Stelladens mysteriosus: A Strange New Mosasaurid (Squamata) from the Maastrichtian (Late Cretaceous) of Morocco †
Abstract
:1. Introduction
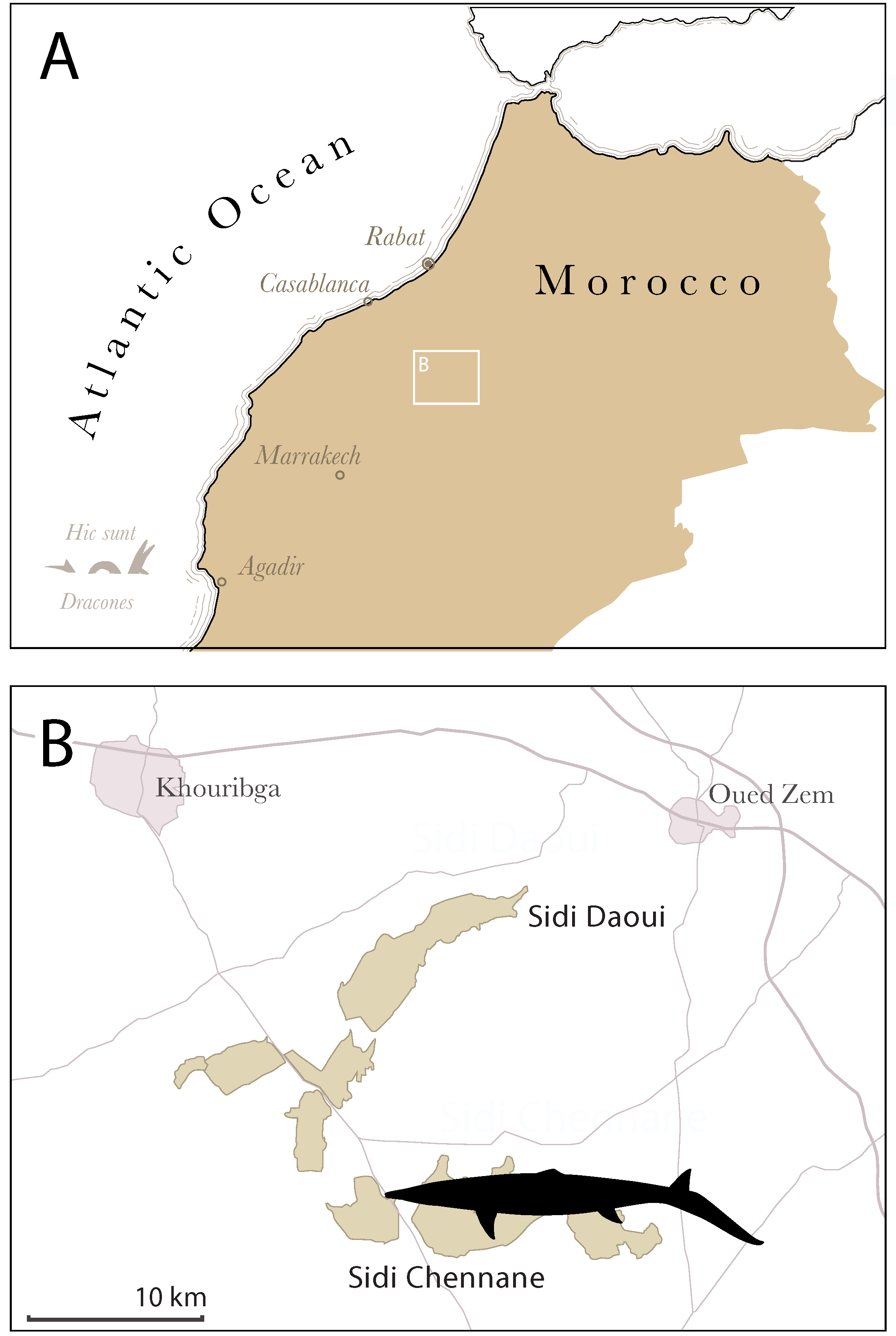
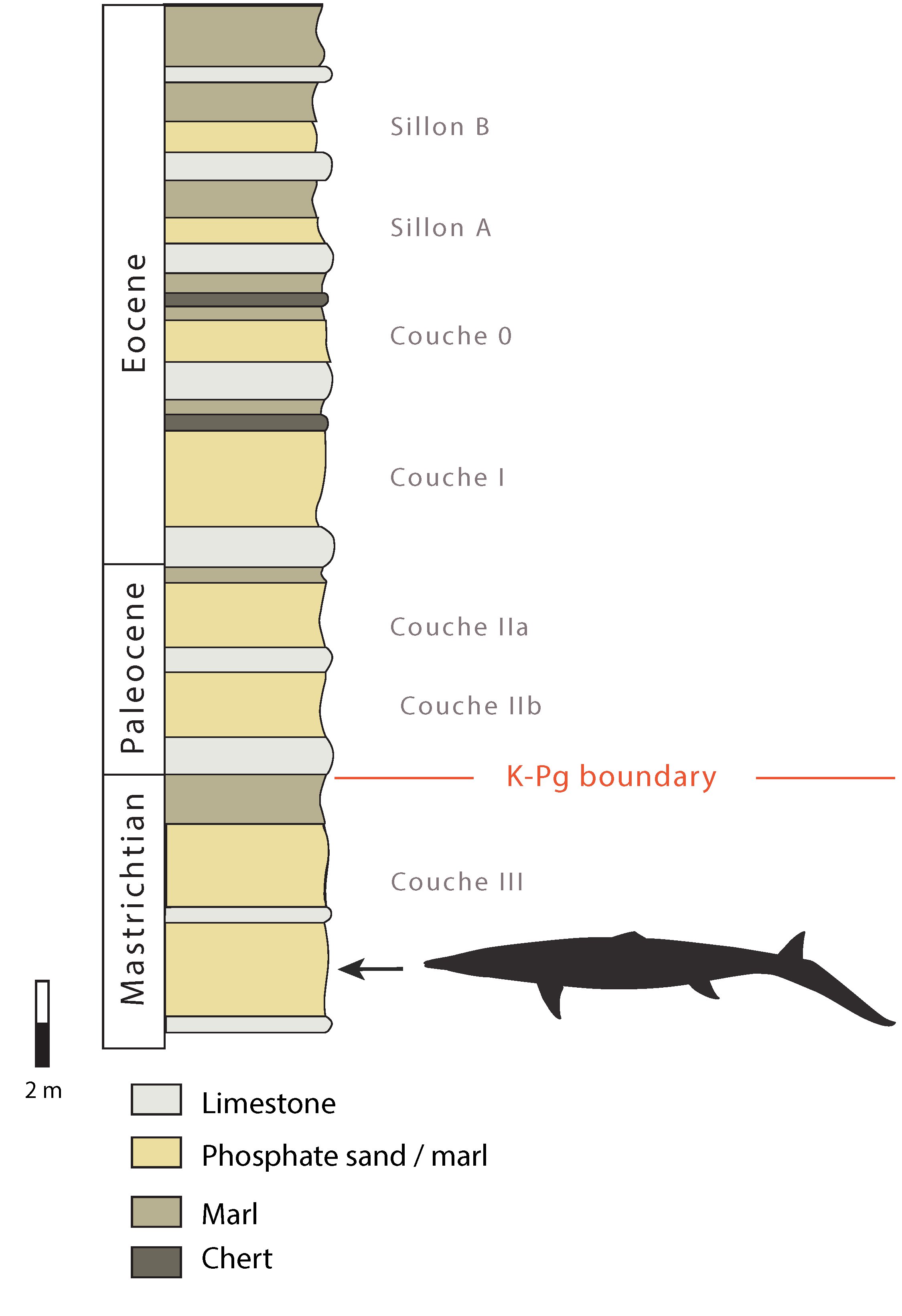
2. Geological Setting
3. Results
3.1. Systematic Paleontology


3.2. Comparative Description
3.2.1. General Remarks
3.2.2. Dentary
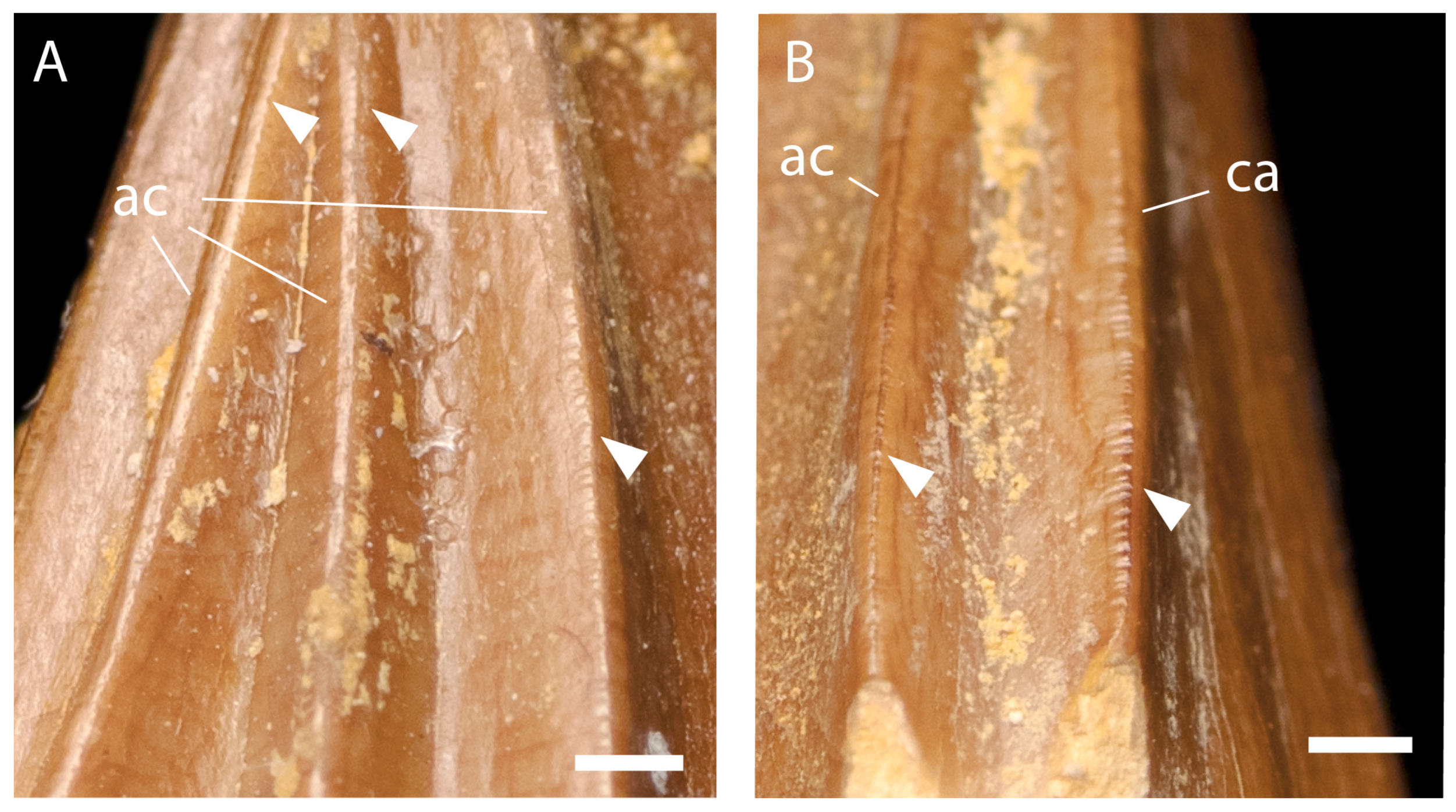
3.2.3. Teeth

4. Discussion
4.1. Comparison and Identification
4.2. The Diet of Stelladens
4.3. Implications for Mosasaurid Evolution
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Polcyn, M.J.; Jacobs, L.L.; Araújo, R.; Schulp, A.S.; Mateus, O. Physical drivers of mosasaur evolution. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 400, 17–27. [Google Scholar] [CrossRef]
- Longrich, N.R.; Jalil, N.-E.; Khaldoune, F.; Yazami, O.K.; Pereda-Suberbiola, X.; Bardet, N. Thalassotitan atrox, a giant predatory mosasaurid (Squamata) from the Upper Maastrichtian Phosphates of Morocco. Cretac. Res. 2022, 140, 105315. [Google Scholar] [CrossRef]
- Zverkov, N.G.; Pervushov, E.M. A gigantic pliosaurid from the Cenomanian (Upper Cretaceous) of the Volga Region, Russia. Cretac. Res. 2020, 110, 104419. [Google Scholar] [CrossRef]
- Fischer, V.; Bardet, N.; Benson, R.B.; Arkhangelsky, M.S.; Friedman, M. Extinction of fish-shaped marine reptiles associated with reduced evolutionary rates and global environmental volatility. Nat. Commun. 2016, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bardet, N.; Pereda-Suberbiola, X.; Jalil, N.-E. A new mosasauroid (Squamata) from the Late Cretaceous (Turonian) of Morocco. Comptes Rendus Palevol. 2003, 2, 607–616. [Google Scholar] [CrossRef]
- Bardet, N.; Falconnet, J.; Fischer, V.; Houssaye, A.; Jouve, S.; Pereda-Suberbiola, X.; Perez-García, A.; Rage, J.-C.; Vincent, P. Mesozoic marine reptile palaeobiogeography in response to drifting plates. Gondwana Res. 2014, 26, 869–887. [Google Scholar] [CrossRef]
