4.1. Inter-Individual Variation in Stable Isotope Values
The δ
13C and δ
15N values of the Raschoille population as a whole range from −22.5‰ to −20.2‰ and from 7.6‰ to 11.9‰, respectively. Although differences in the diet are largely responsible for variation in individual stable isotope values, variation can also result from non-dietary factors including developmental stage and physiological stress related to disease or pregnancy [
51,
52,
53].
The dietary timespan reflected in bone collagen stable isotope values largely depends on the age-at-death of the individual and the skeletal element sampled [
54,
55]. In infants and young children, bone growth and collagen turnover are very rapid, such that bone collagen isotope values in younger children generally reflect the average diet over less than a year before death [
55]. During adolescence, there is rapid bone growth, collagen synthesis and remodeling; it is estimated that complete collagen turnover may occur in cortical bone (e.g., the femoral shaft) in as little as two years [
55,
56]. Moreover, increased protein requirements may result in reduced discrimination, or fractionation, of dietary
14N/
15N [
51,
52]. It is widely assumed that in adults collagen turnover is much slower and stable isotope signatures reflect average dietary intake over at least ten and as much as thirty years before death [
54,
57,
58]. However, a study of collagen turnover in cortical bone of femora shafts of present-day large mammals [
59] found that the isotopic composition of cortical bone collagen reflects that of the diet
during the period of skeletal growth with the implication that collagen recovered from the femoral cortical bone of adults (irrespective of age-at-death) represents isotopic values during adolescence. In comparison, the turnover of collagen in cancellous bone, such as ribs and vertebrae, is relatively rapid and continuous [
54,
56]. For example, adult rib collagen can be completely replaced in as little as one year and up to five years, and therefore represents the shorter-term dietary intake [
56].
It follows that comparing the isotopic values of different skeletal elements in separate individuals, as well as individuals of differing ages (i.e., adult vs. sub-adult), may lead to the assumption of variation in dietary intake where none exists. Detailed evaluation of the significance of the differences in δ
13C and δ
15N between individuals requires the demography of the sampled population, as well as the skeletal element analyzed, to be known. Unfortunately, the specific skeletal element sampled and associated demographic information were not documented for all the analyzed Raschoille Cave remains. Those specimens for which the information was recorded include both adults and sub-adults. The majority (12/14) of the Series A samples were taken from the cortical tissue of a limb bone. However, in one individual a cervical vertebra was sampled, while in another individual a rib was analyzed. The following discussion of individual differences in the diet focuses on those samples in Series A for which both age-at-death and the skeletal element sampled are known (
Table 1).
Although there is no statistically significant difference between the adult and sub-adult stable isotope values (Mann–Whitney U-test δ
15N,
p = 0.688921; δ
13C,
p = 0.378478), the sub-adults have a wider spread of values, with δ
15N ranging from 7.6‰ to 11.9‰. This relatively larger range may relate in part to
15N enrichment associated with nursing [
60] and to the use of lower trophic level foods, such as cereals, during weaning [
55].
No statistical correlation between age and δ
15N value (Spearman Rank Correlation Test
rs = −0.23424,
p = 0.61315) was evident in the Raschoille Cave population (n.b., for the Spearman Rank analysis, the age of the sub-adults was taken as the mid-point of the range quoted in
Table 1). However, sample RC.01, which has the highest δ
15N value of 11.9‰ is from the only individual identified as an infant, approximately 1–2 years old at death. The
15N enrichment may therefore reflect a nursing signal. Fuller et al. [
60] demonstrated that nursing infants had δ
15N values that were elevated by 2–3‰ over those of their mothers. The δ
13C values also exhibited a slight enrichment of c. 1‰ during breastfeeding but decreased rapidly when solid foods were introduced to the infant diet. The Raschoille Cave infant (RC.01) had a δ
15N value that was 2.7‰ above the mean value of the adults from the site. In contrast, the infant’s δ
13C value of −21.8‰ was lower than the adult mean δ
13C value of −20.8‰. The combination of relatively elevated δ
15N and depleted δ
13C suggests the child was in the process of being weaned, with solid foods introduced to complement breastfeeding.
