Acute CrossFit® Workout Session Impacts Blood Redox Marker Modulation
Abstract
:1. Introduction
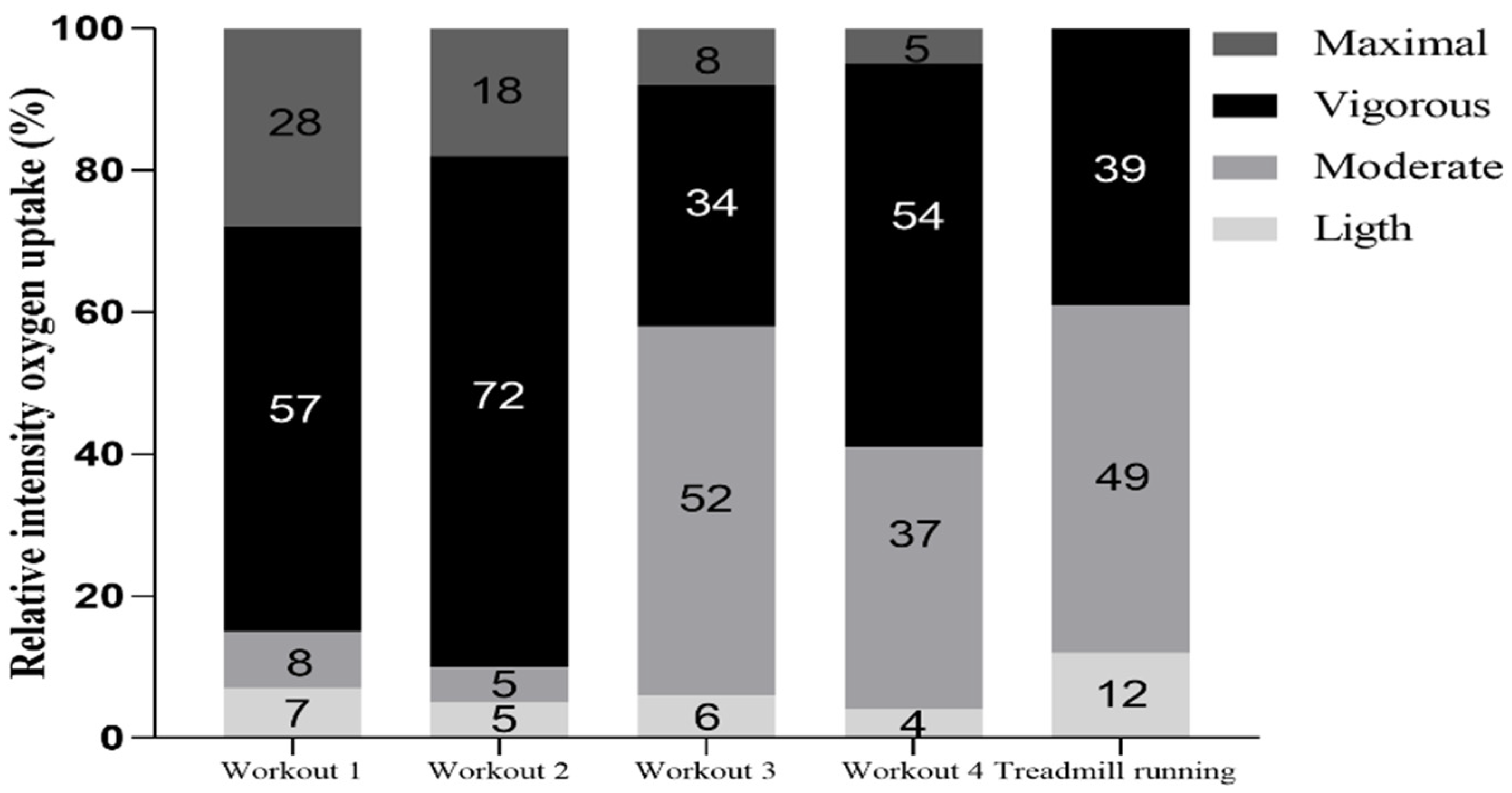
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Practical Applications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thompson, W.R. Worldwide survey of fitness trends for 2019. ACSMs Heal. Fit. J. 2018, 22, 10–17. [Google Scholar] [CrossRef]
- Claudino, J.G.; Gabbett, T.J.; Bourgeois, F.; de Sá Souza, H.; Miranda, R.C.; Mezêncio, B.; Soncin, R.; Filho, C.A.C.; Bottaro, M.; Hernandez, A.J.; et al. CrossFit Overview: Systematic Review and Meta-analysis. Sports Med. Open 2018, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.F.; Solana, R.S.; Moya, D.; Marin, J.M.S.; Ramón, M.M. Acute physiological responses during Crossfit® workouts. Eur. J. Sport Sci. 2015, 35, 114–124. [Google Scholar]
- Schlegel, P. CrossFit® Training Strategies from the Perspective of Concurrent Training: A Systematic Review. J. Sports Sci. Med. 2020, 19, 670. [Google Scholar]
- Murawska-Cialowicz, E.; Wojna, J.; Zuwala-Jagiello, J. Crossfit training changes brain-derived neurotrophic factor and irisin levels at rest, after wingate and progressive tests, and improves aerobic capacity and body composition of young physically active men and women. J. Physiol. Pharmacol. 2015, 66, 811–821. [Google Scholar] [PubMed]
- Kliszczewicz, B.; Quindry, C.J.; Blessing, L.D.; Oliver, D.G.; Esco, R.M.; Taylor, J.K. Acute exercise and oxidative stress: CrossFit™ vs. treadmill bout. J. Hum. Kinet. 2015, 47, 81–90. [Google Scholar] [CrossRef] [Green Version]
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef] [Green Version]
- Radak, Z.; Ishihara, K.; Tekus, E.; Varga, C.; Posa, A.; Balogh, L.; Boldogh, I.; Koltai, E. Exercise, oxidants, and antioxidants change the shape of the bell-shaped hormesis curve. Redox Biol. 2017, 12, 285–290. [Google Scholar] [CrossRef] [Green Version]
- Tryfidou, D.V.; McClean, C.; Nikolaidis, M.G. DNA damage following acute aerobic exercise: A systematic review and meta-analysis. Sports Med. 2020, 50, 103–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magalhães, J.; Ferreira, R.; Marques, F.; Olivera, E.; Soares, J.; Ascensao, A. Indoor climbing elicits plasma oxidative stress. Med. Sci. Sports Exerc. 2007, 39, 955–963. [Google Scholar] [CrossRef]
- Ascensão, A.; Ferreira, R.; Marques, F.; Oliveira, E.; Azevedo, V.; Soares, J.; Magalhães, J. Effect of off-road competitive motocross race on plasma oxidative stress and damage markers. Br. J. Sports Med. 2007, 41, 101–105. [Google Scholar] [CrossRef] [Green Version]
- Ascensão, A.; Rebelo, A.; Oliveira, E.; Marques, F.; Pereira, L.; Magalhães, J. Biochemical impact of a soccer match—analysis of oxidative stress and muscle damage markers throughout recovery. Clin. Biochem. 2008, 41, 841–851. [Google Scholar] [CrossRef]
- Cheng, A.J.; Jude, B.; Lanner, J.T. Intramuscular mechanisms of overtraining. Redox Biol. 2020, 35, 101480. [Google Scholar] [CrossRef]
