Precision Medicine Revolutionizing Esophageal Cancer Treatment: Surmounting Hurdles and Enhancing Therapeutic Efficacy through Targeted Drug Therapies
Abstract
:Simple Summary
Abstract
1. Introduction
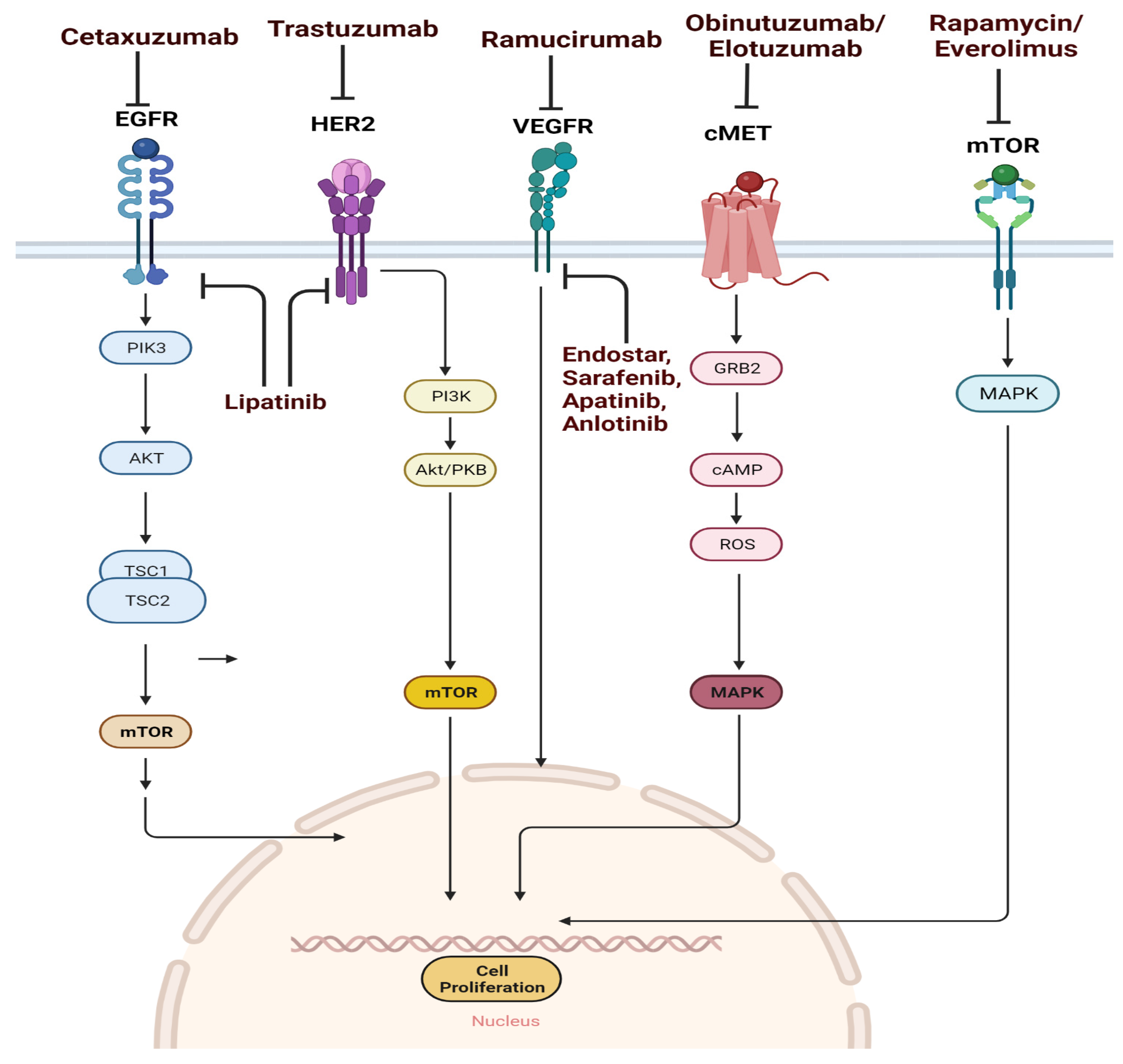
2. Drugs Targeting the Key Signaling Pathway
3. Drugs Targeting the EGFR Pathway
4. Anti-EGFR Monoclonal Antibodies
5. Cetuximab
6. Nimotuzumab
7. Anti-EGFR Tyrosine Kinase Inhibitors
8. Gefitinib
9. Icotinib
10. Drugs Targeting the HER2 Pathway
11. Trastuzumab
12. Lapatinib
13. Drugs Targeting the VEGF/VEGFR Pathway
14. Ramucirumab
15. HGF/c-MET Pathway
16. Rilotumumab and Obinutuzumab
17. mTOR Pathway
18. Factors Associated with Targeted Therapy
18.1. Cancer Heterogeneity
18.2. Drug Resistance
19. Cellular Components Involved in Drug Resistance
19.1. Cancer Stem Cells
19.2. Cancer-Associated Fibroblasts
19.3. Inflammatory Immune Cells
20. Non-Cellular Components
20.1. Cytokines
20.2. Hypoxia
20.3. Growth Factors
20.4. Biomarkers
21. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Global Burden of Disease Cancer Collaboration. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017, 3, 524–548. [Google Scholar] [CrossRef] [Green Version]
- Liu, K.; Zhao, T.; Wang, J.; Chen, Y.; Zhang, R.; Lan, X.; Que, J. Etiology, cancer stem cells and potential diagnostic biomarkers for esophageal cancer. Cancer Lett. 2019, 458, 21–28. [Google Scholar] [CrossRef]
- Yang, Y.-M.; Hong, P.; Xu, W.W.; He, Q.-Y.; Li, B. Advances in targeted therapy for esophageal cancer. Signal Transduct. Target. Ther. 2020, 5, 229. [Google Scholar] [CrossRef] [PubMed]
- Edgren, G.; Adami, H.-O.; Weiderpass, E.; Nyrén, O. A global assessment of the oesophageal adenocarcinoma epidemic. Gut 2013, 62, 1406. [Google Scholar] [CrossRef]
- Xie, S.-H.; Lagergren, J. A global assessment of the male predominance in esophageal adenocarcinoma. Oncotarget 2016, 7, 38876–38883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagergren, J.; Lagergren, P. Oesophageal cancer. BMJ 2010, 341, c6280. [Google Scholar] [CrossRef] [PubMed]
- Smyth, E.C.; Lagergren, J.; Fitzgerald, R.C.; Lordick, F.; Shah, M.A.; Lagergren, P.; Cunningham, D. Oesophageal cancer. Nat. Rev. Dis. Prim. 2017, 3, 17048. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Cao, H.; Xu, Y.; Li, D.; Zhang, H.; Xu, M.; Luo, Y.; Wu, J. Causes, Risk Factors and Outcomes of Patients Readmitted to the Intensive Care Unit After Esophageal Cancer Surgery: A Retrospective Cohort Study. World J. Surg. 2021, 45, 2167–2175. [Google Scholar] [CrossRef] [PubMed]
- Lagergren, J.; Smyth, E.; Cunningham, D.; Lagergren, P. Oesophageal cancer. Lancet 2017, 390, 2383–2396. [Google Scholar] [CrossRef] [Green Version]
- Secrier, M.; Li, X.; de Silva, N.; Eldridge, M.D.; Contino, G.; Bornschein, J.; MacRae, S.; Grehan, N.; O’Donovan, M.; Miremadi, A.; et al. Mutational signatures in esophageal adenocarcinoma define etiologically distinct subgroups with therapeutic relevance. Nat. Genet. 2016, 48, 1131–1141. [Google Scholar] [CrossRef] [Green Version]
- Rahmani, H.; Sarabi Asiabar, A.; Niakan, S.; Hashemi, S.Y.; Faramarzi, A.; Manuchehri, S.; Rajabi Vasokolaei, G. Burden of esophageal cancer in Iran during 1995-2015: Review of findings from the Global Burden of Disease studies. Med. J. Islam. Repub. Iran 2018, 32, 55. [Google Scholar] [CrossRef] [Green Version]
- Schweigert, M.; Dubecz, A.; Stein, H.J. Oesophageal cancer--an overview. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 230–244. [Google Scholar] [CrossRef] [PubMed]
- Fatehi Hassanabad, A.; Chehade, R.; Breadner, D.; Raphael, J. Esophageal carcinoma: Towards targeted therapies. Cell. Oncol. 2020, 43, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Hu, S.; Zhang, W.; Zhang, D.; Zhang, Y.; Yu, D.; Peng, J.; Xu, J.; Wei, Y. Better Prognosis and Survival in Esophageal Cancer Survivors After Comorbid Second Primary Malignancies: A SEER Database-Based Study. Front Surg. 2022, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- van Hagen, P.; Hulshof, M.C.; van Lanschot, J.J.; Steyerberg, E.W.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.; Richel, D.J.; Nieuwenhuijzen, G.A.; Hospers, G.A.; Bonenkamp, J.J.; et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N. Engl. J. Med. 2012, 366, 2074–2084. [Google Scholar] [CrossRef] [Green Version]
- Tong, Y.; Yang, L.; Yu, C.; Zhu, W.; Zhou, X.; Xiong, Y.; Wang, W.; Ji, F.; He, D.; Cao, X. Tumor-Secreted Exosomal lncRNA POU3F3 Promotes Cisplatin Resistance in ESCC by Inducing Fibroblast Differentiation into CAFs. Mol. Ther. Oncolytics 2020, 18, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.; Zeng, X.; Zhang, C.; Yang, Y.; Xiao, X.; Luan, S.; Zhang, Y.; Yuan, Y. Mechanisms of Pharmaceutical Therapy and Drug Resistance in Esophageal Cancer. Front. Cell Dev. Biol. 2021, 9, 612451. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Ding, Z.-Y. PD-1 Inhibitors in the Advanced Esophageal Cancer. Front. Pharmacol. 2019, 10, 1418. [Google Scholar] [CrossRef]
