Abstract
Background/Objectives: The relative efficacy of 8 mg aflibercept compared to 2 mg in treating neovascular age-related macular degeneration (nAMD) has not been fully established. This study aims to compare the visual and anatomical outcomes of aflibercept 8 mg versus 2 mg in patients with nAMD in both treatment-naïve individuals with no history of anti-VEGF treatment and those previously treated with intravitreal injections. Methods: This retrospective study included 13 eyes treated with aflibercept 8 mg and 14 eyes with aflibercept 2 mg in treatment-naïve patients, along with 15 eyes switched to aflibercept 8 mg previously treated with other intravitreal injections and 15 eyes continued on aflibercept 2 mg in patients. Baseline and one-month post-injection changes in best-corrected visual acuity (BCVA) and central macular thickness (CMT) were assessed. Results: In treatment-naïve patients, the aflibercept 8 mg group showed a significant improvement in BCVA (logMAR 0.19 ± 0.23 to 0.13 ± 0.20, p = 0.0156), while the 2 mg group did not. Both doses reduced CMT significantly, with a greater reduction in the 8 mg group (dCMT 28.60% vs. 24.08%, p = 0.0220). In previously treated patients, no significant changes in BCVA were noted in either group; however, both groups showed significant reductions in CMT. Conclusions: Real-world data demonstrated that aflibercept 8 mg led to substantial improvements in anatomical outcomes one month after injection, irrespective of previous intravitreal injection history. However, significant improvements in visual outcomes were observed exclusively in treatment-naïve patients. Further large-scale, long-term studies are required to determine the proportion of patients who experience improvement and to assess whether these improvements are maintained over time.
1. Introduction
Approximately 6–7 million intravitreal injections of anti-vascular endothelial growth factor (VEGF) agents are performed each year in the United States [1,2]. The global prevalence of age-related macular degeneration (AMD) is projected to rise to around 288 million by 2040, and the use of anti-VEGF agents is expected to continue increasing accordingly [3]. VEGF plays a crucial role in the development of macular neovascularization (MNV) and in the regulation of vascular permeability [4]. The implementation of anti-VEGF therapy has resulted in a significant reduction in both the frequency and severity of visual impairment in patients with neovascular age-related macular degeneration (nAMD), thereby greatly enhancing visual prognosis [5]. Recent regulatory approvals of new anti-VEGF agents have expanded the therapeutic arsenal for nAMD, now including bevacizumab, ranibizumab, aflibercept, brolucizumab, and faricimab.
To reduce treatment burden and facilitate extended dosing intervals in patients with nAMD and diabetic macular edema (DME), a high-dose intravitreal formulation of aflibercept (8 mg) was developed [6]. This formulation delivers a higher molar concentration than the standard 2 mg dose and was engineered in a 70-μL volume with optimized solubility, viscosity, stability, and tolerability [7]. Initial evaluation of aflibercept 8 mg in the treatment of nAMD was conducted in the phase 2 CANDELA trial. At 16 weeks, a higher proportion of eyes treated with the 8 mg dose were free of central subfield fluid compared to those treated with the standard 2 mg dose (51% vs. 34%) [7]. Following the phase 3 PULSAR trial, which demonstrated that aflibercept 8 mg administered every 12 or 16 weeks was non-inferior to aflibercept 2 mg administered every 8 weeks in terms of best-corrected visual acuity (BCVA) and central macular thickness (CMT) outcomes, aflibercept 8 mg was introduced in 2023 as a novel anti-VEGF therapy [8].
Aflibercept 8 mg is a relatively recent addition to anti-VEGF therapies, and evidence from prospective and retrospective comparative studies evaluating its effectiveness in real-world clinical settings remains limited. In this study, we aimed to compare visual and anatomical outcomes one month after intravitreal injection of aflibercept 8 mg versus 2 mg in patients with nAMD, including both treatment-naïve eyes and those with a history of prior intravitreal injections.
2. Materials and Methods
This study was approved by the Ethics Committee of Fuchu Hospital (ID number: 2024009, Date 9 July 2024) and adhered to the tenets of the Declaration of Helsinki. Due to the retrospective approach of this analysis, no informed consent was obtained from the patients. Informed consent is not required as per local legislation, “Ethical Guidelines for Life Science and Medical Research Involving Human Subjects”. The Fuchu Hospital’s website allowed participants to withdraw from the study.