- Kurutas, E.B. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: Current state. J. Nutr. 2015, 15, 71. [Google Scholar] [CrossRef] [Green Version]
- Powers, S.K.; Deminice, R.; Ozdemir, M.; Yoshihara, T.; Bomkamp, M.P.; Hyatt, H. Exercise-induced oxidative stress: Friend or foe? J. Sport Health Sci. 2020, 9, 415–425. [Google Scholar] [CrossRef]
- Quan, H.; Koltai, E.; Suzuki, K.; Aguiar-Júnior, A.S.; Pinho, R.; Boldogh, I.; Berkes, I.; Radak, Z. Exercise, redox system and neurodegenerative diseases. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165778. [Google Scholar] [CrossRef]
- Radak, Z.; Zhao, Z.; Koltai, E.; Ohno, H.; Atalay, M. Oxygen consumption and usage during physical exercise: The balance between oxidative stress and ROS-dependent adaptive signaling. Antioxid. Redox Signal. 2013, 18, 1208–1246. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef] [Green Version]
- Magalhães, J.; Rebelo, A.; Oliveira, E.; Silva, J.R.; Marques, F.; Ascensão, A. Impact of Loughborough Intermittent Shuttle Test versus soccer match on physiological, biochemical and neuromuscular parameters. Eur. J. Appl. Physiol. 2010, 108, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.R.; Ascensão, A.; Marques, F.; Seabra, A.; Rebelo, A.; Magalhães, J. Neuromuscular function, hormonal and redox status and muscle damage of professional soccer players after a high-level competitive match. Eur. J. Appl. Physiol. 2013, 113, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Pingitore, A.; Lima, G.P.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef]
- Powers, S.K.; Radak, Z.; Ji, L.L. Exercise-induced oxidative stress: Past, present and future. J. Physiol. 2016, 594, 5081–5092. [Google Scholar] [CrossRef] [Green Version]
- Sohail, M.U.; Al-Mansoori, L.; Al-Jaber, H. Assessment of Serum Cytokines and Oxidative Stress Markers in Elite Athletes Reveals Unique Profiles Associated With Different Sport Disciplines. Front. Physiol. 2020, 11, 600888. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Fridovich, I. Superoxide radicals, superoxide dismutases and the aerobic lifestyle. J. Photochem. Photobiol. 1978, 28, 33–741. [Google Scholar] [CrossRef]
- Pizzino, G.; Irrera, N.; Cucinotta, M. Oxidative stress: Harms and benefits for human health. Oxidative Med. Cell Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef]
- Quindry, J.C.; Stone, W.L.; King, J.; Broeder, C.E. The effects of acute exercise on neutrophils and plasma oxidative stress. Med. Sci. Sports Exerc. 2003, 35, 1139–1145. [Google Scholar] [CrossRef]
- He, F.; Chuang, C.C.; Zhou, T.; Sedlock, D.A.; Zuo, L. Redox correlation in muscle lengthening and immune response in eccentric exercise. PLoS ONE 2018, 13, e0208799. [Google Scholar] [CrossRef] [PubMed]