Bayesian dietary modelling is presented only for those individuals in Series A identified as adults because of the influence of developmental stage on non-dietary related variations in stable isotope values.
4.2. Diet Reconstruction Using Linear Mixing Models
Linear mixing models (LMMs) have been widely employed in archaeology to estimate past human diets from stable isotope data. The results of a simple, one proxy (δ
13C), two food source (terrestrial vs. marine) LMM of west-central Scottish Mesolithic and Neolithic groups are presented in
Table 7. Dietary endpoints used for the model were δ
13C = −20.0‰ for 100% terrestrial diets and δ
13C = −12.0‰ for 100% marine diet [
61].
LMMs have limited applicability. Using LMMs to evaluate dietary constituents from two variables (i.e., δ
13C and δ
15N) can distinguish the contributions of, at most, three isotopically distinct food sources [
65].
Conventionally, it has been widely accepted that in individuals consuming protein-adequate diets, dietary protein will be directly routed to bone collagen. Consequently, bone collagen δ
13C and δ
15N values reflect dietary protein sources [
66,
67]. Where protein intake is inadequate, there is de novo synthesis of non-essential amino acids from dietary carbohydrates and/or lipids. In such instances, the carbon isotope ratios of bone collagen reflect ‘whole’ diet, i.e., protein, carbohydrate and lipid sources [
62,
67].
The effects of nutrient scrambling on collagen isotope values may not only be problematic in individuals with low protein diets. Fernandes et al. [
68] demonstrated that in mammals a proportion of collagen carbon is derived from dietary lipids and/or carbohydrates and that this is uniform, c. 26 ± 4%, irrespective of dietary intake.
Differential routing of nutrients (or nutrient scrambling) undermines the notional linear correlation of δ
13C and δ
15N assumed in LMMs and may mask the contribution of certain food sources to the diet in conventional LMMs. Milner et al. [
6] observed that Neolithic groups with a low-protein diet could have derived up to 20% of their dietary protein from marine resources without any evident impact on the δ
13C value. Hedges [
62] contrasted models of a low protein diet, in which 20–30% of the protein was derived from marine sources, with a high protein 100% terrestrial diet. The difference in the δ
13C values between the two hypothetical diets was just 0.3‰ [
62].
4.3. Diet Reconstruction Using FRUITS
Both protein-routed and nutrient-scrambled Bayesian mixing models were generated, and the results are discussed below. Bayesian mixing models (BMMs) potentially offer higher-resolution reconstructions of the diet compared to simple LMMs with two fixed endpoints. The Bayesian mixing model Food Reconstruction Using Isotopic Transferred Signals (FRUITS) can accurately evaluate the contribution of multiple food sources and allows users to incorporate uncertainty in trophic level offsets and food source isotope values into dietary reconstructions [
36,
69].
The diets of the Raschoille Cave individuals were reconstructed using FRUITS (version 3.0 Beta). Since the accuracy of dietary reconstructions may be affected by the developmental stage of the individual (as discussed above), only individuals thought to be adults or adolescents (age-at-death >10 years) were included in the analysis (
Table 1, RC.07–12). Children plus individuals for whom
reliable demographic information is lacking (
Table 1, RC.1–6, 13–28) were excluded.
The existence of offsets between the diet and consumer collagen stable isotope ratios is well-established [
20,
70]. The δ
13C
diet-collagen offset is widely acknowledged to be c. +5‰ [
16,
36]. However, the size of the δ
15N
diet-collagen offset has been the subject of some debate [
22,
23,
24]. Here we use the diet to collagen offsets set out in [
69]—δ
13C
diet-collagen = +4.8 ± 0.5‰ and δ
15N
diet-collagen = +5.5 ± 0.5‰.
There are also uncertainties in the stable isotope values of consumers (largely reflecting instrument measurement error); these were set at 0.5‰ for both δ
13C and δ
15N [
36,
69].
Uncertainties in food-source stable isotope values (reflecting differences in preparation methods and seasonal and physiological variations in animal metabolism) should also be incorporated into dietary reconstructions. Following [
36,
69] this uncertainty was set at a minimum of 1‰ for all food sources.
