A Review of Monkeypox: The New Global Health Emergency
Abstract
:1. History and Introduction
2. Virology and Pathogenesis
3. Epidemiology—Current Status
- Contact with one or more individuals with a similar rash who received a probable or confirmed monkeypox diagnosis.
- Close or intimate personal contact with individuals in a social network where monkeypox is present, including MSM who meet partners on the web or on mobile applications or at social events including parties.
- Travel outside the US to a country with confirmed monkeypox cases or to a monkeypox-endemic country.
- Contact with a live or dead wild animal or exotic pet that is an African-endemic species or the use of a product derived from such animals, including lotions, cream, powder, and game meat.
4. Transmission
5. Clinical Manifestations and Diagnosis
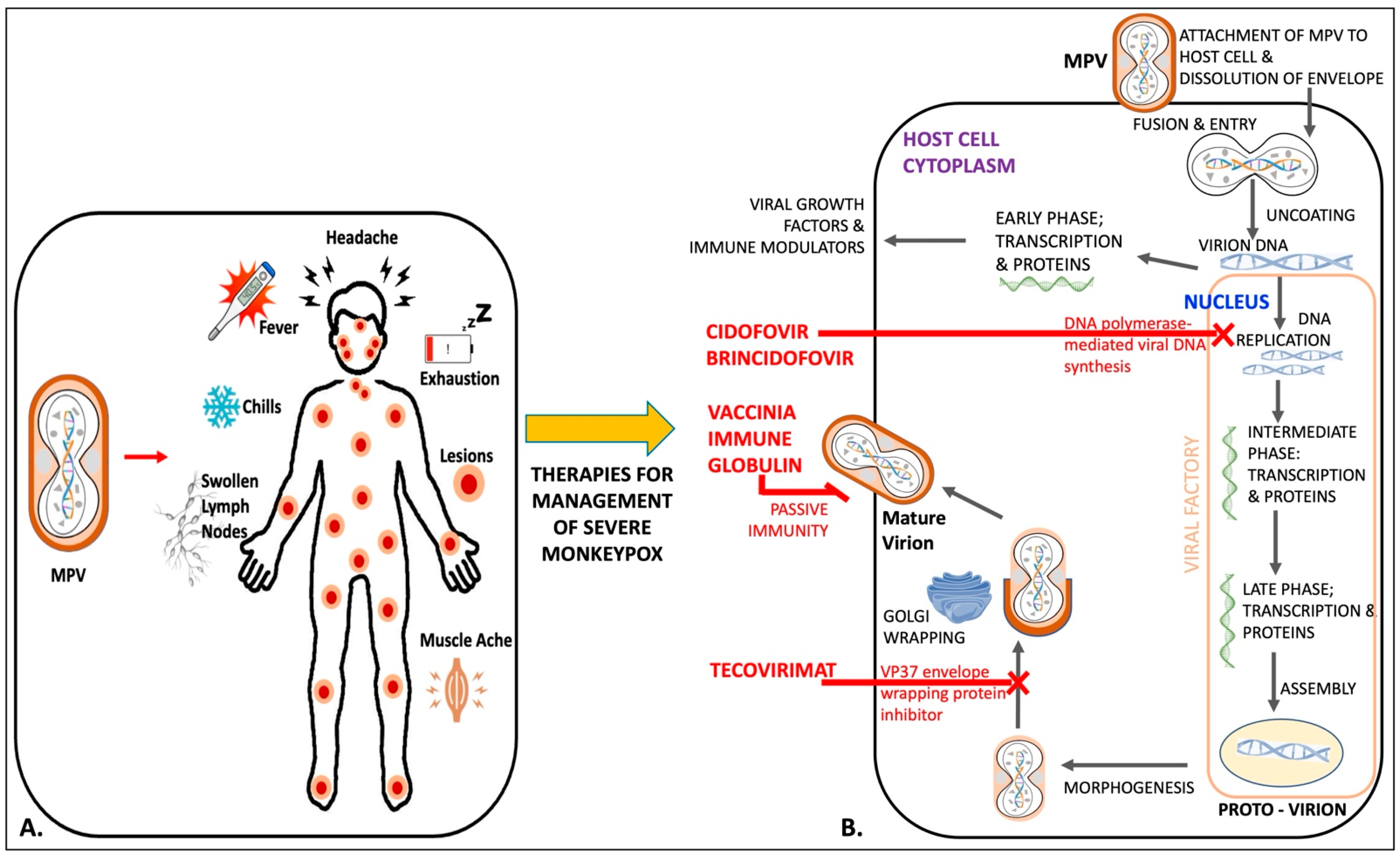
6. Management
6.1. Tecovirimat
6.2. Brincidofovir
6.3. Cidofovir
6.4. Vaccinia Immune Globulin Intravenous (VIGIV)
7. Prevention
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Learned, L.A.; Reynolds, M.G.; Wassa, D.W.; Li, Y.; Olson, V.A.; Karem, K.; Stempora, L.L.; Braden, Z.H.; Kline, R.; Likos, A.; et al. Extended Interhuman Transmission of Monkeypox in a Hospital Community in the Republic of the Congo, 2003. Am. J. Trop. Med. Hyg. 2005, 73, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Nolen, L.D.; Osadebe, L.; Katomba, J.; Likofata, J.; Mukadi, D.; Monroe, B.; Doty, J.; Hughes, C.M.; Kabamba, J.; Malekani, J.; et al. Extended Human-to-Human Transmission during a Monkeypox Outbreak in the Democratic Republic of the Congo. Emerg. Infect. Dis. 2016, 22, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- von Magnus, P.; Andersen, E.K.; Petersen, K.B.; Birch-Andersen, A. A Pox-Like Disease in Cynomolgus Monkeys. Acta Pathol. Microbiol. Scand. 1959, 46, 156–176. [Google Scholar] [CrossRef]
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A Human Infection Caused by Monkeypox Virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593–597. [Google Scholar] [PubMed]
- Cho, C.T.; Wenner, H.A. Monkeypox Virus. Bacteriol. Rev. 1973, 37, 1–18. [Google Scholar] [CrossRef]
