NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications
Abstract
:Simple Summary
Abstract
1. Introduction
2. NUP98: A Commonly Translocated Gene in AML
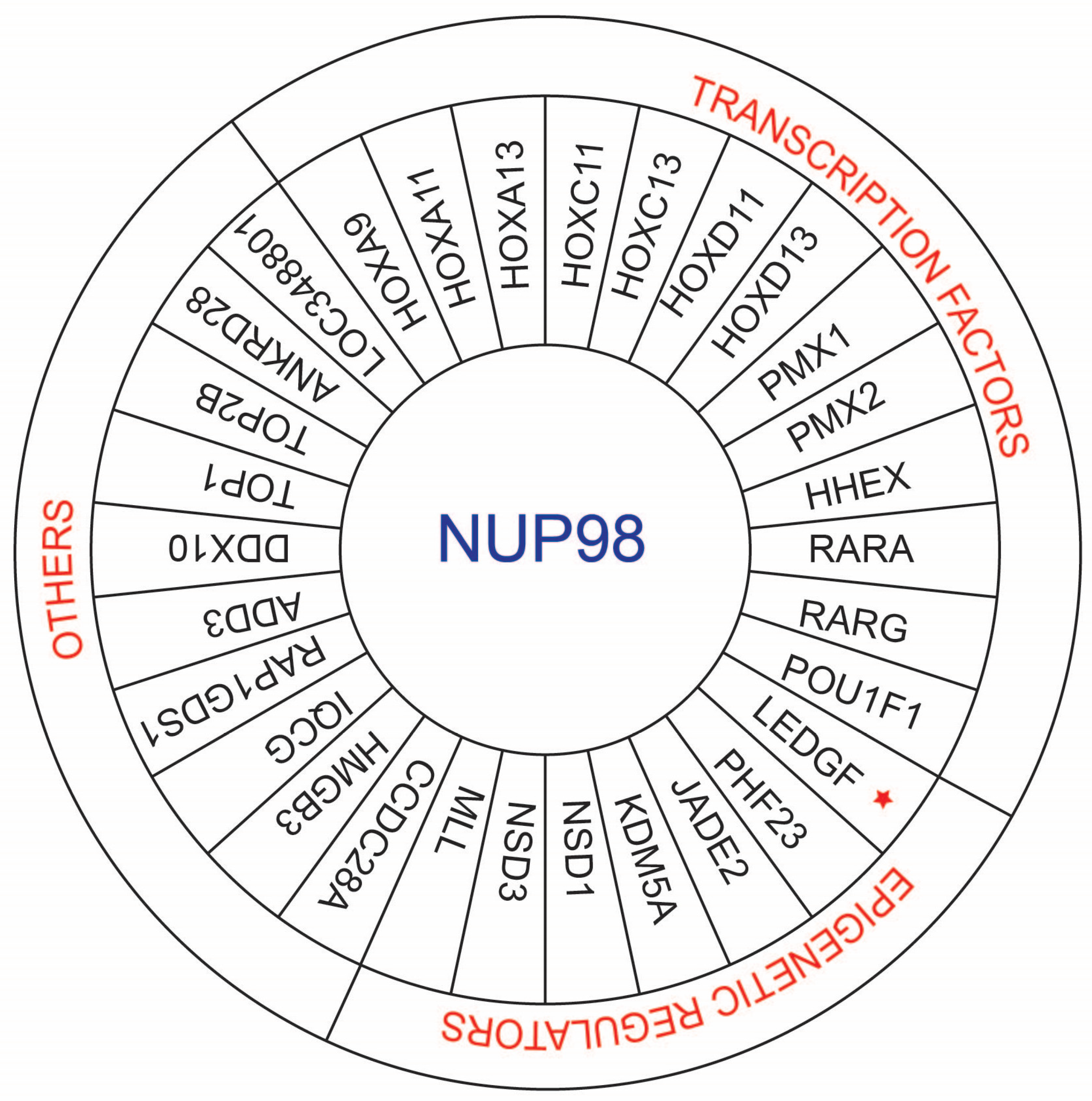
3. Fusion Partners of NUP98 in NUP98-Rearranged AML
| Fusion Partner | Functional Category | AML Subtype | Chromosome Rearrangement | Refs. |
|---|---|---|---|---|
| HOXA9 | Transcription factor | M2, M4 | t(7;11)(p15;p15) | [35,36] |
| HOXA11 | Transcription factor | M2 | t(7;11)(p15;p15) | [37] |
| HOXA13 | Transcription factor | M2 | t(7;11)(p15;p15) | [38] |
| HOXC11 | Transcription factor | M1, M2, M5 | t(11;12)(p15;q13) | [39,40] |
| HOXC13 | Transcription factor | M2, M4 | t(11;12)(p15;q13) | [41,42] |
| HOXD11 | Transcription factor | M4 | t(2;11)(q31;p15) | [43] |
| HOXD13 | Transcription factor | t-AML, M4 | t(2;11)(q31;p15) | [44,45] |
| PMX1 | Transcription factor | M2 | t(1;11)(q23;p15) | [46] |
| PMX2 | Transcription factor | t-AML | t(9;11)(q34;p15) | [[47] |
| HHEX | Transcription factor | M1, M2 | t(10;11)(q23;p15) | [48,49] |
| RARA | Transcription factor | M3 or APL | t(11;17) | [32] |
| RARG | Transcription factor | M3 or APL | t(11;12)(p15;q13) | [33,50] |
| POU1F1 | Transcription factor | t-AML | t(3;11)(p11;p15) | [51] |
| LEDGF/PSIP1 | Transcription coactivator | M1, M2 | t(9;11)(p22;p15) | [34,52,53] |
| PHF23 | Epigenetic modifier | M0, M1, M4, M5 | t(11;17)(p15;p13) | [54,55] |
| JADE2/PHF15 | Epigenetic modifier | M3 or APL | t(5;11)(q31;p15) | [56] |
| JARID1A/KDM5A | Epigenetic modifier | M0-M7 | t(11;15)(p15;q35) | [57,58] |
| NSD1 | Epigenetic modifier | M1, M2, M4, M5, M6 | t(5;11)(q35;p15.5) | [27,59,60,61] |
| NSD3 | Epigenetic modifier | M1 | t(8;11)(p11.2;p15) | [62] |
| MLL/KMT2A | Epigenetic modifier | M1, M2 | inv(11)(p15q23) | [63] |
| C6orf80/CCDC28A | Unknown | M7 | t(6;11)(q24.1;p15.5) | [64] |
| HMGB3 | High-mobility group (HMG) protein | t-AML | t(X;11)(q28;p15) | [65] |
| IQCG | Calcium signaling | AML (Unknown) | t(3;11)(q29q13;p15) | [66] |
| RAP1GDS1 | GTPase activity | AML (Unknown) | unknown | [67] |
| ADD3 | Cytoskeletal protein | AML (Unknown) | t(10;11) | [68] |
| DDX10 | RNA helicase | M6 | inv(11)(p15q22) | [69,70] |
| TOP1 | DNA Topoisomerase | M4, M5 | t(11;20)(p15;q11) | [27,71] |
| TOP2B | DNA Topoisomerase | M5 | t(3;11)(p24;p15) | [72] |
| ANKRD28 | Signaling protein | AML (Unknown) | t(3;5;11)(p25;q35;p15) | [73] |
| LOC348801 | Unknown | M2 | t(3;11)(q12;p15) | [74] |
4. NUP98 Fusions Represent a Poor Prognostic and Chemoresistant AML Subgroup
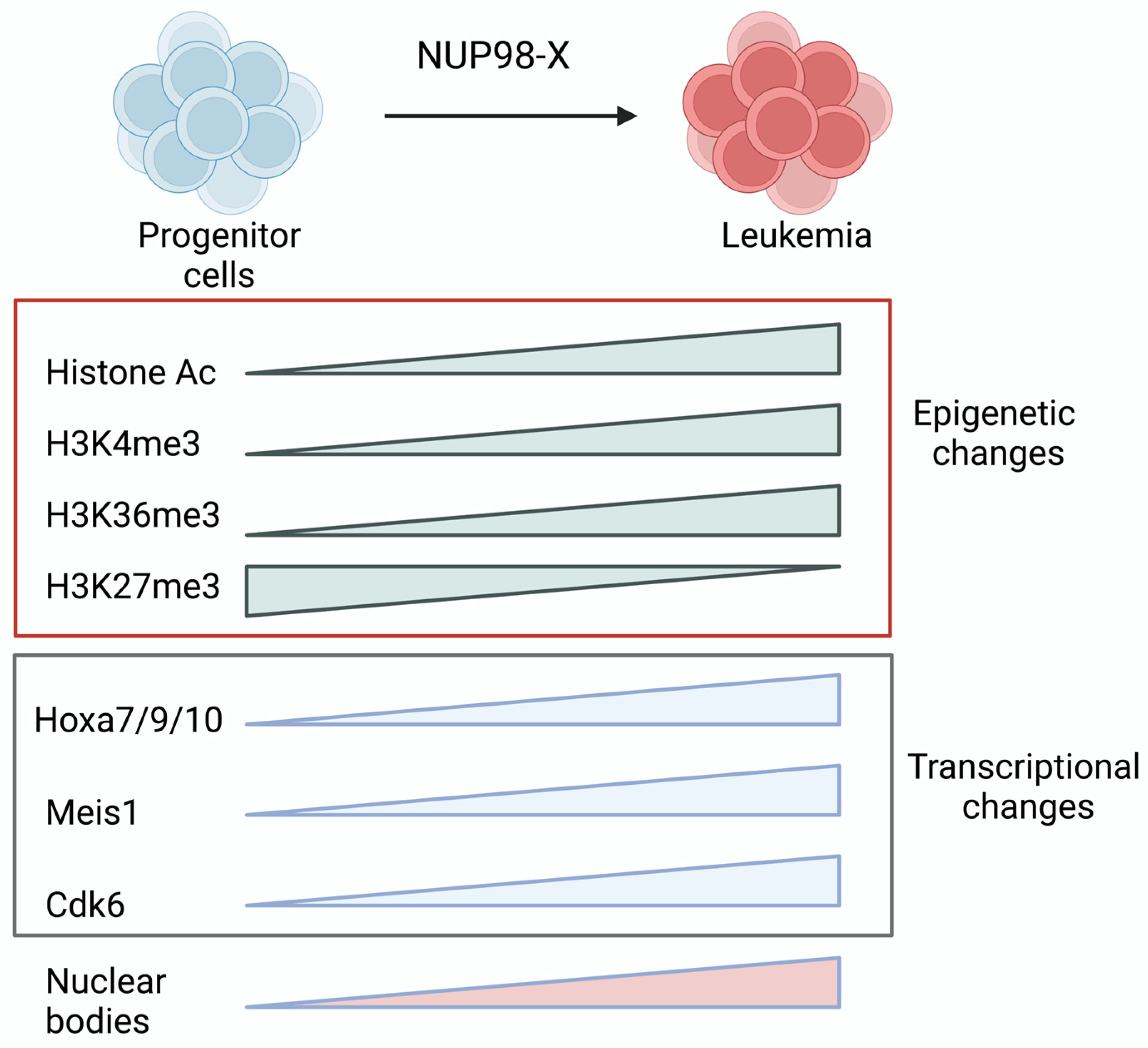
5. Mechanism of NUP98 Fusion Mediated AML
6. Cooperating Abnormalities in NUP98 Rearranged AML
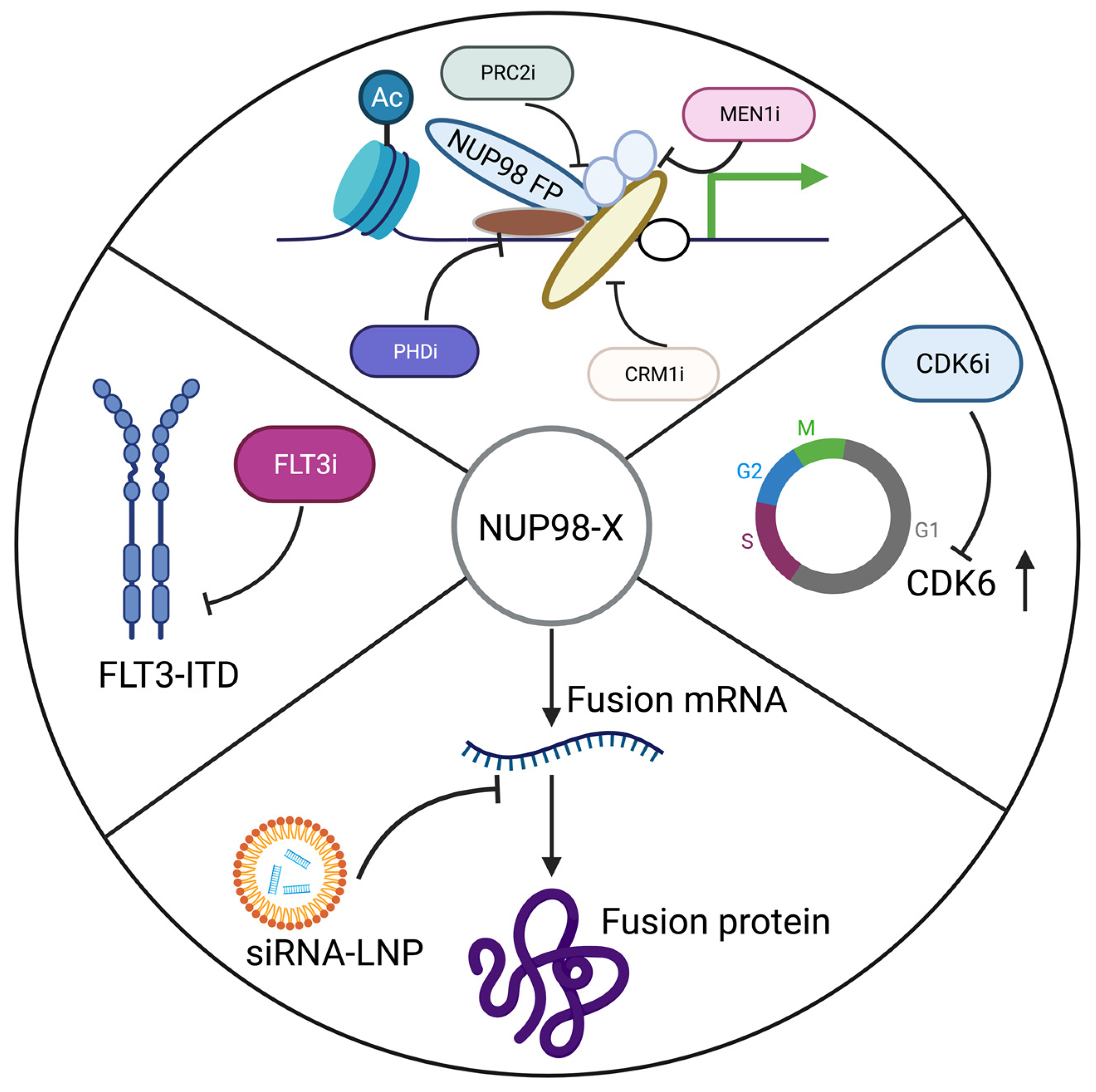
7. Therapeutic Strategies to Treat NUP98 Fusion Positive AML Patients
8. Concluding Remarks
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [Google Scholar] [CrossRef]
- Taniue, K.; Akimitsu, N. Fusion Genes and RNAs in Cancer Development. Non-Coding RNA 2021, 7. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Liang, W.W.; Foltz, S.M.; Mutharasu, G.; Jayasinghe, R.G.; Cao, S.; Liao, W.W.; Reynolds, S.M.; Wyczalkowski, M.A.; Yao, L.; et al. Driver Fusions and Their Implications in the Development and Treatment of Human Cancers. Cell Rep. 2018, 23, 227–238.e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, J.J.; Rohan, S.; Kao, J.; Kitabayashi, N.; Mathew, S.; Chen, Y.-T. Gene fusions between TMPRSS2 and ETS family genes in prostate cancer: Frequency and transcript variant analysis by RT-PCR and FISH on paraffin-embedded tissues. Mod. Pathol. 2007, 20, 921–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Persson, M.; Andrén, Y.; Mark, J.; Horlings, H.M.; Persson, F.; Stenman, G. Recurrent fusion of MYB and NFIB transcription factor genes in carcinomas of the breast and head and neck. Proc. Natl. Acad. Sci. USA 2009, 106, 18740–18744. [Google Scholar] [CrossRef]
- Annala, M.J.; Parker, B.C.; Zhang, W.; Nykter, M. Fusion genes and their discovery using high throughput sequencing. Cancer Lett. 2013, 340, 192–200. [Google Scholar] [CrossRef] [Green Version]
- Engvall, M.; Cahill, N.; Jonsson, B.-I.; Höglund, M.; Hallböök, H.; Cavelier, L. Detection of leukemia gene fusions by targeted RNA-sequencing in routine diagnostics. BMC Med. Genom. 2020, 13, 106. [Google Scholar] [CrossRef]
- Heydt, C.; Wölwer, C.B.; Velazquez Camacho, O.; Wagener-Ryczek, S.; Pappesch, R.; Siemanowski, J.; Rehker, J.; Haller, F.; Agaimy, A.; Worm, K.; et al. Detection of gene fusions using targeted next-generation sequencing: A comparative evaluation. BMC Med. Genom. 2021, 14, 62. [Google Scholar] [CrossRef]
- Padella, A.; Simonetti, G.; Paciello, G.; Giotopoulos, G.; Baldazzi, C.; Righi, S.; Ghetti, M.; Stengel, A.; Guadagnuolo, V.; De Tommaso, R.; et al. Novel and Rare Fusion Transcripts Involving Transcription Factors and Tumor Suppressor Genes in Acute Myeloid Leukemia. Cancers 2019, 11, 1951. [Google Scholar] [CrossRef] [Green Version]
- Druker, B.J.; Tamura, S.; Buchdunger, E.; Ohno, S.; Segal, G.M.; Fanning, S.; Zimmermann, J.; Lydon, N.B. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat. Med. 1996, 2, 561–566. [Google Scholar] [CrossRef]
- Stengel, A.; Shahswar, R.; Haferlach, T.; Walter, W.; Hutter, S.; Meggendorfer, M.; Kern, W.; Haferlach, C. Whole transcriptome sequencing detects a large number of novel fusion transcripts in patients with AML and MDS. Blood Adv. 2020, 4, 5393–5401. [Google Scholar] [CrossRef]
