Development of a Cloud Point Extraction Technique Based on Lecithin for the Recovery of Carotenoids from Liquid Tomato Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and LTW Sample
2.2. Determination of Physicochemical Parameters
2.3. Total Carotenoid Content Determination
2.4. CPE Procedure
2.5. Carotenoid Recovery by CPE
2.6. Determination of the Antioxidant Activity
2.7. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Parameters of the LTW
3.2. Optimization of the Extraction Procedure
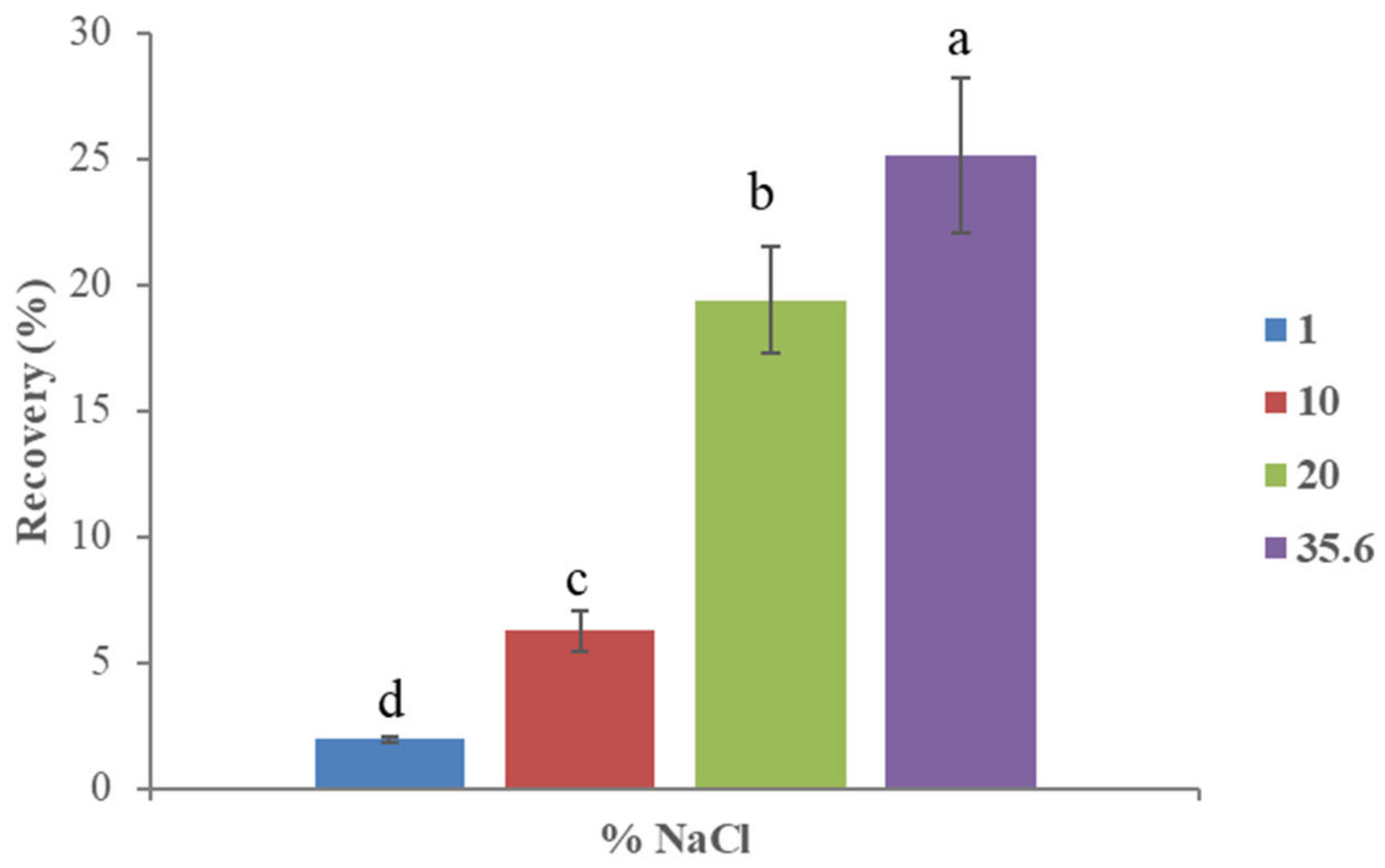
3.2.1. Effect of Ionic Strength
3.2.2. Effect of Sample pH
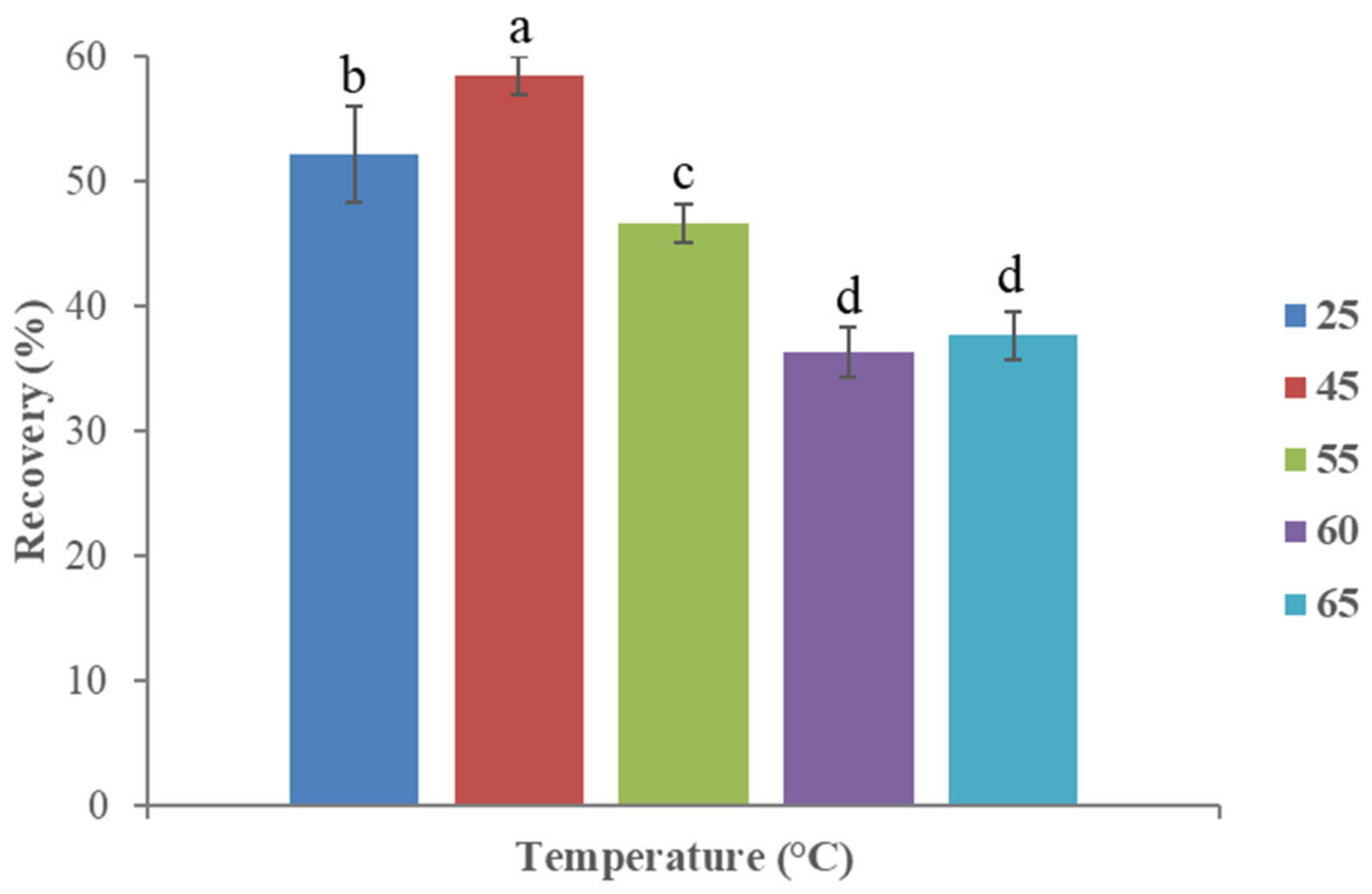
3.2.3. Effect of Temperature
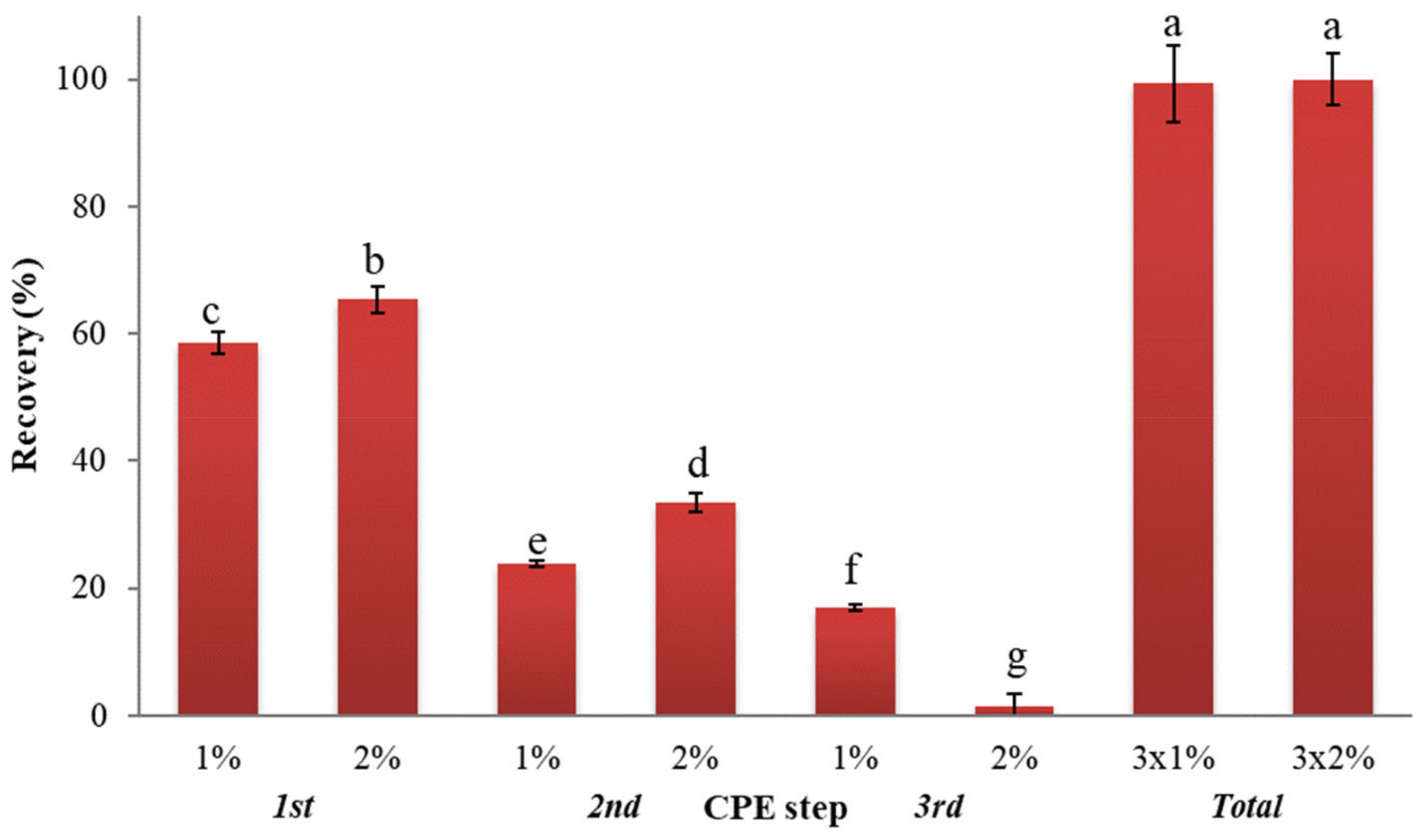
3.2.4. Effect of Surfactant Concentration and Number of Extractions
3.3. Antioxidant Activity of the Recovered Carotenoids
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Felter, S.P.; Zhang, X.; Thompson, C. Butylated hydroxyanisole: Carcinogenic food additive to be avoided or harmless antioxidant important to protect food supply? Regul. Toxicol. Pharmacol. 2021, 121, 104887. [Google Scholar] [CrossRef] [PubMed]
- Caseiro, M.; Ascenso, A.; Costa, A.; Creagh-Flynn, J.; Johnson, M.; Simões, S. Lycopene in human health. LWT 2020, 127, 109323. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maoka, T. Carotenoids as natural functional pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saini, R.K.; Keum, Y.S. Carotenoid extraction methods: A review of recent developments. Food Chem. 2018, 240, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Huang, C.; Chen, Z. A review for the pharmacological effect of lycopene in central nervous system disorders. Biomed. Pharmacother. 2019, 111, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Luengo, E.; Álvarez, I.; Raso, J. Improving Carotenoid Extraction from Tomato Waste by Pulsed Electric Fields. Front. Nutr. 2014, 1, 12. [Google Scholar] [CrossRef] [Green Version]