This was a single-center, retrospective observational study without a predefined control group. Patients were stratified into subgroups based on prior treatment status and the dose of aflibercept received, and comparative analyses were conducted between these groups. Treatment-naïve eyes were defined as eyes with no history of intravitreal anti-VEGF injection before the study period, whereas previously treated eyes were defined as eyes that had received at least one intravitreal anti-VEGF injection before enrollment. This study enrolled 13 eyes from 12 treatment-naïve patients receiving aflibercept 8 mg, 14 eyes from 13 treatment-naïve patients receiving aflibercept 2 mg, 15 eyes from 13 patients previously treated with other intravitreal injections and switched to aflibercept 8 mg, and 15 eyes from 14 patients previously treated with aflibercept 2 mg who continued on the same dosage for nAMD. All patients included in this study were of Japanese ethnicity and received intravitreal injections between January 2023 and September 2024, with a follow-up period of one month. Patients with a history of intravitreal injections of other agents who were switched to aflibercept 8 mg were identified as those in whom the treating physician determined a reduction in %CMT of less than 10% or insufficient improvement in BCVA under prior anti-VEGF therapy.
All patients were administered intravitreal injections of either 2.0 mg/0.05 mL of aflibercept (Eyler™, Bayer HealthCare, Osaka, Japan) or 8.0 mg/0.07 mL of aflibercept (Eyler 8 mg™, Bayer HealthCare, Osaka, Japan) [8]. Following the application of a topical anesthetic (0.4% oxybuprocaine hydrochloride; Benoxil™; Santen Pharmaceutical Co., Tokyo, Japan), the injections were carried out via the standard pars plana technique, positioned 3.5 mm posterior to the limbus, using a 30-gauge needle under sterile conditions in a dedicated proc, Osaka edure room.
The inclusion criteria were as follows: (1) age over 50 years, (2) a diagnosis of MNV due to nAMD, (3) no prior history of intravitreal injections for the treatment-naïve cases, (4) a minimum interval of eight weeks since the last injection for the switch cases and aflibercept 2 mg continuous cases, and (5) followed up one month after injection. The exclusion criteria included: (1) the presence of other retinal or optic nerve disorders, (2) a history of vitrectomy, (3) previous photodynamic therapy, and (4) inflammatory or hereditary conditions known to cause MNV.
Anatomical outcomes, represented by CMT, and visual outcomes, assessed by BCVA, were evaluated at two time points: immediately before and one month after the intravitreal injections. The diagnosis of polypoidal choroidal vasculopathy (PCV) was primarily based on findings from indocyanine green angiography (ICGA). In cases where ICGA was unavailable, PCV was identified using optical coherence tomography (OCT) features following previously reported diagnostic consensus guidelines [9].
Outcome measures included:
- BCVA was recorded in decimal format and transformed into a logarithm of the minimum angle of resolution (logMAR) units for statistical analysis.
- CMT (μm) was measured using OCT (DRI OCT Triton, Topcon Inc., Tokyo, Japan) and defined as the distance from the internal limiting membrane to Bruch’s membrane at the fovea. To evaluate the relative changes in the CMT, we used the dCMT, expressed as the rate of change from the baseline values; dCMT was defined as follows:
dCMT (%) = (CMT at baseline − CMT at 1 month)/CMT
at baseline × 100
- We defined complete fluid resolution on OCT as the absence of subretinal fluid (SRF), intraretinal fluid (IRF), and pigment epithelial detachment (PED). We investigated the proportion of patients who achieved complete fluid resolution at one month after the injection.
Statistical analyses were performed to compare baseline characteristics using the Mann–Whitney U test for continuous variables and Fisher’s exact test for categorical variables. The Wilcoxon signed-rank test was applied to assess differences in measurements obtained before and one month after the injections. The Mann–Whitney U test was used to compare the changes in BCVA and CMT between the treatment-naïve groups treated with 2 mg and 8 mg of aflibercept, as well as between the groups with a history of prior intravitreal injections. The sample was characterized using descriptive statistics, including mean and standard deviation (SD). In all analyses, a p < 0.05 was regarded as indicative of statistical significance. All statistical analyses were conducted with JMP Pro 17 software (SAS Institute, Cary, NC, USA).