- Aref, D.; Croul, S. Medulloblastoma: Recurrence and metastasis. CNS Oncol. 2013, 2, 377–385. [Google Scholar] [CrossRef]
- Kang, X.; Chen, K.; Li, Y.; Li, J.; D’Amico, T.A.; Chen, X. Personalized targeted therapy for esophageal squamous cell carcinoma. World J. Gastroenterol. 2015, 21, 7648–7658. [Google Scholar] [CrossRef]
- Lyons, T.G.; Ku, G.Y. Systemic therapy for esophagogastric cancer: Targeted therapies. Chin. Clin. Oncol. 2017, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Chong, C.R.; Jänne, P.A. The quest to overcome resistance to EGFR-targeted therapies in cancer. Nat. Med. 2013, 19, 1389–1400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ciardiello, F.; Tortora, G. EGFR Antagonists in Cancer Treatment. N. Engl. J. Med. 2008, 358, 1160–1174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinelli, E.; De Palma, R.; Orditura, M.; De Vita, F.; Ciardiello, F. Anti-epidermal growth factor receptor monoclonal antibodies in cancer therapy. Clin. Exp. Immunol. 2009, 158, 1–9. [Google Scholar] [CrossRef]
- Verma, H.K.; Kampalli, P.K.; Lakkakula, S.; Chalikonda, G.; Bhaskar, L.V.K.S.; Pattnaik, S. A Retrospective Look at Anti-EGFR Agents in Pancreatic Cancer Therapy. Curr. Drug Metab. 2019, 20, 958–966. [Google Scholar] [CrossRef]
- Maron, S.B.; Alpert, L.; Kwak, H.A.; Lomnicki, S.; Chase, L.; Xu, D.; O’Day, E.; Nagy, R.J.; Lanman, R.B.; Cecchi, F.; et al. Targeted Therapies for Targeted Populations: Anti-EGFR Treatment for EGFR-Amplified Gastroesophageal Adenocarcinoma. Cancer Discov. 2018, 8, 696–713. [Google Scholar] [CrossRef] [Green Version]
- Baselga, J.; Pfister, D.; Cooper, M.R.; Cohen, R.; Burtness, B.; Bos, M.; D'Andrea, G.; Seidman, A.; Norton, L.; Gunnett, K.; et al. Phase I studies of anti-epidermal growth factor receptor chimeric antibody C225 alone and in combination with cisplatin. J. Clin. Oncol. 2000, 18, 904–914. [Google Scholar] [CrossRef]
- Lv, Z.-C.; Ning, J.-Y.; Chen, H.-B. Efficacy and toxicity of adding cetuximab to chemotherapy in the treatment of metastatic colorectal cancer: A meta-analysis from 12 randomized controlled trials. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2014, 35, 11741–11750. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-H.; Ma, X.-W.; Zhang, J.; Li, X.; Lai, N.-L.; Zhang, S.-X. Cetuximab for esophageal cancer: An updated meta-analysis of randomized controlled trials. BMC Cancer 2018, 18, 1170. [Google Scholar] [CrossRef]
- Sotelo, M.J.; García-Paredes, B.; Aguado, C.; Sastre, J.; Díaz-Rubio, E. Role of cetuximab in first-line treatment of metastatic colorectal cancer. World J. Gastroenterol. 2014, 20, 4208–4219. [Google Scholar] [CrossRef]
- Cunningham, D.; Humblet, Y.; Siena, S.; Khayat, D.; Bleiberg, H.; Santoro, A.; Bets, D.; Mueser, M.; Harstrick, A.; Verslype, C.; et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N. Engl. J. Med. 2004, 351, 337–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, S.; Ramos-Suzarte, M.; Bai, X.; Xu, B. Treatment outcome of nimotuzumab plus chemotherapy in advanced cancer patients: A single institute experience. Oncotarget. 2016, 7, 33391–33407. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Lu, N.; Pan, Y.; Xu, J. Nimotuzumab Combined with Chemotherapy is a Promising Treatment for Locally Advanced and Metastatic Esophageal Cancer. Med. Sci. Monit. 2017, 23, 412–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subramanian, S.; Sridharan, N.; Balasundaram, V.; Chaudhari, S. Effectiveness and tolerability of nimotuzumab in unresectable, locally advanced/metastatic esophageal cancer: Indian hospital-based retrospective evidence. South Asian J. Cancer 2019, 8, 112–115. [Google Scholar] [CrossRef]
- Zhao, K.-l.; Hu, X.-c.; Wu, X.-h.; Fu, X.-l.; Fan, M.; Jiang, G.-L. A phase I dose escalation study of Nimotuzumab in combination with concurrent chemoradiation for patients with locally advanced squamous cell carcinoma of esophagus. Investig. New Drugs 2012, 30, 1585–1590. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.-X.; Zhang, R.-R.; Chen, W.-H.; Dong, H.-M.; Wang, G.; Li, X.-K.; Wang, W. Clinical efficacy and safety of nimotuzumab plus chemotherapy in patients with advanced colorectal cancer: A retrospective analysis. J. Int. Med. Res. 2020, 48, 300060519895858. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Guan, H.; Jiang, L.; Li, X.; Xing, L.; Sun, X. Nimotuzumab, an EGFR-targeted antibody, promotes radiosensitivity of recurrent esophageal squamous cell carcinoma. Int. J. Oncol. 2020, 56, 945–956. [Google Scholar] [CrossRef]