- de Andrade Lima, M.; Kestekoglou, I.; Charalampopoulos, D.; Chatzifragkou, A. Supercritical Fluid Extraction of Carotenoids from Vegetable Waste Matrices. Molecules 2019, 24, 466. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Liu, X.; Zhang, L.; Shao, P.; Wu, W.; Chen, Z.; Li, J.; Renard, C.M.G.C. An overview of carotenoid extractions using green solvents assisted by Z-isomerization. Trends Food Sci. Technol. 2022, 123, 145–160. [Google Scholar] [CrossRef]
- Al_Saadi, M.R.; Al-Garawi, Z.S.; Thani, M.Z. Promising technique, cloud point extraction: Technology & applications. J. Phys. Conf. Ser. 2021, 1853, 012064. [Google Scholar] [CrossRef]
- Carabias-Martínez, R.; Rodríguez-Gonzalo, E.; Moreno-Cordero, B.; Pérez-Pavón, J.L.; García-Pinto, C.; Fernández Laespada, E. Surfactant cloud point extraction and preconcentration of organic compounds prior to chromatography and capillary electrophoresis. J. Chromatogr. A 2000, 902, 251–265. [Google Scholar] [CrossRef] [PubMed]
- Sosa Ferrera, Z.; Padrón Sanz, C.; Mahugo Santana, C.; Santana Rodríguez, J.J. The use of micellar systems in the extraction and pre-concentration of organic pollutants in environmental samples. TrAC-Trends Anal. Chem. 2004, 23, 469–479. [Google Scholar] [CrossRef]
- Katsoyannos, E.; Gortzi, O.; Chatzilazarou, A.; Athanasiadis, V.; Tsaknis, J.; Lalas, S. Evaluation of the suitability of low hazard surfactants for the separation of phenols and carotenoids from red-flesh orange juice and olive mill wastewater using cloud point extraction. J. Sep. Sci. 2012, 35, 2665–2670. [Google Scholar] [CrossRef] [PubMed]
