Changes in SpO2 on Room Air for 34 Severe COVID-19 Patients after Ivermectin-Based Combination Treatment: 62% Normalization within 24 Hours
Abstract
:1. Introduction
IVM for COVID-19 Treatment
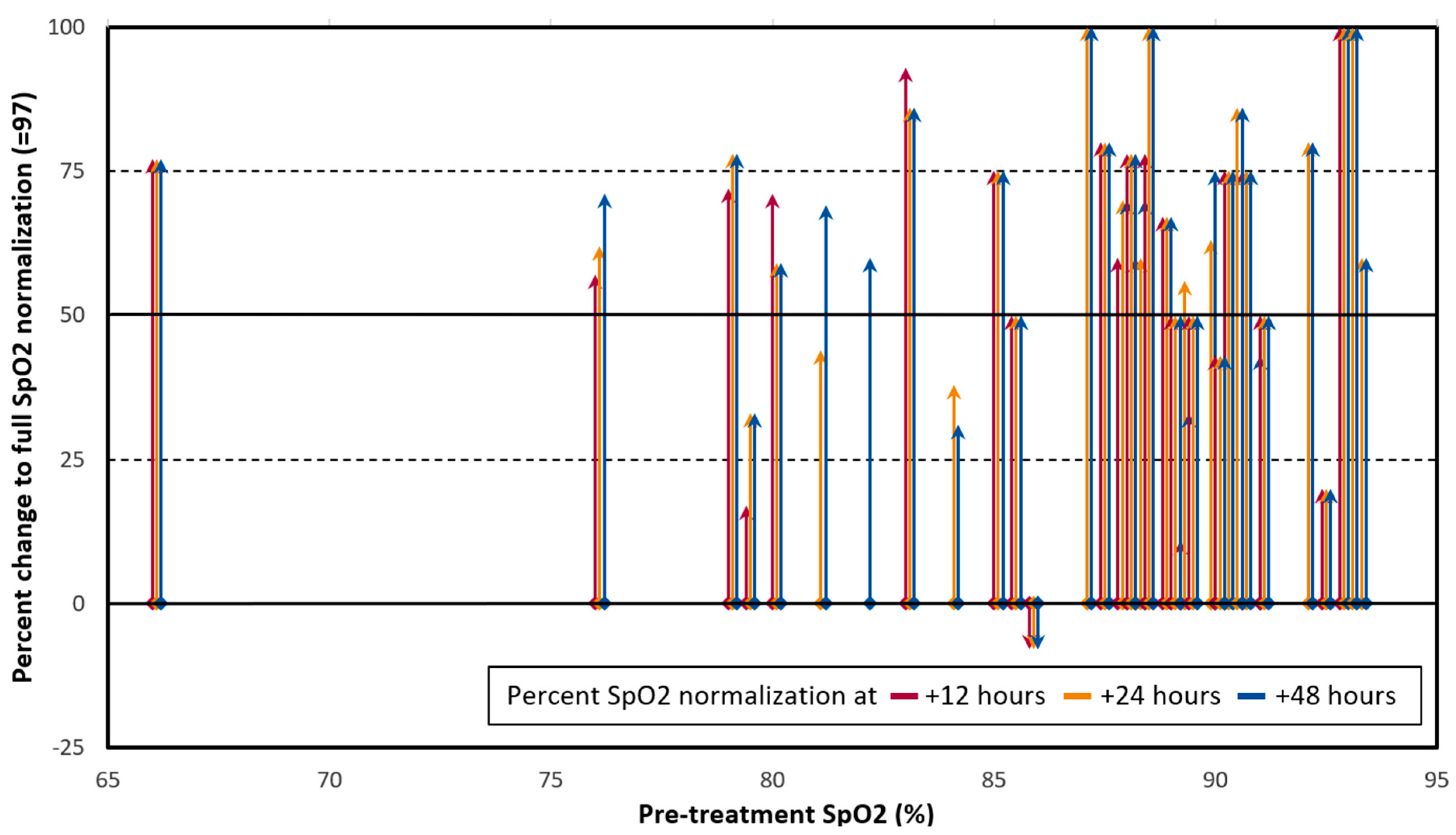
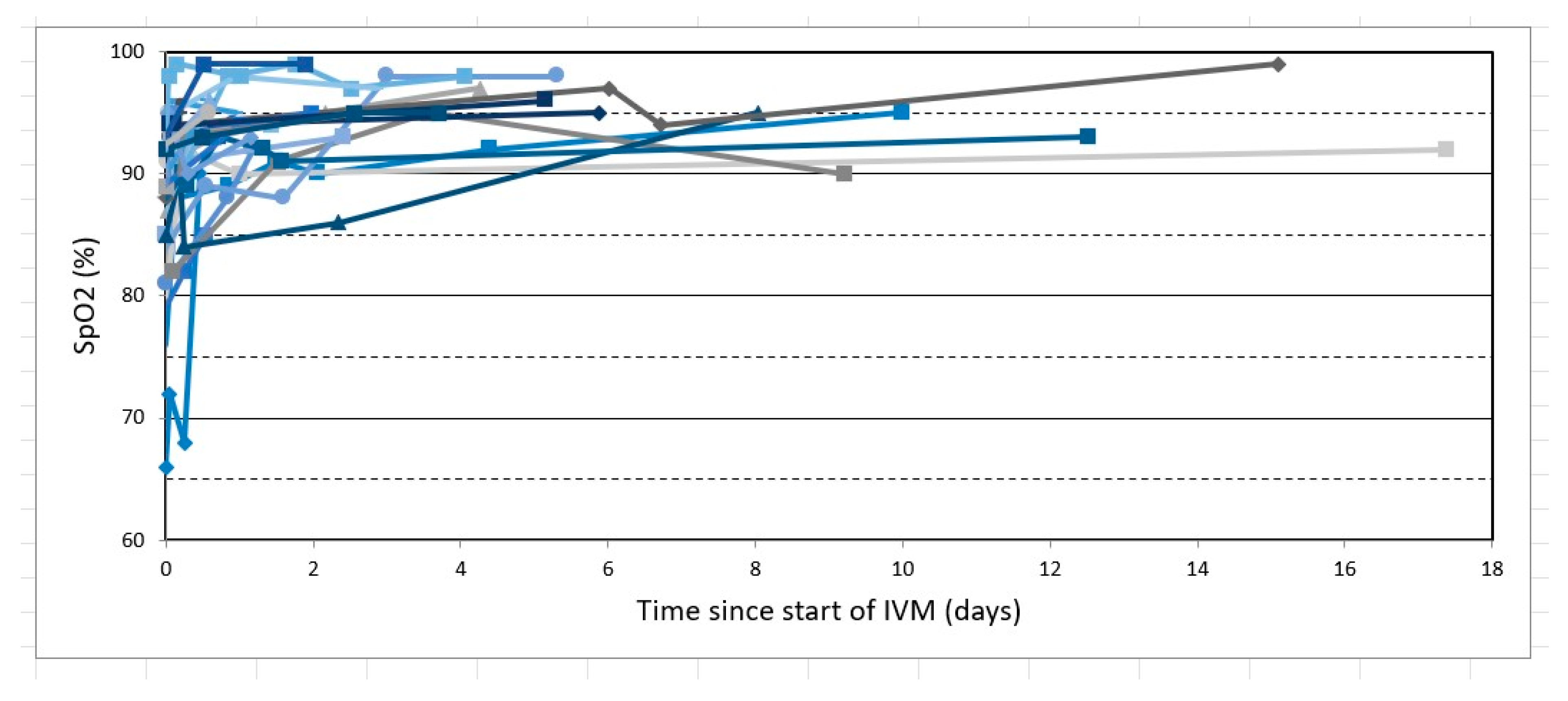
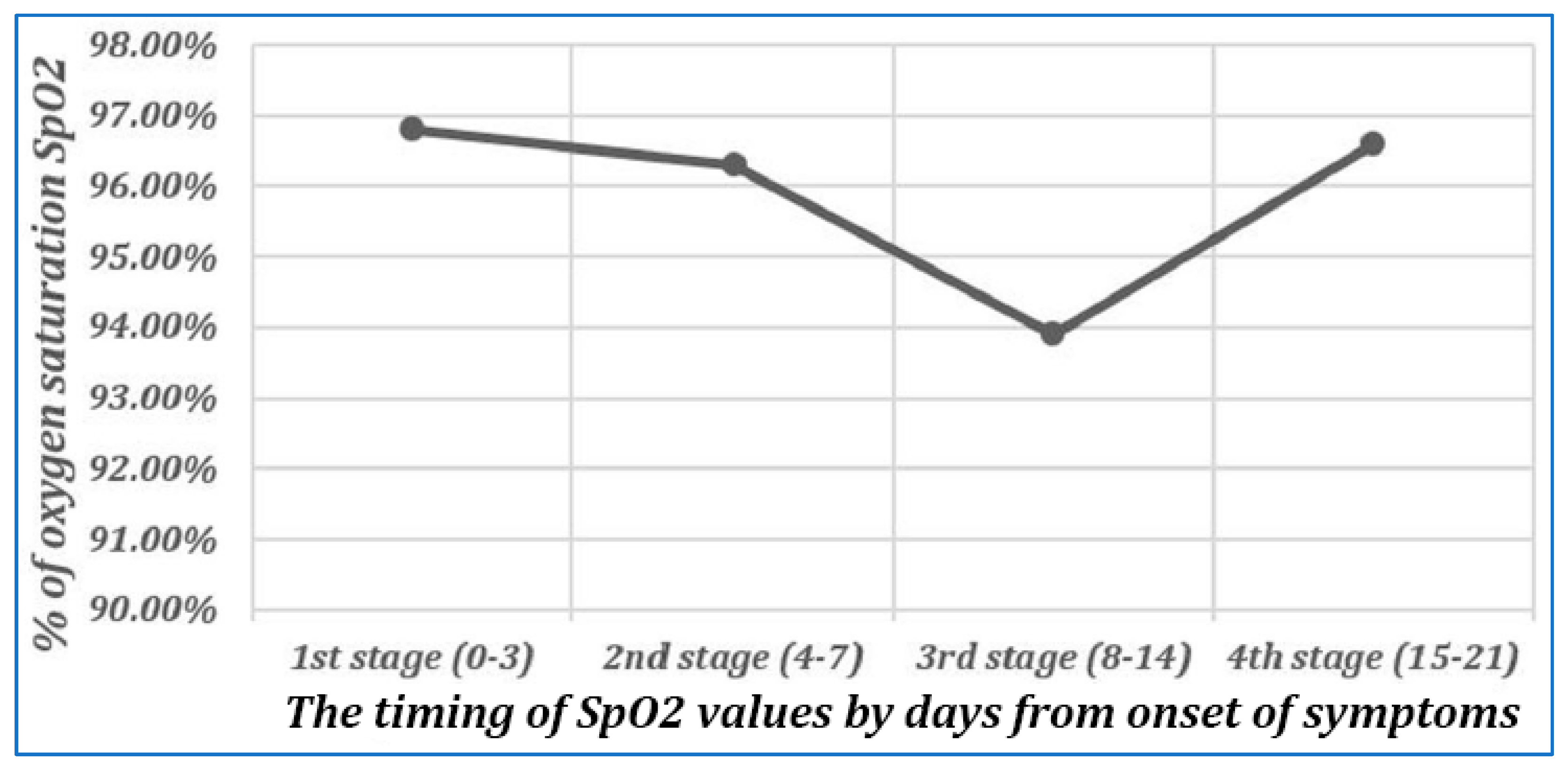
2. Results
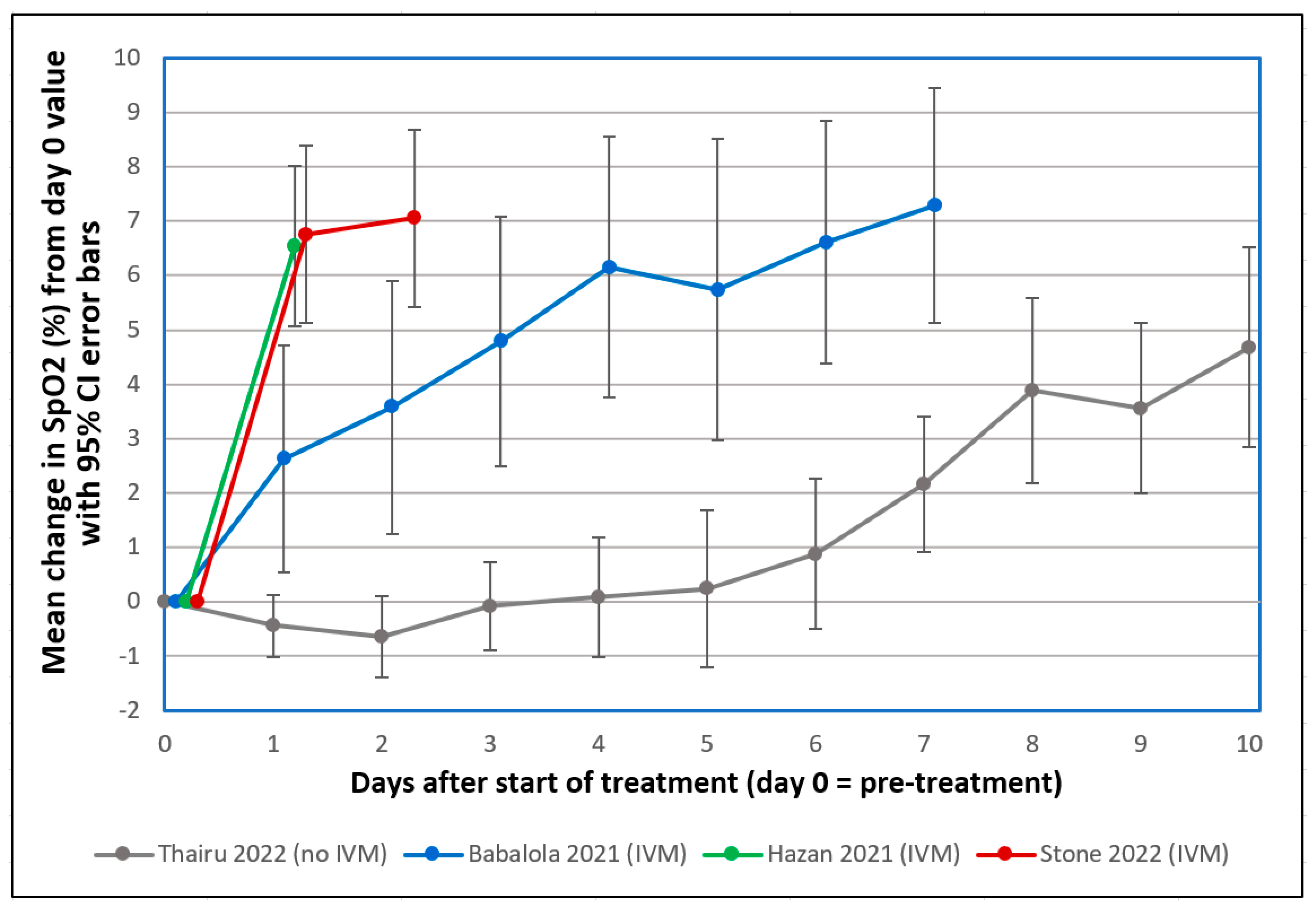
3. Discussion
4. Materials and Methods
4.1. Outcomes
4.2. Participants
4.3. Inclusion and Exclusion Criteria
4.4. Treatment
4.5. Data Collection
4.6. Analytical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AE | Adverse effect |
| CPCPZ | Primary Care Physicians of Zimbabwe |
| COVID-19 | coronavirus disease 2019 |
| IVM | ivermectin |
| RCT | randomized clinical trial |
References
