Rapid Assessment of Protein Structural Changes from Frost Damage: A Proof-of-Concept Study Using Pittosporum spinescens (Apiales) †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection, Treatment and Spectra Acquisition
2.2. Data Analysis
3. Results and Discussion
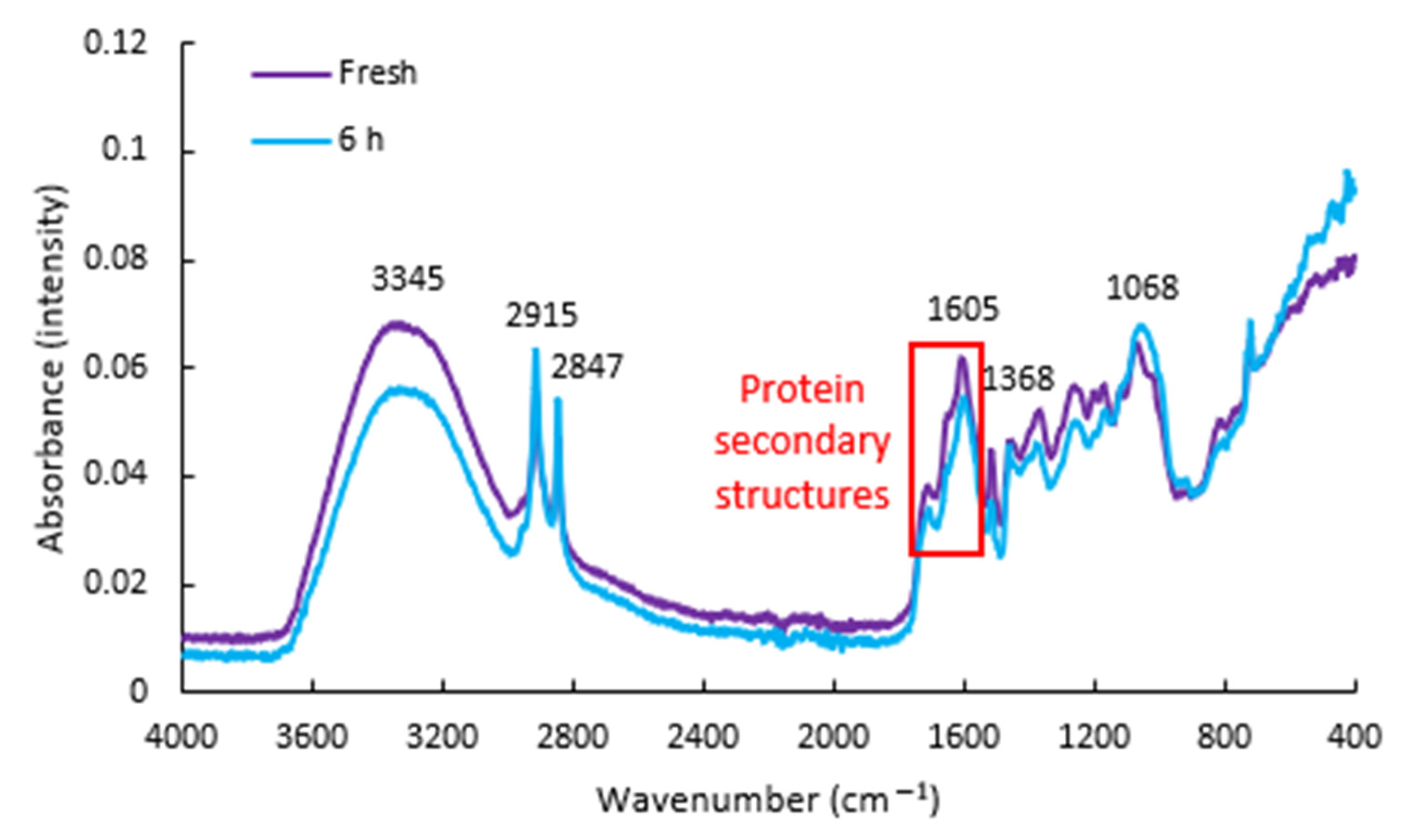
3.1. FTIR Spectra
3.2. Protein Secondary Structure
4. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Inouye, D.W. The ecological and evolutionary significance of frost in the context of climate change. Ecol. Lett. 2000, 3, 457–463. [Google Scholar] [CrossRef]
- Busby, J.R. A biogeoclimatic analysis of Nothofagus cunninghamii (Hook.) Oerst. in southeastern Australia. Aust. J. Ecol. 1986, 11, 1–7. [Google Scholar] [CrossRef]
- Barnes, R.W.; Hill, R.S.; Bradford, J.C. The history of Cunoniaceae in Australia from macrofossil evidence. Aust. J. Bot. 2001, 49, 301–320. [Google Scholar] [CrossRef]
- Bureau of Meteorology. Annual and Monthly Potential Frost Days. Available online: http://www.bom.gov.au/jsp/ncc/climate_averages/frost/index.jsp (accessed on 21 January 2021).