- Martin, G. Surfactant Systems: Their Chemistry, Pharmacy and Biology. Biochem. Soc. Trans. 1984, 12, 719–720. [Google Scholar] [CrossRef]
- Arya, S.S.; Kaimal, A.M.; Chib, M.; Sonawane, S.K.; Show, P.L. Novel, energy efficient and green cloud point extraction: Technology and applications in food processing. J. Food Sci. Technol. 2019, 56, 524–534. [Google Scholar] [CrossRef]
- van Nieuwenhuyzen, W. Lecithin and Other Phospholipids. In Surfactants from Renewable Resources; John Wiley & Sons, Ltd.: Chichester, UK, 2010; pp. 191–212. ISBN 9780470760413. [Google Scholar]
- Chatzilazarou, A.; Katsoyannos, E.; Lagopoulou, M.; Tsaknis, J. Application of cloud point extraction with the aid of Genapol X-080 in the pre-concentration of lycopene and total carotenoids from red fleshed orange. Ernahrung 2011, 35, 5–13. [Google Scholar]
- Baaka, N.; El Ksibi, I.; Mhenni, M.F. Optimisation of the recovery of carotenoids from tomato processing wastes: Application on textile dyeing and assessment of its antioxidant activity. Nat. Prod. Res. 2017, 31, 196–203. [Google Scholar] [CrossRef]
- Biswas, A.K.; Sahoo, J.; Chatli, M.K. A simple UV-Vis spectrophotometric method for determination of β-carotene content in raw carrot, sweet potato and supplemented chicken meat nuggets. LWT-Food Sci. Technol. 2011, 44, 1809–1813. [Google Scholar] [CrossRef]
- Chatzilazarou, A.; Katsoyannos, E.; Gortzi, O.; Lalas, S.; Paraskevopoulos, Y.; Dourtoglou, E.; Tsaknis, J. Removal of polyphenols from wine sludge using cloud point extraction. J. Air Waste Manag. Assoc. 2010, 60, 454–459. [Google Scholar] [CrossRef] [Green Version]
