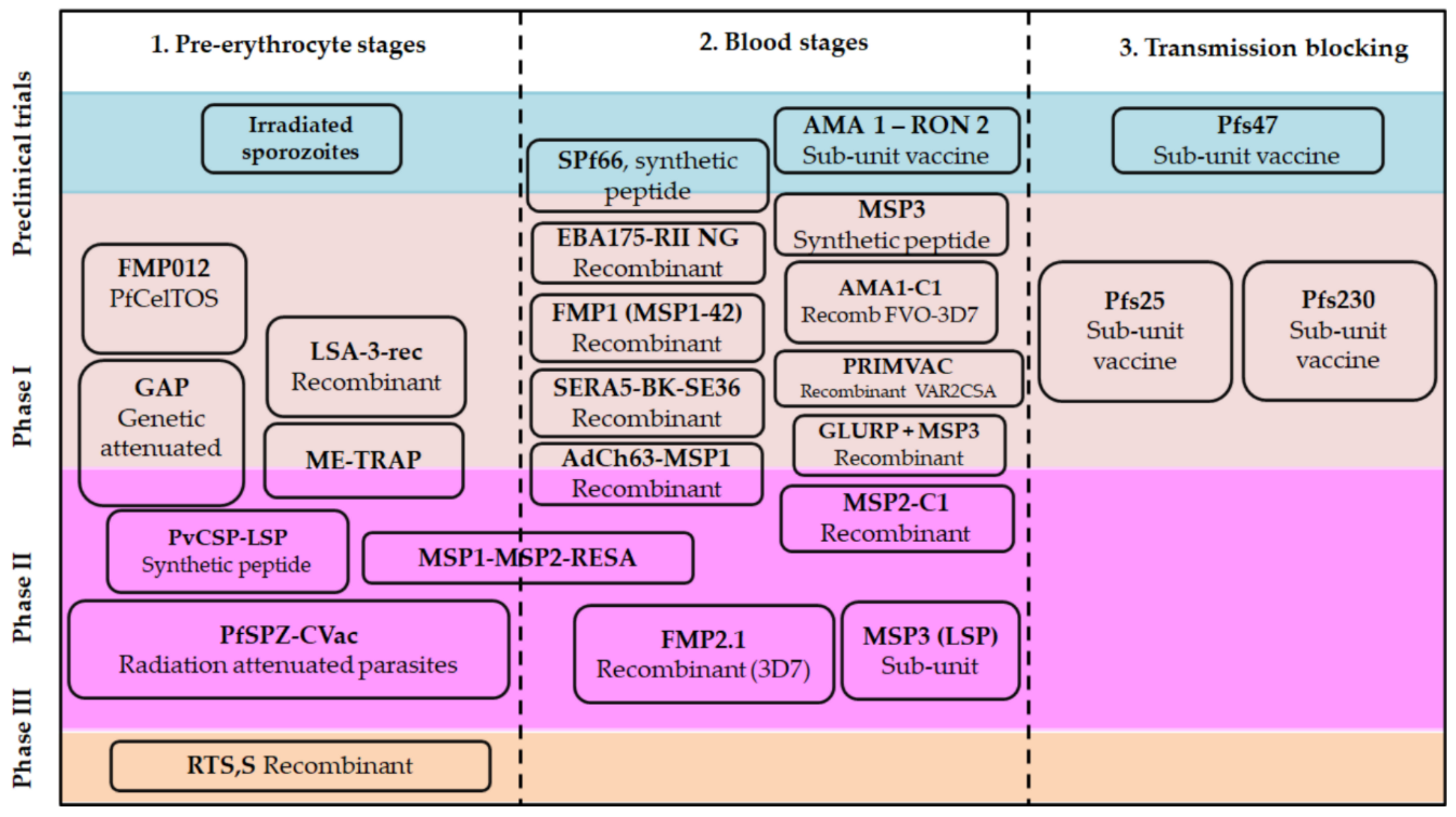
3. Classical Approaches for Malarial Vaccines
3.1. Adjuvants and Vaccine Formulations
- Immuno-potentiators (IP): Lymphocyte stimulation depends on the antigen presentation in the regional lymph nodes by antigen presenting cells.
- Delivery systems (DS): Delivery systems for vaccines are combinations of an immunogen with some carrier-like compounds that lead to forming particulate systems thereof such as emulsions, microparticles, ISCOMs, and liposomes among others, and mainly function to target associated antigens into antigen-presenting cells (APC), including macrophages and dendritic cells. DS require the release of cytokines in soluble form or of membrane co-stimulatory molecules in antigen-presenting cells.
- Immuno-polarizing agents (IPz): Adjuvants that polarize the immune response in a direction required for protection, towards a given Th1, Th2, Th9, TfH, Th17, Th22, or Treg pattern, determined by the released cytokine profile among other factors [81].
3.2. Plasmodium spp. Antigens Regarded as the Basis for a Vaccine Candidate
3.2.1. Candidates Based on Pre-Erythrocyte Stages
3.2.2. Plasmodium Blood Stages Antigens
3.2.3. Plasmodium Antigens Expressed in Merozoite Rhoptries and Micronems
3.2.4. Transmission-Blocking Vaccines
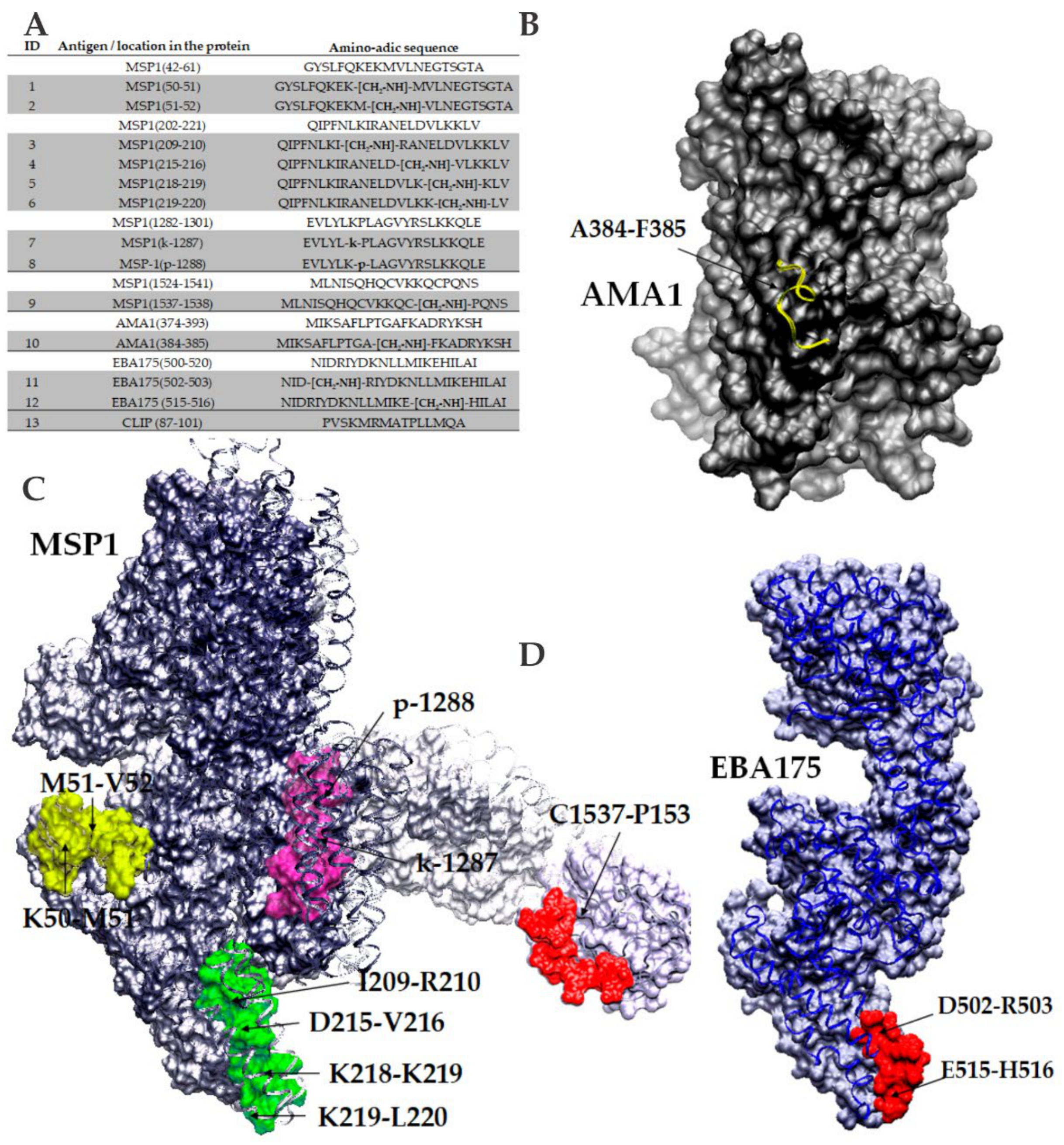
3.2.5. New Strategies for Immunopotentiation Antigenic Targets in Malaria
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. World Malaria Report 2020: 20 Years of Global Progress and Challenges; World Health Organization: Geneva, Switzerland, 2020; ISBN 978-92-4-001579-1. (electronic version). [Google Scholar]
- Santos, M.; Bouma, M.; Kohli, V.; Pascual, M. Population Density, Climate Variables and Poverty Synergistically Structure Spatial Risk in Urban Malaria in India. PLoS Negl. Trop. Dis. 2016, 10, e0005155. [Google Scholar] [CrossRef]
- Sonko, S.T.; Jaiteh, M.; Jafali, J.; Jarju, L.B.S.; D’Alessandro, U.; Camara, A.; Komma-Bah, M.; Saho, A. Does socio-economic status explain the differentials in malaria parasite prevalence? Evidence from The Gambia. Malar. J. 2014, 13, 449. [Google Scholar] [CrossRef]
- Tu, Y. The discovery of artemisinin (qinghaosu) and gifts from Chinese medicine. Nat. Med. 2011, 17, 1217–1220. [Google Scholar] [CrossRef]
- Esposito, S.; Bonanni, P.; Maggic, S.; Tand, L.; Ansaldi, F.; Lopalco, P.; Dagan, R.; Michel, J.; Damme, P.; Gaillat, J.; et al. Recommended immunization schedules for adults: Clinical practice guidelines by the Escmid Vaccine Study Group (EVASG), European Geriatric Medicine Society (EUGMS) and the World Association for Infectious Diseases and Immunological Disorders (WAidid). Hum. Vaccines Immunother. 2016, 12, 1777–1794. [Google Scholar] [CrossRef]
- Takashima, E.; Morita, M.; Tsuboi, T. Vaccine candidates for malaria: What’s new? Expert Rev. Vaccines 2016, 15, 1–3. [Google Scholar] [CrossRef][Green Version]
- Hoffman, S.; Vekemans, J.; Richie, T.; Duffy, P. The march toward malaria vaccines. Vaccine 2015, 33, D13–D23. [Google Scholar] [CrossRef]
- Dinko, B.; Pradel, G. Immune Evasion by Plasmodium falciparum Parasites: Converting a Host Protection Mechanism for the Parasite’s Benefit. Adv. Infect. Dis. 2016, 6, 82–95. [Google Scholar] [CrossRef][Green Version]
- Mueller, I.; Shakri, A.; Chitnis, C. Development of vaccines for Plasmodium vivax malaria. Vaccine 2015, 33, 7489–7495. [Google Scholar] [CrossRef] [PubMed]
- Payne, R.; Silk, S.; Elias, S.; Milne, K.; Rawlinson, T.; Llewellyn, D.; Shakri, R.; Jin, J.; Labbé, G.; Edwards, N.; et al. Human vaccination against Plasmodium vivax Duffy-binding protein induces strain-transcending antibodies. JCI Insight 2017, 2, 93683. [Google Scholar] [CrossRef]
- Tham, W.; Beeson, J.; Rayner, J. Plasmodium vivax vaccine research we’ve only just begun. Int. J. Parasitol. 2017, 47, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Grau, G.; Craig, A. Cerebral malaria pathogenesis: Revisiting parasite and host contributions. Future Microbiol. 2012, 7, 291–302. [Google Scholar] [CrossRef]
- World Health Organization. Available online: https://www.who.int/health-topics/malaria#tab=tab_1 (accessed on 28 December 2020).
- Battle, K.; Karhunen, M.; Bhatt, S.; Gething, P.; Howes, R.; Golding, N.; Van Boeckel, T.; Messina, J.; Shanks, D.; Smith, D.; et al. Geographical variation in Plasmodium vivax relapse. Malar. J. 2014, 13, 144. [Google Scholar] [CrossRef] [PubMed]
- Howes, R.; Battle, K.; Mendis, K.; Smith, D.; Cibulskis, R.; Baird, K.; I Hay, S. Global Epidemiology of Plasmodium vivax. Am. J. Trop. Med. Hyg. 2016, 95, 15–34. [Google Scholar] [CrossRef] [PubMed]
- Beeson, J.; Brown, G. Pathogenesis of Plasmodium falciparum malaria: The roles of parasite adhesion and antigenic variation. Cell. Mol. Life Sci. 2002, 59, 258–271. [Google Scholar] [CrossRef] [PubMed]
- Farooq, F.; Bergmann-Leitner, E. Immune Escape Mechanisms are Plasmodium’s Secret Weapons Foiling the Success of Potent and Persistently Efficacious Malaria Vaccines. J. Clin. Immunol. 2015, 161, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Aminake, M.; Pradel, G. Microbial pathogens and strategies for combating them: Science, technology and education. In Chapter: Antimalarial Drugs Resistance in Plasmodium Falciparum and the Current Strategies to Overcome Them; Vilas, A.M., Ed.; Formatex Research Center: Badajoz, Spain, 2013; pp. 269–282. ISBN 8493984396. [Google Scholar]