- Worldometer Coronovirus Statistics. Available online: https://www.worldometers.info/coronavirus/#countries (accessed on 22 September 2021).
- Yagisawa, M.; Foster, P.J.; Hanaki, H.; Omura, S. Global trends in clinical studies of ivermectin in COVID-19. Jpn. J. Antibiot. 2021, 74, 44–95. [Google Scholar]
- Kory, P.; Meduri, G.U.; Varon, J.; Iglesias, J.; Marik, P.E. Review of the emerging evidence demonstrating the efficacy of ivermectin in the prophylaxis and treatment of COVID-19. Am. J. Ther. 2021, 28, e299–e318. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Garratt, A.; Levi, J.; Falconer, J.; Ellis, L.; McCann, K.; Pilkington, V.; Qavi, A.; Wang, J.; Wentzel, H. Meta-analysis of randomized trials of ivermectin to treat SARS-CoV-2 infection. Open Forum Infect. Dis. 2021, 8, ofab358. [Google Scholar] [CrossRef] [PubMed]
- Santin, A.D.; Scheim, D.E.; McCullough, P.A.; Yagisawa, M.; Borody, T.J. Ivermectin: A multifaceted drug of Nobel prize-honored distinction with indicated efficacy against a new global scourge, COVID-19. N. Microbes N. Infect. 2021, 43, 100924. [Google Scholar] [CrossRef]
- López-Medina, E.; López, P.; Hurtado, I.C.; Dávalos, D.M.; Ramirez, O.; Martínez, E.; Díazgranados, J.A.; Oñate, J.M.; Chavarriaga, H.; Herrera, S.; et al. Effect of ivermectin on time to resolution of symptoms among adults with mild COVID-19: A randomized clinical trial. JAMA 2021, 325, 1426–1435. [Google Scholar] [CrossRef]
- Lim, S.C.L.; Hor, C.P.; Tay, K.H.; Mat Jelani, A.; Tan, W.H.; Ker, H.B.; Chow, T.S.; Zaid, M.; Cheah, W.K.; Lim, H.H.; et al. Efficacy of ivermectin treatment on disease progression among adults with mild to moderate COVID-19 and comorbidities: The I-TECH randomized clinical trial. JAMA Intern. Med. 2022, 182, 426–435. [Google Scholar] [CrossRef]
- Reis, G.; Silva, E.A.S.M.; Silva, D.C.M.; Thabane, L.; Milagres, A.C.; Ferreira, T.S.; dos Santos, C.V.Q.; Campos, V.H.S.; Nogueira, A.M.R.; de Almeida, A.P.F.G.; et al. Effect of early treatment with ivermectin among patients with COVID-19. N. Engl. J. Med. 2022, 386, 1721–1731. [Google Scholar] [CrossRef]
- U.S. National Institutes of Health. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines: Therapeutic Management of Patients with COVID-19; U.S. National Institutes of Health: Bethesda, MD, USA, 2021. Available online: https://www.covid19treatmentguidelines.nih.gov/ (accessed on 12 May 2022).
