From Forest to Table: Optimizing the Nutritional Value of Acorns through Effective Tannin Extraction †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
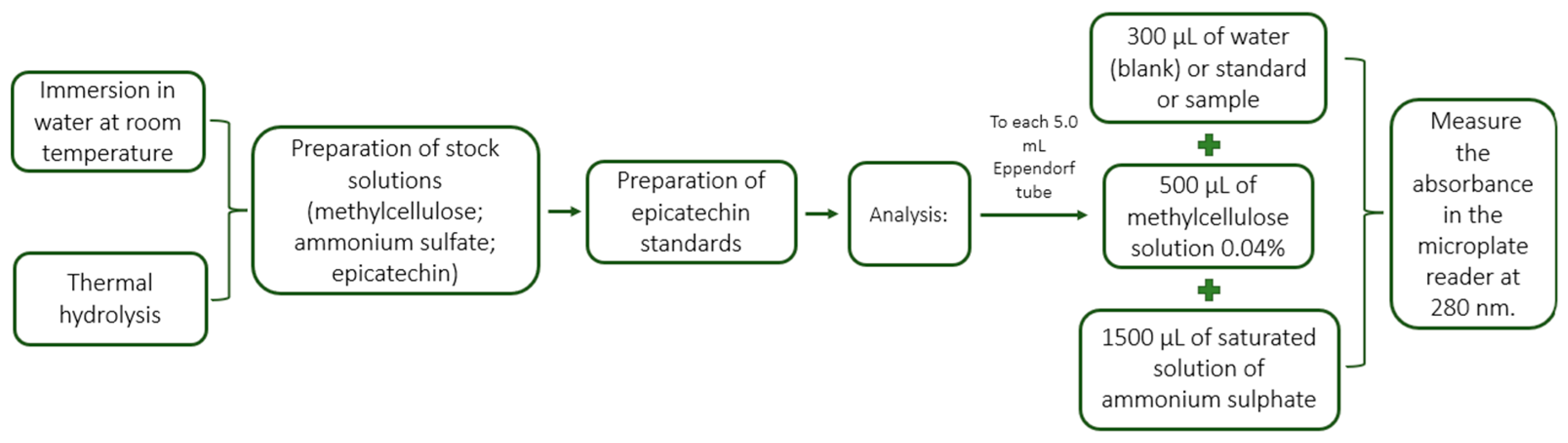
2.2. Tannin Extraction Techniques
2.3. Mineral Analysis
2.4. Statistical Analysis
3. Results and Discussion
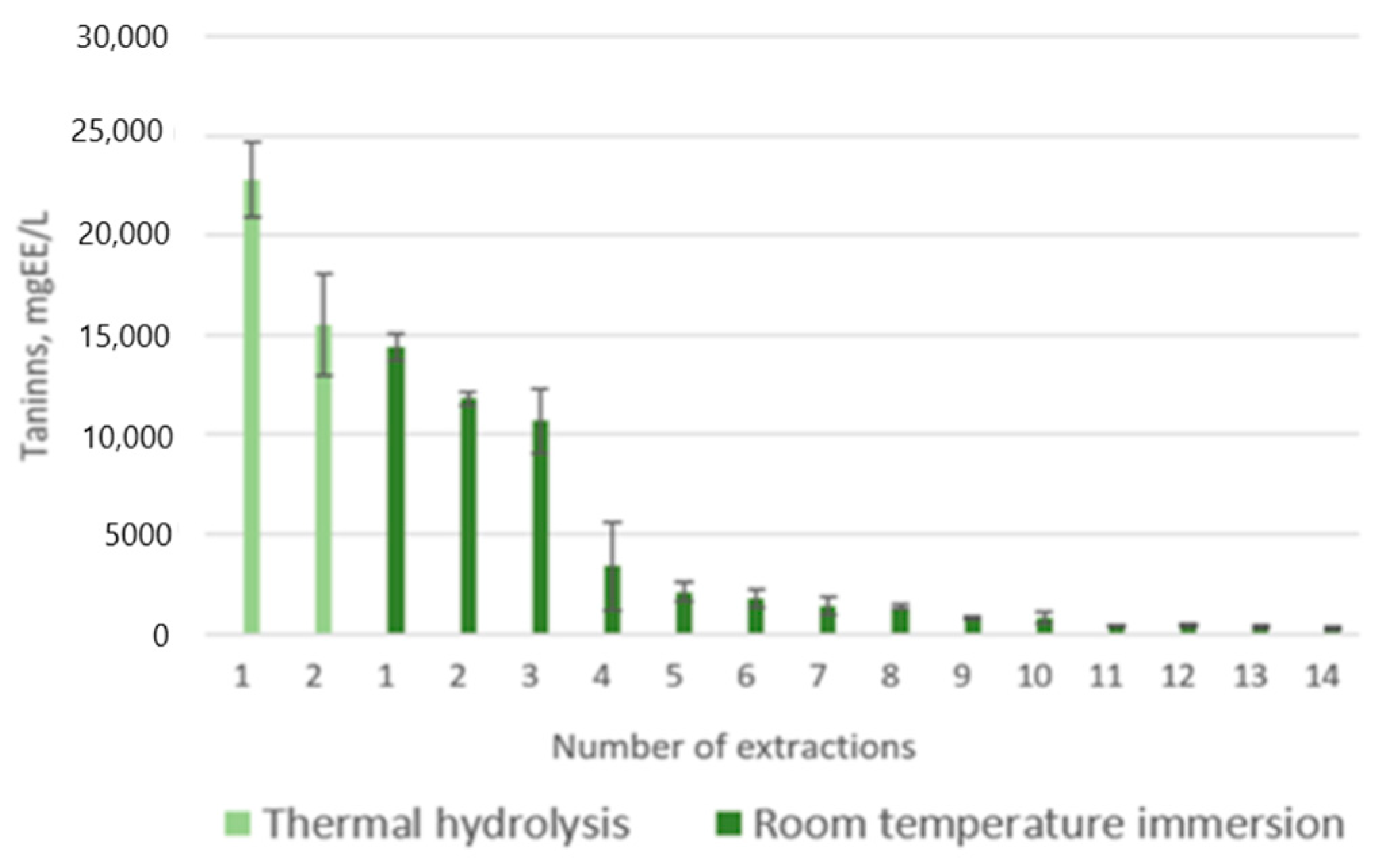
3.1. Condensed Taninns
3.2. Minerals
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Quercus: A Grande Diversidade de Carvalhos em Portugal. Available online: https://florestas.pt/conhecer/quercus-a-grande-diversidade-de-carvalhos-em-portugal/ (accessed on 9 November 2022).