- Paaijmansa, P.; Blanford, S.; Bell, A.; Blanford, J.; Read, A.; Thomas, M. Influence of climate on malaria transmission depends on daily temperature variation. Proc. Natl. Acad. Sci. USA 2010, 107, 15135–15139. [Google Scholar] [CrossRef]
- Mota, M.; Pradel, G.; Vanderberg, J.; Hafalla, J.; Frevert, U.; Nussenzweig, R.; Nussenzweig, V.; Rodríguez, A. Migration of Plasmodium sporozoites through cells before infection. Science 2001, 291, 141–144. [Google Scholar] [CrossRef]
- Mishra, S.; Nussenzweig, R.; Nussenzweig, V. Antibodies to Plasmodium circumsporozoite protein (CSP) inhibit sporozoite’s cell traversal activity. J. Immunol. Methods 2012, 377, 47–52. [Google Scholar] [CrossRef]
- Doolan, D.; Hoffman, S. DNA-based vaccines against malaria: Status and promise of the multi-stage malaria DNA Vaccine Operation. Int. J. Parasitol. 2001, 31, 753–762. [Google Scholar] [CrossRef]
- O’Donnell, R.; Blackman, M. The role of malaria merozoite proteases in red blood cell invasion. Curr. Opin. Microbiol. 2005, 8, 422–427. [Google Scholar] [CrossRef]
- Beeson, J.; Drew, D.; Boyle, M.; Feng, G.; Fowkes, F.; Richards, J. Merozoite surface proteins in red blood cell invasion, immunity and vaccines against malaria. FEMS Microbiol. Rev. 2016, 40, 343–372. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.; Arumugam, T.; Reiling, L.; Heales, J.; Hooder, A.; Fowkes, F.; Cross, N.; Langer, C.; Takeo, S.; Uboldi, A.; et al. Identification and prioritization of merozoíte antigens as targets of protective human immunity to Plasmodium falciparum malaria for vaccine and biomarker development. J. Immunol. 2013, 191, 795–809. [Google Scholar] [CrossRef] [PubMed]
- Bannister, L.; Hopkins, J.; Fowler, R.; Krishna, S.; Mitchell, G. A brief illustrated guide to the ultrastructure of Plasmodium falciparum asexual blood stages. Parasitol. Today 2000, 16, 427–433. [Google Scholar] [CrossRef]
- Richie, T.; Saul, A. Progress and challenges for malaria vaccines. Nature 2002, 415, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Trager, W.; Jensen, J. Human malaria in continuous culture. Science 1976, 193, 673–675. [Google Scholar] [CrossRef]
- Billker, O.; Shaw, M.K.; Margos, G.; Sinden, R.E. The roles of temperature, pH and mosquito factors as triggers of male and female gametogenesis of Plasmodium berghei in vitro. Parasitology 1997, 115, 1–7. [Google Scholar] [CrossRef]
- Vlachou, D.; Zimmermann, T.; Cantera, R.; Janse, C.J.; Waters, A.P.; Kafatos, F.C. Real-time, in vivo analysis of malaria ookinete locomotion and mosquito midgut invasion. Cell. Microbiol. 2004, 6, 671–685. [Google Scholar] [CrossRef]
- Han, Y.S.; Thompson, J.; Kafatos, F.C.; Barillas-Mury, C. Molecular interactions between Anopheles stephensi midgut cells and Plasmodium berghei: The time bomb theory of ookinete invasion of mosquitoes. EMBO J. 2000, 19, 6030–6040. [Google Scholar] [CrossRef]
- Abraham, E.; Jacobs, M. Mosquito midgut barriers to malaria parasite development. Insect Biochem. Mol. Biol. 2004, 34, 667–671. [Google Scholar] [CrossRef]
- Smith, R.; Vega-Rodríguez, J.; Jacobs-Lorena, M. The Plasmodium bottleneck: Malaria parasite losses in the mosquito vector. Mem. Inst. Oswaldo Cruz 2014, 109, 644–661. [Google Scholar] [CrossRef]
- Coelho, C.H.; Jore, M.M.; Canepa, G.E.; Barillas-Mury, C.; Bousema, T.; Duffy, P.E. Antibody Therapy Goes to Insects: Monoclonal Antibodies Can Block Plasmodium Transmission to Mosquitoes. Trends Parasitol. 2020, 36, 880–883. [Google Scholar] [CrossRef] [PubMed]
- Aly, A.; Vaughan, A.; Kappe, S. Malaria Parasite Development in the Mosquito and Infection of the Mammalian Host. Annu. Rev. Microbiol. 2009, 63, 195–221. [Google Scholar] [CrossRef] [PubMed]
- Lopaticki, S.; Yang, A.; John, A.; Scott, N.; Lingford, J.; O’Neill, M. Protein O-fucosylation in Plasmodium falciparum ensures efficient infection of mosquito and vertebrate hosts. Nat. Commun. 2017, 8, 561. [Google Scholar] [CrossRef] [PubMed]
- Ukegbu, C.; Akinosoglou, K.; Christophides, G.; Vlachou, D. Plasmodium berghei PIMMS2 promotes ookinete invasion of the Anopheles gambiae mosquito midgut. Infect. Immun. 2017, 85, e00139-17. [Google Scholar] [CrossRef]
- Pimenta, P.; Orfano, A.; Bahia, A.; Duarte, A.; Ríos-Velásquez, C.; Melo, F. An overview of malaria transmission from the perspective of Amazon Anopheles vectors. Mem. Inst. Oswaldo Cruz 2015, 110, 23–47. [Google Scholar] [CrossRef]
- Arrighi, B.; Lycett, G.; Mahairaki, V.; Siden-Kiamos, I.; Louis, C. Laminin and the malaria parasite’s journey through the mosquito midgut. J. Exp. Biol. 2005, 208, 2497–2502. [Google Scholar] [CrossRef]
- Carter, V.; Nacer, A.; Underhill, A.; Sinden, R.; Hillary, H. Minimum requirements for ookinete to oocyst transformation in Plasmodium. Int. J. Parasitol. 2007, 37, 1221–1232. [Google Scholar] [CrossRef]
- Srinivasan, P.; Fujioka, H.; Jacobs-Lorena, M. PbCap380, a novel oocyst capsule protein, is essential for malaria parasite survival in the mosquito. Cell Microbiol. 2008, 10, 1304–1312. [Google Scholar] [CrossRef]
- Smith, C.; Barillas-Mury, C. Plasmodium Oocysts: Overlooked Targets of Mosquito Immunity. Trends Parasitol. 2016, 32, 979–990. [Google Scholar] [CrossRef]
- Sinka, M.; Rubio-Palis, Y.; Manguin, S.; Patil, A.; Temperley, W.; Gething, P.; Van Boeckel, T.; Kabaria, C.; Harbach, R.; I Hay, S. The dominant Anopheles vectors of human malaria in the Americas: Occurrence data, distribution maps and bionomic précis. Parasites Vectors 2010, 3. [Google Scholar] [CrossRef]