- Meyer, H.; Perrichot, M.; Stemmler, M.; Emmerich, P.; Schmitz, H.; Varaine, F.; Shungu, R.; Tshioko, F.; Formenty, P. Outbreaks of Disease Suspected of Being Due to Human Monkeypox Virus Infection in the Democratic Republic of Congo in 2001. J. Clin. Microbiol. 2002, 40, 2919–2921. [Google Scholar] [CrossRef]
- R Pebody Human Monkeypox in Kasai Oriental, Democratic Republic of Congo, February 1996–October 1997: Preliminary Report. Wkly. Releases 1997, 1, 1015. [CrossRef]
- Heymann, D.L.; Szczeniowski, M.; Esteves, K. Re-Emergence of Monkeypox in Africa: A Review of the Past Six Years. Br. Med. Bull. 1998, 54, 693–702. [Google Scholar] [CrossRef]
- Mukinda, V.; Mwema, G.; Kilundu, M.; Heymann, D.; Khan, A.; Esposito, J. Re-Emergence of Human Monkeypox in Zaire in 1996. Lancet 1997, 349, 1449–1450. [Google Scholar] [CrossRef]
- Hutin, Y.J.F.; Williams, R.J.; Malfait, P.; Pebody, R.; Loparev, V.N.; Ropp, S.L.; Rodriguez, M.; Knight, J.C.; Tshioko, F.K.; Khan, A.S.; et al. Outbreak of Human Monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg. Infect. Dis. 2001, 7, 434–438. [Google Scholar] [CrossRef]
- Doty, J.; Malekani, J.; Kalemba, L.; Stanley, W.; Monroe, B.; Nakazawa, Y.; Mauldin, M.; Bakambana, T.; Liyandja Dja Liyandja, T.; Braden, Z.; et al. Assessing Monkeypox Virus Prevalence in Small Mammals at the Human–Animal Interface in the Democratic Republic of the Congo. Viruses 2017, 9, 283. [Google Scholar] [CrossRef] [PubMed]
- Yinka-Ogunleye, A.; Aruna, O.; Dalhat, M.; Ogoina, D.; McCollum, A.; Disu, Y.; Mamadu, I.; Akinpelu, A.; Ahmad, A.; Burga, J.; et al. Outbreak of Human Monkeypox in Nigeria in 2017–18: A Clinical and Epidemiological Report. Lancet Infect. Dis. 2019, 19, 872–879. [Google Scholar] [CrossRef]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The Detection of Monkeypox in Humans in the Western Hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Center for Disease Control and Prevention. Past U.S. Cases and Outbreaks Monkeypox Poxvirus. Available online: https://www.cdc.gov/poxvirus/monkeypox/outbreak/us-outbreaks.html (accessed on 25 July 2022).