- Bardet, N.; Houssaye, A.; Vincent, P.; Pereda-Suberbiola, X.; Amaghzaz, M.; Jourani, E.; Meslouh, S. Mosasaurids (Squamata) from the Maastrichtian phosphates of Morocco: Biodiversity, palaeobiogeography and palaeoecology based on tooth morphoguilds. Gondwana Res. 2015, 27, 1068–1078. [Google Scholar] [CrossRef]
- Martin, J.E.; Vincent, P.; Tacail, T.; Khaldoune, F.; Jourani, E.; Bardet, N.; Balter, V. Calcium isotopic evidence for vulnerable marine ecosystem structure prior to the K/Pg extinction. Curr. Biol. 2017, 27, 1641–1644.e1642. [Google Scholar] [CrossRef]
- Vincent, P.; Bardet, N.; Pereda Suberbiola, X.; Bouya, B.; Amaghzaz, M.; Meslouh, S. Zarafasaura oceanis, a new elasmosaurid (Reptilia: Sauropterygia) from the Maastrichtian Phosphates of Morocco and the palaeobiogeography of latest Cretaceous plesiosaurs. Gondwana Res. 2011, 19, 1062–1073. [Google Scholar] [CrossRef]
- Vincent, P.; Bardet, N.; Houssaye, A.; Amaghzaz, M.; Meslouh, S. New plesiosaur specimens from the Maastrichtian Phosphates of Morocco and their implications for the ecology of the latest Cretaceous marine apex predators. Gondwana Res. 2013, 24, 796–805. [Google Scholar] [CrossRef]
- Bardet, N.; Jalil, N.-E.; de Lapparent de Broin, F.; Germain, D.; Lambert, O.; Amaghzaz, M. A Giant Chelonioid Turtle from the Late Cretaceous of Morocco with a Suction Feeding Apparatus Unique among Tetrapods. PLoS ONE 2013, 8, e63586. [Google Scholar] [CrossRef]
- Lapparent de Broin, F.d.; Bardet, N.; Amaghzaz, M.; Meslouh, S. A strange new chelonioid turtle from the Latest Cretaceous phosphates of Morocco. Comptes Rendus Palevol. 2013, 13, 87–95. [Google Scholar] [CrossRef]
- Jouve, S.; Bardet, N.; Jalil, N.-E.; Pereda-Suberbiola, X.; Bouya, B.; Amaghzaz, M. The oldest African crocodylian: Phylogeny, paleobiogeography, and differential survivorship of marine reptiles through the Cretaceous-Tertiary boundary. J. Vertebr. Paleontol. 2008, 28, 409–421. [Google Scholar] [CrossRef]
- LeBlanc, A.R.H.; Caldwell, M.W.; Bardet, N. A new mosasaurine from the Maastrichtian (Upper Cretaceous) phosphates of Morocco and its implications for mosasaurine systematics. J. Vertebr. Paleontol. 2012, 32, 82–104. [Google Scholar] [CrossRef]