- Vallejo, A.; Rubiano, K.; Amado, A.; Krystosik, A.; Herrera, S.; Arévalo-Herrera, M. Optimization of a Membrane Feeding Assay for Plasmodium vivax Infection in Anopheles albimanus. PLoS Negl. Trop. Dis. 2016, 10, e0004807. [Google Scholar] [CrossRef] [PubMed]
- Sinden, R.; Carter, R.; Drakeley, C.; Leroy, D. The biology of sexual development of Plasmodium: The design and implementation of transmission-blocking strategies. Malar. J. 2012, 11. [Google Scholar] [CrossRef] [PubMed]
- Baton, L.; Warr, E.; Hoffman, S.A.; Dimopoulos, G. Programmed Cell Death during Malaria Parasite Infection of the Vertebrate Host and Mosquito Vector. In Programmed Cell Death in Protozoa. Molecular Biology Intelligence Unit; Springer: New York, NY, USA, 2008. [Google Scholar] [CrossRef]
- Hernández-Martínez, S.; Lanz-Mendoza, H.; Martínez-Barnetche, J.; Rodríguez, M. Antimicrobial properties of Anopheles albimanus pericardial cells. Cell Tissue Res. 2013, 351, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Hempel, P. Evolutionary ecology of insect immune defenses. Annu. Rev. Entomol. 2005, 50, 529–551. [Google Scholar] [CrossRef]
- Hernández-Martínez, S.; Cardoso-Jaime, V.; Nouzova, M.; Michalkova, V.; Ramirez, C.; Fernandez-Lima, F.; Noriega, F. Juvenile hormone controls ovarian development in female Anopheles albimanus mosquitoes. Sci. Rep. 2019, 9, 2127. [Google Scholar] [CrossRef]
- Erdman, L.; Finney, C.; Liles, W.; Kain, K. Inflammatory pathways in malaria infection: TLRs share the stage with other components of the innate immunity. Mol. Biochem. Parasitol. 2008, 162, 105–111. [Google Scholar] [CrossRef]
- Miller, J.; Brandon, S.; Baldwin, M.; Vaughan, A.; Kappe, S. Interferon-Mediated Innate Immune Responses against Malaria Parasite Liver Stages. Cell Rep. 2014, 7, 436–447. [Google Scholar] [CrossRef]
- Riley, E.; Stewart, A. Immune mechanisms in malaria: New insights in vaccine development. Nat. Med. 2013, 19, 168–178. [Google Scholar] [CrossRef]
- Sedegah, M.; Peters, B.; Hollingdale, M.R.; Ganeshan, H.D.; Huang, J.; Farooq, F.; Belmonte, M.N.; Belmonte, A.D.; Limbach, K.J.; Diggs, C.; et al. Vaccine Strain-Specificity of Protective HLA-Restricted Class 1 P. falciparum Epitopes. PLoS ONE 2016, 11, e0163026. [Google Scholar] [CrossRef]
- Bergmann-Leitner, E.; Duncan, E.; Angov, E. The impact of immune responses on the erythrocytic stages of Plasmodium and the implication for vaccine development. In Malaria Parasites; Okwa, O., Ed.; Intech-Open Access Publisher: Rijeka, Croatia, 2012; pp. 202–230. [Google Scholar] [CrossRef]
- Teo, A.; Feng, G.; Brown, G.; Beeson, J.; Rogerson, S. Functional Antibodies and Protection against Blood-stage Malaria. Trends Parasitol. 2016, 32, 887–898. [Google Scholar] [CrossRef]
- Malaguarnera, L.; Musumeci, S. The Immune Response to Plasmodium falciparum Malaria. Lancet Infect. Dis. 2002, 2, 472–478. [Google Scholar] [CrossRef]
- Kengne-Ouafo, J.A.; Sutherland, C.J.; Binka, F.N.; Awandare, G.A.; Urban, B.C.; Dinko, B. Immune Responses to the Sexual Stages of Plasmodium falciparum Parasites. Front. Immunol. 2019, 10, 136. [Google Scholar] [CrossRef] [PubMed]
- Coppi, A.; Pinzon-Ortiz, C.; Sinnis, H. The Plasmodium circumsporozoite protein is proteolytically processed during cell invasion. J. Exp. Med. 2005, 201, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Kurup, S.; Butler, N.; Harty, J. T cell-mediated immunity to malaria. Nat. Rev. Immunol. 2019, 19, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Van Braeckel-Budimir, N.; Kurup, S.; Harty, J.T. Regulatory issues in immunity to liver and blood-stage malaria. Curr. Opin. Immunol. 2016, 42, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Rénia, L.; Mattei, D.; Goma, J.; Pied, S.; Dubois, P.; Miltgen, F.; Nüssler, A.; Matile, H.; Menégaux, F.; Gentilini, M. A malaria heat-shock-like determinant expressed on the infected hepatocyte surface is the target of antibody-dependent cell-mediated cytotoxic mechanisms by nonparenchymal liver cells. Eur. J. Immunol. 1990, 20, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Belnoue, E.; Costa, F.; Frankenberg, T.; Vigário, A.; Voza, T.; Leroy, N.; Rodrigues, M.; Landau, I.; Snounou, G.; Rénia, L. Protective T cell immunity against malaria liver stage after vaccination with live sporozoites under chloroquine treatment. J. Immunol. 2004, 172, 2487–2495. [Google Scholar] [CrossRef]
- Rénia, M.; Marussig, M.; Grillot, D.; Pied, S.; Corradin, G.; Miltgen, F.; Del Giudice, G.; Mazier, D. In vitro activity of CD4+ and CD8+ T lymphocytes from mice immunized with a synthetic malaria peptide. Proc. Natl. Acad. Sci. USA 1991, 88, 7963–7967. [Google Scholar] [CrossRef]
- Gardner, M.; Hall, N.; Fung, E.; White, O.; Berriman, M.; Hyman, R.; Carlton, J.; Pain, A.; Nelson, K.; Bowman, S.; et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 2002, 419, 498–511. [Google Scholar] [CrossRef]
- Egan, J.E.; Hoffman, S.L.; Haynes, J.D.; Sadoff, J.C.; Schneider, I.; Grau, G.E.; Hollingdale, M.R.; Ballou, W.R.; Gordon, D.M. Humoral immune responses in volunteers immunized with irradiated Plasmodium falciparum sporozoites. Am. J. Trop. Med. Hyg. 1993, 49, 166–173. [Google Scholar] [CrossRef]