A further consideration for dietary reconstructions that rely on the isotope values of archaeological animal bones as a proxy for food web values is the offset between bone collagen and tissue (e.g., muscle, fat) consumed. These offsets are:
terrestrial herbivores Δ
13C
protein-collagen = −2.0‰, and Δ
15N
protein-collagen = 0‰, Δ
13C
lipid-protein = −8.0‰ [
71];
fish Δ
13C
protein-collagen = −1.0‰, and Δ
15N
protein-collagen = +1.5‰, Δ
13C
lipid-protein = −7.0‰ [
71].
The offset for seals was taken from Pickard and Bonsall [
72].
4.4. Food Sources
There are five categories of food sources that may have contributed to the diets of the Early Neolithic individuals from Raschoille Cave: (i) plant foods, including domesticated cereals and wild plants; (ii) meat or dairy from terrestrial mammals; (iii) shellfish; (iv) fish; and (v) sea mammals.
The δ
13C and δ
15N values for plant foods were drawn from Fernandes et al. [
69] and based on data originally published by Bogaard et al. [
73]. The δ
13C and δ
15N values of carbonized plant remains have been established to be appropriate proxies for the raw plant stable isotope ratios [
74,
75]. Non-manured plants had mean energy δ
13C of −23.5‰, protein δ
13C of −26.0 ± 1.0‰ and δ
15N of 2.0 ± 1.0‰, while manured plants had an elevated mean protein δ
15N of 4.5 ± 1.0‰ [
69]. Evidence for manuring in Neolithic Scotland is limited. High levels of soil phosphate in a paleo land surface at North Mains, Strathallan, preserved beneath a Bronze Age mound, indicated the presence of manure. However, it is not certain that this relates to the manuring of arable land; it may reflect animal grazing or penning [
76]. Consequently, non-manured plant values were used for the models evaluated below.
A review of published stable isotope data for terrestrial fauna from Scottish Mesolithic and Neolithic sites reveals significant regional variability in the δ
13C and δ
15N of both wild game and domestic livestock. Therefore, for this study, we have attempted to reconstruct the