- Henríquez-Olguín, C.; Boronat, S.; Cabello-Verrugio, C.; Jaimovich, E.; Hidalgo, E.; Jensen, T.E. The emerging roles of nicotinamide adenine dinucleotide phosphate oxidase 2 in skeletal muscle redox signaling and metabolism. Antioxid. Redox Signal. 2019, 31, 1371–1410. [Google Scholar] [CrossRef]
- Banh, S.; Treberg, J.R. The pH sensitivity of H2O2 metabolism in skeletal muscle mitochondria. FEBS Lett. 2013, 587, 1799–1804. [Google Scholar] [CrossRef] [Green Version]
- Patwell, D.M.; McArdle, A.; Morgan, J.E.; Patridge, T.A.; Jackson, M.J. Release of reactive oxygen and nitrogen species from contracting skeletal muscle cells. Free. Radic. Biol. Med. 2004, 37, 1064–1072. [Google Scholar] [CrossRef]
- Espinosa, A.; Leiva, A.; Peña, M.; Müller, M.; Debandi, A.; Hidalgo, C.; Carrasco, M.A.; Jaimovich, E. Myotube depolarization generates reactive oxygen species through NAD (P) H oxidase; ROS-elicited Ca2+ stimulates ERK, CREB, early genes. J. Cell. Physiol. 2006, 209, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Henríquez-Olguin, C.; Knudsen, J.R.; Raun, S.H.; Li, Z.; Dalbram, E.; Treebak, J.T.; Sylow, L.; Holmdahl, R.; Richter, E.A.; Jaimovich, E.; et al. Cytosolic ROS production by NADPH oxidase 2 regulates muscle glucose uptake during exercise. Nat. Commun. 2019, 10, 4623. [Google Scholar] [CrossRef] [Green Version]
- Henríquez-Olguín, C.; Renani, L.B.; Arab-Ceschia, L.; Raun, S.H.; Bhatia, A.; Li, Z.; Knudsen, J.R.; Holmdahl, R.; Jensen, T.E. Adaptations to high-intensity interval training in skeletal muscle require NADPH oxidase 2. Redox Biol. 2019, 24, 101188. [Google Scholar] [CrossRef]
- Kliszczewicz, B.M.; Esco, M.R.; Quindry, J.C.; Blessing, D.L.; Oliver, G.D.; Taylor, K.J.; Price, B.M. Autonomic responses to an acute bout of high-intensity body weight resistance exercise vs. treadmill running. J. Strength Cond. Res. 2016, 30, 1050–1058. [Google Scholar] [CrossRef]
- Bogdanis, G.C.; Stavrinou, P.; Fatouros, I.G.; Philippou, A.; Chatzinikolaou, A.; Draganidis, D.; Ermidis, G.; Maridaki, M. Short-term high-intensity interval exercise training attenuates oxidative stress responses and improves antioxidant status in healthy humans. Food Chem. Toxicol. 2013, 61, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Shing, C.M.; Peake, J.M.; Ahern, S.M.; Strobel, N.A.; Wilson, G.; Jenkins, D.G.; Coombes, J.S. The effect of consecutive days of exercise on markers of oxidative stress. Appl. Physiol. Nutr. Metab. 2007, 32, 677–685. [Google Scholar] [CrossRef]
- Nocella, C.; Cammisotto, V.; Pigozzi, F.; Borrione, P.; Fossati, C.; D’Amico, A.; Cangemi, R.; Peruzzi, M.; Gobbi, G.; Ettorre, E. Impairment between oxidant and antioxidant systems: Short-and long-term implications for athletes’ health. Nutrients 2019, 11, 1353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolagal, V.; Karanam, S.A.; Dharmavarapu, P.K.; D’Souza, R.; Upadhya, S.; Kumar, V.; Kedage, V.; Muttigi, M.S.; Shetty, J.K.; Prakash, M. Determination of oxidative stress markers and their importance in early diagnosis of uremia-related complications. Indian J. Nephrol. 2009, 19, 8. [Google Scholar]