3. Results
3.1. Patients’ Characteristics
The baseline demographics and clinical information for treatment-naïve patients are presented in Table 1, while data for previously treated patients are shown in Table 2. In the treatment-naïve group, patients who received aflibercept 8 mg had a mean age of 76.31 ± 9.03 years, with 5 women (41.67%) and 7 men (58.33%). In the group that received aflibercept 2 mg, the mean age was 75.29 ± 10.34 years, with 5 women (38.46%) and 7 men (61.54%). In patients with a history of intravitreal injections, those who received aflibercept 8 mg had a mean age of 79.73 ± 7.34 years, with 5 women (38.46%) and 8 men (61.54%), while those treated with aflibercept 2 mg had a mean age of 73.67 ± 8.98 years, comprising 5 women (35.71%) and 9 men (64.29%). Both the treatment-naïve group and the group with a history of prior intravitreal injections showed no statistically significant differences in baseline data between the 8 mg and 2 mg aflibercept treatment arms.

Table 1.
Baseline characteristics of treatment-naïve patients or eyes immediately before injection.

Table 2.
Baseline characteristics of patients or eyes with a history of prior injection immediately before injection.
3.2. Treatment-Naïve Patients
For treatment-naïve patients, the mean baseline BCVA (logMAR) was 0.19 ± 0.23 in the aflibercept 8 mg group, which improved to 0.13 ± 0.20 at one-month post-injection (p = 0.0156). In contrast, the aflibercept 2 mg group showed a mean baseline BCVA of 0.23 ± 0.25, with a post-injection value of 0.20 ± 0.24 at one month, indicating no significant improvement (p = 0.3906) (Figure 1). The improvement in BCVA was 0.053 ± 0.064 in the aflibercept 8 mg group and 0.021 ± 0.089 in the aflibercept 2 mg group, with no statistically significant difference in the degree of improvement between the two groups (p = 0.2808).
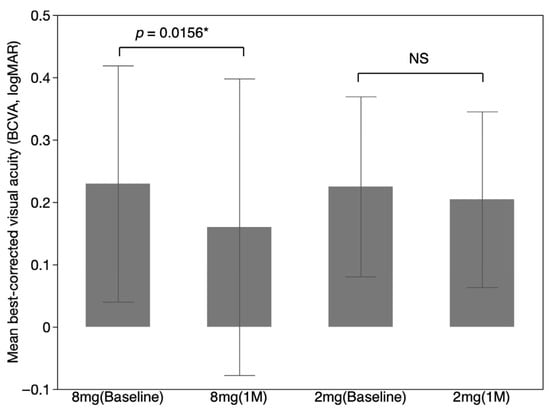
Figure 1.
Mean best-corrected visual acuity (BCVA) before and after administration of aflibercept in treatment-naïve patients. For treatment-naïve patients, the mean baseline BCVA (logMAR) was 0.19 ± 0.23 in the aflibercept 8 mg group, which improved to 0.13 ± 0.20 at one-month post-injections (p = 0.0156). In contrast, the aflibercept 2 mg group showed a mean baseline BCVA of 0.23 ± 0.25, with a post-injection value of 0.20 ± 0.24 at one month, indicating no significant improvement (p = 0.3906). Error bars indicate 95% confidence intervals. 1M, 1 Month. NS, Not Significant. * p < 0.05.
The mean CMT (μm) was 309.15 ± 139.35 in the aflibercept 8 mg group, which improved to 207.62 ± 43.99 at one-month post-injection (p = 0.0002), representing an absolute reduction of 101.53 μm. In contrast, the aflibercept 2 mg group showed a mean baseline CMT of 311.14 ± 61.84, with a post-injection value of 256.57 ± 50.51 at one month (p = 0.0001), corresponding to an absolute reduction of 54.57 μm (Figure 2). The dCMT (%) was slightly greater in the aflibercept 8 mg group (28.60 ± 14.53) compared to the aflibercept 2 mg group (24.08 ± 13.39) (p = 0.0220).
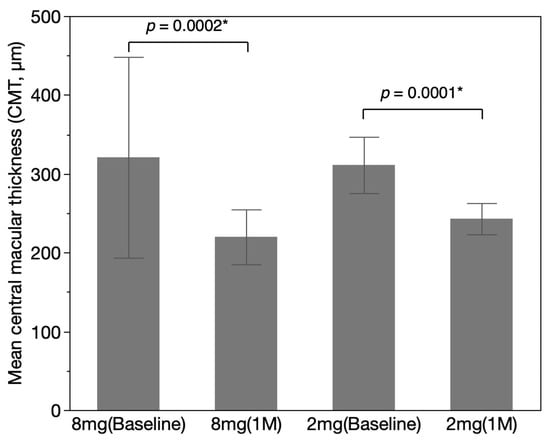
Figure 2.
