Peptides in COVID-19 Clinical Trials—A Snapshot
Abstract
1. Introduction
2. Clinical Trials
2.1. Immunomodulatory
2.2. Regain Homeostasis
2.3. Diagnostics/Biomarkers
2.4. Vaccination
2.5. Antiviral Activity
3. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Dardenne, M.; Safieh-Garabedian, B.; Pleau, J.-M. Thymic Peptides: Transmitters between the Neuroendocrine and the Immune System. In Pain Neuroimmune Interact; Springer: Boston, MA, USA, 2000; pp. 127–137. [Google Scholar] [CrossRef]
- Wolf, E.; Milazzo, S.; Boehm, K.; Zwahlen, M.; Horneber, M. Thymic peptides for treatment of cancer patients. Cochrane Database Syst. Rev. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Safety and Efficacy of Thymic Peptides in the Treatment of Hospitalized COVID-19 Patients in Honduras-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04771013 (accessed on 25 August 2021).
- Sjogren, M.H. Thymalfasin: An immune system enhancer for the treatment of liver disease. J. Gastroenterol. Hepatol. 2004, 19, S69–S72. [Google Scholar] [CrossRef] [PubMed]
- Thymosin Alpha 1 to Prevent COVID-19 Infection in Renal Dialysis Patients-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04428008?term=peptide&cond=COVID-19&draw=1&rank=16 (accessed on 27 August 2021).
- Thymalfasin (Thymosin Alpha 1) to Treat COVID-19 Infection-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04487444?term=peptide&cond=Covid19&draw=2&rank=25 (accessed on 6 September 2021).
- Choudhury, S.R.; Babes, L.; Rahn, J.J.; Ahn, B.Y.; Goring, K.A.R.; King, J.C.; Lau, A.; Petri, B.; Hao, X.; Chojnacki, A.K.; et al. Dipeptidase-1 Is an Adhesion Receptor for Neutrophil Recruitment in Lungs and Liver. Cell 2019, 178, 1205–1221. [Google Scholar] [CrossRef] [PubMed]
- LSALT Peptide vs. Placebo to Prevent ARDS and Acute Kidney Injury in Patients Infected with SARS-CoV-2 (COVID-19)-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04402957?term=peptide&cond=Covid19&draw=2&rank=6 (accessed on 26 August 2021).
- COVID-19 Specific T Cell Derived Exosomes (CSTC-Exo)-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04389385?term=peptide&cond=Covid19&draw=2&rank=54 (accessed on 7 September 2021).
- Sturrock, B.R.; Milne, K.M.; Chevassut, T.J. The renin–angiotensin system—A therapeutic target in COVID-19? Clin. Med. 2020, 20, e72–e75. [Google Scholar] [CrossRef] [PubMed]
- Treatment of Angiotensin Peptide (1-7) for COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04375124?term=peptide&cond=COVID-19&draw=2&rank=2 (accessed on 25 August 2021).
- Angiotensin-(1,7) Treatment in COVID-19: The ATCO Trial-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04332666?term=peptide&cond=COVID-19&draw=1&rank=18 (accessed on 27 August 2021).
- Use of Angiotensin-(1-7) in COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04633772?term=peptide&cond=Covid19&draw=2&rank=58 (accessed on 7 September 2021).
- RAS and Coagulopathy in COVID19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04419610?term=peptide&cond=COVID-19&draw=2&rank=34 (accessed on 26 August 2021).
- Novel Experimental COVID-19 Therapies Affecting Host Response-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04924660?term=peptide&cond=Covid19&draw=2&rank=49 (accessed on 7 September 2021).
- Recombinant Human Angiotensin-Converting Enzyme 2 (rhACE2) as a Treatment for Patients with COVID-19-Study Results-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/results/NCT04335136?term=angiotensine&cond=COVID-19&draw=2&rank=8#all (accessed on 25 August 2021).
- Lelievre, V.; Favrais, G.; Abad, C.; Adle-Biassette, H.; Lu, Y.; Germano, P.M.; Cheung-Lau, G.; Pisegna, J.R.; Gressens, P.; Lawson, G.; et al. Gastrointestinal dysfunction in mice with a targeted mutation in the gene encoding vasoactive intestinal polypeptide: A model for the study of intestinal ileus and Hirschsprung’s disease. Peptides 2007, 28, 1688–1699. [Google Scholar] [CrossRef] [PubMed][Green Version]
- ACTIV-3b: Therapeutics for Severely Ill Inpatients with COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04843761?term=peptide&cond=COVID-19&draw=1&rank=15 (accessed on 27 August 2021).