- Bardet, N.; Pereda-Suberbiola, X.; Iarochène, M.; Bouyahyaoui, F.; Bouya, B.; Amaghzaz, M. Mosasaurus beaugei Arambourg, 1952 (Squamata, Mosasauridae) from the Late Cretaceous phosphates of Morocco. Geobios 2004, 37, 315–324. [Google Scholar] [CrossRef]
- Longrich, N.R.; Bardet, N.; Schulp, A.S.; Jalil, N.-E. Xenodens calminechari gen. et sp. nov., a bizarre mosasaurid (Mosasauridae, Squamata) with shark-like cutting teeth from the upper Maastrichtian of Morocco, North Africa. Cretac. Res. 2021, 123, 104764. [Google Scholar] [CrossRef]
- Schulp, A.S.; Bardet, N.; Bouya, B. A new species of the durophagous mosasaur Carinodens (Squamata, Mosasauridae) and additional material of Carinodens belgicus from the Maastrichtian phosphates of Morocco. Neth. J. Geosci. 2009, 88, 161–167. [Google Scholar] [CrossRef]
- Bardet, N.; Pereda-Suberbiola, X.; Iarochène, M.; Bouya, B.; Amaghzaz, M. A new species of Halisaurus from the Late Cretaceous phosphates of Morocco, and the phylogenetical relationships of the Halisaurinae (Squamata: Mosasauridae). Zool. J. Linn. Soc. 2005, 143, 447–472. [Google Scholar] [CrossRef]
- Longrich, N.R.; Bardet, N.; Khaldoune, F.; Yazami, O.K.; Jalil, N.-E. Pluridens serpentis, a new mosasaurid (Mosasauridae: Halisaurinae) from the Maastrichtian of Morocco and implications for mosasaur diversity. Cretac. Res. 2021, 126, 104882. [Google Scholar] [CrossRef]
- Rempert, T.H.; Martens, B.P.; Melchers, A.P.V. First Record of a Tylosaurine Mosasaur from the Latest Cretaceous Phosphates of Morocco. Open J. Geol. 2022, 12, 883–906. [Google Scholar] [CrossRef]
- Strong, C.R.; Caldwell, M.W.; Konishi, T.; Palci, A. A new species of longirostrine plioplatecarpine mosasaur (Squamata: Mosasauridae) from the Late Cretaceous of Morocco, with a re-evaluation of the problematic taxon ‘Platecarpus’ ptychodon. J. Syst. Palaeontol. 2020, 18, 1769–1804. [Google Scholar] [CrossRef]