- Vinha, A.F.; Barreira, J.C.M.; Costa, A.S.; Oliveira, M.B.P.P. A New Age for Quercus spp. Fruits: Review on Nutritional and Phytochemical Composition and Related Biological Activities of Acorns. Compr. Rev. Food Sci. Food Saf. 2016, 15, 947–981. [Google Scholar] [CrossRef] [PubMed]
- Pasqualone, A.; Makhlouf, F.Z.; Barkat, M.; Difonzo, G.; Summo, C.; Squeo, G.; Caponio, F. Effect of acorn flour on the physico-chemical and sensory properties of biscuits. Heliyon 2019, 5, e02242. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, M.M.; Ali, E.; Gamal, M.; Farag, M.A. How do coffee substitutes compare to coffee? A comprehensive review of its quality characteristics, sensory characters, phytochemicals, health benefits and safety. Food Biosci. 2021, 43, 101290. [Google Scholar] [CrossRef]
- Lema Costa, C. Manual de Cozinha com Bolota Para a era Pós Petrolífera, 1st ed.; 5livros.pt: Porto, Portugal, 2018. [Google Scholar]
- Castroviejo, S.; Aedo, C.; Cirujano, S.; Laínz, M.; Montserrat, P.; Morales, R.; Muñoz Garmendia, F.; Navarro, C.; Paiva, J.; Soriano, C. (Eds.) Flora Ibérica II; Real Jardim Botânico, CSIC: Madrid, Spain, 1990. [Google Scholar]
- Flora-On Portugal Continental|Flora de Portugal Interactiva. Available online: https://flora-on.pt/?q=Quercus+robur (accessed on 15 July 2023).
- Sarneckis, C.J.; Dambergs, R.G.; Jones, P.; Mercurio, M.; Herderich, M.J.; Smith, P.A. Quantification of condensed tannins by precipitation with methyl cellulose. Aust. J. Grape Wine Res. 2006, 12, 39–49. [Google Scholar] [CrossRef]
- Soares, C.; Tenreiro Machado, J.A.; Lopes, A.M.; Vieira, E.; Delerue-Matos, E.C. Electrochemical impedance spectroscopy characterization of beverages. Food Chem. 2020, 302, 125345. [Google Scholar] [CrossRef] [PubMed]
- Jimoh, W.A.; Fagbenro, O.A.; Adeparusi, E.O. Effect of processing on some minerals, anti-nutrients and nutritional composition of sesame (Sesamum indicum) seed meals. Electron. J. Environ. Agric. Food Chem. 2011, 10, 1858–1864. [Google Scholar]
- Petroski, W.; Minich, D.M. Is There Such a Thing as “Anti-Nutrients”? A Narrative Review of Perceived Problematic Plant Compounds. Nutrients 2020, 12, 2929. [Google Scholar] [CrossRef] [PubMed]
- Nath, H.; Samtiya, M.; Dhewa, T. Beneficial attributes and adverse effects of major plant-based foods anti-nutrients on health: A review. Hum. Nutr. Metab. 2022, 28, 200147. [Google Scholar] [CrossRef]
- Łuczaj, L.; Adamczak, A.; Duda, M. Tannin content in acorns (Quercus spp.) from Poland. Dendrobiology 2014, 72, 103–111. [Google Scholar] [CrossRef]
- Zhang, X.-D.; Thai, P.N.; Lieu, D.K.; Chiamvimonvat, E.N. Cardiac small-conductance calcium-activated potassium channels in health and disease. Pflugers Arch. 2021, 473, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Tarasova, N.V.; Vishnyakova, P.A.; Logashina, Y.A.; Elchaninov, E.A.V. Mitochondrial calcium uniporter structure and function in different types of muscle tissues in health and disease. Int. J. Mol. Sci. 2019, 20, 4823. [Google Scholar] [CrossRef]
- Cormick, G.; Belizán, J.M. Calcium Intake and Health. Nutrients 2019, 11, 1606. [Google Scholar] [CrossRef] [PubMed]
- Ginos, B.N.; Engberink, R.H.O. Estimation of Sodium and Potassium Intake: Current Limitations and Future Perspectives. Nutrients 2020, 12, 3275. [Google Scholar] [CrossRef] [PubMed]