- Gortzi, O.; Lalas, S.; Chatzilazarou, A.; Katsoyannos, E.; Papaconstandinou, S.; Dourtoglou, E. Recovery of natural antioxidants from olive mill wastewater using Genapol-X080. JAOCS J. Am. Oil Chem. Soc. 2008, 85, 133–140. [Google Scholar] [CrossRef]
- Katsoyannos, E.; Chatzilazarou, A.; Gortzi, O.; Lalas, S.; Konteles, S.; Tataridis, P. Application of cloud point extraction using surfactants in the isolation of physical antioxidants (phenols) from olive mill wastewater. Fresenius Environ. Bull. 2006, 15, 1122–1125. [Google Scholar]
- Tsaknis, J.; Lalas, S. Extraction and identification of natural antioxidant from Sideritis euboea (mountain tea). J. Agric. Food Chem. 2005, 53, 6375–6381. [Google Scholar] [CrossRef] [PubMed]
- Kotíková, Z.; Hejtmánková, A.; Lachman, J. Determination of the Influence of Variety and Level of Maturity on the Content and Development of Carotenoids in Tomatoes. Czech J. Food Sci. 2009, 27, S200–S203. [Google Scholar] [CrossRef] [Green Version]
- Chatzimitakos, T.G.; Stalikas, C.D. Melamine sponge decorated with copper sheets as a material with outstanding properties for microextraction of sulfonamides prior to their determination by high-performance liquid chromatography. J. Chromatogr. A 2018, 1554, 28–36. [Google Scholar] [CrossRef]
- Chatzimitakos, T.G.; Pierson, S.A.; Anderson, J.L.; Stalikas, C.D. Enhanced magnetic ionic liquid-based dispersive liquid-liquid microextraction of triazines and sulfonamides through a one-pot, pH-modulated approach. J. Chromatogr. A 2018, 1571, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Santalad, A.; Burakham, R.; Srijaranai, S.; Srijaranai, S.; Deming, R.L. Role of different salts on cloud-point extraction of isoprocarb and promecarb insecticides followed by high-performance liquid chromatography. J. Chromatogr. Sci. 2012, 50, 523–530. [Google Scholar] [CrossRef]