- A Clinical Study Evaluating Inhaled Aviptadil on COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04844580?term=peptide&cond=Covid19&draw=2&rank=51 (accessed on 7 September 2021).
- Intravenous Aviptadil for Critical COVID-19 with Respiratory Failure-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04311697?term=peptide&cond=Covid19&draw=2&rank=62 (accessed on 7 September 2021).
- FX06 to Rescue Acute Respiratory Distress Syndrome during Covid-19 Pneumonia-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04618042?term=peptide&cond=Covid19&draw=2&rank=44 (accessed on 6 September 2021).
- Bhutta, M.S.; Sausen, D.G.; Reed, K.M.; Gallo, E.S.; Hair, P.S.; Lassiter, B.P.; Krishna, N.K.; Cunnion, K.M.; Borenstein, R. Peptide Inhibitor of Complement C1, RLS-0071, Reduces Zosteriform Spread of Herpes Simplex Virus Type 1 Skin Infection and Promotes Survival in Infected Mice. Viruses 2021, 13, 1422. [Google Scholar] [CrossRef] [PubMed]
- A Study of RLS-0071 in Patients with Acute Lung Injury Due to COVID-19 Pneumonia in Early Respiratory Failure-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04574869?term=peptide&cond=Covid19&draw=2&rank=76 (accessed on 7 September 2021).
- BNP, Serum Troponin and D-Dimer as Risk Factors in Patients with COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04433026?term=peptide&cond=Covid19&draw=2&rank=3 (accessed on 26 August 2021).
- N-Terminal Pro B-type Natriuretic Peptide and Vitamin D Levels as Prognostic Markers in COVID-19 Pneumonia-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04487951?term=peptide&cond=Covid19&draw=2&rank=8 (accessed on 26 August 2021).
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Raharusun, P.; Priambada, S.; Budiarti, C.; Agung, E.; Budi, C. Patterns of COVID-19 Mortality and Vitamin D: An Indonesian Study. SSRN Electron. J. 2020, 3585561. [Google Scholar] [CrossRef]
- Vimaleswaran, K.S.; Forouhi, N.G.; Khunti, K. Vitamin D and covid-19. BMJ 2021, 372, 735–736. [Google Scholar] [CrossRef]
- Clinical Outcomes of High Dose Vitamin D Versus Standard Dose in COVID-19 Egyptian Patients-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04738760?term=peptide&cond=Covid19&draw=2&rank=47 (accessed on 7 September 2021).
- Cardiac Biomarkers and Mortality in Critically Ill Patients with SARS-CoV-2 (COVID-19)-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04641988?term=peptide&cond=COVID-19&draw=1&rank=23 (accessed on 27 August 2021).
- Paediatric Inflammatory Multisystem Syndrome during COVID-19 Pandemic-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04761913?term=peptide&cond=Covid19&draw=2&rank=22 (accessed on 6 September 2021).
- T Cells Response to SARS COV 2 Peptides-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04573348?term=peptide&cond=COVID-19&draw=1&rank=10 (accessed on 27 August 2021).
- Hausner, S.H.; Bold, R.J.; Cheuy, L.Y.; Chew, H.K.; Daly, M.E.; Davis, R.A.; Foster, C.C.; Kim, E.J.; Sutcliffe, J.L. Preclinical Development and First-in-Human Imaging of the Integrin αvβ6 with [18F]αvβ6-Binding Peptide in Metastatic Carcinoma. Clin. Cancer Res. 2019, 25, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- PET/CT Imaging in COVID-19 Patients-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04376593?term=peptide&cond=COVID-19&draw=1&rank=12 (accessed on 27 August 2021).
- RGD PET/CT Imaging in COVID-19 Patients-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04596943?term=peptide&cond=COVID-19&draw=1&rank=19 (accessed on 27 August 2021).
- Pulmonary Fibrosis during Severe COVID-19 Pneumonia-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04987528?term=peptide&cond=Covid19&draw=2&rank=21 (accessed on 6 September 2021).
- Safety and Immunogenicity Trial of Multi-peptide Vaccination to Prevent COVID-19 Infection in Adults-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04546841 (accessed on 26 August 2021).