- Dragovich, T.; Campen, C. Anti-EGFR-Targeted Therapy for Esophageal and Gastric Cancers: An Evolving Concept. J. Oncol. 2009, 2009, 804108. [Google Scholar] [CrossRef]
- Cui, G.; Cui, M.; Li, Y.; Liang, Y.; Li, W.; Guo, H.; Zhao, S. Galectin-3 knockdown increases gefitinib sensitivity to the inhibition of EGFR endocytosis in gefitinib-insensitive esophageal squamous cancer cells. Med. Oncol. 2015, 32, 124. [Google Scholar] [CrossRef]
- Petty, R.D.; Dahle-Smith, A.; Stevenson, D.A.J.; Osborne, A.; Massie, D.; Clark, C.; Murray, G.I.; Dutton, S.J.; Roberts, C.; Chong, I.Y.; et al. Gefitinib and EGFR Gene Copy Number Aberrations in Esophageal Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2017, 35, 2279–2287. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Hsu, D.K.; Chen, H.-Y.; Yang, R.-Y.; Carraway, K.L.; Isseroff, R.R.; Liu, F.-T. Galectin-3 regulates intracellular trafficking of EGFR through Alix and promotes keratinocyte migration. J. Investig. Derm. 2012, 132, 2828–2837. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Xie, Z.; Shi, Y.; Zhang, M.; Pan, J.; Li, Y.; Lu, H. Gefitinib single drug in treatment of advanced esophageal cancer. J. Cancer Res. Ther. 2016, 12, C295–C297. [Google Scholar] [CrossRef]
- Drenckhan, A.; Grob, T.; Dupree, A.; Dohrmann, T.; Mann, O.; Izbicki, J.R.; Gros, S.J. Esophageal carcinoma cell line with high EGFR polysomy is responsive to gefitinib. Langenbeck’s Arch. Surg. 2014, 399, 879–888. [Google Scholar] [CrossRef]
- Huang, J.; Fan, Q.; Lu, P.; Ying, J.; Ma, C.; Liu, W.; Liu, Y.; Tan, F.; Sun, Y. Icotinib in Patients with Pretreated Advanced Esophageal Squamous Cell Carcinoma with EGFR Overexpression or EGFR Gene Amplification: A Single-Arm, Multicenter Phase 2 Study. J. Thorac. Oncol. 2016, 11, 910–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Jiang, W.; Tian, D.; Guo, Q.; Shen, Z. Icotinib inhibits the proliferation of hepatocellular carcinoma cells in vitro and in vivo dependently on EGFR activation and PDL1 expression. OncoTargets Ther. 2018, 11, 8227–8237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Niu, H.; Fan, Q.; Lu, P.; Ma, C.; Liu, W.; Liu, Y.; Li, W.; Hu, S.; Ling, Y.; et al. Predictive value of EGFR overexpression and gene amplification on icotinib efficacy in patients with advanced esophageal squamous cell carcinoma. Oncotarget 2016, 7, 24744–24751. [Google Scholar] [CrossRef] [Green Version]
- Luo, H.; Jiang, W.; Ma, L.; Chen, P.; Fang, M.; Ding, L.; Hua, Y.; Du, D.; Jing, Z.; Xie, R.; et al. Icotinib With Concurrent Radiotherapy vs Radiotherapy Alone in Older Adults With Unresectable Esophageal Squamous Cell Carcinoma: A Phase II Randomized Clinical Trial. JAMA Netw. Open 2020, 3, 19440. [Google Scholar] [CrossRef] [PubMed]
- Budi, H.S.; Ahmad, F.N.; Achmad, H.; Ansari, M.J.; Mikhailova, M.V.; Suksatan, W.; Chupradit, S.; Shomali, N.; Marofi, F. Human epidermal growth factor receptor 2 (HER2)-specific chimeric antigen receptor (CAR) for tumor immunotherapy; recent progress. Stem Cell Res. Ther. 2022, 13, 40. [Google Scholar] [CrossRef] [PubMed]
- Won, E.; Janjigian, Y.J.; Ilson, D.H. HER2 directed therapy for gastric/esophageal cancers. Curr. Treat. Options Oncol. 2014, 15, 395–404. [Google Scholar] [CrossRef]
- Swain, S.M.; Shastry, M.; Hamilton, E. Targeting HER2-positive breast cancer: Advances and future directions. Nat. Rev. Drug Discov. 2023, 22, 101–126. [Google Scholar] [CrossRef]
- Gerson, J.N.; Skariah, S.; Denlinger, C.S.; Astsaturov, I. Perspectives of HER2-targeting in gastric and esophageal cancer. Expert Opin. Investig. Drugs 2017, 26, 531–540. [Google Scholar] [CrossRef] [Green Version]
- Swain, S.M.; Baselga, J.; Kim, S.B.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.M.; Schneeweiss, A.; Heeson, S.; et al. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N. Engl. J. Med. 2015, 372, 724–734. [Google Scholar] [CrossRef] [Green Version]
- Bang, Y.J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, J.; Hoff, P.M.; Shen, L.; Ohtsu, A.; Shah, M.A.; Cheng, K.; Song, C.; Wu, H.; Eng-Wong, J.; Kim, K.; et al. Pertuzumab plus trastuzumab and chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer (JACOB): Final analysis of a double-blind, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2018, 19, 1372–1384. [Google Scholar] [CrossRef] [PubMed]