- Houssaye, A.; Bardet, N.; Rage, J.-C.; Pereda-Suberbiola, X.; Bouya, B.; Amaghzaz, M.; Amalik, M. A review of Pachyvaranus crassispondylus Arambourg, 1952, a pachyostotic marine squamate from the latest Cretaceous phosphates of Morocco and Syria. Geol. Mag. 2011, 148, 237–249. [Google Scholar] [CrossRef]
- Barlow, J. Cetacean abundance in Hawaiian waters estimated from a summer/fall survey in 2002. Mar. Mammal Sci. 2006, 22, 446–464. [Google Scholar] [CrossRef]
- Waring, G.T.; Nottestad, L.; Olsen, E.; Skov, H.; Vikingsson, G. Distribution and density estimates of cetaceans along the mid-Atlantic Ridge during summer 2004. J. Cetacean Res. Manag. 2008, 10, 137–146. [Google Scholar] [CrossRef]
- Heide-Jorgensen, M.; Simon, M.; Laidre, K. Estimates of large whale abundance in Greenlandic waters from a ship-based survey in 2005. J. Cetacean Res. Manag. 2007, 9, 95. [Google Scholar] [CrossRef]
- Lucas, J.; Prévôt-Lucas, L. Tethyan phosphates and bioproductites. In The Tethys Ocean; Springer: Boston, MA, USA, 1996; pp. 367–391. [Google Scholar]
- Kocsis, L.; Gheerbrant, E.; Mouflih, M.; Cappetta, H.; Yans, J.; Amaghzaz, M. Comprehensive stable isotope investigation of marine biogenic apatite from the Late Cretaceous–Early Eocene phosphate series of Morocco. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2014, 394, 74–88. [Google Scholar] [CrossRef]
- Cappetta, H. Chondrichthyes II. Mesozoic and Cenozoic Elasmobranchii. Handb. Paleoichthyology 1987, 3, 1–193. [Google Scholar]
- Bardet, N.; Gheerbrant, E.; Noubhani, A.; Cappetta, H.; Jouve, S.; Bourdon, E.; Pereda-Suberbiola, X.; Jalil, N.-E.; Vincent, P.; Houssaye, A.; et al. Les Vertébrés des phosphates crétacés-paléogènes (72, 1–47, 8 Ma) du Maroc. In Paléontologie des vertébrés du Maroc: État des Connaissances; Société géologique de France: Paris, France, 2017; pp. 351–452. [Google Scholar]
- Noubhani, A.; Cappetta, H. Les Orectolobiformes, Carcharhiniformes et Myliobatiformes (Elasmobranchii, Neoselachii) des Bassins à phosphate du Maroc (Maastrichtien-Lutétien basal): Systématique, biostratigraphie, évolution et dynamique des faunes. Palaeo Ichthyol. 1997, 8, 1–327. [Google Scholar]