- B-pVAC-SARS-CoV-2: Study to Prevent COVID-19 Infection in Adults with Bcell/Antibody Deficiency-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04954469?term=peptide&cond=COVID-19&draw=1&rank=14 (accessed on 27 August 2021).
- To Evaluate the Safety, and Immunogenicity of Vaccine Candidate Against COVID-19, in Healthy Adults-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04885361?term=peptide&cond=Covid19&draw=2&rank=33 (accessed on 6 September 2021).
- A Study to Evaluate the Safety, Tolerability, and Immunogenicity of UB-612 COVID-19 Vaccine-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04545749?term=peptide&cond=Covid19&draw=2&rank=37 (accessed on 6 September 2021).
- A Study to Evaluate UB-612 COVID-19 Vaccine in Adolescent, Younger and Elderly Adult Volunteers-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04773067?term=peptide&cond=Covid19&draw=2&rank=45 (accessed on 6 September 2021).
- Characterisation of the Immune Response to SARS-CoV-2-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04729452?term=peptide&cond=Covid19&draw=2&rank=41 (accessed on 6 September 2021).
- Study of the Safety, Reactogenicity and Immunogenicity of EpiVacCorona Vaccine for the Prevention of COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04527575?term=peptide&cond=Covid19&draw=2&rank=7 (accessed on 26 August 2021).
- Study of the EpiVacCorona Vaccine with the Involvement of Volunteers Aged 60 Years and Above-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT05021016?term=peptide&cond=Covid19&draw=2&rank=10 (accessed on 26 August 2021).
- Study of the Tolerability, Safety, Immunogenicity and Preventive Efficacy of the EpiVacCorona Vaccine for the Prevention of COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04780035?term=peptide&cond=Covid19&draw=2&rank=11 (accessed on 26 August 2021).
- Salamov, G.; Holms, R.; Bessler, W.G.; Ataullakhanov, R. Treatment of hepatitis C virus infection with human ezrin peptide one (HEP1) in HIV infected patients. Arzneimittelforschung 2007, 57, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Chulkina, M.; Negmadjanov, U.; Lebedeva, E.; Pichugin, A.; Mazurov, D.; Ataullakhanov, R.; Holmuhamedov, E. Synthetic peptide TEKKRRETVEREKE derived from ezrin induces differentiation of NIH/3T3 fibroblasts. Eur. J. Pharmacol. 2017, 811, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Ezrin Peptide (HEP-1) for Treatment of Coronavirus Disease (COVID-19) Infection-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04627233?term=peptide&cond=Covid19&draw=2&rank=5 (accessed on 26 August 2021).
- Bruni, N.; Capucchio, M.T.; Biasibetti, E.; Pessione, E.; Cirrincione, S.; Giraudo, L.; Corona, A.; Dosio, F. Antimicrobial Activity of Lactoferrin-Related Peptides and Applications in Human and Veterinary Medicine. Molecules 2016, 21, 752. [Google Scholar] [CrossRef] [PubMed]
- Utility of Lactoferrin as an Adjunct Therapeutic Agent for COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04421534?term=peptide&cond=Covid19&draw=2&rank=26 (accessed on 6 September 2021).
- Utility of Lactoferrin as a Preventive Agent for Healthcare Workers Exposed to COVID-19-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04427865?term=peptide&cond=Covid19&draw=2&rank=28 (accessed on 6 September 2021).
- Rathnapala, E.C.N.; Ahn, D.U.; Abeyrathne, S. Functional properties of ovotransferrin from chicken egg white and its derived peptides: A review. Food Sci. Biotechnol. 2021, 30, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Giansanti, F.; Massucci, M.T.; Giardi, M.F.; Nozza, F.; Pulsinelli, E.; Nicolini, C.; Botti, D.; Antonini, G. Antiviral activity of ovotransferrin derived peptides. Biochem. Biophys. Res. Commun. 2005, 331, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Efficacy and Safety of Ovotransferrin in COVID-19 Patients-Full Text View-ClinicalTrials. Available online: https://clinicaltrials.gov/ct2/show/NCT04643054?term=peptide&cond=Covid19&draw=2&rank=31 (accessed on 6 September 2021).