local food web by using baseline indicators from the Oban area. Isotopic signatures for terrestrial mammals were determined from archaeological samples from two sites, Raschoille Cave and Carding Mill Bay I (
Table 2). Not all samples have been directly dated, but they are all assumed to derive from the time range c. 6500–3000 cal BC, corresponding to the later part of the Mesolithic and the Early Neolithic. We detected no significant temporal shifts in mammalian baseline values over this period nor any indications of herbivores grazing on seaweed or salt marsh plants (cf. Britton et al.; Jones et al.) [
77,
78].
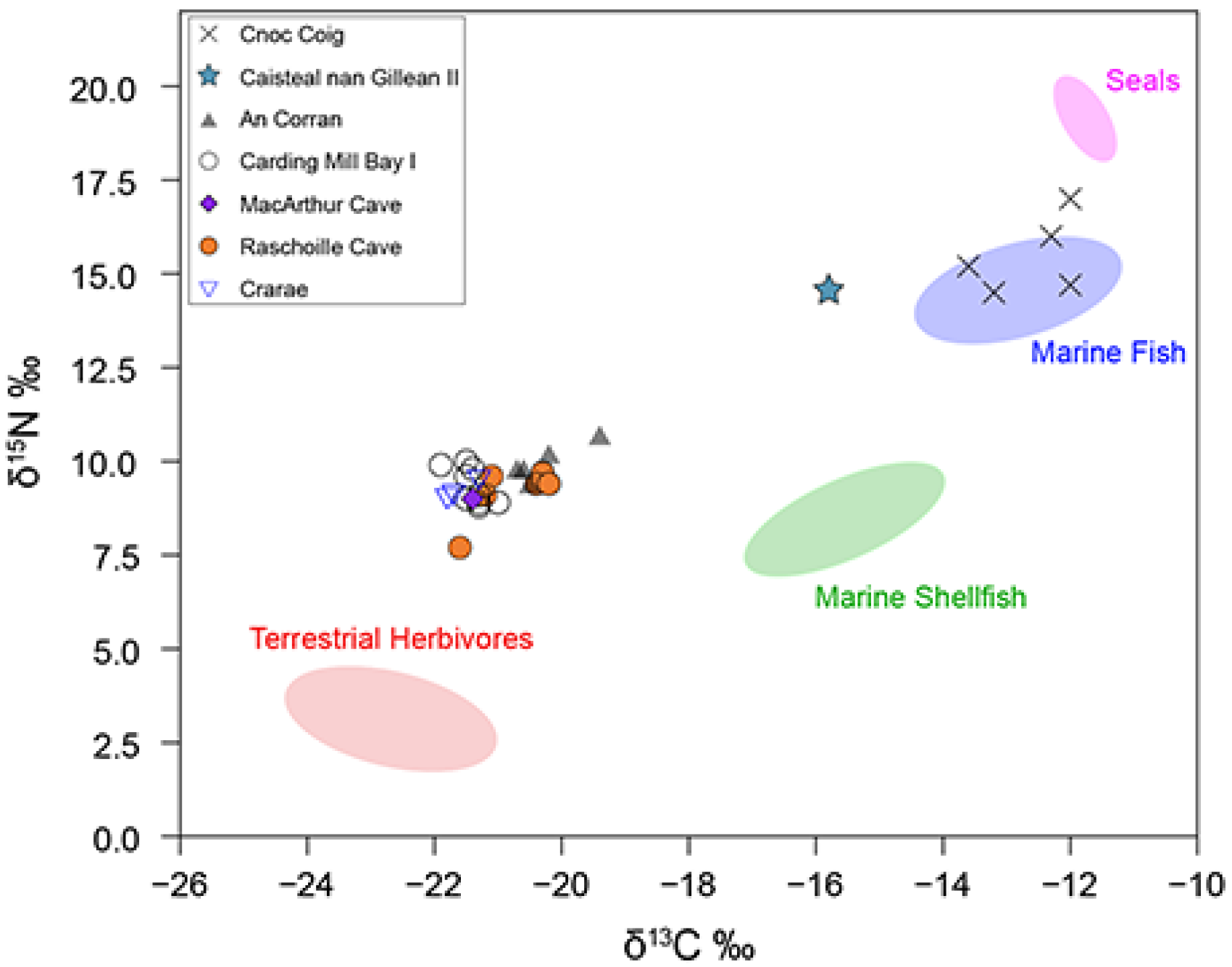
Mean bone collagen δ13C and δ15N values of all terrestrial herbivores and omnivores (n = 28) are −22.7 ± 0.6‰ and 3.1 ± 0.5‰, respectively. There is a small but significant difference in the mean δ13C and δ15N values for red deer (n = 6, −22.2‰, 2.6‰) and domestic cattle (n = 8, −23.1‰, +3.5‰) (Mann–Whitney U test δ13C p = 0.011821, δ15N p= 0.002415). Incorporating the bone collagen to protein tissue offset, the terrestrial animal protein values used for our FRUITS models for Early Neolithic groups were δ13C = −24.7 ± 1.0‰ and δ15N = 3.1 ± 1.0‰. For the Mesolithic groups, wild terrestrial animal protein values were used, δ13C =—22.2 ± 1.0‰ and δ15N = 2.6 ± 1.0‰.
The baseline isotope values of marine resources (represented by fish and sea mammal bones) were drawn primarily from archaeological specimens from central-west Scotland (see
Table 3). Mean carbon and nitrogen stable isotope bone collagen values of archaeological fish were δ
13C = −12.8 ± 0.9‰ and δ
15N = 14.6 ± 0.8‰ (
n = 5), while the mean values for seals were δ
13C = −11.8 ± 0.2‰ and δ
15N = 19.1 ± 0.4‰ (
n = 3). Incorporating the bone collagen to protein tissue offset and food source uncertainties, the fish protein values were δ
13C = −13.8 ± 1.0‰ and δ
15N = 16.1 ± 1.0‰ and seal protein values were δ
13C = −14.1 ± 1.0‰ and δ
15N = 18.5 ± 1.0‰.