- World Health Organization. WHO Recommends against the Use of Remdesivir in COVID-19 Patients; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/news-room/feature-stories/detail/who-recommends-against-the-use-of-remdesivir-in-covid-19-patients (accessed on 12 May 2022).
- Aminpour, M.; Cannariato, M.; Safaeeardebili, M.E.; Preto, J.; Moracchiato, A.; Doria, D.; Donato, F.; Zizzi, E.A.; Deriu, M.A.; Scheim, D.E.; et al. In silico analysis of the multi-targeted mode of action of ivermectin and related compounds. Computation 2022, 10, 51. [Google Scholar] [CrossRef]
- Scheim, D.E. A deadly embrace: Hemagglutination mediated by SARS-CoV-2 spike protein at its 22 N-glycosylation sites, red blood cell surface sialoglycoproteins, and antibody. Int. J. Mol. Sci. 2022, 23, 2558. [Google Scholar] [CrossRef]
- Chirisa, S. Zimbabwe Confirms Its First Case of Coronavirus, 20 March 2020. Available online: https://iharare.com/zimbabwe-confirms-first-case-of-coronavirus-2/ (accessed on 12 May 2022).
- WHO. Coronavirus Disease 2019 (COVID-19), Situation Report—72; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200401-sitrep-72-covid-19.pdf?sfvrsn=3dd8971b_2 (accessed on 12 May 2022).
- Rajter, J.C.; Sherman, M.S.; Fatteh, N.; Vogel, F.; Sacks, J.; Rajter, J.-J. Use of ivermectin is associated with lower mortality in hospitalized patients with COVID-19 (ICON study). Chest 2020, 159, 85–92. [Google Scholar] [CrossRef]
- Campbell, W.C. History of avermectin and ivermectin, with notes on the history of other macrocyclic lactone antiparasitic agents. Curr. Pharm. Biotechnol. 2012, 13, 853–865. [Google Scholar] [CrossRef] [PubMed]
- Crump, A.; Ōmura, S. Ivermectin, ‘wonder drug’ from Japan: The human use perspective. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2011, 87, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Guzzo, C.A.; Furtek, C.I.; Porras, A.G.; Chen, C.; Tipping, R.; Clineschmidt, C.M.; Sciberras, D.G.; Hsieh, J.Y.; Lasseter, K.C. Safety, tolerability, and pharmacokinetics of escalating high doses of ivermectin in healthy adult subjects. J. Clin. Pharmacol. 2002, 42, 1122–1133. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Camprubí, D.; Requena-Méndez, A.; Buonfrate, D.; Giorli, G.; Kamgno, J.; Gardon, J.; Boussinesq, M.; Muñoz, J.; Krolewiecki, A. Safety of high-dose ivermectin: A systematic review and meta-analysis. J. Antimicrob. Chemother. 2020, 75, 827–834. [Google Scholar] [CrossRef]
- Krolewiecki, A.; Lifschitz, A.; Moragas, M.; Travacio, M.; Valentini, R.; Alonso, D.F.; Solari, R.; Tinelli, M.A.; Cimino, R.O.; Álvarez, L.; et al. Antiviral effect of high-dose ivermectin in adults with COVID-19: A proof-of-concept randomized trial. EClinicalMedicine 2021, 37, 100959. [Google Scholar] [CrossRef]