Mean central macular thickness (CMT) (μm) before and after injections of aflibercept in treatment-naïve patients. The mean CMT was 309.15 ± 139.35 in the aflibercept 8 mg group, which improved to 207.62 ± 43.99 at one-month post-injections (p = 0.0002). In contrast, the aflibercept 2 mg group showed a mean baseline CMT of 311.14 ± 61.84, with a post-injection value of 256.57 ± 50.51 at one month (p = 0.0001). Error bars indicate 95% confidence intervals. * p < 0.05.
A complete fluid resolution was achieved in a total of 9 eyes (69.2%) in the aflibercept 8 mg group and 6 eyes (42.9%) in the aflibercept 2 mg group.
3.3. Patients with a History of Intravitreal Injection
The mean number of prior injections was 12.13 ± 7.14 in the aflibercept 8 mg group and 7.87 ± 4.50 in the aflibercept 2 mg group (p = 0.1506). The mean interval from the last injection was 149.40 ± 146.42 days in the aflibercept 8 mg group and 123.13 ± 104.05 days in the aflibercept 2 mg group (p = 0.4646). The mean baseline BCVA (logMAR) was 0.46 ± 0.57 in the aflibercept 8 mg group, and at one-month post-injection, it was 0.44 ± 0.55 (p = 0.5000). In the aflibercept 2 mg group, the mean baseline BCVA was 0.28 ± 0.32, which changed to 0.26 ± 0.32 one-month post-injection. No statistically significant difference was observed (p = 0.5156). Neither group showed a significant improvement in visual outcomes (Figure 3). The improvement in BCVA was 0.019 ± 0.056 in the aflibercept 8 mg group and 0.015 ± 0.091 in the aflibercept 2 mg group, with no statistically significant difference in the degree of improvement between the two groups (p = 0.8821).
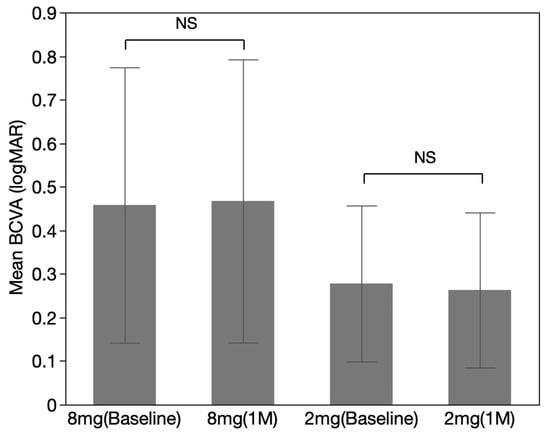
Figure 3.
Mean BCVA before and after administration of aflibercept in patients with a history of intravitreal injection. The mean baseline BCVA (logMAR) was 0.46 ± 0.57 in the aflibercept 8 mg group, and at one-month post-injections, it was 0.44 ± 0.55 (p = 0.5000). In the aflibercept 2 mg group, the mean baseline BCVA was 0.28 ± 0.32, which changed to 0.26 ± 0.32 one month post-injection. Error bars indicate 95% confidence intervals.
The mean CMT was 290.40 ± 82.55 in the aflibercept 8 mg group, which improved to 213.00 ± 38.79 at one-month post-injection (p < 0.0001), representing an absolute reduction of 77.40 μm. The aflibercept 2 mg group showed a mean baseline CMT of 331.07 ± 112.53, with a post-injection value of 248.13 ± 69.35 at one month (p = 0.0015), corresponding to an absolute reduction of 82.94 μm (Figure 4). The dCMT was 24.08 ± 13.39 in the aflibercept 8 mg group and 20.61 ± 20.72 in the aflibercept 2 mg group, with no statistically significant difference in the degree of improvement between the two groups (p = 0.5907).