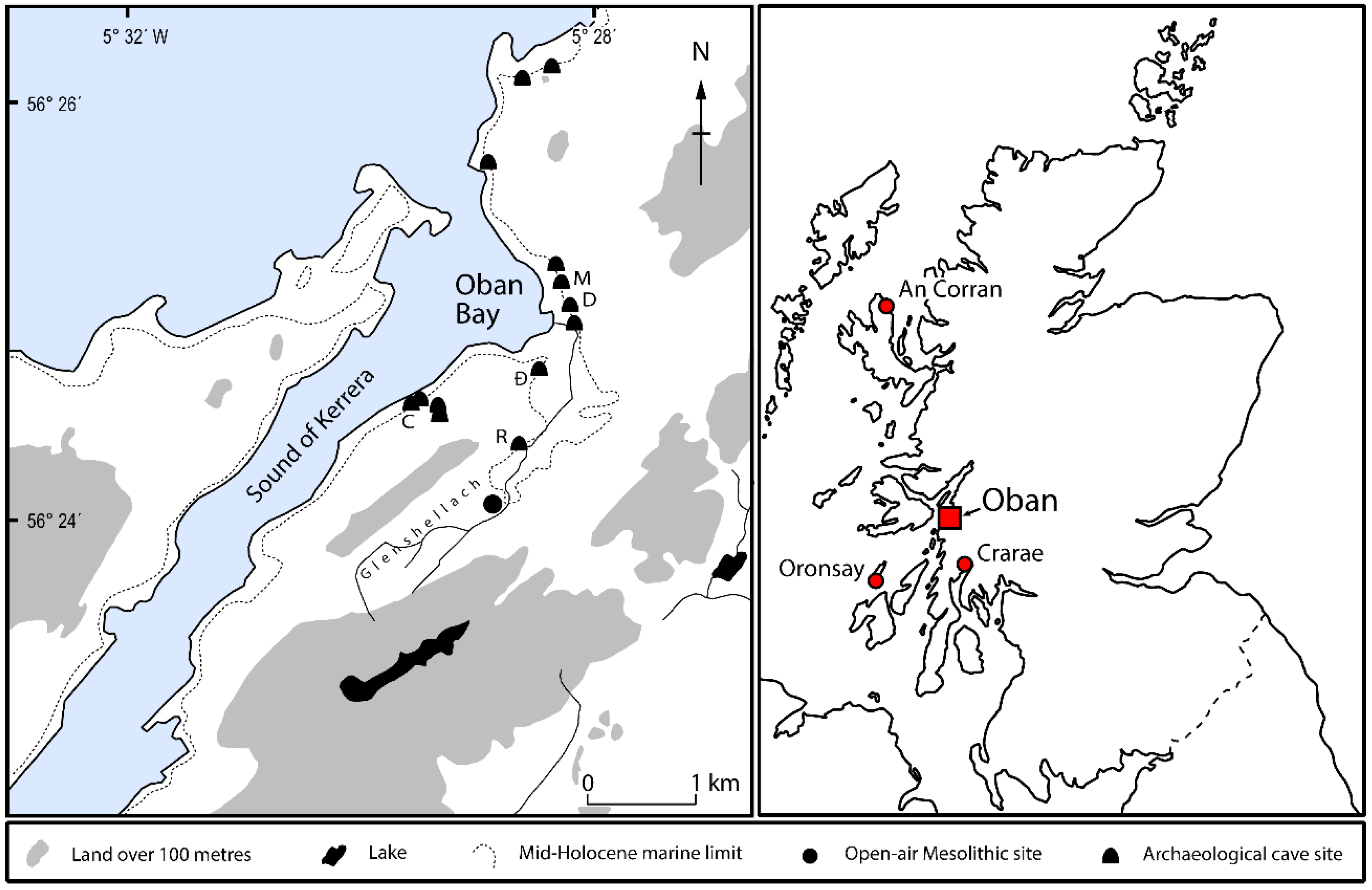
The archaeological evidence for shellfish consumption by Neolithic communities in the Oban Bay area is equivocal. Abundant shellfish remains (mainly periwinkles and bivalves) were recovered from the Neolithic deposits in Raschoille Cave and appear to have been closely associated with concentrations of human bones [
13]. Possible explanations for the co-occurrence of shells with human bones are that the shells are: (1) remains of food offerings to the dead; or (2) food refuse from funeral feasts or external domestic activities (or even pre-existing midden deposits) that were brought into the cave and heaped over human remains as part of the burial ritual. At Carding Mill Bay I, the offset between the
14C ages of human and animal bones from the same contexts [
39] (
Table 7 and
Table 8) suggests the human remains were either inserted into pre-existing midden deposits or that ‘ancient’ midden deposits were transported to the site from elsewhere and heaped over human remains in symbolic acts of burial.