- Hoffman, S.; Goh, M.; Luke, T.; Schneider, I.; Le, T.; Doolan, D.; Sacci, J.; De la Vega, P.; Dowler, M.; Paul, C.; et al. Protection of humans against malaria by immunization with radiation-attenuated Plasmodium falciparum sporozoites. J. Infect. Dis. 2002, 185, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Sabchareon, A.; Burnouf, T.; Ouattara, D.; Attanath, P.; Bouharoun-Tayoun, H.; Chantavanich, P.; Foucault, C.; Chongsuphajaisiddhi, T.; Druilhe, P. Parasitologic and clinical human response to immunoglobulin administration in falciparum malaria. Am. J. Trop. Med. Hyg. 1991, 45, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Baird, J. Age-dependent characteristics of protection vs. susceptibility to Plasmodium falciparum. Ann. Trop. Med. Parasitol. 1998, 92, 367–390. [Google Scholar] [CrossRef] [PubMed]
- Doolan, D.; Dobaño, C.; Baird, K. Acquired Immunity to Malaria. Clin. Microbiol. Rev. 2009, 22, 13–36. [Google Scholar] [CrossRef] [PubMed]
- Vanderberg, J. Reflections on early malaria vaccine studies, the first successful human malaria vaccination, and beyond. Vaccine 2009, 27, 2–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Herrington, D.; Clyde, D.; Losonsky, G.; Cortesia, M.; Murphy, J.R.; Davis, J.; Baqar, S.; Felix, A.M.; Heimer, E.P.; Gillessen, D. Safety and immunogenicity in man of a synthetic peptide malaria vaccine against, Plasmodium falciparum sporozoites. Nature 1987, 328, 257–259. [Google Scholar] [CrossRef]
- Patarroyo, M.E.; Romero, P.; Torres, M.L.; Clavijo, P.; Moreno, A.; Martínez, A.; Rodríguez, R.; Guzman, F.; Cabezas, E. Induction of protective immunity against experimental infection with malaria using synthetic peptides. Nature 1987, 328, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Draper, S.; Angov, E.; Horiic, T.; Miller, L.; Srinivasan, P.; Theisen, M.; Biswas, S. Recent advances in recombinant protein-based malaria vaccines. Vaccine 2015, 33, 7433–7443. [Google Scholar] [CrossRef]
- Long, C.; Zavala, F. Malaria vaccines and human immune responses. Curr. Opin. Microbiol. 2016, 32, 96–102. [Google Scholar] [CrossRef]
- Falgunee, K.; Vasee, S. Plasmodium vivax Vaccine Research: Insights from Colombian Studies. Am. J. Trop. Med. Hyg. 2011, 84, 1–3. [Google Scholar] [CrossRef]
- Birkett, A. Status of vaccine research and development of vaccines for malaria. Vaccine 2016, 34, 2915–2920. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.; Doritchamou, Y.; Zaidi, I.; Duffy, P. Advances in malaria vaccine development: Report from the 2017 malaria vaccine symposium. NPJ Vaccines 2017, 2, 34. [Google Scholar] [CrossRef] [PubMed]
- Moreno, A.; Joyner, C. Malaria vaccine clinical trials: What’s on the horizont. Curr. Opin. Immunol. 2015, 35, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Coffman, R.; Sher, A.; Seder, R. Vaccine Adjuvants: Putting Innate Immunity to Work. Immunity 2010, 33, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Batista-Duharte, A.; Lastre, M.; Pérez, O. Adyuvantes inmunológicos. Determinantes en el balance eficacia-toxicidad de las vacunas contemporáneas. Enferm. Infecc. Microbiol. Clin. 2014, 32, 106–114. [Google Scholar] [CrossRef]
- Pérez, O.; Lastre, M.; Cabrera, O.; del Campo, J.; Bracho, G.; Cuello, M.; Balboa, J.; Acevedo, R.; Zayas, C.; Gil, D. New vaccines require potent adjuvants like AFPL1 and AFCo1. Scand. J. Immunol. 2007, 66, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Ascarateil, S.; Puget, A.; Koziol, M. Safety data of Montanide ISA 51 VG and Montanide ISA 720 VG, two adjuvants dedicated to human therapeutic vaccines. J. Immunother. Cancer 2015, 3, 428. [Google Scholar] [CrossRef]
- Leroux-Roels, G. Unmet needs in modern vaccinology Adjuvants to improve the immune response. Vaccine 2010, 28, C25–C36. [Google Scholar] [CrossRef]
- Pérez, O.; Batista-Duharte, A.; González, E.; Zayas, C.; Balboa, J.; Cuello, M.; Cabrera, O.; Lastre, M.; Schijns, V. Human prophylactic vaccine adjuvants and their determinant role in new vaccine formulations. Braz. J. Med. Biol. Res. 2012, 45, 681–692. [Google Scholar] [CrossRef]
- Hauguel, T.; Hackett, C. Rationally designed vaccine adjuvants: Separating efficacy from toxicity. Front. Biosci. 2008, 13, 2806–2813. [Google Scholar] [CrossRef]
- Pérez, O.; Romeu, B.; Cabrera, O.; González, E.; Batista-Duharte, A. Adjuvants are key factors for the development of future vaccines: Lessons from the Finlay adjuvant platform. Front. Immunol. 2013, 4, 407. [Google Scholar] [CrossRef] [PubMed]
- Guy, B. The perfect mix: Recent progress in adjuvant research. Nat. Rev. Microbiol. 2007, 5, 396–397. [Google Scholar] [CrossRef] [PubMed]
- Bonam, S.; Partidos, C.; Halmuthur, S.; Muller, S. An Overview of Novel Adjuvants Designed for Improving Vaccine Efficacy. Trends Pharmacol. Sci. 2017, 38, 771–793. [Google Scholar] [CrossRef] [PubMed]
- Reddy Bonam, S.; Bhunia, D.; Muller, S.; Goud, S.; Alvala, M.; Halmuthur, S. Novel trisaccharide based phospholipids as immunomodulators. Int. Immunopharmacol. 2019, 74, 105684. [Google Scholar] [CrossRef]
- Schijns, V.; Fernández-Tejada, A.; Barjaktarović, Ž.; Bouzalas, I.; Brimnes, J.; Chernysh, S.; Gizurarson, S.; Gursel, I.; Jakopin, Z.; Lawrenz, M.; et al. Modulation of immune responses using adjuvants to facilitate therapeutic vaccination. Immunol. Rev. 2020, 296, 169–190. [Google Scholar] [CrossRef]
- Lawson, L.; Norton, E.; Clements, J. Defending the mucosa: Adjuvant and carrier formulations for mucosal immunity. Curr. Opin. Immunol. 2011, 23, 414–420. [Google Scholar] [CrossRef]