- Mimura, K.; Kono, K.; Maruyama, T.; Watanabe, M.; Izawa, S.; Shiba, S.; Mizukami, Y.; Kawaguchi, Y.; Inoue, M.; Kono, T.; et al. Lapatinib inhibits receptor phosphorylation and cell growth and enhances antibody-dependent cellular cytotoxicity of EGFR- and HER2-overexpressing esophageal cancer cell lines. Int. J. Cancer 2011, 129, 2408–2416. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, K.; Mimura, K.; Izawa, S.; Inoue, A.; Shiba, S.; Maruyama, T.; Watanabe, M.; Kawaguchi, Y.; Inoue, M.; Fujii, H.; et al. Lapatinib acts on gastric cancer through both antiproliferative function and augmentation of trastuzumab-mediated antibody-dependent cellular cytotoxicity. Gastric Cancer Off. J. Int. Gastric Cancer Assoc. Jpn. Gastric Cancer Assoc. 2012, 16, 571–580. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.-F.; Li, S.-S.; Zhu, X.-F.; Dou, Q.-H.; Liu, D. Lapatinib in combination with paclitaxel plays synergistic antitumor effects on esophageal squamous cancer. Cancer Chemother. Pharmacol. 2018, 82, 383–394. [Google Scholar] [CrossRef]
- Hassan, M.S.; Williams, F.; Awasthi, N.; Schwarz, M.A.; Schwarz, R.E.; Li, J.; von Holzen, U. Combination effect of lapatinib with foretinib in HER2 and MET co-activated experimental esophageal adenocarcinoma. Sci. Rep. 2019, 9, 019–54129. [Google Scholar] [CrossRef] [Green Version]
- Chai, J.; Jones, M.K.; Tarnawski, A.S. Serum response factor is a critical requirement for VEGF signaling in endothelial cells and VEGF-induced angiogenesis. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2004, 18, 1264–1266. [Google Scholar] [CrossRef] [Green Version]
- Meng, L.; Fuhao, Z.; Xiaoming, Z.; Yuxiu, Z.; Zhaojun, D.; Bingcheng, L.; Meiling, X.; Shuyan, S. Survivin is critically involved in VEGFR2 signaling-mediated esophageal cancer cell survival. Biomed. Pharmacother. 2018, 107, 139–145. [Google Scholar] [CrossRef]
- Whittaker, S.; Marais, R.; Zhu, A.X. The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene 2010, 29, 4989–5005. [Google Scholar] [CrossRef]
- Cheung, A.; Xu, W.; Li, B. The potential of targeted antiangiogenesis therapies in the treatment of esophageal cancer. Gastrointest. Cancer Targets Ther. 2015, 2015, 79–88. [Google Scholar] [CrossRef] [Green Version]
- Khan, U.; Shah, M.A. Ramucirumab for the treatment of gastric or gastro-esophageal junction cancer. Expert Opin. Biol. Ther. 2019, 19, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Spratlin, J.L.; Cohen, R.B.; Eadens, M.; Gore, L.; Camidge, D.R.; Diab, S.; Leong, S.; O’Bryant, C.; Chow, L.Q.; Serkova, N.J.; et al. Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 780–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilke, H.; Muro, K.; Van Cutsem, E.; Oh, S.C.; Bodoky, G.; Shimada, Y.; Hironaka, S.; Sugimoto, N.; Lipatov, O.; Kim, T.Y.; et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): A double-blind, randomised phase 3 trial. Lancet Oncol. 2014, 15, 1224–1235. [Google Scholar] [CrossRef]
- Konstorum, A.; Lowengrub, J.S. Activation of the HGF/c-Met axis in the tumor microenvironment: A multispecies model. J. Biol. 2018, 439, 86–99. [Google Scholar] [CrossRef]
- Boromand, N.; Hasanzadeh, M.; ShahidSales, S.; Farazestanian, M.; Gharib, M.; Fiuji, H.; Behboodi, N.; Ghobadi, N.; Hassanian, S.M.; Ferns, G.A.; et al. Clinical and prognostic value of the C-Met/HGF signaling pathway in cervical cancer. J. Cell. Physiol. 2018, 233, 4490–4496. [Google Scholar] [CrossRef]
- Granito, A.; Guidetti, E.; Gramantieri, L. c-MET receptor tyrosine kinase as a molecular target in advanced hepatocellular carcinoma. J. Hepatocell. Carcinoma 2015, 2, 29–38. [Google Scholar] [CrossRef] [Green Version]
- Arnold, L.; Enders, J.; Thomas, S.M. Activated HGF-c-Met Axis in Head and Neck Cancer. Cancers 2017, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Stanley, A.; Ashrafi, G.H.; Seddon, A.M.; Modjtahedi, H. Synergistic effects of various Her inhibitors in combination with IGF-1R, C-MET and Src targeting agents in breast cancer cell lines. Sci. Rep. 2017, 7, 3964. [Google Scholar] [CrossRef]
- Lam, B.Q.; Dai, L.; Qin, Z. The role of HGF/c-MET signaling pathway in lymphoma. J. Hematol. Oncol. 2016, 9, 135. [Google Scholar] [CrossRef] [Green Version]
- Krause, D.S.; Van Etten, R.A. Tyrosine kinases as targets for cancer therapy. N. Engl. J. Med. 2005, 353, 172–187. [Google Scholar] [CrossRef] [Green Version]
- Van Der Steen, N.; Garajova, I.; Rolfo, C.; Cavazzoni, A.; Giovannetti, E. Chapter 2—Targeting the Hepatocyte Growth Factor Receptor to Overcome Resistance to Targeted Therapies. In Targeting Cell Survival Pathways to Enhance Response to Chemotherapy; Johnson, D.E., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 3, pp. 25–60. [Google Scholar]