- Mortada, W.I.; Hassanien, M.M.; El-Asmy, A.A. Cloud point extraction of some precious metals using Triton X-114 and a thioamide derivative with a salting-out effect. Egypt. J. Basic Appl. Sci. 2014, 1, 184–191. [Google Scholar] [CrossRef]
- Bell, T.; Alamzad, R.; Graf, B.A. Effect of pH on the chemical stability of carotenoids in juice. Proc. Nutr. Soc. 2016, 75, E94. [Google Scholar] [CrossRef] [Green Version]
- Alibade, A.; Batra, G.; Bozinou, E.; Salakidou, C.; Lalas, S. Optimization of the extraction of antioxidants from winery wastes using cloud point extraction and a surfactant of natural origin (lecithin). Chem. Pap. 2020, 74, 4517–4524. [Google Scholar] [CrossRef]
- Cristea, E.; Ghendov-Mosanu, A.; Patras, A.; Socaciu, C.; Pintea, A.; Tudor, C.; Sturza, R. The influence of temperature, storage conditions, ph, and ionic strength on the antioxidant activity and color parameters of rowan berry extracts. Molecules 2021, 26, 3786. [Google Scholar] [CrossRef]

| Water content (%, w/w) | 88.7 ± 0.3 |
| Total Solids (%, w/w) | 10.2 ± 0.3 |
| Total Fat (%, w/w) | 1.0 ± 0.1 |
| Total Carotenoids (%, w/w) | 0.034 ± 0.002 |
| pH | 5.6 ± 0.3 |
| CPE Step | % Cs | Vs/Vw | Fc | As |
|---|---|---|---|---|
| 1st | 1% | 0.021 ± 0.001 f | 7.25 ± 0.17 a | 5.21 ± 0.23 a |
| 2% | 0.055 ± 0.001 d | 6.46 ± 0.25 b | 4.63 ± 0.26 b | |
| 2nd | 1% | 0.036 ± 0.002 e | 4.41 ± 0.10 c | 1.65 ± 0.06 e |
| 2% | 0.069 ± 0.002 c | 2.75 ± 0.10 d | 2.47 ± 0.05 d | |