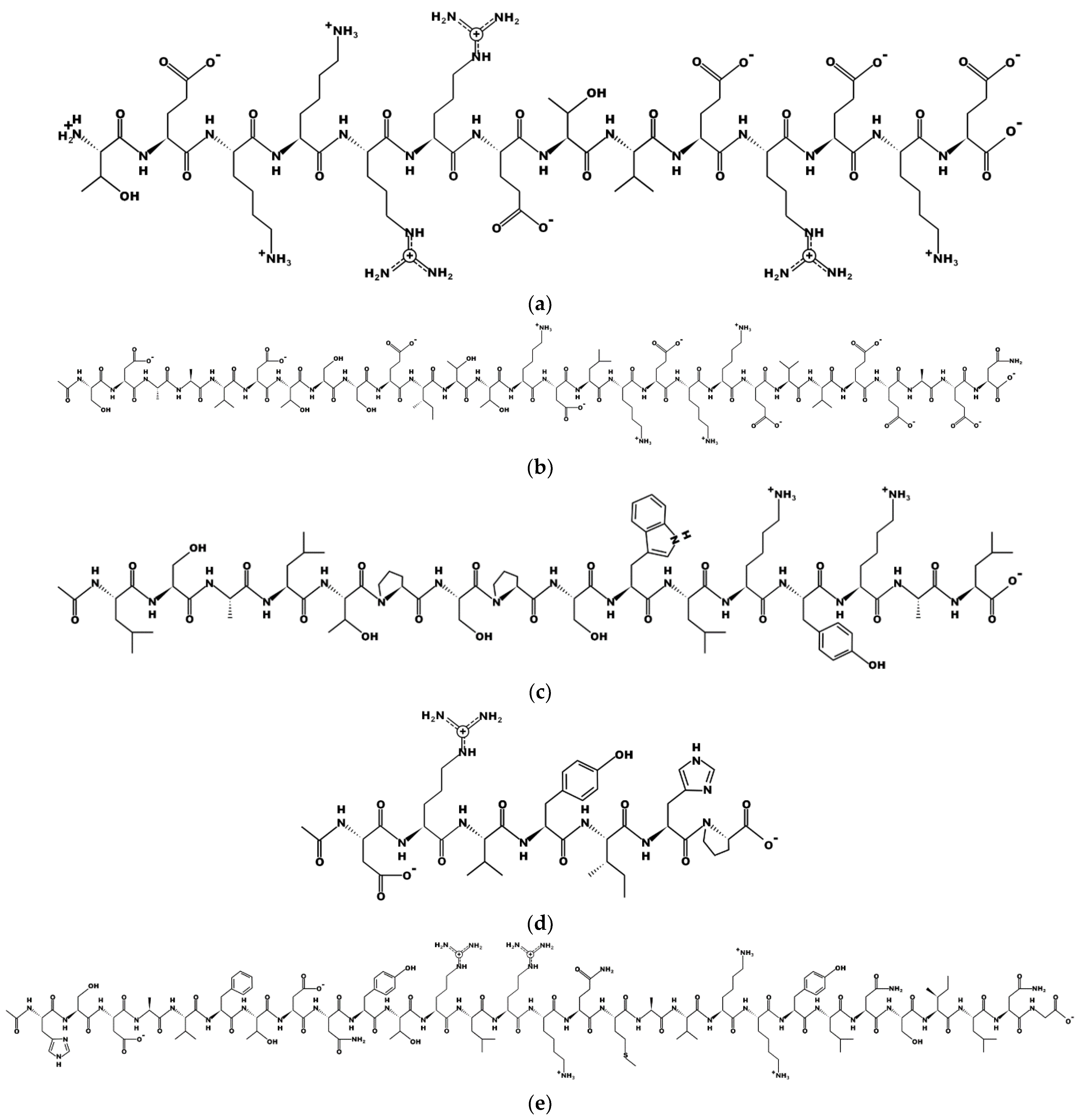
| Title | Peptide | Function | Status | Country | Reference |
|---|---|---|---|---|---|
| Efficacy and Safety of Lactoferrin as an Adjunct Therapeutic Agent for COVID-19 | Peptides derived by proteolytic cleavage of lactoferrin | Antiviral and immunomodulatory | Estimated completion: November 2020, no update | Egypt | [50] |
| Efficacy of Lactoferrin as a Preventive Agent for Healthcare Workers Exposed to COVID-19 | Peptides derived by proteolytic cleavage of lactoferrin | Antiviral and immunomodulatory | Estimated completion: November 2020, no update | Egypt | [51] |
| The Clinical Trial of Application of Ezrin Peptide (HEP-1) for Treatment of Coronavirus Disease (COVID-19) Infection | Human ezrin peptide 324-337, HEP1 (TEKKRRETVEREKE) | Antiviral and immunomodulatory | Estimated completion: April 2021, no update | Iran | [48] |
| Efficacy and Safety of Ovotransferrin in COVID-19 Patients With Mild-to-Moderate Disease | Peptides derived by proteolytic cleavage of ovotransferrin | Antiviral and immunomodulatory | Estimated completion: March 2021, no update | Italy | [54] |
| B-natriuretic Peptide (BNP), Serum Troponin-I, and D-dimer as Risk Factors for In-hospital Death in Patients With COVID-19 | N-terminal pro B type natriuretic peptide (NTproBNP, MDPQTAPSRALLLLLFLHLAFLGGRSHPLGSPGSASDLETSGLQEQRNHLQGKLSELQVEQTSLEPLQESPRPTGV) | Diagnostic/biomarker | Estimated completion: September 2021, no update | Egypt | [24] |
| N-terminal Pro B-type Natriuretic Peptide and Vitamin D Levels as Prognostic Markers in COVID-19 Pneumonia | N-terminal pro B type natriuretic peptide (NTproBNP, MDPQTAPSRALLLLLFLHLAFLGGRSHPLGSPGSASDLETSGLQEQRNHLQGKLSELQVEQTSLEPLQESPRPTGV) | Diagnostic/biomarker | Estimated completion: January 2021, no update | Egypt | [25] |
| Cardiac Biomarkers and Mortality in Critically Ill Patients With SARS-CoV-2 (COVID-19): COROBIOCHIC | N-terminal pro B type natriuretic peptide (NTproBNP, MDPQTAPSRALLLLLFLHLAFLGGRSHPLGSPGSASDLETSGLQEQRNHLQGKLSELQVEQTSLEPLQESPRPTGV) | Diagnostics/biomarker | Completed in June 2020, no update | France | [30] |
| Incidence, Risk Factors and Prognosis of Pulmonary Fibrosis During Severe COVID-19 Pneumonia | amino-terminal type I (PINP) and type III (PIIINP) peptides of procollagen | Diagnostics/biomarker | Estimated completion: December 2021 | France | [36] |
| Prospective, One Center, Four Groups, Open, Comparative, Controlled Study to Explore T Cells Response to SARS COV 2 Peptides by Metabolic Activity Method in Convalesce and Healthy Individuals Versus Antibody Response | undefined SARS-CoV-2 peptides | Diagnostics/biomarker | Estimated completion: October 2021 | Israel | [32] |
| [68Ga]Ga-DOTA-(RGD)2 PET/CT Imaging of Activated Endothelium in Lung Parenchyma of COVID-19 Patients | [68Ga]Ga-DOTA-(RGD)2 | Diagnostics/biomarker | Estimated completion: May 2022 | Netherlands | [35] |