- Shah, M.; Bang, Y.-J.; Lordick, F.; Tabernero, J.; Chen, M.; Hack, S.; Phan, S.C.; Shames, D.S.; Cunningham, D. METGastric: A phase III study of onartuzumab plus mFOLFOX6 in patients with metastatic HER2-negative (HER2-) and MET-positive (MET+) adenocarcinoma of the stomach or gastroesophageal junction (GEC). J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2015, 33, 4012. [Google Scholar] [CrossRef]
- Catenacci, D.V.T.; Tebbutt, N.C.; Davidenko, I.; Murad, A.M.; Al-Batran, S.E.; Ilson, D.H.; Tjulandin, S.; Gotovkin, E.; Karaszewska, B.; Bondarenko, I.; et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2017, 18, 1467–1482. [Google Scholar] [CrossRef]
- Pópulo, H.; Lopes, J.M.; Soares, P. The mTOR signalling pathway in human cancer. Int. J. Mol. Sci. 2012, 13, 1886–1918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirashima, K.; Baba, Y.; Watanabe, M.; Karashima, R.-I.; Sato, N.; Imamura, Y.; Hiyoshi, Y.; Nagai, Y.; Hayashi, N.; Iyama, K.-i.; et al. Phosphorylated mTOR Expression is Associated with Poor Prognosis for Patients with Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2010, 17, 2486–2493. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef] [Green Version]
- Hasskarl, J. Everolimus. Recent Results Cancer Res. 2018, 211, 101–123. [Google Scholar] [CrossRef]
- Rades, D.; Bartscht, T.; Hunold, P.; Schmidberger, H.; König, L.; Debus, J.; Belka, C.; Homann, N.; Spillner, P.; Petersen, C.; et al. Radiochemotherapy with or without cetuximab for unresectable esophageal cancer: Final results of a randomized phase 2 trial (LEOPARD-2). Strahlenther. Onkol. Organ Dtsch. Rontgenges. 2020, 196, 795–804. [Google Scholar] [CrossRef]
- Yang, X.; Zhai, Y.; Bi, N.; Zhang, T.; Deng, L.; Wang, W.; Wang, X.; Chen, D.; Zhou, Z.; Wang, L.; et al. Radiotherapy combined with nimotuzumab for elderly esophageal cancer patients: A phase II clinical trial. Chin. J. Cancer Res. 2021, 33, 53–60. [Google Scholar] [CrossRef]
- Qi, S.; Mao, Y.; Jiang, M. A phase I study evaluating combined nimotuzumab and neoadjuvant chemoradiotherapy followed by surgery in locally advanced esophageal cancer. Cancer Chemother. Pharmacol. 2019, 84, 1115–1123. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zheng, Y.; Sun, X.; Yu, X.; Gu, J.; Wu, W.; Zhang, G.; Hu, J.; Sun, W.; Mao, W. Concurrent radiotherapy with gefitinib in elderly patients with esophageal squamous cell carcinoma: Preliminary results of a phase II study. Oncotarget 2015, 6, 38429–38439. [Google Scholar] [CrossRef] [Green Version]
- Guan, Y.-S.; He, Q.; Li, M. Icotinib: Activity and clinical application in Chinese patients with lung cancer. Expert Opin Pharm. 2014, 15, 717–728. [Google Scholar] [CrossRef]
- Wang, J.; Peng, J.; Luo, H.; Song, Y. Development and internal validation of a nomogram for predicting survival of nonoperative EGFR-positive locally advanced elderly esophageal cancers. Front. Oncol. 2023, 13, 1097907. [Google Scholar] [CrossRef] [PubMed]
- Soularue, É.; Cohen, R.; Tournigand, C.; Zaanan, A.; Louvet, C.; Bachet, J.-B.; Hentic, O.; Chibaudel, B.; Gramont, A.; André, T. Efficacy and safety of trastuzumab in combination with oxaliplatin and fluorouracil-based chemotherapy for patients with HER2-positive metastatic gastric and gastro-oesophageal junction adenocarcinoma patients: A retrospective study. Bull. Cancer 2015, 102, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Minchev, V.T.; Research, C. Toxic and Adverse Effects of Chemotherapy with 5-Fluoropyrimidine Drugs. Could Dihydropyrimidine Dehydrogenase Enzyme Screening Serve as a Prerequisite to Successful Chemotherapy? J. Biomed. Clin. Res. 2020, 13, 87–99. [Google Scholar] [CrossRef]
- Nolting, M.; Schneider-Merck, T.; Trepel, M. Lapatinib. Small Mol. Oncol. 2014, 201, 125–143. [Google Scholar] [CrossRef]
- Smyth, E.C.; Rowley, S.; Cafferty, F.H.; Allum, W.; Grabsch, H.I.; Stenning, S.; Wotherspoon, A.; Alderson, D.; Crosby, T.; Mansoor, W.; et al. Safety and Efficacy of the Addition of Lapatinib to Perioperative Chemotherapy for Resectable HER2-Positive Gastroesophageal Adenocarcinoma: A Randomized Phase 2 Clinical Trial. JAMA Oncol. 2019, 5, 1181–1187. [Google Scholar] [CrossRef] [Green Version]
- Kawai, S.; Fukuda, N.; Yamamoto, S.; Mitani, S.; Omae, K.; Wakatsuki, T.; Kato, K.; Kadowaki, S.; Takahari, D.; Boku, N.; et al. Retrospective observational study of salvage line ramucirumab monotherapy for patients with advanced gastric cancer. BMC Cancer 2020, 20, 338. [Google Scholar] [CrossRef] [Green Version]
- Han, H.S.; Kim, B.J.; Jee, H.J.; Ryu, M.H.; Park, S.H.; Rha, S.Y.; Kim, J.G.; Bae, W.K.; Lee, K.W.; Oh, D.Y.; et al. Ramucirumab plus paclitaxel as second-line treatment in patients with advanced gastric or gastroesophageal junction adenocarcinoma: A nationwide real-world outcomes in Korea study (KCSG-ST19-16). Ther. Adv. Med. Oncol. 2021, 13, 17588359211042812. [Google Scholar] [CrossRef]