- de Castro, C.G., Jr.; Gregianin, L.J.; Burger, J.A. Continuous high-dose ivermectin appears to be safe in patients with acute myelogenous leukemia and could inform clinical repurposing for COVID-19 infection. Leuk Lymphoma 2020, 61, 2536–2537. [Google Scholar] [CrossRef]
- Chung, K.; Yang, C.C.; Wu, M.L.; Deng, J.F.; Tsai, W.J. Agricultural avermectins: An uncommon but potentially fatal cause of pesticide poisoning. Ann. Emerg. Med. 1999, 34, 51–57. [Google Scholar] [CrossRef]
- Scheim, D.E.; Hibberd, J.A.; Chamie-Quintero, J.J. Protocol Violations in López-Medina et al.: 38 Switched Ivermectin (IVM) and Placebo Doses, Failure of Blinding, Ubiquitous IVM Use OTC in Cali, and Nearly Identical AEs for the IVM and Control Groups. Available online: https://doi.org/10.31219/osf.io/u7ewz (accessed on 13 February 2022). [CrossRef]
- Scheim, D.E. The Drug Used in Lim et al. 2022, Source Not Specified, Had <1% Incidence of AEs Distinctive and Common for Ivermectin at This Study’s Very High Dose, 2 mg/kg. Available online: https://doi.org/10.31219/osf.io/5cwmr (accessed on 12 May 2022). [CrossRef]
- de Melo, G.D.; Lazarini, F.; Levallois, S.; Hautefort, C.; Michel, V.; Larrous, F.; Verillaud, B.; Aparicio, C.; Wagner, S.; Gheusi, G.; et al. COVID-19-related anosmia is associated with viral persistence and inflammation in human olfactory epithelium and brain infection in hamsters. Sci. Transl. Med. 2021, 13, eabf8396. [Google Scholar]
- Arévalo, A.P.; Pagotto, R.; Pórfido, J.L.; Daghero, H.; Segovia, M.; Yamasaki, K.; Varela, B.; Hill, M.; Verdes, J.M.; Duhalde Vega, M.; et al. Ivermectin reduces in vivo coronavirus infection in a mouse experimental model. Sci. Rep. 2021, 11, 7132. [Google Scholar] [CrossRef]
- Mahmud, R.; Rahman, M.M.; Alam, I.; Ahmed, K.G.U.; Kabir, A.K.M.H.; Sayeed, S.K.J.B.; Rassel, M.A.; Monayem, F.B.; Islam, M.S.; Islam, M.M.; et al. Ivermectin in combination with doxycycline for treating COVID-19 symptoms: A randomized trial. J. Int. Med. Res. 2021, 49, 03000605211013550. [Google Scholar] [CrossRef]
- Hashim, H.A.; Maulood, M.F.; Rasheed, A.M.; Fatak, D.F.; Kabah, K.K.; Abdulamir, A.S. Controlled randomized clinical trial on using Ivermectin with Doxycycline for treating COVID-19 patients in Baghdad, Iraq. medRxiv 2020. [Google Scholar] [CrossRef]
- George, L.L.; Borody, T.J.; Andrews, P.; Devine, M.; Moore-Jones, D.; Walton, M.; Brandl, S. Cure of duodenal ulcer after eradication of Helicobacter pylori. Med. J. Aust. 1990, 153, 145–149. [Google Scholar] [CrossRef] [PubMed]
- U.S. Centers for Disease Control and Prevention (CDC). Coronavirus Disease 2019 (COVID-19). The Basics of Oxygen Monitoring and Oxygen Therapy during the COVID-19 Pandemic. Available online: https://www.cdc.gov/coronavirus/2019-ncov/videos/oxygen-therapy/Basics_of_Oxygen_Monitoring_and_Oxygen_Therapy_Transcript.pdf (accessed on 23 September 2021).