| Investigating Cytokine Storm Biomarkers in Children Presenting to Acute Paediatric Services (Non-intensive Care) With Paediatric Inflammatory Multisystem Syndrome During the Covid-19 Pandemic. An Observation Study | N-terminal pro B type natriuretic peptide (NTproBNP, MDPQTAPSRALLLLLFLHLAFLGGRSHPLGSPGSASDLETSGLQEQRNHLQGKLSELQVEQTSLEPLQESPRPTGV) | Diagnostics/biomarker | Estimated completion: December 2021 | UK | [31] |
| 18F-αvβ6-binding-peptide PET/CT in Patients Post SARS CoV2 Infection | [18F]-αvβ6 binding peptide | Diagnostics/biomarker | Estimated completion: May 2022 | USA | [34] |
| A Single-arm, Open-Label, Phase II Clinical Study to Evaluate the Safety and Efficacy of Thymic Peptides in the Treatment of Hospitalized COVID-19 Patients in Honduras | thymic peptides, composition and source unknown | Immunomodulatory | Completed in May 2021, no update | Honduras | [3] |
| Aerosol Inhalation of the Exosomes Derived From Allogenic COVID-19 T Cell in the Treatment of Early Stage Novel Coronavirus Pneumonia | peptide fragments of the COVID-19 proteome | Immunomodulatory | Estimated completion: May 2021, no update | Turkey | [9] |
| A Pilot Trial of Thymalfasin (Ta1) to Prevent COVID-19 Infection in Renal Dialysis Patients) | thymosin-alpha 1 (Ac-SDAAVDTSSEITTKDLKEKKEVVEEAEN) | Immunomodulatory | Estimated completion: May 2022 | USA | [5] |
| A Pilot Trial of Thymalfasin (Ta1) to Treat COVID-19 Infection in Patients With Lymphocytopenia | thymosin-alpha 1 (Ac-SDAAVDTSSEITTKDLKEKKEVVEEAEN) | Immunomodulatory | Estimated completion: June 2021 | USA | [6] |
| Multicenter, Randomized, Double-Blind, Placebo-Controlled, Proof of Concept Study of LSALT Peptide as Prevention of Acute Respiratory Distress Syndrome (ARDS) and Acute Kidney Injury in Patients Infected With SARS-CoV-2 (COVID-19) | LSALT (LSALTPSPSWLKYKAL) | Immunomodulatory | Estimated completion: June 2021, no update | USA, Canada, Turkey | [8] |
| Angiotensin-(1,7) Treatment in COVID-19: the ATCO Trial | angiotensin peptide (1-7) (DRVYIHP) | Restoring homeostasis | Estimated completion: June 2023 | Belgium | [12] |
| Randomized Clinical Trial Phase I/II for the Use of Angiotensin-(1-7) in the Treatment of Severe Infection by Sars-CoV-2 | angiotensin peptide (1-7) (DRVYIHP) | Restoring homeostasis | Estimated completion: December 2021 | Brazil | [13] |
| FX06 to Rescue Acute Respiratory Distress Syndrome During Covid-19 Pneumonia | fibrin-derived peptide beta15-42 | Restoring homeostasis | Completed in June 2021, no update | France | [21] |
| Evaluation of the Possible Role of Angiotensin Peptide (1-7) on Treatment of COVID-19 | angiotensin peptide (1-7) (DRVYIHP) | Restoring homeostasis | Estimated completion: September 2020, no update | Turkey | [11] |