- Arambourg, C. Les vertébrés fossiles des gisements de phosphates (Maroc-Algérie-Tunisie). Notes Et MÉMoires Du Serv. GÉOlogique Du Maroc. 1952, 92, 1–372. [Google Scholar]
- Pereda-Suberbiola, X.; Bardet, N.; Jouve, S.; Iarochène, M.; Bouya, B.; Amaghzaz, M. A new azhdarchid pterosaur from the Late Cretaceous phosphates of Morocco. Geol. Soc. Lond. Spec. Publ. 2003, 217, 79–90. [Google Scholar] [CrossRef]
- Longrich, N.R.; Martill, D.M.; Andres, B. Late Maastrichtian pterosaurs from North Africa and mass extinction of Pterosauria at the Cretaceous-Paleogene boundary. PLoS Biol. 2018, 16, e2001663. [Google Scholar]
- Pereda-Suberbiola, X.; Bardet, N.; Iarochène, M.; Bouya, B.; Amaghzaz, M. The first record of a sauropod dinosaur from the Late Cretaceous phosphates of Morocco. J. Afr. Earth Sci. 2004, 40, 81–88. [Google Scholar] [CrossRef]
- Longrich, N.R.; Suberbiola, X.P.; Pyron, R.A.; Jalil, N.-E. The first duckbill dinosaur (Hadrosauridae: Lambeosaurinae) from Africa and the role of oceanic dispersal in dinosaur biogeography. Cretac. Res. 2021, 120, 104678. [Google Scholar] [CrossRef]
- Longrich, N.R.; Pereda-Suberbiola, X.; Jalil, N.-E.; Khaldoune, F.; Jourani, E. An abelisaurid from the latest Cretaceous (late Maastrichtian) of Morocco, North Africa. Cretac. Res. 2017, 76, 40–52. [Google Scholar] [CrossRef]
- Oppel, M. Die Ordnungen, Familien und Gattungen der Reptilien als Prodrom Einer Naturgeschichte Derselben; Lindauer: München, Germany, 1811; p. 87. [Google Scholar]
- Gervais, P. Zoologie et Paléontologie Françaises (Animaux Vertébrés): Nouvelles Recherches sur les Animaux Vivants del la France; Arthus Bertrand: Paris, France, 1852. [Google Scholar]
- Bell, G.L., Jr. A phylogenetic revision of North American and Adriatic Mosasauroidea. In Ancient marine reptiles; Elsevier: Amsterdam, The Netherlands, 1997; pp. 293–332. [Google Scholar]