- ElHalawani, H.; Abdel-Rahman, O. Critical evaluation of ramucirumab in the treatment of advanced gastric and gastroesophageal cancers. Ther. Clin. Risk Manag. 2015, 11, 1123–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ling, Y.; Yang, Y.; Lu, n.; Wang, S.; Gao, Y.; Chen, Y.; Guo, Q.-L. Endostar, a novel recombinant human endostatin, exerts antiangiogenic effect via blocking VEGF-induced tyrosine phosphorylation of KDR/Flk-1 of endothelial cells. Biochem. Biophys. Res. Commun. 2007, 361, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Huang, X.-E.; Yan, P.-W.; Jiang, Y.; Xiang, J. Efficacy and Safety of Endostar Combined with Chemotherapy in Patients with Advanced Solid Tumors. Asian Pac. J. Cancer Prev. APJCP 2010, 11, 1119–1123. [Google Scholar]
- Adnane, L.; Trail, P.A.; Taylor, I.; Wilhelm, S.M. Sorafenib (BAY 43-9006, Nexavar), a dual-action inhibitor that targets RAF/MEK/ERK pathway in tumor cells and tyrosine kinases VEGFR/PDGFR in tumor vasculature. Methods Enzymol. 2006, 407, 597–612. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.; Vakiani, E.; Ku, G.; Herrera, J.; Tang, L.; Bouvier, N.; Viale, A.; Socci, N.; Capanu, M.; Berger, M.; et al. Phase II Trial of Sorafenib in Patients with Chemotherapy Refractory Metastatic Esophageal and Gastroesophageal (GE) Junction Cancer. PLoS ONE 2015, 10, e0134731. [Google Scholar] [CrossRef] [Green Version]
- Sun, W.; Powell, M.; O’Dwyer, P.J.; Catalano, P.; Ansari, R.H.; Benson, A.B., 3rd. Phase II study of sorafenib in combination with docetaxel and cisplatin in the treatment of metastatic or advanced gastric and gastroesophageal junction adenocarcinoma: ECOG 5203. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 2947–2951. [Google Scholar] [CrossRef]
- Roviello, G.; Ravelli, A.; Polom, K.; Petrioli, R.; Marano, L.; Marrelli, D.; Roviello, F.; Generali, D. Apatinib: A novel receptor tyrosine kinase inhibitor for the treatment of gastric cancer. Cancer Lett. 2016, 372, 187–191. [Google Scholar] [CrossRef]
- Hu, L.; Sun, F.; Sun, Z.; Ni, X.; Wang, J.; Wang, J.; Zhou, M.; Feng, Y.; Kong, Z.; Hua, Q.; et al. Apatinib enhances the radiosensitivity of the esophageal cancer cell line KYSE-150 by inducing apoptosis and cell cycle redistribution. Oncol. Lett. 2019, 17, 1609–1616. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Lei, J.; Yu, J.; Zhang, C.; Song, X.; Zhang, N.; Wang, Y.; Zhang, S. Clinical efficacy and safety of apatinib combined with S-1 in advanced esophageal squamous cell carcinoma. Investig. New Drugs 2020, 38, 500–506. [Google Scholar] [CrossRef] [Green Version]
- Metibemu, D.S.; Akinloye, O.A.; Akamo, A.J.; Ojo, D.A.; Okeowo, O.T.; Omotuyi, I.O. Exploring receptor tyrosine kinases-inhibitors in Cancer treatments. Egypt. J. Med. Hum. Genet. 2019, 20, 35. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Xiao, J.; Fang, W.; Lu, P.; Fan, Q.; Shu, Y.; Feng, J.; Zhang, S.; Ba, Y.; Zhao, Y.; et al. Anlotinib for previously treated advanced or metastatic esophageal squamous cell carcinoma: A double-blind randomized phase 2 trial. Cancer Med. 2021, 10, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Zheng, F.; Ren, D.; Du, F.; Dong, Q.; Wang, Z.; Zhao, F.; Ahmad, R.; Zhao, J. Anlotinib: A novel multi-targeting tyrosine kinase inhibitor in clinical development. J. Hematol. Oncol. 2018, 11, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, N.; Wu, T.; Hong, Y.-G.; Guo, Y.-Z.; Cheng, Y.-F.; Ma, Y.-J.; Bie, L.-Y.; Cui, D.-H.; Gao, X.-H.; Tan, B.-X.; et al. A multi-center, single-arm, phase II study of anlotinib plus paclitaxel and cisplatin as the first-line therapy of recurrent/advanced esophageal squamous cell carcinoma. BMC Med. 2022, 20, 472. [Google Scholar] [CrossRef] [PubMed]
- Paplomata, E.; Zelnak, A.; O’Regan, R. Everolimus: Side effect profile and management of toxicities in breast cancer. Breast Cancer Res. Treat. 2013, 140, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Wang, X.; Proud, C.G. mTOR inhibitors in cancer therapy. F1000Res 2016, 5, F1000. [Google Scholar] [CrossRef] [Green Version]
- Tabernero, J.; Rojo, F.; Calvo, E.; Burris, H.; Judson, I.; Hazell, K.; Martinelli, E.; Ramon y Cajal, S.; Jones, S.; Vidal, L.; et al. Dose- and schedule-dependent inhibition of the mammalian target of rapamycin pathway with everolimus: A phase I tumor pharmacodynamic study in patients with advanced solid tumors. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 1603–1610. [Google Scholar] [CrossRef]