| A Comparative, Multicenter, Placebo-Controlled, Double-Blind Phase II Clinical Trial Evaluating the Efficacy, Safety and Tolerability of Inhaled Aviptadil in Patients 18 Years and Older With COVID-19 Pulmonary Involvement - HOPE | Aviptadil, a vasoactive intestinal peptide (VIP), analogue, inhalable | Restoring homeostasis | Estimated completion: December 2022 | Turkey | [19] |
| Investigating the Relationship Between the Renin Angiotensin System and the Coagulopathy Associated With COVID-19 | TRV027, a modified angiotensin peptide (1-7) (XRVYIHPA; X stands for sarcosine (Sar), and the C-terminal alanine residue has a hydroxy group) | Restoring homeostasis | Completed in May 2021, no data available yet | UK | [14] |
| CONNECTS Master Protocol for Clinical Trials Targeting Macro-, Micro-immuno-thrombosis, Vascular Hyperinflammation, and Hypercoagulability and Renin-angiotensin-aldosterone System (RAAS) in Hospitalized Patients With COVID-19 (ACTIV-4 Host Tissue) | TRV027 and TXA127, (TRV027:XRVYIHPA; X stands for sarcosine (Sar), and the C-terminal alanine residue has a hydroxy group), TXA127: pharmaceutical formulation of the natural angiotensin peptide (1-7) DRVYIHP) | Restoring homeostasis | Estimated completion: December 2022 | USA | [15] |
| ZYESAMI (Aviptadil) for the Treatment of Critical COVID-19 With Respiratory Failure | Aviptadil (HSDAVFTDNYTRLRKQMAVKKYLNSILN), a vasoactive intestinal peptide (VIP), analogue, intravenously administered | Restoring homeostasis | Completion: February 2021 | USA | [20] |
| A Randomized, Double-Blind, Placebo-Controlled, Two-Part Study to Evaluate the Safety, Tolerability, Preliminary Efficacy, PK, & PD of RLS-0071 in Patients With Acute Lung Injury Due to COVID-19 Pneumonia in Early Respiratory Failure | RLS-0071 (IALILEPICCQERAA-dPEG24) | Restoring homeostasis | Estimated completion: July 2021 | USA | [23] |
| A Multicenter, Adaptive, Randomized, Blinded Controlled Trial of the Safety and Efficacy of Investigational Therapeutics for Hospitalized Patients With Acute Respiratory Distress Syndrome Associated With COVID-19 | vasoactive intestinal peptide (VIP: HSDAVFTDNYTRLRKQMAVKKYLNSILNG and other drugs) | Restoring homeostasis | Estimated completion: April 2023 | Various | [18] |
| A Randomized, Open Label, Phase 1 Study to Evaluate the Safety, Reactogenicity and Immunogenicity of OSE-13E, a Multiepitope-based Vaccine Candidate Against COVID-19, in Healthy Adults (COVEPIT-3) | T-cell epitopes from 11 different proteins of SARS-CoV-2 | Vaccination | Estimated completion: March 2022 | Belgium | [39] |
| P-pVAC-SARS-CoV-2: Phase I Single-center Safety and Immunogenicity Trial of Multi-peptide Vaccination to Prevent COVID-19 Infection in Adults | a mixture of undefined peptides | Vaccination | Estimated completion: December 2021 | Germany | [37] |