Since shellfish flesh is not recovered from archaeological sites, the isotope signatures of modern shellfish may be used in diet reconstructions [
63]. However, using modern samples as proxies for prehistoric resources introduces further uncertainties into dietary reconstructions and therefore modern comparanda should be used cautiously. Carbon isotope ratios of modern specimens do not directly mirror the values of prehistoric resources. The use of fossil fuels in the industrial era has resulted in
13C-depleted atmospheric CO
2—known as the ‘Suess effect’ [
79,
80,
81]. Modern atmospheric CO
2 carbon isotope values are lower than pre-industrial values [
82]. Therefore, a correction factor needs to be applied to the δ
13C values of modern samples when used as proxies for prehistoric food webs. Keeling et al. [
82] described a c. 2‰ difference between modern (i.e., AD 2014) and pre-industrial δ
13C values of atmospheric CO
2 and noted that this depletion may be larger in terrestrial plants and by inference in terrestrial food webs. The Suess effect has had a smaller impact on oceanic δ
13C; in the North Atlantic,
13C-depletion of up to 0.8‰ is evident [
83].
The effects of lipid removal pre-treatments on the δ
15N values of modern samples are also problematic. Lipid δ
13C is
13C-depleted in comparison to bone collagen values—by up to 7‰—and it has been recommended that lipids should be removed from samples prior to isotope ratio measurements [
84]. However, the solvents used to remove lipids have been demonstrated to alter δ
15N values [
85,
86]. Sotiropoulos et al. [
87] recommended a two-fold approach—measurement of two separate portions of a single sample, δ
13C from a lipid-extracted portion of a sample and δ
15N from an untreated portion of the sample.
Additionally, regional-scale spatial variation in intra-species δ
13C and δ
15N values of marine organisms are evident in British coastal waters, largely reflecting the extent of mixing of waters, as well as differences in phytoplankton communities, microalgae and cyanobacteria [
88]. Where possible, the modern comparanda should be sourced locally to the archaeological sites under investigation. The shellfish flesh isotope values used for this study were selected from data published in [
30]. The measurements from two genera of shellfish, limpets and periwinkles (the most frequently recovered species from archaeological shell middens and the most common species in the intertidal zone along the west coast of Scotland) were utilized. Only those specimens with C:N ratios in the range 2.9–3.6 after lipid extraction were used as proxies for prehistoric shellfish. Additionally, the δ
13C of lipid-extracted samples were combined with the δ
15N values from the untreated portion of the sample. Mean carbon and nitrogen stable isotope values of shellfish flesh were δ
13C = −14.6 ± 0.7‰ and δ
15N = 7.4 ± 1.6‰ (
n = 11).