- Meacham, C.E.; Morrison, S.J. Tumour heterogeneity and cancer cell plasticity. Nature 2013, 501, 328–337. [Google Scholar] [CrossRef] [Green Version]
- Dinh, H.Q.; Pan, F.; Wang, G.; Huang, Q.-F.; Olingy, C.E.; Wu, Z.-Y.; Wang, S.-H.; Xu, X.; Xu, X.-E.; He, J.-Z.; et al. Integrated single-cell transcriptome analysis reveals heterogeneity of esophageal squamous cell carcinoma microenvironment. Nat. Commun. 2021, 12, 7335. [Google Scholar] [CrossRef]
- He, W.; Leng, X.; Yang, Y.; Peng, L.; Shao, Y.; Li, X.; Han, Y. Genetic Heterogeneity of Esophageal Squamous Cell Carcinoma with Inherited Family History. Onco Targets Ther. 2020, 13, 8795–8802. [Google Scholar] [CrossRef] [PubMed]
- Alfarouk, K.O.; Stock, C.-M.; Taylor, S.; Walsh, M.; Muddathir, A.K.; Verduzco, D.; Bashir, A.H.H.; Mohammed, O.Y.; Elhassan, G.O.; Harguindey, S.; et al. Resistance to cancer chemotherapy: Failure in drug response from ADME to P-gp. Cancer Cell Int. 2015, 15, 71. [Google Scholar] [CrossRef] [Green Version]
- Luan, S.; Zeng, X.; Zhang, C.; Qiu, J.; Yang, Y.; Mao, C.; Xiao, X.; Zhou, J.; Zhang, Y.; Yuan, Y. Advances in Drug Resistance of Esophageal Cancer: From the Perspective of Tumor Microenvironment. Front. Cell Dev. Biol. 2021, 9, 664816. [Google Scholar] [CrossRef]
- Yang, W.; Ma, J.; Zhou, W.; Zhou, X.; Cao, B.; Zhang, H.; Zhao, Q.; Fan, D.; Hong, L. Molecular mechanisms and clinical implications of miRNAs in drug resistance of esophageal cancer. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adhikari, S.; Bhattacharya, A.; Adhikary, S.; Singh, V.; Gadad, S.S.; Roy, S.; Das, C. The paradigm of drug resistance in cancer: An epigenetic perspective. Biosci. Rep. 2022, 42, BSR20211812. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Pestell, T.G.; Lisanti, M.P.; Pestell, R.G. Cancer stem cells. Int. J. Biochem. Cell Biol. 2012, 44, 2144–2151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walcher, L.; Kistenmacher, A.-K.; Suo, H.; Kitte, R.; Dluczek, S.; Strauß, A.; Blaudszun, A.-R.; Yevsa, T.; Fricke, S.; Kossatz-Boehlert, U. Cancer Stem Cells—Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front. Immunol. 2020, 11, 1280. [Google Scholar] [CrossRef] [PubMed]
- Batlle, E.; Clevers, H. Cancer stem cells revisited. Nat. Med. 2017, 23, 1124–1134. [Google Scholar] [CrossRef]
- Zhao, J. Cancer stem cells and chemoresistance: The smartest survives the raid. Pharmacol. Ther. 2016, 160, 145–158. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Gao, Q.; Guo, L.; Lu, S.H. The PTEN/PI3K/Akt pathway regulates stem-like cells in primary esophageal carcinoma cells. Cancer Biol. 2011, 11, 950–958. [Google Scholar] [CrossRef] [Green Version]
- Tong, D.; Sun, J.; Huang, P.; Li, M.; Zhang, F. p75 neurotrophin receptor: A potential surface marker of tongue squamous cell carcinoma stem cells. Mol. Med. Rep. 2017, 15, 2521–2529. [Google Scholar] [CrossRef] [Green Version]
- Galluzzi, L.; Senovilla, L.; Vitale, I.; Michels, J.; Martins, I.; Kepp, O.; Castedo, M.; Kroemer, G. Molecular mechanisms of cisplatin resistance. Oncogene 2012, 31, 1869–1883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Miyata, H.; Sugimura, K.; Fukuda, S.; Kanemura, T.; Yamashita, K.; Miyazaki, Y.; Takahashi, T.; Kurokawa, Y.; Yamasaki, M.; et al. miR-27 is associated with chemoresistance in esophageal cancer through transformation of normal fibroblasts to cancer-associated fibroblasts. Carcinogenesis 2015, 36, 894–903. [Google Scholar] [CrossRef] [Green Version]
- Qiao, Y.; Zhang, C.; Li, A.; Wang, D.; Luo, Z.; Ping, Y.; Zhou, B.; Liu, S.; Li, H.; Yue, D.; et al. IL6 derived from cancer-associated fibroblasts promotes chemoresistance via CXCR7 in esophageal squamous cell carcinoma. Oncogene 2018, 37, 873–883. [Google Scholar] [CrossRef]
- Verma, H.K.; Falco, G.; Bhaskar, L.V.K.S. Molecular Signaling Pathways Involved in Gastric Cancer Chemoresistance. In Theranostics Approaches to Gastric and Colon Cancer; Raju, G.S.R., Bhaskar, L.V.K.S., Eds.; Springer: Singapore, 2020; pp. 117–134. [Google Scholar] [CrossRef]
- Juneja, V.R.; McGuire, K.A.; Manguso, R.T.; LaFleur, M.W.; Collins, N.; Haining, W.N.; Freeman, G.J.; Sharpe, A.H. PD-L1 on tumor cells is sufficient for immune evasion in immunogenic tumors and inhibits CD8 T cell cytotoxicity. J. Exp. Med. 2017, 214, 895–904. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Huang, J. Breast cancer immunology and immunotherapy: Targeting the programmed cell death protein-1/programmed cell death protein ligand-1. Chin. Med. J. 2020, 133, 853–862. [Google Scholar] [CrossRef]
- Huang, T.-X.; Fu, L. The immune landscape of esophageal cancer. Cancer Commun. 2019, 39, 79. [Google Scholar] [CrossRef] [Green Version]