- Ley, T.J.; Miller, C.; Ding, L.; Raphael, B.J.; Mungall, A.J.; Robertson, A.; Hoadley, K.; Triche, T.J., Jr.; Laird, P.W.; Baty, J.D.; et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 2013, 368, 2059–2074. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Kasper, L.H.; Mantcheva, R.T.; Mantchev, G.T.; Springett, M.J.; van Deursen, J.M. Disruption of the FG nucleoporin NUP98 causes selective changes in nuclear pore complex stoichiometry and function. Proc. Natl. Acad. Sci. USA 2001, 98, 3191–3196. [Google Scholar] [CrossRef] [PubMed]
- Fontoura, B.M.; Blobel, G.; Matunis, M.J. A conserved biogenesis pathway for nucleoporins: Proteolytic processing of a 186-kilodalton precursor generates Nup98 and the novel nucleoporin, Nup96. J. Cell Biol. 1999, 144, 1097–1112. [Google Scholar] [CrossRef]
- Paci, G.; Caria, J.; Lemke, E.A. Cargo transport through the nuclear pore complex at a glance. J. Cell Sci. 2021, 134, jcs247874. [Google Scholar] [CrossRef] [PubMed]
- Nofrini, V.; Di Giacomo, D.; Mecucci, C. Nucleoporin genes in human diseases. Eur. J. Hum. Genet. EJHG 2016, 24, 1388–1395. [Google Scholar] [CrossRef] [Green Version]
- Griffis, E.R.; Xu, S.; Powers, M.A. Nup98 localizes to both nuclear and cytoplasmic sides of the nuclear pore and binds to two distinct nucleoporin subcomplexes. Mol. Biol. Cell 2003, 14, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Chatel, G.; Desai, S.H.; Mattheyses, A.L.; Powers, M.A.; Fahrenkrog, B. Domain topology of nucleoporin Nup98 within the nuclear pore complex. J. Struct. Biol. 2012, 177, 81–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwamoto, M.; Asakawa, H.; Hiraoka, Y.; Haraguchi, T. Nucleoporin Nup98: A gatekeeper in the eukaryotic kingdoms. Genes Cells Devoted Mol. Cell. Mech. 2010, 15, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Oka, M.; Asally, M.; Yasuda, Y.; Ogawa, Y.; Tachibana, T.; Yoneda, Y. The mobile FG nucleoporin Nup98 is a cofactor for Crm1-dependent protein export. Mol. Biol. Cell 2010, 21, 1885–1896. [Google Scholar] [CrossRef] [Green Version]
- Pritchard, C.E.; Fornerod, M.; Kasper, L.H.; van Deursen, J.M. RAE1 is a shuttling mRNA export factor that binds to a GLEBS-like NUP98 motif at the nuclear pore complex through multiple domains. J. Cell Biol. 1999, 145, 237–254. [Google Scholar] [CrossRef]
- Ren, Y.; Seo, H.S.; Blobel, G.; Hoelz, A. Structural and functional analysis of the interaction between the nucleoporin NUP98 and the mRNA export factor Rae1. Proc. Natl. Acad. Sci. USA 2010, 107, 10406–10411. [Google Scholar] [CrossRef] [PubMed]
- Griffis, E.R.; Altan, N.; Lippincott-Schwartz, J.; Powers, M.A. NUP98 is a mobile nucleoporin with transcription-dependent dynamics. Mol. Biol. Cell 2002, 13, 1282–1297. [Google Scholar] [CrossRef] [PubMed]
- Kasper, L.H.; Brindle, P.K.; Schnabel, C.A.; Pritchard, C.E.; Cleary, M.L.; van Deursen, J.M. CREB binding protein interacts with nucleoporin-specific FG repeats that activate transcription and mediate NUP98-HOXA9 oncogenicity. Mol. Cell Biol. 1999, 19, 764–776. [Google Scholar] [CrossRef] [Green Version]
- Kalverda, B.; Pickersgill, H.; Shloma, V.V.; Fornerod, M. Nucleoporins directly stimulate expression of developmental and cell-cycle genes inside the nucleoplasm. Cell 2010, 140, 360–371. [Google Scholar] [CrossRef] [Green Version]
- Michmerhuizen, N.L.; Klco, J.M.; Mullighan, C.G. Mechanistic insights and potential therapeutic approaches for NUP98-rearranged hematologic malignancies. Blood 2020, 136, 2275–2289. [Google Scholar] [CrossRef]
- Struski, S.; Lagarde, S.; Bories, P.; Puiseux, C.; Prade, N.; Cuccuini, W.; Pages, M.P.; Bidet, A.; Gervais, C.; Lafage-Pochitaloff, M.; et al. NUP98 is rearranged in 3.8% of pediatric AML forming a clinical and molecular homogenous group with a poor prognosis. Leukemia 2017, 31, 565–572. [Google Scholar] [CrossRef]
- Bisio, V.; Zampini, M.; Tregnago, C.; Manara, E.; Salsi, V.; Di Meglio, A.; Masetti, R.; Togni, M.; Di Giacomo, D.; Minuzzo, S.; et al. NUP98-fusion transcripts characterize different biological entities within acute myeloid leukemia: A report from the AIEOP-AML group. Leukemia 2017, 31, 974–977. [Google Scholar] [CrossRef]
- Bertrums, E.J.M.; Smith, J.L.; Ries, R.E.; Alonzo, T.A.; Ostronoff, F.; Kaspers, G.J.L.; Hasle, H.; Zwaan, C.M.; Hirsch, B.A.; Raimondi, S.C.; et al. The Molecular Characteristics and Clinical Relevance of NUP98-Other Translocations in Pediatric Acute Myeloid Leukemia. Blood 2020, 136, 36–37. [Google Scholar] [CrossRef]
- Bertrums, E.J.M.; Smith, J.L.; Harmon, L.; Ries, R.E.; Wang, Y.J.; Alonzo, T.A.; Menssen, A.J.; Chisholm, K.M.; Leonti, A.R.; Tarlock, K.; et al. Comprehensive molecular and clinical characterization of NUP98 fusions in pediatric acute myeloid leukemia. Haematologica 2023. [Google Scholar] [CrossRef]
- Gough, S.M.; Slape, C.I.; Aplan, P.D. NUP98 gene fusions and hematopoietic malignancies: Common themes and new biologic insights. Blood 2011, 118, 6247–6257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, H.H.; Yang, M.C.; Wang, F.; Lou, Y.J.; Jin, J.; Li, K.; Zhang, S.Z. Identification of a novel NUP98-RARA fusion transcript as the 14th variant of acute promyelocytic leukemia. Am. J. Hematol. 2020, 95, E184–E186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Such, E.; Cervera, J.; Valencia, A.; Barragán, E.; Ibañez, M.; Luna, I.; Fuster, Ó.; Perez-Sirvent, M.L.; Senent, L.; Sempere, A.; et al. A novel NUP98/RARG gene fusion in acute myeloid leukemia resembling acute promyelocytic leukemia. Blood 2011, 117, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Gallego Hernanz, M.P.; Torregrosa Diaz, J.M.; Sorel, N.; Bobin, A.; Dindinaud, E.; Bouyer, S.; Desmier, D.; Brizard, F.; Leleu, X.; Maillard, N.; et al. Long-term molecular remission in a patient with acute myeloid leukemia harboring a new NUP98-LEDGF rearrangement. Cancer Med. 2019, 8, 1765–1770. [Google Scholar] [CrossRef]