- Osman, A.M.; Farouk, S.; Osman, N.M.; Abdrabou, A.M. Longitudinal assessment of chest computerized tomography and oxygen saturation for patients with COVID-19. Egypt. J. Radiol. Nucl. Med. 2020, 51, 255. [Google Scholar] [CrossRef]
- Metwally, M.I.; Basha, M.A.A.; Zaitoun, M.M.A.; Abdalla, H.M.; Nofal, H.A.E.; Hendawy, H.; Manajrah, E.; Hijazy, R.f.; Akbazli, L.; Negida, A.; et al. Clinical and radiological imaging as prognostic predictors in COVID-19 patients. Egypt. J. Radiol. Nucl. Med. 2021, 52, 100. [Google Scholar] [CrossRef]
- Aoki, R.; Iwasawa, T.; Hagiwara, E.; Komatsu, S.; Utsunomiya, D.; Ogura, T. Pulmonary vascular enlargement and lesion extent on computed tomography are correlated with COVID-19 disease severity. Jpn. J. Radiol. 2021, 39, 451–458. [Google Scholar] [CrossRef]
- Ding, X.; Xu, J.; Zhou, J.; Long, Q. Chest CT findings of COVID-19 pneumonia by duration of symptoms. Eur. J. Radiol. 2020, 127, 109009. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, C.; Hu, Y.; Li, C.; Ren, Q.; Zhang, X.; Shi, H.; Zhou, M. Temporal changes of CT findings in 90 patients with COVID-19 pneumonia: A longitudinal study. Radiology 2020, 296, E55–E64. [Google Scholar] [CrossRef]
- Quispe-Cholan, A.; Anticona-De-La-Cruz, Y.; Cornejo-Cruz, M.; Quispe-Chirinos, O.; Moreno-Lazaro, V.; Chavez-Cruzado, E. Tomographic findings in patients with COVID-19 according to evolution of the disease. Egypt. J. Radiol. Nucl. Med. 2020, 51, 215. [Google Scholar] [CrossRef]
- Annunziata, A.; Coppola, A.; Carannante, N.; Simioli, F.; Lanza, M.; Di Micco, P.; Fiorentino, G. Home management of patients with moderate or severe respiratory failure secondary to COVID-19, using remote monitoring and oxygen with or without HFNC. Pathogens 2021, 10, 413. [Google Scholar] [CrossRef]
- Thairu, Y.; Babalola, O.E.; Ajayi, A.A.; Ndanusa, Y.; Ogedengbe, J.O.; Omede, O. A comparison of Ivermectin and non Ivermectin based regimen for COVID-19 in Abuja: Effects on virus clearance, days-to-discharge and mortality. Res. Sq. 2022. [Google Scholar] [CrossRef]
- Babalola, O.E.; Ndanusa, Y.; Adesuyi, A.; Ogedengbe, O.J.; Thairu, Y.; Ogu, O. A randomized controlled trial of ivermectin monotherapy versus HCQ, IVM, and AZ combination therapy in COVID-19 patients in Nigeria. J. Infect. Dis. Epidemiol. 2021, 7, 233. [Google Scholar] [CrossRef]
- Ayerbe, L.; Risco, C.; Ayis, S. The association between treatment with heparin and survival in patients with Covid-19. J. Thromb. Thrombolysis 2020, 50, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Negri, E.M.; Piloto, B.M.; Morinaga, L.K.; Jardim, C.V.P.; Lamy, S.A.E.-D.; Ferreira, M.A.; D’Amico, E.A.; Deheinzelin, D. Heparin therapy improving hypoxia in COVID-19 patients—A case series. Front. Physiol. 2020, 11, 1341. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Huang, M.; Li, D.; Tang, N. Difference of coagulation features between severe pneumonia induced by SARS-CoV2 and non-SARS-CoV2. J. Thromb. Thrombolysis 2020, 51, 1107–1110. [Google Scholar] [CrossRef] [PubMed]
- Dayer, M. Coronavirus (2019-nCoV) deactivation via spike glycoprotein shielding by old drugs, bioinformatic study. Preprints.org 2020. [Google Scholar] [CrossRef]
- Babalola, O.E.; (Bingham University, New Karu, Nigeria). Personal communication, 2022. This communication provided deidentified patient level data for SpO2 changes post-treatment from two studies which he coauthored (per references [38] and [39] as cited above), one using standard treatment excluding IVM (#38) and the other including IVM in the treatment regimen (#39), for those values obtained exclusively on room air and with pre-treatment SpO2 values ≤ 93%.
- Hazan, S.; Dave, S.; Gunaratne, A.W.; Dolai, S.; Clancy, R.L.; McCullough, P.A.; Borody, T.J. Effectiveness of ivermectin-based multidrug therapy in severely hypoxic, ambulatory COVID-19 patients. Future Microbiol. 2022, 17, 339–350. [Google Scholar] [CrossRef]
- Rajter, J.J.; (Broward Health Medical Center, Fort Lauderdale, FL, USA). Personal communication, 2020.