- Lingham-Soliar, T. Anatomy and functional morphology of the largest marine reptile known, Mosasaurus hoffmanni (Mosasauridae, Reptilia) from the Upper Cretaceous, Upper Maastrichtian of the Netherlands. Philos. Trans. R. Soc. London Ser. B Biol. Sci. 1995, 347, 155–180. [Google Scholar]
- Russell, D.A. Systematics and morphology of American mosasaurs. Bull. Peabody Mus. Nat. Hist. 1967, 23, 1–240. [Google Scholar]
- Street, H.; LeBlanc, A.; Caldwell, M. A histological investigation of dental crown characters used in mosasaur phylogenetic analyses. Vertebr. Anat. Morphol. Palaeontol. 2021, 9, 83–94. [Google Scholar] [CrossRef]
- Longrich, N.R. A new species of Pluridens (Mosasauridae: Halisaurinae) from the upper Campanian of Southern Nigeria. Cretac. Res. 2016, 64, 36–44. [Google Scholar] [CrossRef]
- McCurry, M.R.; Evans, A.R.; Fitzgerald, E.M.; McHenry, C.R.; Bevitt, J.; Pyenson, N.D. The repeated evolution of dental apicobasal ridges in aquatic-feeding mammals and reptiles. Biol. J. Linn. Soc. 2019, 127, 245–259. [Google Scholar] [CrossRef]
- Massare, J.A. Tooth morphology and prey preference of Mesozoic marine reptiles. J. Vertebr. Paleontol. 1987, 7, 121–137. [Google Scholar] [CrossRef]
- Konishi, T.; Newbrey, M.G.; Caldwell, M.W. A small, exquisitely preserved specimen of Mosasaurus missouriensis (Squamata, Mosasauridae) from the upper Campanian of the Bearpaw Formation, western Canada, and the first stomach contents for the genus. J. Vertebr. Paleontol. 2014, 34, 802–819. [Google Scholar] [CrossRef]
- Bardet, N.; Pereda-Suberbiola, X.; Iarochène, M.; Amalik, M.; Bouya, B. Durophagous Mosasauridae (Squamata) from the Upper Cretaceous phosphates of Morocco, with description of a new species of Globidens. Neth. J. Geosci. 2005, 84, 167–175. [Google Scholar] [CrossRef]
- Bardet, N.; Pereda-Suberbiola, X.; Schulp, A.S.; Bouya, B. New material of Carinodens (Squamata, Mosasauridae) from the Maastrichtian (Late Cretaceous) phosphates of Morocco. In Proceedings of the Second Mosasaur Meeting; Fort Hays Studies, Specialp; Sternberg Museum of Natural History: Hays, KS, USA, 2008; pp. 29–36. [Google Scholar]