- Vaughan, A.; Aarons, E.; Astbury, J.; Brooks, T.; Chand, M.; Flegg, P.; Hardman, A.; Harper, N.; Jarvis, R.; Mawdsley, S.; et al. Human-to-Human Transmission of Monkeypox Virus, United Kingdom, October 2018. Emerg. Infect. Dis. 2020, 26, 782–785. [Google Scholar] [CrossRef] [PubMed]
- Erez, N.; Achdout, H.; Milrot, E.; Schwartz, Y.; Wiener-Well, Y.; Paran, N.; Politi, B.; Tamir, H.; Israely, T.; Weiss, S.; et al. Diagnosis of Imported Monkeypox, Israel, 2018. Emerg. Infect. Dis. 2019, 25, 980–983. [Google Scholar] [CrossRef]
- Yong, S.E.F.; Ng, O.T.; Ho, Z.J.M.; Mak, T.M.; Marimuthu, K.; Vasoo, S.; Yeo, T.W.; Ng, Y.K.; Cui, L.; Ferdous, Z.; et al. Imported Monkeypox, Singapore. Emerg. Infect. Dis. 2020, 26, 1826–1830. [Google Scholar] [CrossRef]
- World Health Organization. Monkeypox Key Facts. Available online: https://www.who.int/news-room/fact-sheets/detail/monkeypox (accessed on 25 July 2022).
- Center for Disease Control and Prevention. 2022 Monkeypox Outbreak Global Map. Poxvirus. Available online: https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html (accessed on 21 July 2022).
- World Health Organization. Director-General Declares the Ongoing Monkeypox Outbreak a Public Health Emergency of International Concern. Available online: https://www.who.int/europe/news/item/23-07-2022-who-director-general-declares-the-ongoing-monkeypox-outbreak-a-public-health-event-of-international-concern (accessed on 25 July 2022).
- Shchelkunov, S.N.; Totmenin, A.V.; Babkin, I.V.; Safronov, P.F.; Ryazankina, O.I.; Petrov, N.A.; Gutorov, V.V.; Uvarova, E.A.; Mikheev, M.V.; Sisler, J.R.; et al. Human Monkeypox and Smallpox Viruses: Genomic Comparison. FEBS Lett. 2001, 509, 66–70. [Google Scholar] [CrossRef]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef]
- Orthopoxvirus. ViralZone. Available online: https://viralzone.expasy.org/149?outline=all_by_species (accessed on 26 July 2022).
- Henderson, D.A.; Borio, L.L. Chapter 58—Smallpox and Monkeypox. In Tropical Infectious Diseases, 2nd ed.; Guerrant, R.L., Walker, D.H., Weller, P.F., Eds.; Churchill Livingstone: Philadelphia, PA, USA, 2006; pp. 621–636. ISBN 978-0-443-06668-9. [Google Scholar]
- Srinivasan, K.; Rao, M. Poxvirus Driven Human Diseases and Emerging Therapeutics. Med. Phamacol. 2022, 2022070300. [Google Scholar] [CrossRef]
- Law, M.; Carter, G.C.; Roberts, K.L.; Hollinshead, M.; Smith, G.L. Ligand-Induced and Nonfusogenic Dissolution of a Viral Membrane. Proc. Natl. Acad. Sci. USA 2006, 103, 5989–5994. [Google Scholar] [CrossRef] [Green Version]
- ClinicalKey. Available online: https://www.clinicalkey.com/#!/content/book/3-s2.0-B9780323482554001326 (accessed on 27 July 2022).