- Huang, S.Y.; Tang, J.L.; Liang, Y.J.; Wang, C.H.; Chen, Y.C.; Tien, H.F. Clinical, haematological and molecular studies in patients with chromosome translocation t(7;11): A study of four Chinese patients in Taiwan. Br. J. Haematol. 1997, 96, 682–687. [Google Scholar] [CrossRef]
- Lahortiga, I.; Belloni, E.; Vázquez, I.; Agirre, X.; Larrayoz, M.J.; Vizmanos, J.L.; Valgañón, M.; Zudaire, I.; Sáez, B.; Mateos, M.C.; et al. NUP98 is fused to HOXA9 in a variant complex t(7;11;13;17) in a patient with AML-M2. Cancer Genet. Cytogenet. 2005, 157, 151–156. [Google Scholar] [CrossRef]
- Suzuki, A.; Ito, Y.; Sashida, G.; Honda, S.; Katagiri, T.; Fujino, T.; Nakamura, T.; Ohyashiki, K. t(7;11)(p15;p15) Chronic myeloid leukaemia developed into blastic transformation showing a novel NUP98/HOXA11 fusion. Br. J. Haematol. 2002, 116, 170–172. [Google Scholar] [CrossRef]
- Taketani, T.; Taki, T.; Ono, R.; Kobayashi, Y.; Ida, K.; Hayashi, Y. The chromosome translocation t(7;11)(p15;p15) in acute myeloid leukemia results in fusion of the NUP98 gene with a HOXA cluster gene, HOXA13, but not HOXA9. Genes Chromosomes Cancer 2002, 34, 437–443. [Google Scholar] [CrossRef]
- Taketani, T.; Taki, T.; Shibuya, N.; Kikuchi, A.; Hanada, R.; Hayashi, Y. Novel NUP98-HOXC11 Fusion Gene Resulted from a Chromosomal Break within Exon 1 of HOXC11 in Acute Myeloid Leukemia with t(11;12)(p15;q13)1. Cancer Res. 2002, 62, 4571–4574. [Google Scholar]
- Gu, B.W.; Wang, Q.; Wang, J.M.; Xue, Y.Q.; Fang, J.; Wong, K.F.; Chen, B.; Shi, Z.Z.; Shi, J.Y.; Bai, X.T.; et al. Major form of NUP98/HOXC11 fusion in adult AML with t(11;12)(p15;q13) translocation exhibits aberrant trans-regulatory activity. Leukemia 2003, 17, 1858–1864. [Google Scholar] [CrossRef] [Green Version]
- Panagopoulos, I.; Isaksson, M.; Billström, R.; Strömbeck, B.; Mitelman, F.; Johansson, B. Fusion of the NUP98 gene and the homeobox gene HOXC13 in acute myeloid leukemia with t(11;12)(p15;q13). Genes Chromosomes Cancer 2003, 36, 107–112. [Google Scholar] [CrossRef]
- Tosić, N.; Stojiljković, M.; Colović, N.; Colović, M.; Pavlović, S. Acute myeloid leukemia with NUP98-HOXC13 fusion and FLT3 internal tandem duplication mutation: Case report and literature review. Cancer Genet. Cytogenet. 2009, 193, 98–103. [Google Scholar] [CrossRef]
- Taketani, T.; Taki, T.; Shibuya, N.; Ito, E.; Kitazawa, J.; Terui, K.; Hayashi, Y. The HOXD11 Gene Is Fused to the NUP98 Gene in Acute Myeloid Leukemia with t(2;11)(q31;p15)1. Cancer Res. 2002, 62, 33–37. [Google Scholar] [PubMed]
- Raza-Egilmez, S.Z.; Jani-Sait, S.N.; Grossi, M.; Higgins, M.J.; Shows, T.B.; Aplan, P.D. NUP98-HOXD13 gene fusion in therapy-related acute myelogenous leukemia. Cancer Res. 1998, 58, 4269–4273. [Google Scholar] [PubMed]
- Arai, Y.; Kyo, T.; Miwa, H.; Arai, K.; Kamada, N.; Kita, K.; Ohki, M. Heterogeneous fusion transcripts involving the NUP98 gene and HOXD13 gene activation in a case of acute myeloid leukemia with the t(2;11)(q31;p15) translocation. Leukemia 2000, 14, 1621–1629. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, T.; Yamazaki, Y.; Hatano, Y.; Miura, I. NUP98 is fused to PMX1 homeobox gene in human acute myelogenous leukemia with chromosome translocation t(1;11)(q23;p15). Blood 1999, 94, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Gervais, C.; Mauvieux, L.; Perrusson, N.; Hélias, C.; Struski, S.; Leymarie, V.; Lioure, B.; Lessard, M. A new translocation t(9;11)(q34;p15) fuses NUP98 to a novel homeobox partner gene, PRRX2, in a therapy-related acute myeloid leukemia. Leukemia 2005, 19, 145–148. [Google Scholar] [CrossRef]
- Jankovic, D.; Gorello, P.; Liu, T.; Ehret, S.; La Starza, R.; Desjobert, C.; Baty, F.; Brutsche, M.; Jayaraman, P.-S.; Santoro, A.; et al. Leukemogenic mechanisms and targets of a NUP98/HHEX fusion in acute myeloid leukemia. Blood 2008, 111, 5672–5682. [Google Scholar] [CrossRef]
- Sorel, N.; Raimbault, A.; Brizard, F.; Depaire, T.; Pierini, V.; Dupraz, C.; Millot, F.; Mecucci, C.; Chomel, J.C. Identification and genetic characterization of a NUP98-HHEX molecular rearrangement in a pediatric acute myeloid leukemia. Leuk. Lymphoma 2021, 62, 3531–3535. [Google Scholar] [CrossRef]
- Tao, S.; Song, L.; Deng, Y.; Chen, Y.; Shi, Y.; Gan, Y.; Deng, Z.; Ding, B.; He, Z.; Wang, C.; et al. Acute Myeloid Leukemia with NUP98-RARG Gene Fusion Similar to Acute Promyelocytic Leukemia: Case Report and Literature Review. OncoTargets Ther. 2020, 13, 10559–10566. [Google Scholar] [CrossRef]
- Lisboa, S.; Cerveira, N.; Bizarro, S.; Correia, C.; Vieira, J.; Torres, L.; Mariz, J.M.; Teixeira, M.R. POU1F1 is a novel fusion partner of NUP98 in acute myeloid leukemia with t(3;11)(p11;p15). Mol. Cancer 2013, 12, 5. [Google Scholar] [CrossRef] [Green Version]
- Ahuja, H.G.; Hong, J.; Aplan, P.D.; Tcheurekdjian, L.; Forman, S.J.; Slovak, M.L. t(9;11)(p22;p15) in acute myeloid leukemia results in a fusion between NUP98 and the gene encoding transcriptional coactivators p52 and p75-lens epithelium-derived growth factor (LEDGF). Cancer Res. 2000, 60, 6227–6229. [Google Scholar]
- Hussey, D.J.; Moore, S.; Nicola, M.; Dobrovic, A. Fusion of the NUP98 gene with the LEDGF/p52 gene defines a recurrent acute myeloid leukemia translocation. BMC Genet. 2001, 2, 20. [Google Scholar] [CrossRef]
- Reader, J.C.; Meekins, J.S.; Gojo, I.; Ning, Y. A novel NUP98-PHF23 fusion resulting from a cryptic translocation t(11;17)(p15;p13) in acute myeloid leukemia. Leukemia 2007, 21, 842–844. [Google Scholar] [CrossRef] [PubMed]