- Gilmore, C.W. A new mosasauroid reptile from the Cretaceous of Alabama. Proc. United States Natl. Mus. 1912, 41, 479–484. [Google Scholar] [CrossRef]
- Owocki, K.; Madzia, D. Predatory behaviour in mosasaurid squamates inferred from tooth microstructure and mineralogy. Cretac. Res. 2020, 111, 104430. [Google Scholar] [CrossRef]
- Herrera-Flores, J.A.; Stubbs, T.L.; Benton, M.J. Ecomorphological diversification of squamates in the Cretaceous. R. Soc. Open Sci. 2021, 8, 201961. [Google Scholar] [CrossRef]
- Longrich, N.R.; Bhullar, B.-A.S.; Gauthier, J. Mass extinction of lizards and snakes at the Cretaceous-Paleogene boundary. Proc. Natl. Acad. Sci. USA 2012, 109, 21396–21401. [Google Scholar] [CrossRef]
- Lafuma, F.; Corfe, I.J.; Clavel, J.; Di-Poï, N. Multiple evolutionary origins and losses of tooth complexity in squamates. Nat. Commun. 2021, 12, 6001. [Google Scholar] [CrossRef]
- Erickson, G.M. Split carinae on tyrannosaurid teeth and implications of their development. J. Vertebr. Paleontol. 1995, 15, 268–274. [Google Scholar] [CrossRef]
- Beatty, B.L.; Heckert, A.B. A large archosauriform tooth with multiple supernumerary carinae from the Upper Triassic of New Mexico (USA), with comments on carina development and anomalies in the Archosauria. Hist. Biol. 2009, 21, 57–65. [Google Scholar] [CrossRef]
- Welsh, E.; Boyd, C.A.; Spearing, K. Split carinae on a specimen of false saber-toothed cat (Carnivora: Nimravidae) and the implications for analogous tooth abnormality formation in mammals and theropod dinosaurs. In Proceedings of the South Dakota Academy of Science; South Dakota Academy of Science: Madison, SD, USA, 2020. [Google Scholar]
- Nydam, R.L.; Gauthier, J.A.; Chiment, J.J. The mammal-like teeth of the Late Cretaceous lizard Peneteius aquilonius Estes 1969. J. Vertebr. Paleontol. 2000, 20, 628–631. [Google Scholar] [CrossRef]
- Bunker, G.; Martill, D.M.; Smith, R.; Zourhi, S.; Longrich, N.R. Plesiosaurs from the fluvial Kem Kem Group (mid-Cretaceous) of eastern Morocco and a review of non-marine plesiosaurs. Cretac. Res. 2022, 140, 105310. [Google Scholar] [CrossRef]