| B-pVAC-SARS-CoV-2: Phase I/II Multicenter Safety and Immunogenicity Trial of Multi-peptide Vaccination to Prevent COVID-19 Infection in Adults With Bcell/ Antibody Deficiency | a mixture of undefined peptides | Vaccination | Estimated completion: December 2021 | Germany | [38] |
| Simple, Blind, Placebo-controlled, Randomized Study of the Safety, Reactogenicity and Immunogenicity of Vaccine Based on Peptide Antigens for the Prevention of COVID-19 (EpiVacCorona), in Volunteers Aged 18-60 Years (I-II Phase) | chemically synthesised peptides are antigens of SARS-CoV-2 proteins, undefined | Vaccination | Estimated completion: May 2021, no update | Russian Federation | [43] |
| An Open Study of the Safety, Reactogenicity and Immunogenicity of the Vaccine Based on Peptide Antigens for the Prevention of COVID-19 (EpiVacCorona), With the Involvement of Volunteers Aged 60 Years and Above (Phase III-IV) | chemically synthesised peptides are antigens of SARS-CoV-2 proteins, undefined | Vaccination | Estimated completion: January 2021, no update | Russian Federation | [44] |
| Multicenter Double-blind Placebo-controlled Comparative Randomized Study of the Tolerability, Safety, Immunogenicity and Prophylactic Efficacy of the EpiVacCorona Peptide Antigen-based Vaccine for the Prevention of COVID-19, With the Participation of 3000 Volunteers Aged 18 Years and Above (Phase III-IV) | chemically synthesised peptides are antigens of SARS-CoV-2 proteins, undefined | Vaccination | Estimated completion: January 2021, no update | Russian Federation | [45] |
| A Phase I, Open-label Study to Evaluate the Safety, Tolerability, and Immunogenicity of UB-612 Vaccine in Healthy Adult Volunteers | Th/CTL epitope peptide pool | Vaccination | Estimated completion: May 2021, no update | Taiwan | [40] |
| A Phase II, Placebo-controlled, Randomized, Observer-blind Study to Evaluate the Immunogenicity, Safety, and Tolerability of UB-612 Vaccine Against COVID-19 in Adolescent, Younger and Elderly Adult Volunteers | Th/CTL epitope peptide pool | Vaccination | Estimated completion: May 2021, no update | Taiwan | [41] |
| Characterisation of the Immune Response to SARS-CoV-2 Infection | overlapping peptides from the SARS-CoV-2 proteome | Vaccination | Estimated completion: December 2023 | UK | [42] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hilpert, K. Peptides in COVID-19 Clinical Trials—A Snapshot. Biologics 2021, 1, 300-311. https://doi.org/10.3390/biologics1030018
Hilpert K. Peptides in COVID-19 Clinical Trials—A Snapshot. Biologics. 2021; 1(3):300-311. https://doi.org/10.3390/biologics1030018
Chicago/Turabian StyleHilpert, Kai. 2021. "Peptides in COVID-19 Clinical Trials—A Snapshot" Biologics 1, no. 3: 300-311. https://doi.org/10.3390/biologics1030018
APA StyleHilpert, K. (2021). Peptides in COVID-19 Clinical Trials—A Snapshot. Biologics, 1(3), 300-311. https://doi.org/10.3390/biologics1030018