- Togni, M.; Masetti, R.; Pigazzi, M.; Astolfi, A.; Zama, D.; Indio, V.; Serravalle, S.; Manara, E.; Bisio, V.; Rizzari, C.; et al. Identification of the NUP98-PHF23 fusion gene in pediatric cytogenetically normal acute myeloid leukemia by whole-transcriptome sequencing. J. Hematol. Oncol. 2015, 8, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.-K.; Chan, H.-Y.; Yung, Y.-L.; Wan, T.S.K.; Leung, A.W.K.; Li, C.-K.; Tian, K.; Chan, N.P.H.; Cheung, J.S.; Ng, M.H.L. A novel NUP98-JADE2 fusion in a patient with acute myeloid leukemia resembling acute promyelocytic leukemia. Blood Adv. 2022, 6, 410–415. [Google Scholar] [CrossRef] [PubMed]
- de Rooij, J.D.E.; Hollink, I.H.I.M.; Arentsen-Peters, S.T.C.J.M.; van Galen, J.F.; Berna Beverloo, H.; Baruchel, A.; Trka, J.; Reinhardt, D.; Sonneveld, E.; Zimmermann, M.; et al. NUP98/JARID1A is a novel recurrent abnormality in pediatric acute megakaryoblastic leukemia with a distinct HOX gene expression pattern. Leukemia 2013, 27, 2280–2288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noort, S.; Wander, P.; Alonzo, T.A.; Smith, J.; Ries, R.E.; Gerbing, R.B.; Dolman, M.E.M.; Locatelli, F.; Reinhardt, D.; Baruchel, A.; et al. The clinical and biological characteristics of NUP98-KDM5A in pediatric acute myeloid leukemia. Haematologica 2021, 106, 630–634. [Google Scholar] [CrossRef]
- Jaju, R.J.; Fidler, C.; Haas, O.A.; Strickson, A.J.; Watkins, F.; Clark, K.; Cross, N.C.P.; Cheng, J.-F.; Aplan, P.D.; Kearney, L.; et al. A novel gene, NSD1, is fused to NUP98 in the t(5;11)(q35;p15.5) in de novo childhood acute myeloid leukemia. Blood 2001, 98, 1264–1267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iacobucci, I.; Wen, J.; Meggendorfer, M.; Choi, J.K.; Shi, L.; Pounds, S.B.; Carmichael, C.L.; Masih, K.E.; Morris, S.M.; Lindsley, R.C.; et al. Genomic subtyping and therapeutic targeting of acute erythroleukemia. Nat. Genet. 2019, 51, 694–704. [Google Scholar] [CrossRef]
- Wang, T.; Ni, J.B.; Wang, X.Y.; Dai, Y.; Ma, X.L.; Su, Y.C.; Gao, Y.Y.; Chen, X.; Yuan, L.L.; Liu, H.X. [Genetic characteristics and clinical outcomes of pediatric acute myeloid leukemia with NUP98-NSD1 fusion gene]. Zhonghua Yi Xue Za Zhi 2019, 99, 2820–2825. [Google Scholar] [CrossRef] [PubMed]
- Rosati, R.; La Starza, R.; Veronese, A.; Aventin, A.; Schwienbacher, C.; Vallespi, T.; Negrini, M.; Martelli, M.F.; Mecucci, C. NUP98 is fused to the NSD3 gene in acute myeloid leukemia associated with t(8;11)(p11.2;p15). Blood 2002, 99, 3857–3860. [Google Scholar] [CrossRef] [Green Version]
- Kaltenbach, S.; Soler, G.; Barin, C.; Gervais, C.; Bernard, O.A.; Penard-Lacronique, V.; Romana, S.P. NUP98-MLL fusion in human acute myeloblastic leukemia. Blood 2010, 116, 2332–2335. [Google Scholar] [CrossRef]
- Tosi, S.; Ballabio, E.; Teigler-Schlegel, A.; Boultwood, J.; Bruch, J.; Harbott, J. Characterization of 6q abnormalities in childhood acute myeloid leukemia and identification of a novel t(6;11)(q24.1;p15.5) resulting in a NUP98-C6orf80 fusion in a case of acute megakaryoblastic leukemia. Genes Chromosomes Cancer 2005, 44, 225–232. [Google Scholar] [CrossRef]
- Petit, A.; Ragu, C.; Della-Valle, V.; Mozziconacci, M.J.; Lafage-Pochitaloff, M.; Soler, G.; Schluth, C.; Radford, I.; Ottolenghi, C.; Bernard, O.A.; et al. NUP98–HMGB3: A novel oncogenic fusion. Leukemia 2010, 24, 654–658. [Google Scholar] [CrossRef] [Green Version]
- Pan, Q.; Zhu, Y.-J.; Gu, B.-W.; Cai, X.; Bai, X.-T.; Yuan, H.-Y.; Zhu, J.; Chen, Z.; Xue, Y.-Q.; Chen, S.-J. A New Fusion Gene NUP98-IQCG Identified in an Acute T/Myeloid Leukemia with t(3;11)(q29q13;p15) Translocation. Blood 2007, 110, 1828. [Google Scholar] [CrossRef]
- Umeda, M.; Michmerhuizen, N.; Ma, J.; Westover, T.; Walsh, M.P.; Song, G.; Mecucci, C.; Giacomo, D.D.; Locatelli, F.; Masetti, R.; et al. AML-283 The Genetic Landscape of NUP98-Rearranged Pediatric Leukemia. Clin. Lymphoma Myeloma Leuk. 2022, 22, S233. [Google Scholar] [CrossRef]
- Bisio, V.; Pigazzi, M.; Manara, E.; Masetti, R.; Togni, M.; Astolfi, A.; Mecucci, C.; Zappavigna, V.; Salsi, V.; Merli, P.; et al. NUP98 Fusion Proteins Are Recurrent Aberrancies in Childhood Acute Myeloid Leukemia: A Report from the AIEOP AML-2001-02 Study Group. Blood 2014, 124, 1025. [Google Scholar] [CrossRef]
- Arai, Y.; Hosoda, F.; Kobayashi, H.; Arai, K.; Hayashi, Y.; Kamada, N.; Kaneko, Y.; Ohki, M. The inv(11)(p15q22) chromosome translocation of de novo and therapy-related myeloid malignancies results in fusion of the nucleoporin gene, NUP98, with the putative RNA helicase gene, DDX10. Blood 1997, 89, 3936–3944. [Google Scholar] [CrossRef] [PubMed]
- Hollink, I.H.I.M.; van den Heuvel-Eibrink, M.M.; Arentsen-Peters, S.T.C.J.M.; Pratcorona, M.; Abbas, S.; Kuipers, J.E.; van Galen, J.F.; Beverloo, H.B.; Sonneveld, E.; Kaspers, G.-J.J.L.; et al. NUP98/NSD1 characterizes a novel poor prognostic group in acute myeloid leukemia with a distinct HOX gene expression pattern. Blood 2011, 118, 3645–3656. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Xue, Y.; Chen, Z.; Guo, Y.; Wu, Y.; Pan, J. Generation of the NUP98-TOP1 fusion transcript by the t(11;20) (p15;q11) in a case of acute monocytic leukemia. Cancer Genet. Cytogenet. 2003, 140, 153–156. [Google Scholar] [CrossRef]
- Nebral, K.; Schmidt, H.H.; Haas, O.A.; Strehl, S. NUP98 Is Fused to Topoisomerase (DNA) IIβ 180 kDa (TOP2B) in a Patient with Acute Myeloid Leukemia with a New t(3;11)(p24;p15). Clin. Cancer Res. 2005, 11, 6489–6494. [Google Scholar] [CrossRef] [Green Version]
- Ishikawa, M.; Yagasaki, F.; Okamura, D.; Maeda, T.; Sugahara, Y.; Jinnai, I.; Bessho, M. A novel gene, ANKRD28 on 3p25, is fused with NUP98 on 11p15 in a cryptic 3-way translocation of t(3;5;11)(p25;q35;p15) in an adult patient with myelodysplastic syndrome/acute myelogenous leukemia. Int. J. Hematol. 2007, 86, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Gorello, P.; Brandimarte, L.; La Starza, R.; Pierini, V.; Bury, L.; Rosati, R.; Martelli, M.F.; Vandenberghe, P.; Wlodarska, I.; Mecucci, C. t(3;11)(q12;p15)/NUP98-LOC348801 fusion transcript in acute myeloid leukemia. Haematologica 2008, 93, 1398–1401. [Google Scholar] [CrossRef] [Green Version]