- Del Giudice, G.; Rappuoli, R.; Didierlaurent, A. Correlates of adjuvanticity: A review on adjuvants in licensed vaccines. In Seminars in Immunology; Academic Press: Cambridge, MA, USA, 2018; Volume 39, pp. 14–21. [Google Scholar] [CrossRef]
- Nussenzweig, R.S.; Vanderberg, J.; Most, H.; Orton, C. Protective immunity produced by the injection of X-irradiated sporozoites of Plasmodium berghei. Nature 1967, 216, 160–162. [Google Scholar] [CrossRef]
- Collins, W.; Contacos, P. Immunization of monkeys against Plasmodium cynomolgi by X-irradiated sporozoites. Nature 1972, 236, 176–177. [Google Scholar] [CrossRef]
- Clyde, D. Immunity to falciparum and vivax malaria induced by irradiated sporozoites: A review of the University of Maryland studies, 1971–1975. Bull. World Health Organ. 1990, 68, 9–12. [Google Scholar]
- Epstein, J.; Tewari, K.; Lyke, K.; Sim, B.; Billingsley, P.; Laurens, M.; Gunasekera, A.; Chakravarty, S.; James, E.R.; Sedegah, M.; et al. Live attenuated malaria vaccine designed to protect through hepatic CD8+ T cell immunity. Science 2011, 334, 475–480. [Google Scholar] [CrossRef]
- Hoffman, S.; Billingsley, P.; James, E.; Richman, A.; Loyevsky, M.; Li, T.; Chakravarty, S.; Gunasekera, A.; Chattopadhyay, R.; Li, M.; et al. Development of a metabolically active, non-replicating sporozoite vaccine to prevent Plasmodium falciparum malaria. Hum. Vaccines 2010, 6, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.; Paolino, K.; Richie, T.; Sedegah, M.; Singer, A.; Ruben, A.; Chakravarty, S.; Stafford, A.; Ruck, R.; Eappen, A.; et al. Protection against Plasmodium falciparum malaria by PfSPZ vaccine. JCI Insight 2017, 2, e89154. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczak, S.; Lakshmanan, V.; Fishbaugher, M.; Camargo, N.; Harupa, A. A next-generation genetically attenuated Plasmodium falciparum parasite created by triple gene deletion. Mol. Ther. 2014, 22, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Kublin, J.; Mikolajczak, S.; Sack, B.; Fishbaugher, M.; Seilie, A.; Shelton, L.; VonGoedert, T.; First, M.; Magee, S.; Fritzen, E.; et al. Complete attenuation of genetically engineered Plasmodium falciparum sporozoites in human subjects. Sci. Transl. Med. 2017, 9, 371. [Google Scholar] [CrossRef]
- Bastiaens, G.; Van Meer, M.P.A.; Scholzen, A.; Obiero, J.; Vatanshenassan, M.; Grinsven, T.; Lee Sim, B.; Billingsley, P.; James, E.; Gunasekera, A.; et al. Safety, Immunogenicity, and Protective Efficacy of Intradermal Immunization with Aseptic, Purified, Cryopreserved Plasmodium falciparum Sporozoites in Volunteers under Chloroquine Prophylaxis: A Randomized Controlled Trial. Am. J. Trop. Med. Hyg. 2016, 94, 663–673. [Google Scholar] [CrossRef][Green Version]
- Mordmüller, B.; Surat, G.; Lagler, H.; Chakravarty, S.; Ishizuka, A. Sterile protection against human malaria by chemoattenuated PfSPZ vaccine. Nature 2017, 542, 445–449. [Google Scholar] [CrossRef]
- Richie, T.; Billingsley, P.; Kim Lee Sim, B.; James, E.; Chakravarty, S.; Epstein, J.; Lyke, K.; Mordmüller, B.; Alonso, P.; Patrick, D.; et al. Progress with Plasmodium falciparum sporozoite (PfSPZ)-based malaria vaccines. Vaccine 2015, 33, 7452–7461. [Google Scholar] [CrossRef]
- Coppi, A.; Natarajan, R.; Pradel, G.; Bennett, B.; James, E.; Roggero, M.; Corradin, G.; Persson, C.; Tewari, R.; Sinnis, P. The malaria circumsporozoite protein has two functional domains, each with distinct roles as sporozoites journey from mosquito to mammalian host. J. Exp. Med. 2011, 208, 341–356. [Google Scholar] [CrossRef]
- Nussenzweig, V.; Nussenzweig, R. Circumsporozoite proteins of malaria parasites. Cell 1985, 42, 401–403. [Google Scholar] [CrossRef]
- Sheehy, S.; Duncan, C.; Elias, S.; Choudhary, P.; Biswas, S.; Halstead, F.; Collins, K.; Edwards, N.; Douglas, A.; Anagnostou, N.; et al. ChAd63-MVA-vectored blood-stage malaria vaccines targeting MSP1 and AMA1: Assessment of efficacy against mosquito bite challenge in humans. Mol. Ther. 2012, 20, 2355–2368. [Google Scholar] [CrossRef]
- Cohen, S.; Mc, G.I.; Carrington, S. Gamma-globulin and acquired immunity to human malaria. Nature 1961, 192, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Cowman, A.; Berry, D.; Baum, J. The cellular and molecular basis for malaria parasite invasion of the human red blood cell. J. Cell Biol. 2012, 198, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.; Mackay, M.; Goman, M.; Scaife, J. Allelic dimorphism in a surface antigen gene of the malaria parasite Plasmodium falciparum. J. Mol. Biol. 1987, 195, 273–287. [Google Scholar] [CrossRef]
- Blackman, M.; Ling, I.; Stephen, N.; Holder, A. Proteolytic processing of the Plasmodium falciparum merozoite surface protein-1 produces a membrane-bound fragment containing two epidermal growth factor-like domains. Mol. Biochem. Parasitol. 1991, 49, 29–34. [Google Scholar] [CrossRef]
- Blackman, M.; Whittle, H.; Holder, A. Processing of the Plasmodium falciparum merozoite surface protein-l: Identification of a 33-kilodalton secondary processing product which is shed prior to erythrocyte invasion. Mol. Biochem. Parasitol. 1991, 49, 35–44. [Google Scholar] [CrossRef]
- Lyon, J.; Angov, E.; Fay, M.; Sullivan, J.; Girourd, A.; Robinson, S.; Bergmann-Leitner, E.; Duncan, E.; Darko, C.; Collins, W.; et al. Protection induced by Plasmodium falciparum MSP1 is strain-specific, antigen and adjuvant dependent, and correlates with antibody responses. PLoS ONE 2008, 3, e2830. [Google Scholar] [CrossRef]