| 3rd | 1% | 0.039 ± 0.001 e | 4.81 ± 0.27 c | 1.58 ± 0.06 e |
| 2% | 0.041 ± 0.001 e | 4.72 ± 0.21 c | 2.71 ± 0.11 d | |
| Total | 3 × 1% | 0.096 ± 0.006 b | 6.07 ± 0.45 b | 4.36 ± 0.31 b |
| 3 × 2% | 0.165 ± 0.009 a | 4.71 ± 0.35 c | 3.42 ± 0.21 c |
| Phase | % Scavenging |
|---|---|
| Initial LTW sample | 47.4 ± 3.1 a |
| Lycopene (10 ppm) | 47.2 ± 2.8 a |
| CPE extract from LTW | 36.3 ± 1.7 b |
| CPE extract from lycopene solution | 46.9 ± 0.9 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giovanoudis, I.; Athanasiadis, V.; Chatzimitakos, T.; Gortzi, O.; Nanos, G.D.; Lalas, S.I. Development of a Cloud Point Extraction Technique Based on Lecithin for the Recovery of Carotenoids from Liquid Tomato Wastewater. Waste 2023, 1, 105-114. https://doi.org/10.3390/waste1010008
Giovanoudis I, Athanasiadis V, Chatzimitakos T, Gortzi O, Nanos GD, Lalas SI. Development of a Cloud Point Extraction Technique Based on Lecithin for the Recovery of Carotenoids from Liquid Tomato Wastewater. Waste. 2023; 1(1):105-114. https://doi.org/10.3390/waste1010008
Chicago/Turabian StyleGiovanoudis, Ioannis, Vassilis Athanasiadis, Theodoros Chatzimitakos, Olga Gortzi, George D. Nanos, and Stavros I. Lalas. 2023. "Development of a Cloud Point Extraction Technique Based on Lecithin for the Recovery of Carotenoids from Liquid Tomato Wastewater" Waste 1, no. 1: 105-114. https://doi.org/10.3390/waste1010008
APA StyleGiovanoudis, I., Athanasiadis, V., Chatzimitakos, T., Gortzi, O., Nanos, G. D., & Lalas, S. I. (2023). Development of a Cloud Point Extraction Technique Based on Lecithin for the Recovery of Carotenoids from Liquid Tomato Wastewater. Waste, 1(1), 105-114. https://doi.org/10.3390/waste1010008