- Blake, J.; Hill, R. An Examination of the Drought and Frost Tolerance of Banksia marginata (Proteaceae) as an Explanation of Its Current Widespread Occurrence in Tasmania. Aust. J. Bot. 1996, 44, 265–281. [Google Scholar] [CrossRef]
- Raymond, C.; Harwood, C.; Owen, J. A Conductivity Method for Screening Populations of Eucalypts for Frost Damage and Frost Tolerance. Aust. J. Bot. 1986, 34, 377–393. [Google Scholar] [CrossRef]
- Percival, G.; Henderson, A. An assessment of the freezing tolerance of urban trees using chlorophyll fluorescence. J. Hortic. Sci. Biotechnol. 2003, 78, 254–260. [Google Scholar] [CrossRef]
- Xin, H.; Zhang, X.; Yu, P. Using synchrotron radiation-based infrared microspectroscopy to reveal microchemical structure characterization: Frost damaged wheat vs. normal wheat. Int. J. Mol. Sci. 2013, 14, 16706–16718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stuart, S.A. Cold Comfort: Diversification and Adaptive Evolution across Latitudinal Gradients. Ph.D. Thesis, University of California, Berkeley, Berkeley, CA, USA, 2011. [Google Scholar]
- Suresh, S.; Karthikeyan, S.; Jayamoorthy, K. FTIR and multivariate analysis to study the effect of bulk and nano copper oxide on peanut plant leaves. J. Sci. Adv. Mater. Dev. 2016, 1, 343–350. [Google Scholar] [CrossRef] [Green Version]
- Kong, J.; Yu, S. Fourier transform infrared spectroscopic analysis of protein secondary structures. Acta Biochim. Biophys. Sin. 2007, 39, 549–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moskal, P.; Wesełucha-Birczyńska, A.; Łabanowska, M.; Filek, M. Adaxial and abaxial pattern of Urtica dioica leaves analyzed by 2DCOS ATR-FTIR as a function of their growth time and impact of environmental pollution. Vib. Spectrosc. 2019, 104, 102948. [Google Scholar] [CrossRef]
- Capron, I.; Robert, P.; Colonna, P.; Brogly, M.; Planchot, V. Starch in rubbery and glassy states by FTIR spectroscopy. Carbohydr. Polym. 2007, 68, 249–259. [Google Scholar] [CrossRef]
- Chen, Y.; Cao, X.; Chang, P.R.; Huneault, M.A. Comparative study on the films of poly (vinyl alcohol)/pea starch nanocrystals and poly (vinyl alcohol)/native pea starch. Carbohydr. Polym. 2008, 73, 8–17. [Google Scholar] [CrossRef]
- Sutinen, M.-L.; Arora, R.; Wisniewski, M.; Ashworth, E.; Strimbeck, R.; Palta, J. Mechanisms of frost survival and freeze-damage in nature. In Conifer Cold Hardiness; Springer: Berlin/Heidelberg, Germany, 2001; pp. 89–120. [Google Scholar]
- Mayland, H.; Cary, J. Frost and chilling injury to growing plants. Adv. Agron. 1970, 22, 203–234. [Google Scholar]
- Kacperska-Palacz, A.; Dlugokecka, E.; Breitenwald, J.; Wciślińska, B. Physiological mechanisms of frost tolerance: Possible role of protein in plant adaptation to cold. Biol. Plant. 1977, 19, 10–17. [Google Scholar] [CrossRef]
- Hiramatsu, H.; Kitagawa, T. FT-IR approaches on amyloid fibril structure. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2005, 1753, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Sundaralingam, M.; Sekharudu, Y. Water-inserted alpha-helical segments implicate reverse turns as folding intermediates. Science 1989, 244, 1333–1337. [Google Scholar] [CrossRef] [PubMed]
- Reingold, N. Cold Weather Agriculture; Science and Technology Division, Library of Congress: Guangzhou, China, 1960. [Google Scholar]