- Moyer, R.W.; Graves, R.L. The Mechanism of Cytoplasmic Orthopoxvirus DNA Replication. Cell 1981, 27, 391–401. [Google Scholar] [CrossRef]
- Vanderplasschen, A.; Hollinshead, M.; Smith, G.L. Intracellular and Extracellular Vaccinia Virions Enter Cells by Different Mechanisms. J. Gen. Virol. 1998, 79, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Shang, J.; Weng, S.; Aliyari, S.R.; Ji, C.; Cheng, G.; Wu, A. Genomic Annotation and Molecular Evolution of Monkeypox Virus Outbreak in 2022. J. Med. Virol. 2022, jmv.28036. [Google Scholar] [CrossRef] [PubMed]
- Isidro, J.; Borges, V.; Pinto, M.; Sobral, D.; Santos, J.D.; Nunes, A.; Mixão, V.; Ferreira, R.; Santos, D.; Duarte, S.; et al. Phylogenomic Characterization and Signs of Microevolution in the 2022 Multi-Country Outbreak of Monkeypox Virus. Nat. Med. 2022, 28, 1569–1572. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.F.; Sohail, M.S.; Quadeer, A.A.; McKay, M.R. Vaccinia Virus Vaccination Is Expected to Elicit Highly Cross-Reactive Immunity to the 2022 Monkeypox Virus. BioRxiv 2022. [Google Scholar] [CrossRef]
- Douglass, N.J.; Dumbell, K.R. DNA Sequence Variation as a Clue to the Phylogenesis of Orthopoxviruses. J. Gen. Virol. 1996, 77, 947–951. [Google Scholar] [CrossRef]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-Rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A Tale of Two Clades: Monkeypox Viruses. J. Gen. Virol. 2005, 86, 2661–2672. [Google Scholar] [CrossRef]
- Chen, N.; Li, G.; Liszewski, M.K.; Atkinson, J.P.; Jahrling, P.B.; Feng, Z.; Schriewer, J.; Buck, C.; Wang, C.; Lefkowitz, E.J.; et al. Virulence Differences between Monkeypox Virus Isolates from West Africa and the Congo Basin. Virology 2005, 340, 46–63. [Google Scholar] [CrossRef]
- Parker, S.; Buller, R.M. A Review of Experimental and Natural Infections of Animals with Monkeypox Virus between 1958 and 2012. Future Virol. 2013, 8, 129–157. [Google Scholar] [CrossRef]
- Zdenek, J.; Fenner, F. Human MonkeyPox; Karger: Basel, Switzerland, 1988. [Google Scholar]
- Gigante, C.M.; Korber, B.; Seabolt, M.H.; Wilkins, K.; Davidson, W.; Rao, A.K.; Zhao, H.; Hughes, C.M.; Minhaj, F.; Waltenburg, M.A.; et al. Multiple Lineages of Monkeypox Virus Detected in the United States, 2021–2022. BioRxiv 2022. [Google Scholar] [CrossRef]
- Kraemer, M.U.G.; Tegally, H.; Pigott, D.M.; Dasgupta, A.; Sheldon, J.; Wilkinson, E.; Schultheiss, M.; Han, A.; Oglia, M.; Marks, S.; et al. Tracking the 2022 Monkeypox Outbreak with Epidemiological Data in Real-Time. Lancet Infect. Dis. 2022, 22, 941–942. [Google Scholar] [CrossRef]
- World Health Organization. 2022 Monkeypox Outbreak: Global Trends—Detailed Case Data. Available online: https://worldhealthorg.shinyapps.io/mpx_global/#3_Detailed_case_data (accessed on 30 August 2022).
- Center for Disease Control and Prevention. Technical Report: Multi-National Monkeypox Outbreak, United States, 2022; CDC: Atlanta, GA, USA, 2022. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/technical-report.html (accessed on 6 August 2022).
- Center for Disease Control and Prevention. Monkeypox in the U.S. Available online: https://www.cdc.gov/poxvirus/monkeypox/index.html (accessed on 20 July 2022).
- Center for Disease Control and Prevention. Monkeypox Cases by Age and Gender, Race/Ethnicity, and Symptoms. Available online: https://www.cdc.gov/poxvirus/monkeypox/response/2022/demographics.html (accessed on 30 August 2022).
- Center for Disease Control and Prevention. Monkeypox in the U.S.—Case Definitions for Use in the 2022 Monkeypox Response. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/case-definition.html (accessed on 9 August 2022).