- Liquori, A.; Ibañez, M.; Sargas, C.; Sanz, M.Á.; Barragán, E.; Cervera, J. Acute Promyelocytic Leukemia: A Constellation of Molecular Events around a Single PML-RARA Fusion Gene. Cancers 2020, 12, 624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurnari, C.; Voso, M.T.; Girardi, K.; Mastronuzzi, A.; Strocchio, L. Acute Promyelocytic Leukemia in Children: A Model of Precision Medicine and Chemotherapy-Free Therapy. Int. J. Mol. Sci. 2021, 22, 642. [Google Scholar] [CrossRef]
- De Rooij, J.D.E.; Masetti, R.; van den Heuvel-Eibrink, M.M.; Cayuela, J.-M.; Trka, J.; Reinhardt, D.; Rasche, M.; Sonneveld, E.; Alonzo, T.A.; Fornerod, M.; et al. Recurrent abnormalities can be used for risk group stratification in pediatric AMKL: A retrospective intergroup study. Blood 2016, 127, 3424–3430. [Google Scholar] [CrossRef] [PubMed]
- Marceau-Renaut, A.; Duployez, N.; Ducourneau, B.; Labopin, M.; Petit, A.; Rousseau, A.; Geffroy, S.; Bucci, M.; Cuccuini, W.; Fenneteau, O.; et al. Molecular Profiling Defines Distinct Prognostic Subgroups in Childhood AML: A Report From the French ELAM02 Study Group. HemaSphere 2018, 2, e31. [Google Scholar] [CrossRef]
- McNeer, N.A.; Philip, J.; Geiger, H.; Ries, R.E.; Lavallée, V.-P.; Walsh, M.; Shah, M.; Arora, K.; Emde, A.-K.; Robine, N.; et al. Genetic mechanisms of primary chemotherapy resistance in pediatric acute myeloid leukemia. Leukemia 2019, 33, 1934–1943. [Google Scholar] [CrossRef]
- Shimada, A.; Iijima-Yamashita, Y.; Tawa, A.; Tomizawa, D.; Yamada, M.; Norio, S.; Watanabe, T.; Taga, T.; Iwamoto, S.; Terui, K.; et al. Risk-stratified therapy for children with FLT3-ITD-positive acute myeloid leukemia: Results from the JPLSG AML-05 study. Int. J. Hematol. 2018, 107, 586–595. [Google Scholar] [CrossRef]
- Shiba, N.; Ichikawa, H.; Taki, T.; Park, M.J.; Jo, A.; Mitani, S.; Kobayashi, T.; Shimada, A.; Sotomatsu, M.; Arakawa, H.; et al. NUP98-NSD1 gene fusion and its related gene expression signature are strongly associated with a poor prognosis in pediatric acute myeloid leukemia. Genes Chromosomes Cancer 2013, 52, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Raess, P.W.; Dunlap, J.; Hoyos, C.M.; Li, H.; Li, P.; Swords, R.; Olson, S.B.; Yang, F.; Anekpuritanang, T.; et al. Adult acute myeloid leukemia patients with NUP98 rearrangement have frequent cryptic translocations and unfavorable outcome. Leuk. Lymphoma 2022, 63, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- Niktoreh, N.; Walter, C.; Zimmermann, M.; von Neuhoff, C.; von Neuhoff, N.; Rasche, M.; Waack, K.; Creutzig, U.; Hanenberg, H.; Reinhardt, D. Mutated WT1, FLT3-ITD, and NUP98-NSD1 Fusion in Various Combinations Define a Poor Prognostic Group in Pediatric Acute Myeloid Leukemia. J. Oncol. 2019, 2019, 1609128. [Google Scholar] [CrossRef] [Green Version]
- Ostronoff, F.; Othus, M.; Gerbing, R.B.; Loken, M.R.; Raimondi, S.C.; Hirsch, B.A.; Lange, B.J.; Petersdorf, S.; Radich, J.; Appelbaum, F.R.; et al. NUP98/NSD1 and FLT3/ITD coexpression is more prevalent in younger AML patients and leads to induction failure: A COG and SWOG report. Blood 2014, 124, 2400–2407. [Google Scholar] [CrossRef] [PubMed]
- Franks, T.M.; Hetzer, M.W. The role of NUP98 in transcription regulation in healthy and diseased cells. Trends Cell Biol. 2013, 23, 112–117. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.G.; Cai, L.; Pasillas, M.P.; Kamps, M.P. NUP98-NSD1 links H3K36 methylation to Hox-A gene activation and leukaemogenesis. Nat. Cell Biol. 2007, 9, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Yung, E.; Sekulovic, S.; Argiropoulos, B.; Lai, C.K.; Leung, M.; Berg, T.; Vollett, S.; Chang, V.C.; Wan, A.; Wong, S.; et al. Delineating domains and functions of NUP98 contributing to the leukemogenic activity of NUP98-HOX fusions. Leuk. Res. 2011, 35, 545–550. [Google Scholar] [CrossRef] [Green Version]
- Gurevich, R.M.; Aplan, P.D.; Humphries, R.K. NUP98-topoisomerase I acute myeloid leukemia-associated fusion gene has potent leukemogenic activities independent of an engineered catalytic site mutation. Blood 2004, 104, 1127–1136. [Google Scholar] [CrossRef] [Green Version]
- Hirose, K.; Abramovich, C.; Argiropoulos, B.; Humphries, R.K. Leukemogenic properties of NUP98-PMX1 are linked to NUP98 and homeodomain sequence functions but not to binding properties of PMX1 to serum response factor. Oncogene 2008, 27, 6056–6067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, M.; Zhang, Q.; Liu, P.; Huang, J.; Wang, Y.; Chen, S. Inhibition of the nuclear export of p65 and IQCG in leukemogenesis by NUP98-IQCG. Front. Med. 2016, 10, 410–419. [Google Scholar] [CrossRef]
- Takeda, A.; Sarma, N.J.; Abdul-Nabi, A.M.; Yaseen, N.R. Inhibition of CRM1-mediated nuclear export of transcription factors by leukemogenic NUP98 fusion proteins. J. Biol. Chem. 2010, 285, 16248–16257. [Google Scholar] [CrossRef] [Green Version]
- Pan, M.M.; Zhang, Q.Y.; Wang, Y.Y.; Liu, P.; Ren, R.B.; Huang, J.Y.; Chen, L.T.; Xi, X.D.; Chen, Z.; Chen, S.J. Human NUP98-IQCG fusion protein induces acute myelomonocytic leukemia in mice by dysregulating the Hox/Pbx3 pathway. Leukemia 2016, 30, 1590–1593. [Google Scholar] [CrossRef]
- Boija, A.; Klein, I.A.; Sabari, B.R.; Dall’Agnese, A.; Coffey, E.L.; Zamudio, A.V.; Li, C.H.; Shrinivas, K.; Manteiga, J.C.; Hannett, N.M.; et al. Transcription Factors Activate Genes through the Phase-Separation Capacity of Their Activation Domains. Cell 2018, 175, 1842–1855.e16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, J.H.; Davis, E.S.; Daugird, T.A.; Zhao, S.; Quiroga, I.Y.; Uryu, H.; Li, J.; Storey, A.J.; Tsai, Y.-H.; Keeley, D.P.; et al. Phase separation drives aberrant chromatin looping and cancer development. Nature 2021, 595, 591–595. [Google Scholar] [CrossRef]