- Zverkov, N.G.; Fischer, V.; Madzia, D.; Benson, R.B. Increased pliosaurid dental disparity across the Jurassic–Cretaceous transition. Palaeontology 2018, 61, 825–846. [Google Scholar] [CrossRef]
- Madzia, D.; Sachs, S.; Lindgren, J. Morphological and phylogenetic aspects of the dentition of Megacephalosaurus eulerti, a pliosaurid from the Turonian of Kansas, USA, with remarks on the cranial anatomy of the taxon. Geol. Mag. 2019, 156, 1201–1216. [Google Scholar] [CrossRef]
- Erickson, G.M.; Sidebottom, M.A.; Kay, D.I.; Turner, K.T.; Ip, N.; Norell, M.A.; Sawyer, W.G.; Krick, B.A. Wear biomechanics in the slicing dentition of the giant horned dinosaur Triceratops. Sci. Adv. 2015, 1, e1500055. [Google Scholar] [CrossRef] [PubMed]
- Erickson, G.M.; Krick, B.A.; Hamilton, M.; Bourne, G.R.; Norell, M.A.; Lilleodden, E.; Sawyer, W.G. Complex dental structure and wear biomechanics in hadrosaurid dinosaurs. Science 2012, 338, 98–101. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Longrich, N.R.; Jalil, N.-E.; Pereda-Suberbiola, X.; Bardet, N. Stelladens mysteriosus: A Strange New Mosasaurid (Squamata) from the Maastrichtian (Late Cretaceous) of Morocco. Foss. Stud. 2023, 1, 2-14. https://doi.org/10.3390/fossils1010002
Longrich NR, Jalil N-E, Pereda-Suberbiola X, Bardet N. Stelladens mysteriosus: A Strange New Mosasaurid (Squamata) from the Maastrichtian (Late Cretaceous) of Morocco. Fossil Studies. 2023; 1(1):2-14. https://doi.org/10.3390/fossils1010002
Chicago/Turabian StyleLongrich, Nicholas R., Nour-Eddine Jalil, Xabier Pereda-Suberbiola, and Nathalie Bardet. 2023. "Stelladens mysteriosus: A Strange New Mosasaurid (Squamata) from the Maastrichtian (Late Cretaceous) of Morocco" Fossil Studies 1, no. 1: 2-14. https://doi.org/10.3390/fossils1010002
APA StyleLongrich, N. R., Jalil, N.-E., Pereda-Suberbiola, X., & Bardet, N. (2023). Stelladens mysteriosus: A Strange New Mosasaurid (Squamata) from the Maastrichtian (Late Cretaceous) of Morocco. Fossil Studies, 1(1), 2-14. https://doi.org/10.3390/fossils1010002