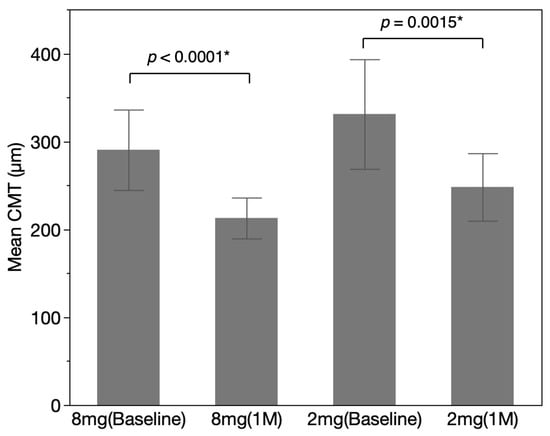
Figure 4.
Mean CMT before and after intravitreal injections of aflibercept in patients with a history of intravitreal injections. The mean CMT was 290.40 ± 82.55 in the aflibercept 8 mg group, which improved to 213.00 ± 38.79 at one-month post-injections (p < 0.0001). The aflibercept 2 mg group showed a mean baseline CMT of 331.07 ± 112.53, with a post-injection value of 248.13 ± 69.35 at one month (p = 0.0015). Error bars indicate 95% confidence intervals. * p < 0.05.
A complete fluid resolution was achieved in a total of 11 eyes (78.6%) in the aflibercept 8 mg group and 9 eyes (60.0%) in the aflibercept 2 mg group.
3.4. Safety
There were no observed instances of endophthalmitis, occlusive vasculitis, intraocular inflammation (IOI), or other ocular complications, and no systemic adverse events were reported post-injection.
4. Discussion
In this study, we retrospectively assessed the efficacy and safety of aflibercept 8 mg in both treatment-naïve patients and those with a prior history of intravitreal injections, comparing it to the standard aflibercept 2 mg dosage. Our findings suggest that, in treatment-naïve patients, aflibercept 8 mg demonstrated marginally greater efficacy in improving anatomic outcomes compared to aflibercept 2 mg; however, this difference did not extend to visual outcomes. In contrast, no significant difference in efficacy was observed between the two dosages in patients with a history of intravitreal injections. To our knowledge, this is the first head-to-head comparison of aflibercept 8 mg versus 2 mg in both treatment-naïve and previously treated nAMD patients in a real-world clinical setting.
Aflibercept 8 mg is a newly developed formulation characterized by improved molecular stability and a higher concentration than the conventional 2 mg dose, designed to reduce treatment burden. This results in a significantly increased molar amount delivered intravitreally, which, combined with aflibercept’s high binding affinity for VEGF and its estimated intraocular half-life, is expected to extend VEGF suppression [6,10]. Consequently, treatment intervals may be extended to as long as 16 weeks after the initial monthly loading phase, offering a promising strategy for decreasing the frequency of intravitreal injections [8]. In this study, we examined whether there is a difference in early treatment response between conventional aflibercept 2 mg and the high-dose aflibercept 8 mg formulation, using real-world clinical data. Aflibercept 8 mg showed slightly greater anatomical efficacy than 2 mg in treatment-naïve patients, with no corresponding difference in visual outcomes; no significant differences were observed in previously treated patients. In treatment-naïve patients, starting therapy with aflibercept 8 mg may provide superior therapeutic efficacy compared to the traditional 2 mg formulation. Beginning treatment with aflibercept 8 mg from the outset may enable patients to perceive greater therapeutic benefit after the first injection. This early anatomical improvement may help boost patient motivation and reduce the likelihood of treatment discontinuation, which is a common concern in real-world settings due to the burden and expense of intravitreal injections. Additionally, as demonstrated in large-scale clinical trials and supported by case series, aflibercept 8 mg has the potential to offer the added benefit of extended dosing intervals over the long term [7,8,11]. In previously treated patients, the absence of significant improvement in BCVA despite a notable reduction in CMT may be due to the inclusion of individuals who were poor responders to prior anti-VEGF therapy. This finding is consistent with previous reports indicating that switching to aflibercept 8 mg in treatment-refractory nAMD eyes in a real-world setting was associated with modest anatomical improvements, but no significant changes in BCVA [12]. These patients may have sustained irreversible damage to the photoreceptor layer or other retinal structures, limiting functional recovery even when anatomical improvements are seen.