- Thorsen, S.M.; Höglind, M. Assessing winter survival of forage grasses in Norway under future climate scenarios by simulating potential frost tolerance in combination with simple agroclimatic indices. Agric. For. Meteorol. 2010, 150, 1272–1282. [Google Scholar] [CrossRef]
- Maqbool, A.; Shafiq, S.; Lake, L. Radiant frost tolerance in pulse crops—A review. Euphytica 2010, 172, 1–12. [Google Scholar] [CrossRef]
- Junttila, O.; Svenning, M.M.; Solheim, B. Effects of temperature and photoperiod on frost resistance of white clover (Trifolium repens) ecotypes. Physiol. Plant. 1990, 79, 435–438. [Google Scholar] [CrossRef]
- Rognli, O.A. Breeding for improved winter survival in forage grasses. In Plant and Microbe Adaptations to Cold in a Changing World; Springer: Berlin/Heidelberg, Germany, 2013; pp. 197–208. [Google Scholar]
| Peak Centre | Protein Structure | Length of Simulated Frost (h) | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| 0 | 0.25 | 0.5 | 1 | 3 | 6 | |||
| Peak Areas | ||||||||
| 1589 cm−1 | Anti-parallel β-sheet ^ & inter-molecular aggregates | 0 a | 0 a | 0 a | 0 a | 0 a | 4.0 b | 0.002 |
| 1594 cm−1 | 10.3 | 8.5 | 11.0 | 12.5 | 7.4 | 6.6 | 0.648 | |
| 1602 cm−1 | 0 a | 11.6 a | 0 a | 0.7 a | 0 a | 53.7 b | <0.001 | |
| Sum | Anti-parallel β-sheet ^ & inter-molecular aggregates | 10.3 a | 20.1 a | 11.0 a | 13.2 a | 7.4 a | 64.3 b | <0.001 |
| 1609 cm−1 | Inter-molecular aggregates | 32.6 a,b,c | 13.9 a,b | 50.1 c | 35.4 a,b,c | 45.6 b,c | 8.1 a | 0.003 |
| 1619 cm−1 | 6.9 | 23.3 | 6.7 | 9.7 | 1.5 | 8.4 | 0.110 | |
| Sum | Inter-molecular aggregates | 39.5 a,b | 37.2 a,b | 56.8 b | 45.1 a,b | 47.1 b | 16.5 a | 0.009 |
| 1629 cm−1 | β-sheets | 0 | 10.7 | 3.1 | 7.3 | 0 | 4.0 | 0.168 |
| 1635 cm−1 | 15.2 | 16.6 | 5.9 | 9.1 | 16.5 | 5.3 | 0.162 | |
| Sum | β-sheets | 15.2 | 27.3 | 9.0 | 16.4 | 16.5 | 9.3 | 0.196 |
| 1648 cm−1 | Random coils | 14.3 | 8.2 | 5.4 | 14.2 | 22.6 | 4.6 | 0.062 |
| 1653 cm−1 | α-helices | 19.6 a | 7.4 a | 17.5 a | 10.4 a | 4.9 a | 4.4 a | 0.035 |
| 1662 cm−1 | 310 helix | 0.4 | 0 | 0 | 1.1 | 0.8 | 0.5 | 0.706 |
| 1667 cm−1 | β-turn | 0.7 | 0 | 0.2 | 0 | 0.7 | 0.4 | 0.662 |
| Peak heights | ||||||||
| 1653 cm−1 | α-helices | 0.0508 b | 0.0299 a | 0.0508 b | 0.0537 b | 0.0808 c | 0.0409 a,b | <0.001 |
| 1635 cm−1 | β-sheets | 0.0526 b | 0.0313 a | 0.0495 b | 0.0587 b | 0.0860 c | 0.0438 a,b | <0.001 |
| Ratio | α-helices: β-sheets | 0.968 b | 0.954 a,b | 1.022 c | 0.916 a | 0.939 a,b | 0.935 a,b | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, J.B. Rapid Assessment of Protein Structural Changes from Frost Damage: A Proof-of-Concept Study Using Pittosporum spinescens (Apiales). Biol. Life Sci. Forum 2022, 11, 2. https://doi.org/10.3390/IECPS2021-11945
Johnson JB. Rapid Assessment of Protein Structural Changes from Frost Damage: A Proof-of-Concept Study Using Pittosporum spinescens (Apiales). Biology and Life Sciences Forum. 2022; 11(1):2. https://doi.org/10.3390/IECPS2021-11945
Chicago/Turabian StyleJohnson, Joel B. 2022. "Rapid Assessment of Protein Structural Changes from Frost Damage: A Proof-of-Concept Study Using Pittosporum spinescens (Apiales)" Biology and Life Sciences Forum 11, no. 1: 2. https://doi.org/10.3390/IECPS2021-11945
APA StyleJohnson, J. B. (2022). Rapid Assessment of Protein Structural Changes from Frost Damage: A Proof-of-Concept Study Using Pittosporum spinescens (Apiales). Biology and Life Sciences Forum, 11(1), 2. https://doi.org/10.3390/IECPS2021-11945