- Chandra, B.; Michmerhuizen, N.L.; Shirnekhi, H.K.; Tripathi, S.; Pioso, B.J.; Baggett, D.W.; Mitrea, D.M.; Iacobucci, I.; White, M.R.; Chen, J.; et al. Phase Separation Mediates NUP98 Fusion Oncoprotein Leukemic Transformation. Cancer Discov. 2022, 12, 1152–1169. [Google Scholar] [CrossRef] [PubMed]
- Terlecki-Zaniewicz, S.; Humer, T.; Eder, T.; Schmoellerl, J.; Heyes, E.; Manhart, G.; Kuchynka, N.; Parapatics, K.; Liberante, F.G.; Müller, A.C.; et al. Biomolecular condensation of NUP98 fusion proteins drives leukemogenic gene expression. Nat. Struct. Mol. Biol. 2021, 28, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, H.J.; Christensen, J.; Fong, S.; Hu, Y.L.; Weissman, I.; Sauvageau, G.; Humphries, R.K.; Largman, C. Loss of expression of the Hoxa-9 homeobox gene impairs the proliferation and repopulating ability of hematopoietic stem cells. Blood 2005, 106, 3988–3994. [Google Scholar] [CrossRef] [Green Version]
- Pineault, N.; Helgason, C.D.; Lawrence, H.J.; Humphries, R.K. Differential expression of Hox, Meis1, and Pbx1 genes in primitive cells throughout murine hematopoietic ontogeny. Exp. Hematol. 2002, 30, 49–57. [Google Scholar] [CrossRef]
- Kroon, E.; Krosl, J.; Thorsteinsdottir, U.; Baban, S.; Buchberg, A.M.; Sauvageau, G. Hoxa9 transforms primary bone marrow cells through specific collaboration with Meis1a but not Pbx1b. EMBO J. 1998, 17, 3714–3725. [Google Scholar] [CrossRef]
- Zhang, T.; Cooper, S.; Brockdorff, N. The interplay of histone modifications—Writers that read. EMBO Rep. 2015, 16, 1467–1481. [Google Scholar] [CrossRef]
- Wang, G.G.; Song, J.; Wang, Z.; Dormann, H.L.; Casadio, F.; Li, H.; Luo, J.L.; Patel, D.J.; Allis, C.D. Haematopoietic malignancies caused by dysregulation of a chromatin-binding PHD finger. Nature 2009, 459, 847–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, A.J.; Cheung, P.; Chen, K.; Zee, B.M.; Kioi, M.; Lauring, J.; Xi, Y.; Park, B.H.; Shi, X.; Garcia, B.A.; et al. NSD2 links dimethylation of histone H3 at lysine 36 to oncogenic programming. Mol. Cell 2011, 44, 609–620. [Google Scholar] [CrossRef] [Green Version]
- Gough, S.M.; Lee, F.; Yang, F.; Walker, R.L.; Zhu, Y.J.; Pineda, M.; Onozawa, M.; Chung, Y.J.; Bilke, S.; Wagner, E.K.; et al. NUP98-PHF23 is a chromatin-modifying oncoprotein that causes a wide array of leukemias sensitive to inhibition of PHD histone reader function. Cancer Discov. 2014, 4, 564–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Guo, Y.; Gough, S.M.; Zhang, J.; Vann, K.R.; Li, K.; Cai, L.; Shi, X.; Aplan, P.D.; Wang, G.G.; et al. Mechanistic insights into chromatin targeting by leukemic NUP98-PHF23 fusion. Nat. Commun. 2020, 11, 3339. [Google Scholar] [CrossRef]
- Kroon, E.; Thorsteinsdottir, U.; Mayotte, N.; Nakamura, T.; Sauvageau, G. NUP98-HOXA9 expression in hemopoietic stem cells induces chronic and acute myeloid leukemias in mice. EMBO J. 2001, 20, 350–361. [Google Scholar] [CrossRef] [Green Version]
- Pineault, N.; Abramovich, C.; Humphries, R.K. Transplantable cell lines generated with NUP98-Hox fusion genes undergo leukemic progression by Meis1 independent of its binding to DNA. Leukemia 2005, 19, 636–643. [Google Scholar] [CrossRef] [Green Version]
- Pineault, N.; Buske, C.; Feuring-Buske, M.; Abramovich, C.; Rosten, P.; Hogge, D.E.; Aplan, P.D.; Humphries, R.K. Induction of acute myeloid leukemia in mice by the human leukemia-specific fusion gene NUP98-HOXD13 in concert with Meis1. Blood 2003, 101, 4529–4538. [Google Scholar] [CrossRef]
- Calvo, K.R.; Sykes, D.B.; Pasillas, M.P.; Kamps, M.P. Nup98-HoxA9 immortalizes myeloid progenitors, enforces expression of Hoxa9, Hoxa7 and Meis1, and alters cytokine-specific responses in a manner similar to that induced by retroviral co-expression of Hoxa9 and Meis1. Oncogene 2002, 21, 4247–4256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salsi, V.; Ferrari, S.; Gorello, P.; Fantini, S.; Chiavolelli, F.; Mecucci, C.; Zappavigna, V. NUP98 Fusion Oncoproteins Promote Aneuploidy by Attenuating the Mitotic Spindle Checkpoint. Cancer Res. 2014, 74, 1079–1090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ricke, R.M.; van Ree, J.H.; van Deursen, J.M. Whole chromosome instability and cancer: A complex relationship. Trends Genet. TIG 2008, 24, 457–466. [Google Scholar] [CrossRef] [Green Version]
- Bharadwaj, R.; Yu, H. The spindle checkpoint, aneuploidy, and cancer. Oncogene 2004, 23, 2016–2027. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salsi, V.; Fantini, S.; Zappavigna, V. NUP98 fusion oncoproteins interact with the APC/C(Cdc20) as a pseudosubstrate and prevent mitotic checkpoint complex binding. Cell Cycle 2016, 15, 2275–2287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taketani, T.; Taki, T.; Nakamura, T.; Kobayashi, Y.; Ito, E.; Fukuda, S.; Yamaguchi, S.; Hayashi, Y. High frequencies of simultaneous FLT3-ITD, WT1 and KIT mutations in hematological malignancies with NUP98-fusion genes. Leukemia 2010, 24, 1975–1977. [Google Scholar] [CrossRef] [Green Version]
- Guan, W.; Zhou, L.; Li, Y.; Yang, E.; Liu, Y.; Lv, N.; Fu, L.; Ding, Y.; Wang, N.; Fang, N.; et al. Profiling of somatic mutations and fusion genes in acute myeloid leukemia patients with FLT3-ITD or FLT3-TKD mutation at diagnosis reveals distinct evolutionary patterns. Exp. Hematol. Oncol. 2021, 10, 27. [Google Scholar] [CrossRef]
- Fang, Y.; Han, X.; Shen, L.; Hou, J. NUP98-HOXA9 Bearing Acute Myeloid Leukemia. Blood 2022, 140 (Suppl. S1), 11614. [Google Scholar] [CrossRef]