- Al-Yaman, F.; Genton, B.; Anders, R.; Falk, M.; Triglia, T.; Lewis, D.; Hii, J.; Beck, H.P.; Alpers, M.P. Relationship between humoral response to Plasmodium falciparum merozoite surface antigen-2 and malaria morbidity in a highly endemic area of Papua New Guinea. Am. J. Trop. Med. Hyg. 1994, 51, 593–602. [Google Scholar] [CrossRef]
- Moorthy, S.; Good, M.; Hill, A. Malaria vaccine developments. Lancet 2004, 363, 150–156. [Google Scholar] [CrossRef]
- Singh, S.; Soe, S.; Mejia, J.; Roussilhon, C.; Theisen, M.; Corradin, G.; Druilhe, P. Identification of a conserved region of Plasmodium falciparum MSP3 targeted by biologically active antibodies to improve vaccine design. J. Infect. Dis. 2004, 190, 1010–1018. [Google Scholar] [CrossRef]
- Marshall, V.; Silva, A.; Foley, M.; Cranmer, S.; Wang, L.; McColl, D.J.; Kemp, D.J.; Coppel, R.L. A second merozoite surface protein (MSP-4) of Plasmodium falciparum that contains an epidermal growth factor-like domain. Infect. Immun. 1997, 65, 4460–4467. [Google Scholar] [CrossRef]
- Boyle, M.; Langer, C.; Chan, J.; Hodderb, A.; Coppel, R.; Anders, F.; Beeson, J. Sequential processing of merozoite surface proteins during and after erythrocyte invasion by Plasmodium falciparum. Infect. Immun. 2014, 82, 924–936. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Black, C.; Wang, L.; Hibbs, A.; Coppel, R. Lack of sequence diversity in the gene encoding merozoite surface protein 5 of Plasmodium falciparum. Mol. Biochem. Parasitol. 1999, 103, 243–250. [Google Scholar] [CrossRef]
- Perraut, R.; Joos, C.; Sokhna, C.; Polson, H.; Trape, J.; Tall, A.; Marrama, L.; Mercereau-Puijalon, O.; Richard, V.; Longacre, S. Association of antibody responses to the conserved Plasmodium falciparum merozoite surface protein 5 with protection against clinical malaria. PLoS ONE 2014, 9, e101737. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J.; Triglia, T.; Hodder, A.; Jackson, D.; Cowman, A.; Anders, R. Plasmodium falciparum merozoite surface protein 6 is a dimorphic antigen. Infect. Immun. 2004, 72, 2321–2328. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pachebat, J.; Ling, I.; Grainger, M.; Trucco, C.; Howell, S.; Fernandez-Reyes, D.; Gunaratne, R.; Holder, A. The 22 kDa component of the protein complex on the surface of Plasmodium falciparum merozoites is derived from a larger precursor, merozoite surface protein 7. Mol. Biochem. Parasitol. 2001, 117, 83–89. [Google Scholar] [CrossRef]
- Black, C.; Wu, T.; Wang, L.; Hibbs, A.; Coppel, R. Merozoite surface protein-8 of Plasmodium falciparum contains two epidermal growth factor-like domains. Mol. Biochem. Parasitol. 2001, 114, 217–226. [Google Scholar] [CrossRef]
- Weber, J.; Lyon, J.; Wolff, R.; Hall, T.; Lowell, G.; Chulay, J. Primary structure of a Plasmodium falciparum malaria antigen located at the merozoite surface a within the parasitophorous vacuole. J. Biol. Chem. 1988, 263, 11421–11425. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Kumar, A.; Singh, B.; Bharadwaj, A.; Sailaja, V.; Adak, T.; Kushwaha, A.; Malhotra, P.; Chauhan, V.S. Characterization of protective epitopes in a highly conserved Plasmodium falciparum antigenic protein containing repeats of acidic and basic residues. Infect. Immun. 1998, 66, 2895–2904. [Google Scholar] [CrossRef]
- Kushwaha, A.; Rao, P.; Suresh, R.; Chauhan, V. Immunogenicity of recombinant fragments of Plasmodium falciparum acidic basic repeat antigen produced in Escherichia coli. Parasite Immunol. 2001, 23, 435–444. [Google Scholar] [CrossRef]
- Black, C.G.; Wang, L.; Wu, T.; Coppel, R.L. Apical location of a novel EGF-like domain-containing protein of Plasmodium falciparum. Mol. Biochem. Parasitol. 2003, 127, 59–68. [Google Scholar] [CrossRef]
- Pacheco, A.; Elango, A.; Rahman, A.; Fisher, D.; Collins, W.; Barnwell, J.; Escalante, A. Evidence of purifying selection on merozoite surface protein 8 (MSP8) and 10 (MSP10) in Plasmodium spp. Infect. Genet. Evol. 2012, 12, 978–986. [Google Scholar] [CrossRef] [PubMed]
- Palacpac, N.; Ntege, E.; Yeka, A.; Balikagala, B.; Suzuki, N.; Shirai, H.; Yagi, M.; Ito, K.; Fukushima, W.; Hirota, Y. Phase 1b randomized trial and follow-up study in Uganda of the bloodstage malaria vaccine candidate BK-SE36. PLoS ONE 2013, 8, e64073. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.; Good, R.; Drew, D.; Delorenzi, M.; Sanders, P.; Hodder, A.; Speed, T.; Cowman, A.; Koning-Ward, T.; Crabb, B. A subset of Plasmodium falciparum SERA genes are expressed and appear to play an important role in the erythrocytic cycle. J. Biol. Chem. 2002, 277, 47524–47532. [Google Scholar] [CrossRef] [PubMed]
- Borre, M.; Dziegiel, M.; Hogh, B.; Petersen, E.; Rieneck, K.; Meis, J.F.; Aikawa, M.; Nakamura, K.; Harada, M. Primary structure and localization of a conserved immunogenic Plasmodium falciparum glutamate rich protein (GLURP) expressed in both the preerythrocytic and erythrocytic stages of the vertebrate life cycle. Mol. Biochem. Parasitol. 1991, 49, 119–132. [Google Scholar] [CrossRef]
- Hermsen, C.; Verhage, D.; Telgt, D.; Teelen, K.; Bousema, J.; Roestenberg, M.; Bolad, A.; Berzins, K.; Corradin, G.; Leroy, O. Glutamate-rich protein (GLURP) induces antibodies that inhibit in vitro growth of Plasmodium falciparum in a phase 1 malaria vaccine trial. Vaccine 2007, 25, 2930–2940. [Google Scholar] [CrossRef]