- Khodakevich, L.; Ježek, Z.; Messinger, D. Monkeypox Virus: Ecology and Public Health Significance. Bull. World Health Organ. 1988, 66, 747–752. [Google Scholar] [PubMed]
- Formenty, P.; Muntasir, M.O.; Damon, I.; Chowdhary, V.; Opoka, M.L.; Monimart, C.; Mutasim, E.M.; Manuguerra, J.-C.; Davidson, W.B.; Karem, K.L.; et al. Human Monkeypox Outbreak Caused by Novel Virus Belonging to Congo Basin Clade, Sudan, 2005. Emerg. Infect. Dis. 2010, 16, 1539–1545. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, A.; Aarons, E.; Astbury, J.; Balasegaram, S.; Beadsworth, M.; Beck, C.R.; Chand, M.; O’Connor, C.; Dunning, J.; Ghebrehewet, S.; et al. Two Cases of Monkeypox Imported to the United Kingdom, September 2018. Eurosurveillance 2018, 23, 1800509. [Google Scholar] [CrossRef]
- Thornhill, J.P.; Barkati, S.; Walmsley, S.; Rockstroh, J.; Antinori, A.; Harrison, L.B.; Palich, R.; Nori, A.; Reeves, I.; Habibi, M.S.; et al. Monkeypox Virus Infection in Humans across 16 Countries—April–June 2022. N. Engl. J. Med. 2022, 387, 679–691. [Google Scholar] [CrossRef]
- Lapa, D.; Carletti, F.; Mazzotta, V.; Matusali, G.; Pinnetti, C.; Meschi, S.; Gagliardini, R.; Colavita, F.; Mondi, A.; Minosse, C.; et al. Monkeypox Virus Isolation from a Semen Sample Collected in the Early Phase of Infection in a Patient with Prolonged Seminal Viral Shedding. Lancet Infect. Dis. 2022, 22, P1267–P1269. [Google Scholar] [CrossRef]
- Alakunle, E.F.; Okeke, M.I. Monkeypox Virus: A Neglected Zoonotic Pathogen Spreads Globally. Nat. Rev. Microbiol. 2022, 20, 507–508. [Google Scholar] [CrossRef]
- Mbala, P.K.; Huggins, J.W.; Riu-Rovira, T.; Ahuka, S.M.; Mulembakani, P.; Rimoin, A.W.; Martin, J.W.; Muyembe, J.-J.T. Maternal and Fetal Outcomes Among Pregnant Women with Human Monkeypox Infection in the Democratic Republic of Congo. J. Infect. Dis. 2017, 216, 824–828. [Google Scholar] [CrossRef]
- Dashraath, P.; Nielsen-Saines, K.; Mattar, C.; Musso, D.; Tambyah, P.; Baud, D. Guidelines for Pregnant Individuals with Monkeypox Virus Exposure. Lancet 2022, 400, 21–22. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention. Monkeypox in the U.S—Clinical Recognition. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html (accessed on 6 August 2022).
- Baetselier, I.D.; Dijck, C.V.; Kenyon, C.; Coppens, J.; den Bossche, D.V.; Smet, H.; Liesenborghs, L.; Vanroye, F.; de Block, T.; Rezende, A.; et al. Asymptomatic Monkeypox Virus Infections among Male Sexual Health Clinic Attendees in Belgium. MedRxiv 2022. [Google Scholar] [CrossRef]
- Patel, A.; Bilinska, J.; Tam, J.C.H.; Fontoura, D.D.S.; Mason, C.Y.; Daunt, A.; Snell, L.B.; Murphy, J.; Potter, J.; Tuudah, C.; et al. Clinical Features and Novel Presentations of Human Monkeypox in a Central London Centre during the 2022 Outbreak: Descriptive Case Series. BMJ 2022, 378, e072410. [Google Scholar] [CrossRef] [PubMed]
- McCollum, A.M.; Damon, I.K. Human Monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Adalja, A.; Inglesby, T. A Novel International Monkeypox Outbreak. Ann. Intern. Med. 2022, 175, 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Girometti, N.; Byrne, R.; Bracchi, M.; Heskin, J.; McOwan, A.; Tittle, V.; Gedela, K.; Scott, C.; Patel, S.; Gohil, J.; et al. Demographic and Clinical Characteristics of Confirmed Human Monkeypox Virus Cases in Individuals Attending a Sexual Health Centre in London, UK: An Observational Analysis. Lancet Infect. Dis. 2022, 22, P1321–P1328. [Google Scholar] [CrossRef]
- Li, Y.; Olson, V.A.; Laue, T.; Laker, M.T.; Damon, I.K. Detection of Monkeypox Virus with Real-Time PCR Assays. J. Clin. Virol. 2006, 36, 194–203. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. HHS Expanding Monkeypox Testing Capacity to Five Commercial Laboratory Companies. Available online: https://www.hhs.gov/about/news/2022/06/22/hhs-expanding-monkeypox-testing-capacity-five-commercial-laboratory-companies.html (accessed on 28 July 2022).
- Center for Disease Control and Prevention. Laboratory Procedures and Biosafety Guidelines—Monkeypox in the U.S. Available online: https://www.cdc.gov/poxvirus/monkeypox/lab-personnel/lab-procedures.html (accessed on 28 July 2022).