- Chou, W.C.; Chen, C.Y.; Hou, H.A.; Lin, L.I.; Tang, J.L.; Yao, M.; Tsay, W.; Ko, B.S.; Wu, S.J.; Huang, S.Y.; et al. Acute myeloid leukemia bearing t(7;11)(p15;p15) is a distinct cytogenetic entity with poor outcome and a distinct mutation profile: Comparative analysis of 493 adult patients. Leukemia 2009, 23, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Lavallée, V.P.; Lemieux, S.; Boucher, G.; Gendron, P.; Boivin, I.; Girard, S.; Hébert, J.; Sauvageau, G. Identification of MYC mutations in acute myeloid leukemias with NUP98–NSD1 translocations. Leukemia 2016, 30, 1621–1624. [Google Scholar] [CrossRef]
- Cui, J.; Xie, J.; Qin, L.; Chen, S.; Zhao, Y.; Wu, D. A unique acute myeloid leukemia patient with cryptic NUP98-NSD1 gene and ASXL1 mutation. Leuk. Lymphoma 2016, 57, 196–198. [Google Scholar] [CrossRef]
- Kelly, L.M.; Gilliland, D.G. Genetics of myeloid leukemias. Annu. Rev. Genom. Hum. Genet. 2002, 3, 179–198. [Google Scholar] [CrossRef]
- Takahashi, S. Current findings for recurring mutations in acute myeloid leukemia. J. Hematol. Oncol. 2011, 4, 36. [Google Scholar] [CrossRef] [Green Version]
- Ahuja, H.G.; Popplewell, L.; Tcheurekdjian, L.; Slovak, M.L. NUP98 gene rearrangements and the clonal evolution of chronic myelogenous leukemia. Genes Chromosomes Cancer 2001, 30, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Nakamura, Y.; Nakamura, Y.; Saito, K.; Furusawa, S. Expression of the NUP98/HOXA9 fusion transcript in the blast crisis of Philadelphia chromosome-positive chronic myelogenous leukaemia with t(7;11)(p15;p15). Br. J. Haematol. 2000, 109, 423–426. [Google Scholar] [CrossRef]
- Dash, A.B.; Williams, I.R.; Kutok, J.L.; Tomasson, M.H.; Anastasiadou, E.; Lindahl, K.; Li, S.; Van Etten, R.A.; Borrow, J.; Housman, D.; et al. A murine model of CML blast crisis induced by cooperation between BCR/ABL and NUP98/HOXA9. Proc. Natl. Acad. Sci. USA 2002, 99, 7622–7627. [Google Scholar] [CrossRef]
- Di Giacomo, D.; Pierini, V.; Barba, G.; Ceccarelli, V.; Vecchini, A.; Mecucci, C. Blast crisis Ph+ chronic myeloid leukemia with NUP98/HOXA13 up-regulating MSI2. Mol. Cytogenet. 2014, 7, 42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, M.; Kakihana, K.; Kurosu, T.; Murakami, N.; Miura, O. Clonal evolution with inv(11)(p15q22) and NUP98/DDX10 fusion gene in imatinib-resistant chronic myelogenous leukemia. Cancer Genet. Cytogenet. 2005, 157, 104–108. [Google Scholar] [CrossRef]
- Mohanty, S.; Heuser, M. Mouse Models of Frequently Mutated Genes in Acute Myeloid Leukemia. Cancers 2021, 13, 6192. [Google Scholar] [CrossRef]
- Greenblatt, S.; Li, L.; Slape, C.; Nguyen, B.; Novak, R.; Duffield, A.; Huso, D.; Desiderio, S.; Borowitz, M.J.; Aplan, P.; et al. Knock-in of a FLT3/ITD mutation cooperates with a NUP98-HOXD13 fusion to generate acute myeloid leukemia in a mouse model. Blood 2012, 119, 2883–2894. [Google Scholar] [CrossRef] [Green Version]
- Mohanty, S.; Jyotsana, N.; Sharma, A.; Kloos, A.; Gabdoulline, R.; Othman, B.; Lai, C.K.; Schottmann, R.; Mandhania, M.; Schmoellerl, J.; et al. Targeted Inhibition of the NUP98-NSD1 Fusion Oncogene in Acute Myeloid Leukemia. Cancers 2020, 12, 2766. [Google Scholar] [CrossRef] [PubMed]
- Thanasopoulou, A.; Tzankov, A.; Schwaller, J. Potent co-operation between the NUP98-NSD1 fusion and the FLT3-ITD mutation in acute myeloid leukemia induction. Haematologica 2014, 99, 1465–1471. [Google Scholar] [CrossRef] [Green Version]
- Matsukawa, T.; Yin, M.; Nigam, N.; Negi, V.; Li, L.; Small, D.; Zhu, Y.J.; Walker, R.L.; Meltzer, P.S.; Aplan, P.D. NUP98::Nsd1 and FLT3-ITD collaborate to generate acute myeloid leukemia. Leukemia 2023, 37, 1545–1548. [Google Scholar] [CrossRef]
- Nakamura, T. Retroviral insertional mutagenesis identifies oncogene cooperation. Cancer Sci. 2005, 96, 7–12. [Google Scholar] [CrossRef]
- Slape, C.; Hartung, H.; Lin, Y.W.; Bies, J.; Wolff, L.; Aplan, P.D. Retroviral insertional mutagenesis identifies genes that collaborate with NUP98-HOXD13 during leukemic transformation. Cancer Res. 2007, 67, 5148–5155. [Google Scholar] [CrossRef] [Green Version]
- Slape, C.; Liu, L.Y.; Beachy, S.; Aplan, P.D. Leukemic transformation in mice expressing a NUP98-HOXD13 transgene is accompanied by spontaneous mutations in Nras, Kras, and Cbl. Blood 2008, 112, 2017–2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stahl, M.; Tallman, M.S. Acute promyelocytic leukemia (APL): Remaining challenges towards a cure for all. Leuk. Lymphoma 2019, 60, 3107–3115. [Google Scholar] [CrossRef] [PubMed]
- Pikman, Y.; Stegmaier, K. Targeted therapy for fusion-driven high-risk acute leukemia. Blood 2018, 132, 1241–1247. [Google Scholar] [CrossRef] [Green Version]
- Jyotsana, N.; Sharma, A.; Chaturvedi, A.; Budida, R.; Scherr, M.; Kuchenbauer, F.; Lindner, R.; Noyan, F.; Sühs, K.W.; Stangel, M.; et al. Lipid nanoparticle-mediated siRNA delivery for safe targeting of human CML in vivo. Ann. Hematol. 2019, 98, 1905–1918. [Google Scholar] [CrossRef]
- Jyotsana, N.; Sharma, A.; Chaturvedi, A.; Scherr, M.; Kuchenbauer, F.; Sajti, L.; Barchanski, A.; Lindner, R.; Noyan, F.; Sühs, K.W.; et al. RNA interference efficiently targets human leukemia driven by a fusion oncogene in vivo. Leukemia 2018, 32, 224–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Issa, H.; Swart, L.E.; Rasouli, M.; Ashtiani, M.; Nakjang, S.; Jyotsana, N.; Schuschel, K.; Heuser, M.; Blair, H.; Heidenreich, O. Nanoparticle-mediated targeting of the fusion gene RUNX1/ETO in t(8;21)-positive acute myeloid leukaemia. Leukemia 2023, 37, 820–834. [Google Scholar] [CrossRef]