- Mohammadifard, N.; Humphries, K.H.; Gotay, C.; Mena-Sánchez, G.; Salas-Salvadó, J.; Esmaillzadeh, A.; Ignaszewski, A.; Sarrafzadegan, N. Trace minerals intake: Risks and benefits for cardiovascular health. Crit. Rev. Food Sci. Nutr. 2019, 59, 1334–1346. [Google Scholar] [CrossRef] [PubMed]
- Chasapis, C.T.; Ntoupa, P.-S.A.; Spiliopoulou, C.A.; Stefanidou, M.E. Recent aspects of the effects of zinc on human health. Arch. Toxicol. 2020, 94, 1443–1460. [Google Scholar] [CrossRef] [PubMed]
- Dehbalaei, M.G.; Ashtary-Larky, D.; Mesrkanlou, H.A.; Talebi, S.; Asbaghi, O. The effects of magnesium and vitamin E co-supplementation on some cardiovascular risk factors: A meta-analysis. Clin. Nutr. ESPEN 2021, 41, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.L.; Yuan, H.Y.; Yin, X.L.; Wu, C.Z.; Wu, S.B.; Zhou, Z.Q. Effects of chemical form of sodium on the product characteristics of alkali lignin pyrolysis. Bioresour. Technol. 2014, 152, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Holland, C.; Ryden, P.; Edwards, C.H.; Grundy, M.M.-L. Plant cell walls: Impact on nutrient bioaccessibility and digestibility. Foods 2020, 9, 201. [Google Scholar] [CrossRef] [PubMed]
| Acorn Samples | Tannins (mg EE/g) |
|---|---|
| No treatment | |
| Raw | 77.1 ± 2.5 c |
| Thermal hydrolysis | |
| 1 boil | 57.7± 12.1 b |
| 2 boils | 34.8 ± 6.5 a |
| Water immersion | |
| 7 days in water | 34.5 ± 1.3 a |
| 10 days in water | 27.0 ± 1.3 a |
| 14 days in water | 20.5 ± 1.0 a |
| Extractions | - | 1 Boil | 2 Boils | |
|---|---|---|---|---|
| Minerals, mg/g | ||||
| Calcium (Ca) | 11.3 ± 0.2 b | 0.688 ± 0.089 a | 0.396 ± 0.086 a | |
| Copper (Cu) | 0.0349 ± 0.0044 b | 0.0227 ± 0.0037 a | 0.0212 ± 0.0036 a | |
| Iron (Fe) | 0.351 ± 0.015 c | 0.0609 ± 0.0080 b | 0.0292 ± 0.0072 a | |
| Magnesium (Mg) | 0.299 ± 0.044 a | 0.278 ± 0.075 a | 0.332 ± 0.068 a | |
| Potassium (K) | 23.4 ± 0.3 c | 19.4 ± 0.3 b | 18.0 ± 0.3 a | |
| Sodium (Na) | 0.142 ± 0.019 a | 0.287 ± 0.033 b | 0.256 ± 0.002 b | |
| Zinc (Zn) | 0.0193 ± 0.0025 b | 0.00462 ± 0.00189 a | 0.00352 ± 0.00179 a | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monteiro, V.; Soares, C.; Grosso, C.; Delerue-Matos, C.; Ramalhosa, M.J. From Forest to Table: Optimizing the Nutritional Value of Acorns through Effective Tannin Extraction. Biol. Life Sci. Forum 2023, 26, 16. https://doi.org/10.3390/Foods2023-15036
Monteiro V, Soares C, Grosso C, Delerue-Matos C, Ramalhosa MJ. From Forest to Table: Optimizing the Nutritional Value of Acorns through Effective Tannin Extraction. Biology and Life Sciences Forum. 2023; 26(1):16. https://doi.org/10.3390/Foods2023-15036
Chicago/Turabian StyleMonteiro, Vanessa, Cristina Soares, Clara Grosso, Cristina Delerue-Matos, and Maria João Ramalhosa. 2023. "From Forest to Table: Optimizing the Nutritional Value of Acorns through Effective Tannin Extraction" Biology and Life Sciences Forum 26, no. 1: 16. https://doi.org/10.3390/Foods2023-15036
APA StyleMonteiro, V., Soares, C., Grosso, C., Delerue-Matos, C., & Ramalhosa, M. J. (2023). From Forest to Table: Optimizing the Nutritional Value of Acorns through Effective Tannin Extraction. Biology and Life Sciences Forum, 26(1), 16. https://doi.org/10.3390/Foods2023-15036