4.5. FRUITS Model Predictions
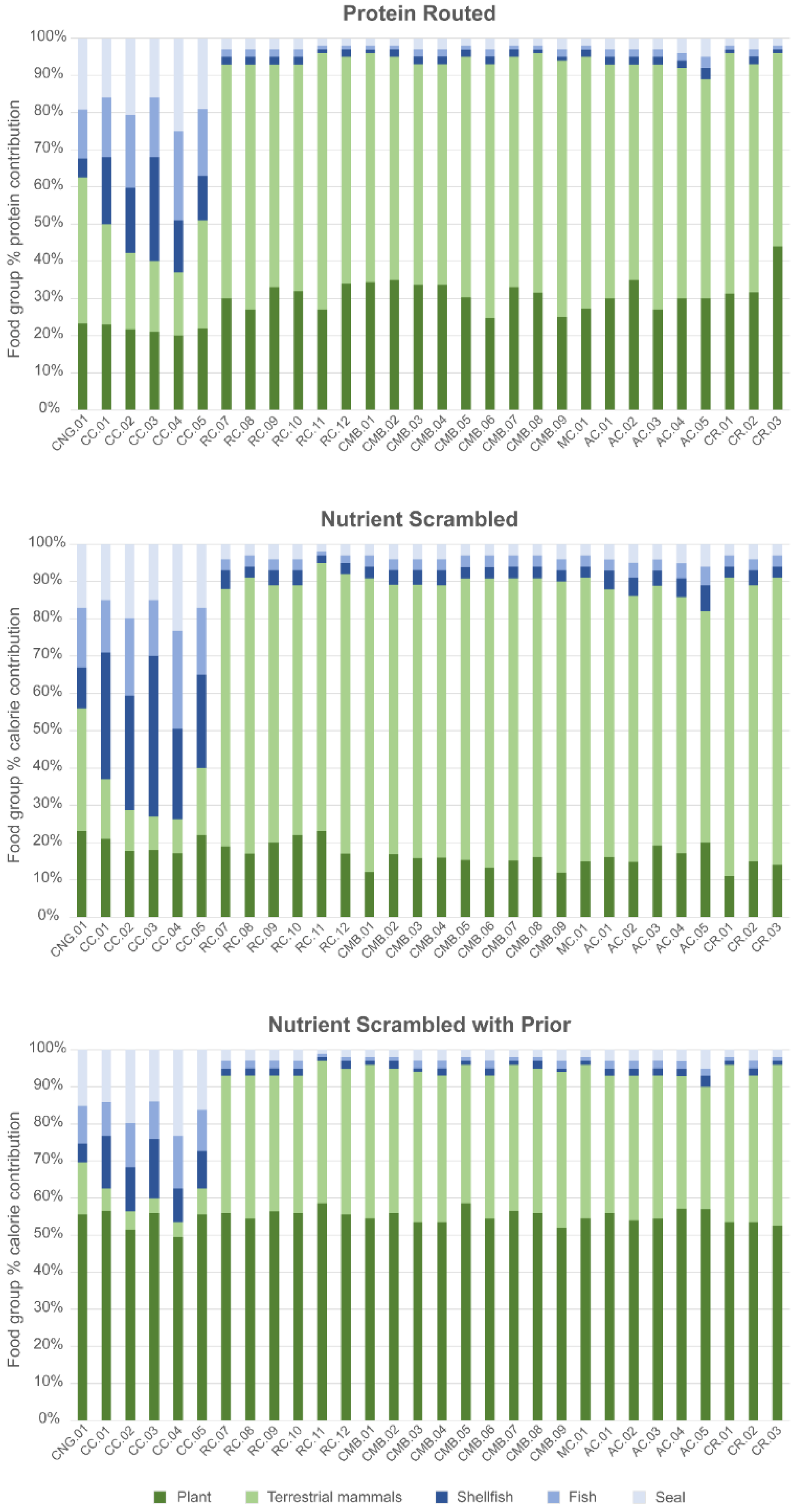
Three models were evaluated using FRUITS. The different models were designed to test the impact of varying parameters to reconstruct the diet and to assess how this might impact our understanding of the role of marine resources in diet:
For each of the three models, FRUITS’ reconstructions indicate that terrestrial food sources (i.e., plant foods and meat and/or dairy from terrestrial mammals) dominated dietary intake at Raschoille Cave.
The nutrient-scrambled model with the prior protein restriction (Model 3) suggests that similar proportions of calories were derived from terrestrial plant and animal sources. However, because food source data (and local plant food isotope values, in particular) are limited, there is uncertainty in the precise contribution these resources made to individual diets.
Both the protein-routed and the nutrient-scrambled models suggest that marine resources were a minor component of the diet with shellfish comprising 0.0–5.9% (95% CI) of protein intake (Model 1) and whole diet calories (Model 3). In the same way as shellfish, fish likely made a very small or no contribution to the diet (0.0–5.9%, 95% CI). Seals may have provided more protein and calories to the diets of the people interred at Raschoille Cave than fish and shellfish, but this contribution would still have been small (protein (Model 1) 0.0–8.9%, 95% CI; and calories (Model 3) 0.0–8.9%, 95% CI).