- Center for Disease Control and Prevention. Monkeypox in the U.S.—Preparation and Collection of Specimens. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/prep-collection-specimens.html (accessed on 9 August 2022).
- Center for Disease Control and Prevention. Monkeypox in the U.S—Guidance for Tecovirimat Use Under Expanded Access Investigational New Drug Protocol during 2022 U.S. Monkeypox Cases. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/Tecovirimat.html (accessed on 9 August 2022).
- SIGA Technologies. A Double-Blind, Randomized, Placebo-Controlled, Multicenter Study to Assess the Safety, Tolerability, and Pharmacokinetics of TPOXX When Administered Orally for 28 Days in Adult Subjects; SIGA Technologies: New York, NY, USA, 2022. [Google Scholar]
- Adler, H.; Gould, S.; Hine, P.; Snell, L.B.; Wong, W.; Houlihan, C.F.; Osborne, J.C.; Rampling, T.; Beadsworth, M.B.; Duncan, C.J.; et al. Clinical Features and Management of Human Monkeypox: A Retrospective Observational Study in the UK. Lancet Infect. Dis. 2022, 22, 1153–1162. [Google Scholar] [CrossRef]
- Rao, A.K.; Schulte, J.; Chen, T.-H.; Hughes, C.M.; Davidson, W.; Neff, J.M.; Markarian, M.; Delea, K.C.; Wada, S.; Liddell, A.; et al. Monkeypox in a Traveler Returning from Nigeria—Dallas, Texas, July 2021. Morb. Mortal. Wkly. Rep. 2022, 71, 509–516. [Google Scholar] [CrossRef]
- Hutson, C.L.; Kondas, A.V.; Mauldin, M.R.; Doty, J.B.; Grossi, I.M.; Morgan, C.N.; Ostergaard, S.D.; Hughes, C.M.; Nakazawa, Y.; Kling, C.; et al. Pharmacokinetics and Efficacy of a Potential Smallpox Therapeutic, Brincidofovir, in a Lethal Monkeypox Virus Animal Model. mSphere 2021, 6, e00927-20. [Google Scholar] [CrossRef]
- Smee, D.F. Progress in the Discovery of Compounds Inhibiting Orthopoxviruses in Animal Models. Antivir. Chem. Chemother. 2008, 19, 115–124. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention. Monkeypox in the U.S.—Treatment Information for Healthcare Professionals. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/treatment.html (accessed on 9 August 2022).
- Center for Disease Control and Prevention. Prevention—Monkeypox in the U.S. Available online: https://www.cdc.gov/poxvirus/monkeypox/prevention.html (accessed on 28 July 2022).
- Hammarlund, E.; Lewis, M.W.; Carter, S.V.; Amanna, I.; Hansen, S.G.; Strelow, L.I.; Wong, S.W.; Yoshihara, P.; Hanifin, J.M.; Slifka, M.K. Multiple Diagnostic Techniques Identify Previously Vaccinated Individuals with Protective Immunity against Monkeypox. Nat. Med. 2005, 11, 1005–1011. [Google Scholar] [CrossRef]
- Center for Disease Control and Prevention. Considerations for Monkeypox Vaccination. Available online: https://www.cdc.gov/poxvirus/monkeypox/considerations-for-monkeypox-vaccination.html (accessed on 28 July 2022).
- Rao, A.K. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 734–742. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration. Research for B.E. and ACAM2000; FDA: Silver Spring, MD, USA, 2019.
- U.S. Food and Drug Administration. Research for B.E. and JYNNEOS; FDA: Silver Spring, MD, USA, 2021.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajsri, K.S.; Rao, M. A Review of Monkeypox: The New Global Health Emergency. Venereology 2022, 1, 199-211. https://doi.org/10.3390/venereology1020014
Rajsri KS, Rao M. A Review of Monkeypox: The New Global Health Emergency. Venereology. 2022; 1(2):199-211. https://doi.org/10.3390/venereology1020014
Chicago/Turabian StyleRajsri, Kritika Srinivasan, and Mana Rao. 2022. "A Review of Monkeypox: The New Global Health Emergency" Venereology 1, no. 2: 199-211. https://doi.org/10.3390/venereology1020014
APA StyleRajsri, K. S., & Rao, M. (2022). A Review of Monkeypox: The New Global Health Emergency. Venereology, 1(2), 199-211. https://doi.org/10.3390/venereology1020014