The incidence of intraocular inflammation (IOI) following intravitreal anti-VEGF therapy is usually less than 1%, with variations in frequency and clinical presentation depending on the specific agent used [13]. In large clinical trials, no significant differences in safety profiles were seen between the 2 mg and 8 mg doses. The occurrence of iritis and vitritis was under 1% in both groups, and there were no reports of retinal vasculitis [8]. However, several case reports have documented instances of retinal vasculitis after treatment with aflibercept 8 mg [14,15,16]. Since previous intravitreal anti-VEGF exposure has not been linked to an increased risk of IOI, inflammation may occur following the very first injection [17]. Although no IOI cases were observed in this study, clinicians should stay alert when using aflibercept 8 mg, considering the potential risk noted in earlier case reports.
Several limitations should be acknowledged in this study. First, the retrospective design and small sample size necessitate validation through larger, prospective investigations. Second, the follow-up period was limited to one month after injection, restricting the assessment of long-term outcomes. Third, as all participants were Japanese patients with nAMD, the generalizability of the findings to other Asian populations or to Caucasian patients may be limited. Additionally, in the treatment-naïve cohort, the decision to initiate aflibercept 8 mg or 2 mg was entirely at the discretion of the treating physician, potentially introducing selection bias. Finally, the switch group included eyes previously treated with aflibercept 2 mg, brolucizumab, or faricimab, leading to heterogeneity in prior treatment history. This variability may have influenced treatment responses and limited comparability within the switch group. Future studies should consider restricting inclusion to patients with a uniform treatment background to minimize potential confounding factors.
5. Conclusions
Real-world evidence demonstrated that aflibercept 8 mg resulted in significant anatomical improvements one month after injection, irrespective of prior intravitreal treatment history. However, notable gains in visual acuity were observed exclusively in treatment-naïve patients. The inclusion of real-world data is critical for gaining a comprehensive and practical understanding of treatment effectiveness in routine clinical practice, where outcomes may differ from those reported in controlled clinical trials. To further clarify the durability and generalizability of these findings, larger prospective studies with extended follow-up periods are needed.
Author Contributions
Conceptualization, T.M. and S.U.; methodology, T.M.; validation, T.M. and S.U.; formal analysis, T.M. and S.U.; investigation, T.M., S.U. and S.M.; data curation, T.M. and S.U.; writing—original draft preparation, T.M. and S.U.; writing—review and editing, S.M.; visualization, T.M.; supervision, Y.S.; project administration, Y.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This study was approved by the Ethics Committee of Fuchu Hospital (ID number: 2024009, Date 9 July 2024) and adhered to the tenets of the Declaration of Helsinki.
Informed Consent Statement
Due to the retrospective approach of this analysis, no informed consent was obtained from the patients. Informed consent is not required as per local legislation, “Ethical Guidelines for Life Science and Medical Research Involving Human Subjects”. The Fuchu Hospital’s website allowed participants to withdraw from the study.
Data Availability Statement
The data used and analyzed for this study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AMD | age-related macular degeneration |
| BCVA | best-corrected visual acuity |
| CMT | central macular thickness |
| DME | diabetic macular edema |
| ICGA | indocyanine green angiography |
| IOI | intraocular inflammation |
| IRF | intraretinal fluid |
| logMAR | logarithm of the minimum angle of resolution |
| MNV | macular neovascularization |
| NA | not applicable |
| NS | not significant |
| nAMD | neovascular age-related macular degeneration |
| OCT | optical coherence tomography |
| PCV | polypoidal choroidal vasculopathy |
| PED | pigment epithelial detachment |
| SD | standard deviation |
| SRF | subretinal fluid |
| VEGF | vascular endothelial growth factor |
References