4.6. Neolithic Diets in Central-West Scotland
Bownes et al. [
39] determined, using Bayesian modelling, that the protein sources of four individuals from Carding Mill Bay contained “modest amounts (15 ± 11 to 21 ± 14%)” of marine foods. However, Bownes et al. [
39] did not factor wild plant foods or cereals into their FRUITS calculations but assumed that 100% of dietary protein was derived from either terrestrial meats or the flesh of aquatic animals. Bownes [
30] (p. 150) acknowledged that plant foods would have been exploited by both Mesolithic and Neolithic groups in Scotland but excluded them from dietary models on the basis that “plants contain very little … protein in relation to animal meat”.
This assumption is problematic for two main reasons:
The protein content of animal meats varies depending on the species and the cut of meat. Some cereals and wild plants have similar protein content to that of certain animal products (see
Table 8). Hazelnuts and barley, which are among the most frequently identified plant foods recovered from Scottish Neolithic sites [
89], have relatively high protein yields;
Deriving dietary protein exclusively from animal products is atypical [
23]. Modern mean protein intake in developed countries is 57% animal protein and 43% plant protein, while in developing countries the relative proportions of animal and plant protein are 30% and 70%, respectively. Animal protein as a proportion of total protein intake may be higher among groups that consume large quantities of dairy products [
23].
Table 8.
Protein content (in grams) per 100g of wild plants, cereals and animal products. The plants listed have all been identified at Scottish Neolithic sites [
89]. All food protein data were drawn from the
USDA Food Composition Database [
90].
Table 8.
Protein content (in grams) per 100g of wild plants, cereals and animal products. The plants listed have all been identified at Scottish Neolithic sites [
89]. All food protein data were drawn from the
USDA Food Composition Database [
90].
| Food | Protein Content (g) per 100 g |
|---|
| Plant products | |
| Barley, hulled | 12.48 |
| Flax seed | 16.67 |
| Hazelnut | 14.95 |
| Oat | 12.50 |
| Rye | 6.98 |
| Wheat—durum | 13.68 |
| Wheat—spelt | 14.57 |
| Animal meats | |
| Beef mince (grass-fed cattle) | 18.75 |
| Beef steak (grass-fed cattle) | 23.01 |
| Lamb shoulder steak | 16.48 |
| Lamb loin | 20.88 |
| Pork carcass | 13.91 |
| Pork loin | 20.71 |
| Venison (grass-fed deer) | 22.32 |
| Dairy products | |
| Sheep’s milk | 5.98 |
| Sheep’s milk cheese | 25.00 |
| Whole cow’s milk | 3.33 |
| Cow’s milk cheese, hard | 25.00 |
| Cow’s milk, cream cheese | 6.15 |
While the use of wild plant foods in Neolithic western Scotland is well documented [
89,
91], cereal cultivation has been suggested to be of minor importance based on a perceived lack of evidence for large-scale vegetation clearance before the Bronze Age [
92]. This argument must be weighed against the sharp increase in cereal (
Hordeum)-type pollen after c. 3800 cal BC recorded in pollen diagrams across the Oban region [
12] (
Figure 4) and archaeobotanical and other evidence for cereal cultivation in the Hebrides from c. 3700 cal BC [
93,
94,
95].
The Raschoille individuals have similar stable isotope signatures, and by inference likely had similar diets, to other west Scottish Early Neolithic populations. Both FRUITS (each model) and LMM reconstructions indicate that terrestrial resources dominated diets at all Neolithic sites (
Figure 4;
Table 4,
Table 5,
Table 6 and
Table 7). At Crarae, there may have been a greater reliance on terrestrial animal food sources than among Early Neolithic populations elsewhere. However, what is perhaps surprising in the FRUITS reconstructions is that at all the sites considered in this study, including both Mesolithic and Neolithic, plant foods made a significant contribution to dietary calories.
The LMM estimations of the proportion of marine resources in Neolithic diet do not exceed uncertainty (i.e., >10% [
62]; see
Table 7). Our FRUITS models confirm that marine resources were likely a minor component in the diets of Early Neolithic groups in western Scotland. The proportion of calories derived from marine food sources modelled slightly higher in some individuals from An Corran than in the individuals from the other Neolithic sites included in this study. As with plant foods, reliance on marine resources may have varied regionally in Neolithic Scotland [
63]. However, these differences are
very small.