- Bélard, S.; Issifou, S.; Hounkpatin, A.; Schaumburg, F.; Ngoa, U.; Esen, M.; Fendel, R.; Martinez de Salazar, P.; Mürbeth, R.; Milligan, P.; et al. A randomized controlled phase Ib trial of the malaria vaccine candidate GMZ2 in African children. PLoS ONE 2011, 6, e22525. [Google Scholar] [CrossRef]
- Sirima, S.; Richert, L.; Chêne, A.; Konate, A.; Campion, C.; Dechavanne, S.; Semblat, J.; Benhamouda, N.; Bahuaud, M.; Loulergue, P.; et al. PRIMVAC vaccine adjuvanted with Alhydrogel or GLA-SE to prevent placental malaria: A first-in-human, randomised, double-blind, placebo-controlled study. Lancet Infect. Dis. 2020, 20, 585–597. [Google Scholar] [CrossRef]
- Silvie, O.; Franetich, J.; Charrin, S.; Mueller, M.; Siau, A.; Bodescot, M.; Rubinstein, E.; Hannoun, L.; Charoenvit, Y.; Kocken, C.; et al. A role for apical membrane antigen during invasion of hepatocytes by Plasmodium falciparum sporozoites. J. Biol. Chem. 2004, 279, 9490–9496. [Google Scholar] [CrossRef]
- Berzins, K.; Perlmann, H.; Wahlin, B.; Carlsson, J.; Wahlgren, M.; Udomsangpetch, R.; Björkman, A.; Patarroyo, M.E.; Perlmann, P. Rabbit and human antibodies to a repeated amino acid sequence of a Plasmodium falciparum antigen, Pf 155, react with the native protein and inhibit merozoite invasion. Proc. Natl. Acad. Sci. USA 1986, 83, 1065–1069. [Google Scholar] [CrossRef]
- Irani, V.; Ramsland, P.; Guy, A.; Siba, P.; Mueller, I.; Richards, J.; Beeson, J. Acquisition of Functional Antibodies That Block the Binding of Erythrocyte-Binding Antigen 175 and Protection Against Plasmodium falciparum Malaria in Children. Clin. Infect. Dis. 2015, 61, 1244–1252. [Google Scholar] [CrossRef]
- Koram, K.; Adu, B.; Ocran, J.; Karikari, Y.; Adu-Amankwah, S.; Ntiri, M.; Abuaku, B.; Dodoo, D.; Gyan, B.; Kronmann, K.; et al. Safety and Immunogenicity of EBA-175 RII-NG Malaria Vaccine Administered Intramuscularly in Semi-Immune Adults: A Phase 1, Double-Blinded Placebo Controlled Dosage Escalation Study. PLoS ONE 2016, 11, e0163066. [Google Scholar] [CrossRef] [PubMed]
- Deans, J.; Knight, A.; Jean, W.; Waters, A.; Cohen, S.; Mitchell, G. Vaccination trials in rhesus monkeys with a minor, invariant, Plasmodium knowlesi 66 kD merozoite antigen. Parasite Immunol. 1988, 10, 535–552. [Google Scholar] [CrossRef] [PubMed]
- Sagara, I.; Dicko, A.; Ellis, R.; Fay, M.; Diawara, S.; Sissoko, M.; Kone, M.; Diallo, A.; Saye, R. A randomized controlled phase 2 trial of the blood stage AMA1-C1/Alhydrogel malaria vaccine in children in Mali. Vaccine 2009, 27, 3090–3098. [Google Scholar] [CrossRef]
- Spring, M.; Cummings, J.; Ockenhouse, C.; Dutta, S.; Reidler, R.; Angov, E.; Bergmann-Leitner, E.; Stewart, V.; Bittner, S.; Juompan, L. Phase I/IIa study of the malaria vaccine candidate apical membrane antigen-1 (AMA-1) administered in adjuvant system AS01B or AS02A. PLoS ONE 2009, 4, e5254. [Google Scholar] [CrossRef] [PubMed]
- Preiser, P.; Kaviratnea, M.; Khana, S.; Bannister, L.; Jarra, W. The apical organelles of malaria merozoites: Host cell selection, invasion, host immunity and immune evasion. Microbes Infect. 2000, 2, 1461–1477. [Google Scholar] [CrossRef]
- Culvenor, J.; Day, K.; Anders, R. Plasmodium falciparum ring-infected erythrocyte surface antigen is released from merozoite dense granules after erythrocyte invasion. Infect. Immun. 1991, 59, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Wahlin, B.; Sjolander, A.; Ahlborg, N.; Udomsangpetch, R.; Scherf, A.; Mattei, D.; Berzins, K.; Perlmann, P. Involvement of Pf155/RESA and cross-reactive antigens in Plasmodium falciparum merozoite invasion in vitro. Infect. Immun. 1992, 60, 443–449. [Google Scholar] [CrossRef]
- Crompton, P.; Pierce, S.; Miller, L. Advances and challenges in malaria vaccine development. J. Clin. Investig. 2010, 120, 4168–4178. [Google Scholar] [CrossRef]
- Miura, K. Progress and prospects for blood-stage malaria vaccines. Expert Rev. Vaccines 2016, 15, 765–781. [Google Scholar] [CrossRef]
- Nikolaeva, D.; Draper, S.; Biswas, S. Toward the development of effective transmission-blocking vaccines for malaria. Expert Rev. Vaccines 2015, 14, 653–680. [Google Scholar] [CrossRef]
- Wu, Y.; Sinden, R.; Churcher, T.; Tsuboi, T.; Yusibov, V. Development of malaria transmission-blocking vaccines: From concept to product. Adv. Parasitol. 2015, 89, 109–152. [Google Scholar] [CrossRef] [PubMed]
- Kapulu, M.; Da, D.; Miura, K.; Li, Y.; Blagborough, A.; Churcher, T.; Nikolaeva, D.; Williams, A.; Goodman, A.; Sangare, I. Comparative Assessment of Transmission-Blocking Vaccine Candidates against Plasmodium falciparum. Sci. Rep. 2015, 5, 11193. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ellis, R.; Shaffer, D.; Fontes, E.; Malkin, E.; Mahanty, S.; Fay, M.; Narum, D.; Rausch, K.; Miles, A. Phase 1 trial of malaria transmission blocking vaccine candidates Pfs25 and Pvs25 formulated with montanide ISA 51. PLoS ONE 2008, 3, e2636. [Google Scholar] [CrossRef] [PubMed]
- Talaat, K.; Ellis, R.; Hurd, J.; Hentrich, A.; Gabriel, E.; Hynes, N.; Rausch, K.; Zhu, D.; Muratova, O.; Herrera, R. Safety and Immunogenicity of Pfs25-EPA/Alhydrogel1, a Transmission Blocking Vaccine against Plasmodium falciparum: An Open Label Study in Malaria Naïve Adults. PLoS ONE 2016, 11, e0163144. [Google Scholar] [CrossRef]
- Williamson, K.; Criscio, M.; Kaslow, D. Cloning and expression of the gene for Plasmodium falciparum transmission blocking target antigen, Pfs230. Mol. Biochem. Parasitol. 1993, 58, 355–358. [Google Scholar] [CrossRef]