- Shigenaga, M.K.; Hagen, T.M.; Ames, B.N. Oxidative damage and mitochondrial decay in aging. Proc. Natl. Acad. Sci. USA 1994, 91, 10771–10778. [Google Scholar] [CrossRef] [Green Version]
- Ortiz-Franco, M.; Planells, E.; Quintero, B.; Acuña-Castroviejo, D.; Rusanova, I.; Escames, G.; Molina-López, J. Effect of melatonin supplementation on antioxidant status and DNA damage in high intensity trained athletes. Int. J. Sports Med. 2017, 38, 1117–1125. [Google Scholar] [CrossRef]
- Palazzetti, S.; Richard, M.J.; Favier, A.; Margaritis, I. Overloaded training increases exercise-induced oxidative stress and damage. Can. J. Appl. Physiol. 2003, 28, 588–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, K.H.; Reichhold, S.; Neubauer, O. Impact of endurance and ultraendurance exercise on DNA damage. Ann. N. Y. Acad. Sci. 2011, 1229, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Aksenov, M.Y.; Markesbery, W.R. Changes in thiol content and expression of glutathione redox system genes in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci. Lett. 2001, 302, 141–145. [Google Scholar] [CrossRef]

| (n = 10) | |
|---|---|
| Age (years) | 30.8 ± 5.6 |
| Training experience (years) | 3.6 ± 0.2 |
| Height (cm) | 169.5 ± 10.3 |
| Weight (kg) | 73.5 ± 13 |
| Body mass index (kg/m2) | 26 ± 2 |
| Fat mass (%) | 19.9 ± 3.9 |
| Lean Mass (kg) | 55 ± 12 |
| Maximal oxygen uptake (mL·kg 1·min−1) | 44.9 ± 7.2 |
| Maximal respiratory quotient | 1.1 ± 0.9 |
| Maximal heart rate (b·min−1) | 181 ± 11 |
| Data are mean standard deviation (SD) |
| Workouts | Duration | Repetitions | Recovery | Load (Men and Women) |
|---|---|---|---|---|
| Rowing ergometer | 4 min | Maximum | 4 min | ---- |
| Air bike | 4 min | Maximum | 8 min | ---- |
| Randy | 6 min | Maximum | 8 min | 35 and 25 kg |
| Deadlift, toes to bar, dumbbell thruster and dumbbell walking lunges | 6 min circuit | 10 each exercise | ---- | Deadlift (80 and 55 kg), dumbbell thruster (30 and 20 kg) and dumbbell walking lunges (30 and 20 kg) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rios, M.; Macan, T.; Stevanović-Silva, J.; Nhusawi, K.; Fernandes, R.J.; Beleza, J.; Ascensão, A.; Magalhães, J. Acute CrossFit® Workout Session Impacts Blood Redox Marker Modulation. Physiologia 2021, 1, 13-21. https://doi.org/10.3390/physiologia1010004
Rios M, Macan T, Stevanović-Silva J, Nhusawi K, Fernandes RJ, Beleza J, Ascensão A, Magalhães J. Acute CrossFit® Workout Session Impacts Blood Redox Marker Modulation. Physiologia. 2021; 1(1):13-21. https://doi.org/10.3390/physiologia1010004
Chicago/Turabian StyleRios, Manoel, Tamires Macan, Jelena Stevanović-Silva, Kamonrat Nhusawi, Ricardo J. Fernandes, Jorge Beleza, António Ascensão, and José Magalhães. 2021. "Acute CrossFit® Workout Session Impacts Blood Redox Marker Modulation" Physiologia 1, no. 1: 13-21. https://doi.org/10.3390/physiologia1010004