- Berkowitz, S.T.; Sternberg, P., Jr.; Feng, X.; Chen, Q.; Patel, S. Analysis of Anti-Vascular Endothelial Growth Factor Injection Claims Data in US Medicare Part B Beneficiaries From 2012 to 2015. JAMA Ophthalmol. 2019, 137, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Glasser, D.B.; Parikh, R.; Lum, F.; Williams, G.A. Intravitreal Anti-Vascular Endothelial Growth Factor Cost Savings Achievable with Increased Bevacizumab Reimbursement and Use. Ophthalmology 2020, 127, 1688–1692. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.G.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Heier, J.S.; Brown, D.M.; Chong, V.; Korobelnik, J.-F.; Kaiser, P.K.; Nguyen, Q.D.; Kirchhof, B.; Ho, A.; Ogura, Y.; Yancopoulos, G.D.; et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 2012, 119, 2537–2548. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, P.J.; Brown, D.M.; Heier, J.S.; Boyer, D.S.; Kaiser, P.; Chung, C.Y.; Kim, R.Y. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 2006, 5, 1419–1431. [Google Scholar] [CrossRef] [PubMed]
- Korobelnik, J.F.; Lanzetta, P.; Wykoff, C.C.; Wong, T.Y.; Zhang, X.; Morgan-Warren, P.; Fitzpatrick, S.; Leal, S.; Brunck, L.; Hasanbasic, Z.; et al. Sustained disease control with aflibercept 8 mg: A new benchmark in the management of retinal neovascular diseases. Eye 2024, 38, 3218–3221. [Google Scholar] [CrossRef] [PubMed]
- Wykoff, C.C.; Brown, D.M.; Reed, K.; Berliner, A.J.; Gerstenblith, A.T.; Breazna, A.; Abraham, P.; Fein, J.G.; Chu, K.W.; Clark, W.L.; et al. Effect of High-Dose Intravitreal Aflibercept, 8 mg, in Patients with Neovascular Age-Related Macular Degeneration: The Phase 2 CANDELA Randomized Clinical Trial. JAMA Ophthalmol. 2023, 1, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Lanzetta, P.; Korobelnik, J.F.; Heier, J.S.; Leal, S.; Holz, F.G.; Clark, W.L.; Eichenbaum, D.; Iida, T.; Sun, X.; Berliner, A.J.; et al. Intravitreal aflibercept 8 mg in neovascular age-related macular degeneration (PULSAR): 48-week results from a randomised, double-masked, non-inferiority, phase 3 trial. Lancet 2024, 23, 1141–1152. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.M.G.; Lai, T.Y.Y.; Teo, K.; Ruamviboonsuk, P.; Chen, S.-J.; Kim, J.E.; Gomi, F.; Koh, A.H.; Kokame, G.; Jordan-Yu, J.M.; et al. Polypoidal Choroidal Vasculopathy: Consensus Nomenclature and Non-Indocyanine Green Angiograph Diagnostic Criteria from the Asia-Pacific Ocular Imaging Society PCV Workgroup. Ophthalmology 2021, 128, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Veritti, D.; Sarao, V.; Di Bin, F.; Lanzetta, P. Pharmacokinetic and Pharmacodynamic Rationale for Extending VEGF Inhibition Increasing Intravitreal Aflibercept Dose. Pharmaceutics 2023, 15, 1416. [Google Scholar] [CrossRef] [PubMed]
- Sambhara, D.; Vakharia, P.; Eichenbaum, D.A. Real-world efficacy and safety of 8 mg aflibercept in neovascular AMD: A case series. BMJ Open Ophthalmol. 2025, 10, e002091. [Google Scholar] [CrossRef] [PubMed]
- Musadiq, M.; Latif, F.; Ng, B.; Azzopardi, M.; Gilead, N.; Needham, A.; Chong, Y.J. Early Real-World Outcomes of Switching to 8 mg Aflibercept for Neovascular Age-Related Macular Degeneration in the United Kingdom. Life 2025, 15, 903. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.D.; Chong, D.; Fuller, T.; Callanan, D. NONINFECTIOUS VITRITIS AFTER INTRAVITREAL INJECTION OF: ANTI-VEGF AGENTS: Variations in Rates and Presentation by Medication. Retina 2016, 36, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, L.; Michels, S.; Eandi, C.; Karam, M.A.; Figueiredo, E.C.O.; Hatz, K. Aflibercept high-dose (8mg) related intraocular inflammation (IOI)—A case series. BMC Ophthalmol. 2024, 2, 520. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Hoshino, J.; Numaga, S.; Mimura, K.; Asatori, Y.; Akiyama, H. Retinal vasculitis after intravitreal aflibercept 8 mg for neovascular age-related macular degeneration. Jpn J. Ophthalmol. 2024, 68, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Sisk, R.A. Occlusive Retinal Vasculitis After Aflibercept 8mg Injection for Wet Macular Degeneration. Retin. Cases Brief Rep. 2025, Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Chong, D.Y.; Anand, R.; Williams, P.D.; Qureshi, J.A.; Callanan, D.G. Characterization of sterile intraocular inflammatory responses after intravitreal bevacizumab injection. Retina 2010, 30, 1432–1440. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).