- Quakyi, I.; Carter, R.; Rener, J.; Kumar, N.; Good, M.; Miller, L. The 230-kDa gamete surface protein of Plasmodium falciparum is also a target for transmission blocking antibodies. J. Immunol. 1987, 139, 4213–4217. [Google Scholar]
- MacDonald, N.; Nguyen, V.; Shimp, R.; Reiter, K.; Herrera, R.; Burkhardt, M.; Muratova, O.; Kumar, K.; Aebig, J.; Rausch, K.; et al. Structural and immunological characterization of recombinant 6-Cysteine domains of the Plasmodium falciparum sexual stage protein Pfs230. J. Biol. Chem. 2016, 291, 19913–19922. [Google Scholar] [CrossRef]
- Bousema, T.; Roeffen, W.; Meijerink, H.; Mwerinde, H.; Mwakalinga, S.; Gemert, G.; Vegte-Bolmer, M.; Mosha, F.; Targett, G.; Riley, E.; et al. The dynamics of naturally acquired immune responses to Plasmodium falciparum sexual stage antigens Pfs230 & Pfs48/45 in a low endemic area in Tanzania. PLoS ONE 2010, 5, e14114. [Google Scholar] [CrossRef]
- Fonseca, J.; Cabrera-Mora, M.; Singh, B.; Oliveira-Ferreira, J.; Lima-Junior, J.; Calvo-Calle, J.M.; Lozano, J.M.; Moreno, A. A chimeric protein-based malaria vaccine candidate induces robust T cell responses against Plasmodium vivax MSP119. Sci. Rep. 2016, 6, 34527. [Google Scholar] [CrossRef]
- Canepa, G.; Molina-Cruz, A.; Yenkoidiok-Douti, L.; Burkhardt, M.; Peng, F.; Narum, D.; Boulanger, M.; Valenzuela, J. Antibody targeting of a specific region of Pfs47 blocks Plasmodium falciparum malaria transmission. NPJ Vaccines 2018, 3, 26. [Google Scholar] [CrossRef]
- Purcell, A.; McCluskey, J.; Ross, J. More than one reason to rethink the use of peptides in vaccine design. Nat. Rev. Drug Discov. 2007, 6, 404–414. [Google Scholar] [CrossRef] [PubMed]
- Cross, S.; Kuttell, M.M.; Stone, J.E.; Gain, J.E. Visualization of cyclic and multi-branched molecules with VMD. J. Mol. Graph. Model. 2009, 28, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Croft, N.; Purcell, A. Peptidomimetics: Modifying peptides in the pursuit of better vaccines. Expert Rev. Vaccines 2011, 10, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Lozano, J.M.; Espejo, F.; Díaz, D.; Salazar, L.; Rodríguez, J.; Pinzón, C.; Calvo, J.C.; Guzmán, F.; Patarroyo, M.E. Reduced amide pseudopeptide analogues of a malaria peptide possess secondary structural elements responsible for induction of functional antibodies which react with native proteins expressed in Plasmodium falciparum erythrocyte stages. J. Pept. Res. 1998, 52, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Lozano, J.M.; Patarroyo, M.E. A rational strategy for a malarial vaccine development. Microbes Infect. 2007, 9, 751–760. [Google Scholar] [CrossRef]
- Lozano, J.M.; Guerrero, Y.; Alba, M.; Lesmes, L.; Escobar, J.; Patarroyo, M.E. Redefining an epitope of a malaria vaccine candidate, with antibodies against the N-terminal MSA-2 antigen of Plasmodium harboring non-natural peptide bonds. Amino Acids 2013, 45, 913–935. [Google Scholar] [CrossRef]
- Vanegas, M.; Bermúdez, A.; Guerrero, Y.; Cortes-Vecino, J.; Curtidor, H.; Patarroyo, M.E.; Lozano, J.M. Protecting capacity against malaria of chemically defined tetramer forms based on the Plasmodium falciparum apical sushi protein as potential vaccine components. Biochem. Biophys. Res. Commun. 2014, 451, 15–23. [Google Scholar] [CrossRef]
- Sissoko, M.; Healy, S.; Katile, A.; Omaswa, F.; Zaidi, I. Safety and efficacy of PfSPZ vaccine against Plasmodium falciparum via direct venous inoculation in healthy malaria-exposed adults in Mali: A randomised, double-blind phase 1 trial. Lancet Infect. Dis. 2017, 17, 498–509. [Google Scholar] [CrossRef]
- Drew, D.R.; Beeson, J.G. PfRH5 as a candidate vaccine for Plasmodium falciparum malaria. Trends Parasitol. 2015, 31, 87–88. [Google Scholar] [CrossRef]
- Protein Data Bank. Available online: https://www.rcsb.org/ (accessed on 28 November 2020).
- LBtope: Prediction of Linear B-cell Epitopes. Available online: http://crdd.osdd.net/raghava/lbtope/ (accessed on 25 November 2020).
- IEDB Analysis Resource. Available online: http://tools.iedb.org/netchop/help/server (accessed on 25 November 2020).
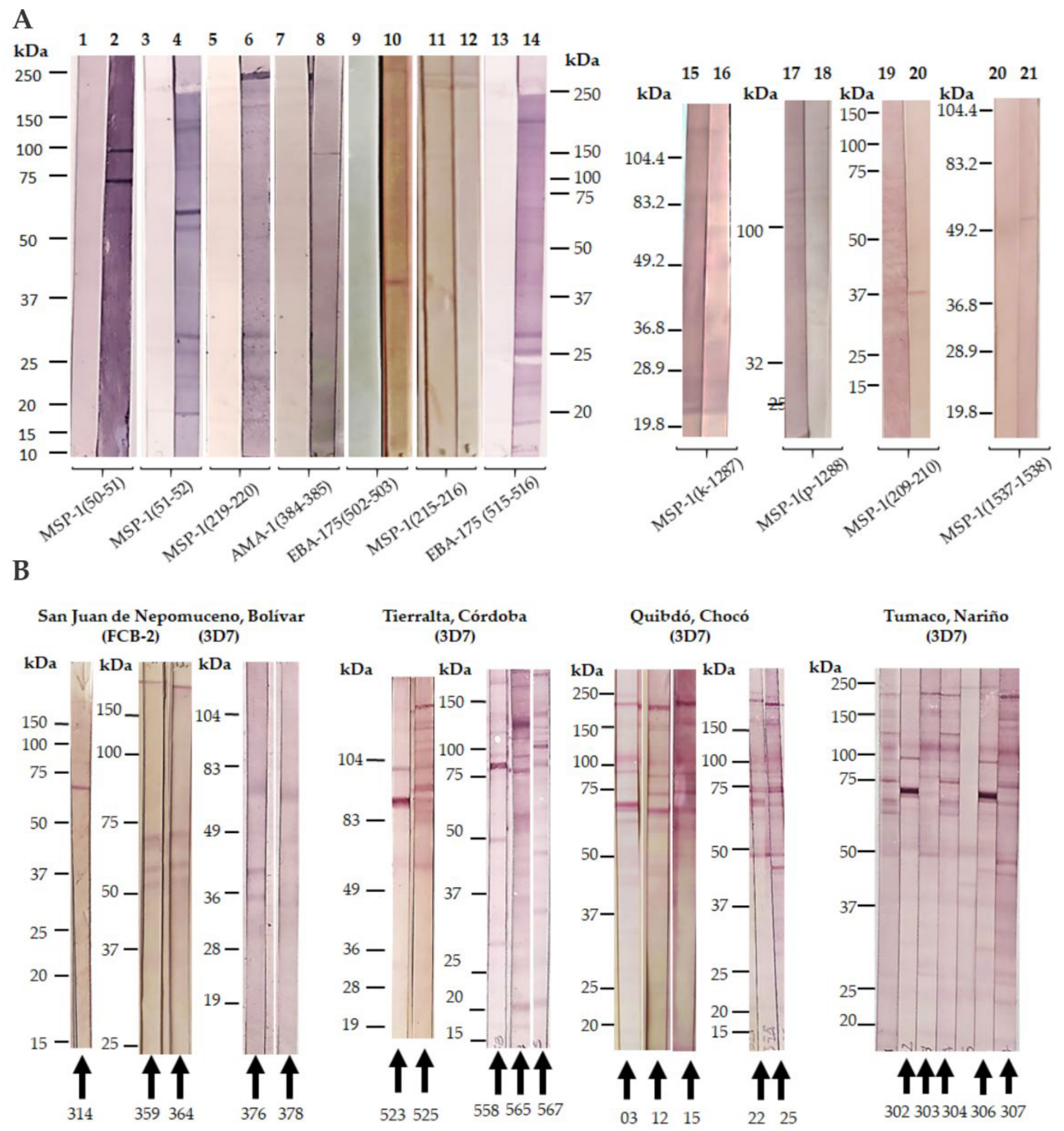
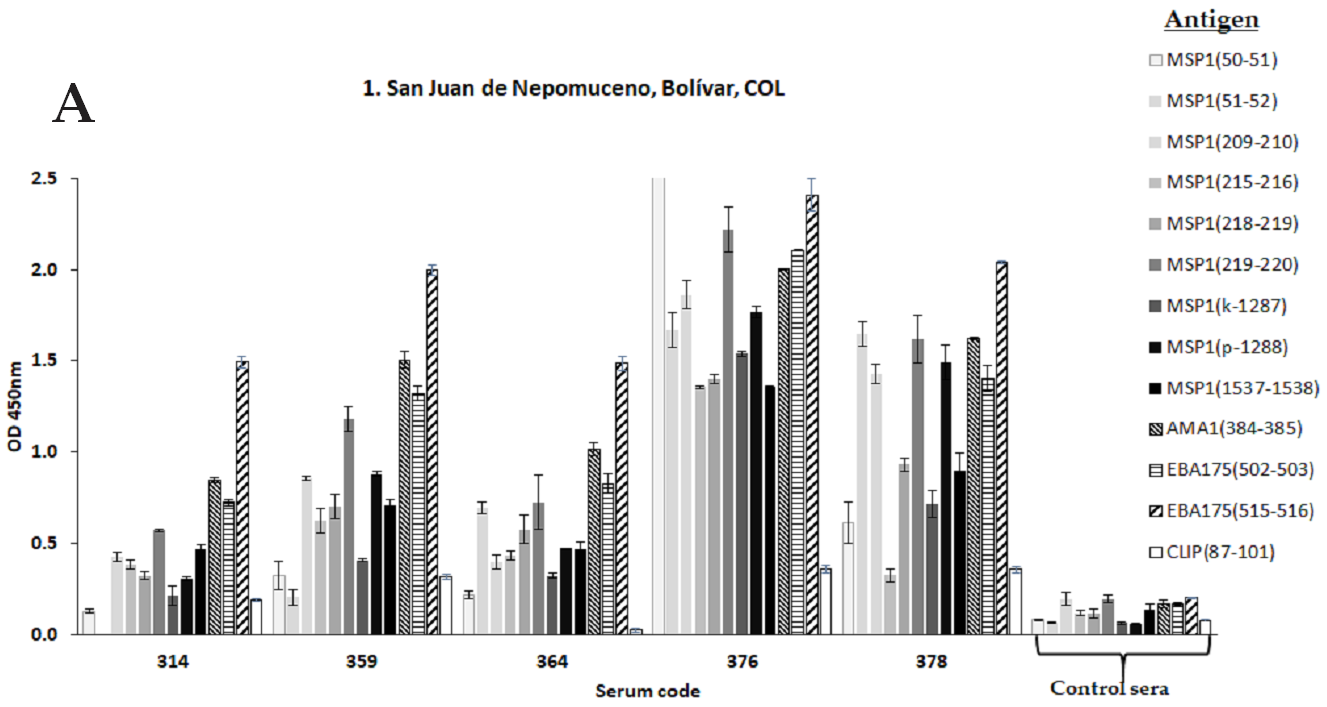
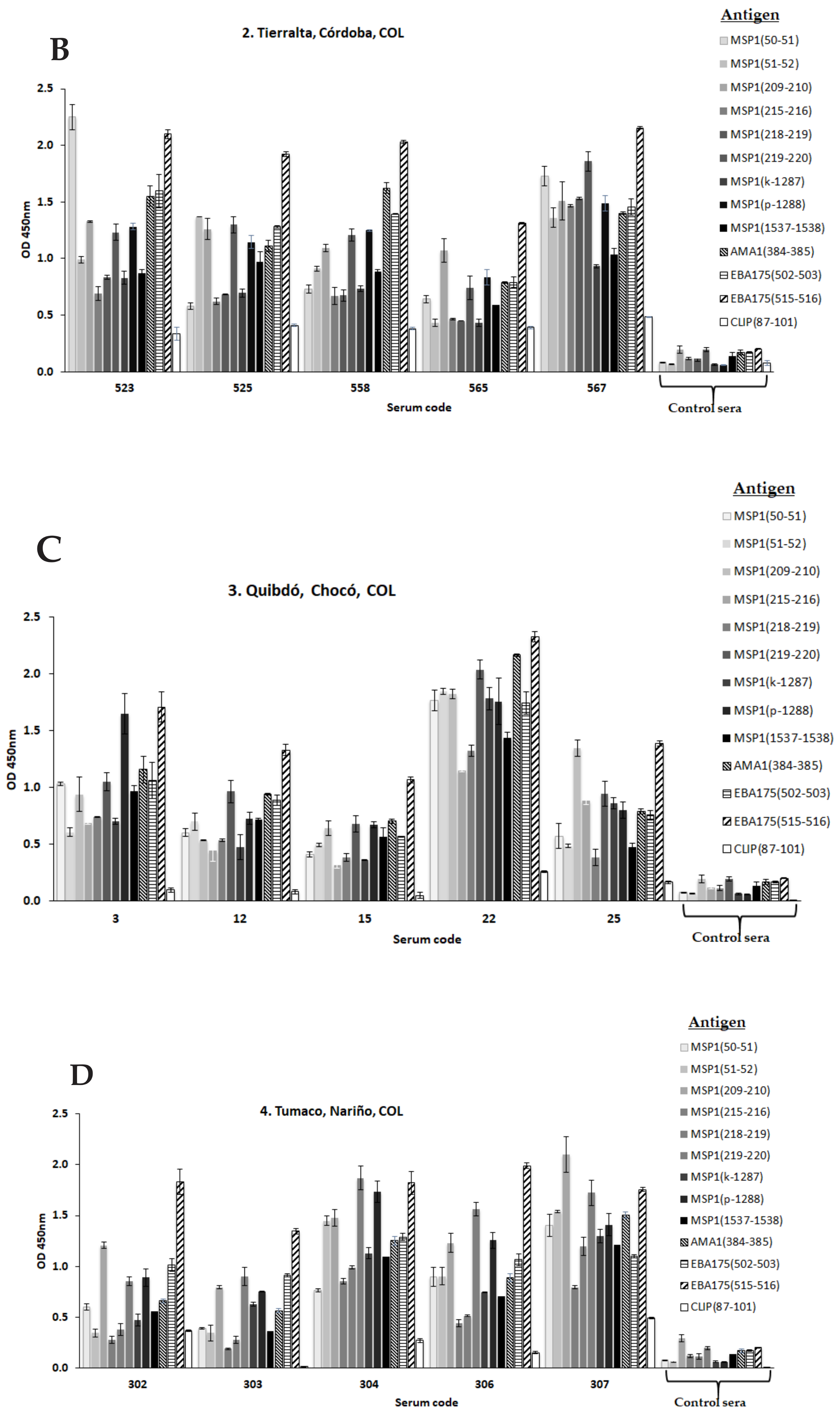
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozano, J.M.; Rodríguez Parra, Z.; Hernández-Martínez, S.; Yasnot-Acosta, M.F.; Rojas, A.P.; Marín-Waldo, L.S.; Rincón, J.E. The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes. Vaccines 2021, 9, 115. https://doi.org/10.3390/vaccines9020115
Lozano JM, Rodríguez Parra Z, Hernández-Martínez S, Yasnot-Acosta MF, Rojas AP, Marín-Waldo LS, Rincón JE. The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes. Vaccines. 2021; 9(2):115. https://doi.org/10.3390/vaccines9020115
Chicago/Turabian StyleLozano, José Manuel, Zully Rodríguez Parra, Salvador Hernández-Martínez, Maria Fernanda Yasnot-Acosta, Angela Patricia Rojas, Luz Stella Marín-Waldo, and Juan Edilberto Rincón. 2021. "The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes" Vaccines 9, no. 2: 115. https://doi.org/10.3390/vaccines9020115
APA StyleLozano, J. M., Rodríguez Parra, Z., Hernández-Martínez, S., Yasnot-Acosta, M. F., Rojas, A. P., Marín-Waldo, L. S., & Rincón, J. E. (2021). The Search of a Malaria Vaccine: The Time for Modified Immuno-Potentiating Probes. Vaccines, 9(2), 115. https://doi.org/10.3390/vaccines9020115