- Stone, J.C.; Ndarukwa, P.; Scheim, D.E.; Dancis, B.M.; Dancis, J.; Gill, M.G.; Aldous, C. Rapid increase of SpO2 on room air for 34 severe COVID-19 patients after ivermectin-based combination treatment: 55–62% normalization within 12–24 hours. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Zaidi, A.K.; Dehgani-Mobaraki, P. The mechanisms of action of ivermectin against SARS-CoV-2—An extensive review. J. Antibiot. 2022, 75, 60–71. [Google Scholar] [CrossRef]
- Wang, H.; Yu, M.; Ochani, M.; Amella, C.A.; Tanovic, M.; Susarla, S.; Li, J.H.; Wang, H.; Yang, H.; Ulloa, L.; et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature 2003, 421, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Krause, R.M.; Buisson, B.; Bertrand, S.; Corringer, P.J.; Galzi, J.L.; Changeux, J.P.; Bertrand, D. Ivermectin: A positive allosteric effector of the alpha7 neuronal nicotinic acetylcholine receptor. Mol. Pharmacol. 1998, 53, 283–294. [Google Scholar] [CrossRef] [Green Version]
- Ren, C.; Tong, Y.L.; Li, J.C.; Lu, Z.Q.; Yao, Y.M. The protective effect of alpha 7 nicotinic acetylcholine receptor activation on critical illness and its mechanism. Int. J. Biol. Sci. 2017, 13, 46–56. [Google Scholar] [CrossRef] [Green Version]
- U.S. National Institutes of Health (NIH). Clinical Spectrum of SARS-CoV-2 Infection. Available online: https://www.covid19treatmentguidelines.nih.gov/overview/clinical-spectrum/ (accessed on 2 March 2022).
- The Nobel Assembly at Karolinska Institutet. The 2015 Nobel Prize in Physiology or Medicine—Press Release; The Nobel Assembly at Karolinska Institutet: Solna, Sweden, 2015; Available online: https://www.nobelprize.org/prizes/medicine/2015/press-release/ (accessed on 12 May 2022).


| Age | Initial SpO2 (%) | ||||
|---|---|---|---|---|---|
| Group * | Sex | 66–84 | 85–89 | 90–93 | Total |
| 25–49 | |||||
| Female | 2 | 2 | 1 | 5 | |
| Male | 2 | 2 | 2 | 6 | |
| Total | 4 | 4 | 3 | 11 | |
| 50–59 | |||||
| Female | 2 | 2 | 1 | 5 | |
| Male | 2 | 2 | 2 | 6 | |
| Total | 4 | 4 | 3 | 11 | |
| 60–69 | |||||
| Female | 0 | 1 | 1 | 2 | |
| Male | 0 | 4 | 2 | 6 | |
| Total | 0 | 5 | 3 | 8 | |
| 70–79 | |||||
| Female | 0 | 1 | 0 | 1 | |
| Male | 1 | 0 | 0 | 1 | |
| Total | 1 | 1 | 0 | 2 | |
| 80+ | |||||
| Female | 0 | 1 | 0 | 1 | |
| Male | 0 | 1 | 0 | 1 | |
| Total | 0 | 2 | 0 | 2 | |
| TOTAL | 9 | 16 | 9 | 34 | |
| SpO2 (%) | |||
|---|---|---|---|
| Pre-Treatment | at +12 h | at +24 h | at +48 h |
| 66–84% | +12.8 ± 6.7 | +11.4 ± 6.1 | +11.7 ± 5.8 |
| 85–89% | +5.4 ± 2.6 | +6.2 ± 2.8 | +6.3 ± 3.0 |
| 90–93% | +2.8 ± 2.7 | +3.7 ± 2.3 | +3.9 ± 1.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stone, J.C.; Ndarukwa, P.; Scheim, D.E.; Dancis, B.M.; Dancis, J.; Gill, M.G.; Aldous, C. Changes in SpO2 on Room Air for 34 Severe COVID-19 Patients after Ivermectin-Based Combination Treatment: 62% Normalization within 24 Hours. Biologics 2022, 2, 196-210. https://doi.org/10.3390/biologics2030015
Stone JC, Ndarukwa P, Scheim DE, Dancis BM, Dancis J, Gill MG, Aldous C. Changes in SpO2 on Room Air for 34 Severe COVID-19 Patients after Ivermectin-Based Combination Treatment: 62% Normalization within 24 Hours. Biologics. 2022; 2(3):196-210. https://doi.org/10.3390/biologics2030015
Chicago/Turabian StyleStone, Jaqueline C., Pisirai Ndarukwa, David E. Scheim, Barry M. Dancis, Jerome Dancis, Martin G. Gill, and Colleen Aldous. 2022. "Changes in SpO2 on Room Air for 34 Severe COVID-19 Patients after Ivermectin-Based Combination Treatment: 62% Normalization within 24 Hours" Biologics 2, no. 3: 196-210. https://doi.org/10.3390/biologics2030015
APA StyleStone, J. C., Ndarukwa, P., Scheim, D. E., Dancis, B. M., Dancis, J., Gill, M. G., & Aldous, C. (2022). Changes in SpO2 on Room Air for 34 Severe COVID-19 Patients after Ivermectin-Based Combination Treatment: 62% Normalization within 24 Hours. Biologics, 2(3), 196-210. https://doi.org/10.3390/biologics2030015