- Schmoellerl, J.; Barbosa, I.A.M.; Eder, T.; Brandstoetter, T.; Schmidt, L.; Maurer, B.; Troester, S.; Pham, H.T.T.; Sagarajit, M.; Ebner, J.; et al. CDK6 is an essential direct target of NUP98 fusion proteins in acute myeloid leukemia. Blood 2020, 136, 387–400. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, L.; Hei, R.; Li, X.; Cai, H.; Wu, X.; Zheng, Q.; Cai, C. CDK inhibitors in cancer therapy, an overview of recent development. Am. J. Cancer Res. 2021, 11, 1913–1935. [Google Scholar]
- Krivtsov, A.V.; Evans, K.; Gadrey, J.Y.; Eschle, B.K.; Hatton, C.; Uckelmann, H.J.; Ross, K.N.; Perner, F.; Olsen, S.N.; Pritchard, T.; et al. A Menin-MLL Inhibitor Induces Specific Chromatin Changes and Eradicates Disease in Models of MLL-Rearranged Leukemia. Cancer Cell 2019, 36, 660–673.e11. [Google Scholar] [CrossRef] [PubMed]
- Uckelmann, H.J.; Kim, S.M.; Wong, E.M.; Hatton, C.; Giovinazzo, H.; Gadrey, J.Y.; Krivtsov, A.V.; Rücker, F.G.; Döhner, K.; McGeehan, G.M.; et al. Therapeutic targeting of preleukemia cells in a mouse model of NPM1 mutant acute myeloid leukemia. Science 2020, 367, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Pommert, L.; Tarlock, K. The evolution of targeted therapy in pediatric AML: Gemtuzumab ozogamicin, FLT3/IDH/BCL2 inhibitors, and other therapies. Hematology 2022, 2022, 603–610. [Google Scholar] [CrossRef]
- Heikamp, E.B.; Henrich, J.A.; Perner, F.; Wong, E.M.; Hatton, C.; Wen, Y.; Barwe, S.P.; Gopalakrishnapillai, A.; Xu, H.; Uckelmann, H.J.; et al. The menin-MLL1 interaction is a molecular dependency in NUP98-rearranged AML. Blood 2022, 139, 894–906. [Google Scholar] [CrossRef]
- Issa, G.C.; Ravandi, F.; DiNardo, C.D.; Jabbour, E.; Kantarjian, H.M.; Andreeff, M. Therapeutic implications of menin inhibition in acute leukemias. Leukemia 2021, 35, 2482–2495. [Google Scholar] [CrossRef]
- Issa, G.C.; Aldoss, I.; DiPersio, J.; Cuglievan, B.; Stone, R.; Arellano, M.; Thirman, M.J.; Patel, M.R.; Dickens, D.S.; Shenoy, S.; et al. The menin inhibitor revumenib in KMT2A-rearranged or NPM1-mutant leukaemia. Nature 2023, 615, 920–924. [Google Scholar] [CrossRef]
- Perner, F.; Stein, E.M.; Wenge, D.V.; Singh, S.; Kim, J.; Apazidis, A.; Rahnamoun, H.; Anand, D.; Marinaccio, C.; Hatton, C.; et al. MEN1 mutations mediate clinical resistance to menin inhibition. Nature 2023, 615, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Oka, M.; Mura, S.; Yamada, K.; Sangel, P.; Hirata, S.; Maehara, K.; Kawakami, K.; Tachibana, T.; Ohkawa, Y.; Kimura, H.; et al. Chromatin-prebound Crm1 recruits Nup98-HoxA9 fusion to induce aberrant expression of Hox cluster genes. eLife 2016, 5, e09540. [Google Scholar] [CrossRef]
- Ren, Z.; Kim, A.; Huang, Y.T.; Pi, W.C.; Gong, W.; Yu, X.; Qi, J.; Jin, J.; Cai, L.; Roeder, R.G.; et al. A PRC2-Kdm5b axis sustains tumorigenicity of acute myeloid leukemia. Proc. Natl. Acad. Sci. USA 2022, 119, e2122940119. [Google Scholar] [CrossRef]
- Gan, L.; Yang, Y.; Li, Q.; Feng, Y.; Liu, T.; Guo, W. Epigenetic regulation of cancer progression by EZH2: From biological insights to therapeutic potential. Biomark. Res. 2018, 6, 10. [Google Scholar] [CrossRef]
- Straining, R.; Eighmy, W. Tazemetostat: EZH2 Inhibitor. J. Adv. Pract. Oncol. 2022, 13, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Kivioja, J.L.; Thanasopoulou, A.; Kumar, A.; Kontro, M.; Yadav, B.; Majumder, M.M.; Javarappa, K.K.; Eldfors, S.; Schwaller, J.; Porkka, K.; et al. Dasatinib and navitoclax act synergistically to target NUP98-NSD1(+)/FLT3-ITD(+) acute myeloid leukemia. Leukemia 2019, 33, 1360–1372. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.; Doki, N.; Aoki, J.; Mori, J.; Machida, S.; Masuko, M.; Uchida, N.; Najima, Y.; Fukuda, T.; Kanamori, H.; et al. Outcomes after allogeneic hematopoietic stem cell transplantation in patients with acute myeloid leukemia harboring t(7;11)(p15;p15). Haematologica 2018, 103, e69–e72. [Google Scholar] [CrossRef] [Green Version]
- Békés, M.; Langley, D.R.; Crews, C.M. PROTAC targeted protein degraders: The past is prologue. Nat. Rev. Drug Discov. 2022, 21, 181–200. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, T.; Song, X.; Liu, B.; Wei, J. Gene fusion neoantigens: Emerging targets for cancer immunotherapy. Cancer Lett. 2021, 506, 45–54. [Google Scholar] [CrossRef]
- Yang, W.; Lee, K.W.; Srivastava, R.M.; Kuo, F.; Krishna, C.; Chowell, D.; Makarov, V.; Hoen, D.; Dalin, M.G.; Wexler, L.; et al. Immunogenic neoantigens derived from gene fusions stimulate T cell responses. Nat. Med. 2019, 25, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K.; Imamura, T.; Tanaka, S.; Urata, T.; Yoshida, H.; Shiba, N.; Iehara, T. The Nup98::Nsd1 fusion gene induces CD123 expression in 32D cells. Int. J. Hematol. 2023, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Thiollier, C.; Lopez, C.K.; Gerby, B.; Ignacimouttou, C.; Poglio, S.; Duffourd, Y.; Guégan, J.; Rivera-Munoz, P.; Bluteau, O.; Mabialah, V.; et al. Characterization of novel genomic alterations and therapeutic approaches using acute megakaryoblastic leukemia xenograft models. J. Exp. Med. 2012, 209, 2017–2031. [Google Scholar] [CrossRef]
- Mohanty, S.; Jyotsana, N.; Sharma, A.; Othman, B.; Kloos, A.; Mandhania, M.; Schottmann, R.; Ramsay, E.; Vornlocher, H.-P.; Ganser, A.; et al. Targeted Inhibition of the NUP98-NSD1 Fusion Oncogene in AML. Blood 2019, 134, 2545. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohanty, S. NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications. Onco 2023, 3, 147-164. https://doi.org/10.3390/onco3030011
Mohanty S. NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications. Onco. 2023; 3(3):147-164. https://doi.org/10.3390/onco3030011
Chicago/Turabian StyleMohanty, Sagarajit. 2023. "NUP98 Rearrangements in AML: Molecular Mechanisms and Clinical Implications" Onco 3, no. 3: 147-164. https://doi.org/10.3390/onco3030011