- Dhupar, R.; Van Der Kraak, L.; Pennathur, A.; Schuchert, M.J.; Nason, K.S.; Luketich, J.D.; Lotze, M.T. Targeting Immune Checkpoints in Esophageal Cancer: A High Mutational Load Tumor. Ann. Thorac. Surg. 2017, 103, 1340–1349. [Google Scholar] [CrossRef] [Green Version]
- Yagi, T.; Baba, Y.; Okadome, K.; Kiyozumi, Y.; Hiyoshi, Y.; Ishimoto, T.; Iwatsuki, M.; Miyamoto, Y.; Yoshida, N.; Watanabe, M.; et al. Tumour-associated macrophages are associated with poor prognosis and programmed death ligand 1 expression in oesophageal cancer. Eur. J. Cancer 2019, 111, 38–49. [Google Scholar] [CrossRef]
- Däster, S.; Eppenberger-Castori, S.; Mele, V.; Schäfer, H.M.; Schmid, L.; Weixler, B.; Soysal, S.D.; Droeser, R.A.; Spagnoli, G.C.; Kettelhack, C.J. Low expression of programmed death 1 (PD-1), PD-1 Ligand 1 (PD-L1), and Low CD8+ T Lymphocyte infiltration identify a subgroup of patients with gastric and esophageal adenocarcinoma with severe prognosis. Front. Med. 2020, 7, 144. [Google Scholar] [CrossRef] [PubMed]
- Bielenberg, D.R.; Zetter, B.R. The contribution of angiogenesis to the process of metastasis. Cancer J. 2015, 21, 267. [Google Scholar] [PubMed] [Green Version]
- Nisar, S.; Hashem, S.; Macha, M.A.; Yadav, S.K.; Muralitharan, S.; Therachiyil, L.; Sageena, G.; Al-Naemi, H.; Haris, M.; Bhat, A.A. Exploring dysregulated signaling pathways in cancer. Curr. Pharm. Des. 2020, 26, 429–445. [Google Scholar] [PubMed]
- Leu, C.-M.; Wong, F.-H.; Chang, C.; Huang, S.-F.; Hu, C. Interleukin-6 acts as an antiapoptotic factor in human esophageal carcinoma cells through the activation of both STAT3 and mitogen-activated protein kinase pathways. Oncogene 2003, 22, 7809–7818. [Google Scholar] [CrossRef] [Green Version]
- Ebbing, E.A.; van der Zalm, A.P.; Steins, A.; Creemers, A.; Hermsen, S.; Rentenaar, R.; Klein, M.; Waasdorp, C.; Hooijer, G.K.J.; Meijer, S.L.; et al. Stromal-derived interleukin 6 drives epithelial-to-mesenchymal transition and therapy resistance in esophageal adenocarcinoma. Proc. Natl. Acad. Sci. USA 2019, 116, 2237–2242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugimura, K.; Miyata, H.; Tanaka, K.; Hamano, R.; Takahashi, T.; Kurokawa, Y.; Yamasaki, M.; Nakajima, K.; Takiguchi, S.; Mori, M.; et al. Let-7 expression is a significant determinant of response to chemotherapy through the regulation of IL-6/STAT3 pathway in esophageal squamous cell carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 5144–5153. [Google Scholar] [CrossRef] [Green Version]
- Hatata, T.; Higaki, K.; Tatebe, S.; Shomori, K.; Ikeguchi, M. Immunohistochemical study of nuclear factor-κB expression in esophageal squamous cell carcinoma: Prognostic significance and sensitivity to treatment with 5-FU. Dis. Esophagus 2012, 25, 716–722. [Google Scholar] [CrossRef]
- Chen, S.; Shen, Z.; Liu, Z.; Gao, L.; Han, Z.; Yu, S.; Kang, M.J. IL-1RA suppresses esophageal cancer cell growth by blocking IL-1α. J. Clin. Lab. Anal. 2019, 33, e22903. [Google Scholar] [CrossRef] [Green Version]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Brown, A.; Kumar, S.; Tchounwou, P.B. Cisplatin-Based Chemotherapy of Human Cancers. J. Cancer Sci. Ther. 2019, 11, 97. [Google Scholar]
- Kimura, S.; Kitadai, Y.; Tanaka, S.; Kuwai, T.; Hihara, J.; Yoshida, K.; Toge, T.; Chayama, K. Expression of hypoxia-inducible factor (HIF)-1alpha is associated with vascular endothelial growth factor expression and tumour angiogenesis in human oesophageal squamous cell carcinoma. Eur. J. Cancer 2004, 40, 1904–1912. [Google Scholar] [CrossRef]
- Macedo-Silva, C.; Miranda-Gonçalves, V.; Henrique, R.; Jerónimo, C.; Bravo, I. The Critical Role of Hypoxic Microenvironment and Epigenetic Deregulation in Esophageal Cancer Radioresistance. Genes 2019, 10, 927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, X.; You, M.; Wang, F.; Wang, Z.; Gao, X.; Jing, C.; Liu, J.; Guo, M.; Li, J.; Luo, A.; et al. Multifunctional Graphdiyne-Cerium Oxide Nanozymes Facilitate MicroRNA Delivery and Attenuate Tumor Hypoxia for Highly Efficient Radiotherapy of Esophageal Cancer. Adv. Mater. 2021, 33, e2100556. [Google Scholar] [CrossRef] [PubMed]
- Iyer, M.K.; Niknafs, Y.S.; Malik, R.; Singhal, U.; Sahu, A.; Hosono, Y.; Barrette, T.R.; Prensner, J.R.; Evans, J.R.; Zhao, S.; et al. The landscape of long noncoding RNAs in the human transcriptome. Nat. Genet. 2015, 47, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.J.; Pang, Y.; Jin, G.; Zhang, H.Y.; Wang, W.H.; Liu, J.W.; Tuo, G.X.; Wu, P.; Yang, Y.; Wang, Z.Q.; et al. Hypoxia induces chemoresistance of esophageal cancer cells to cisplatin through regulating the lncRNA-EMS/miR-758-3p/WTAP axis. Aging 2021, 13, 17155–17176. [Google Scholar] [CrossRef]
- Li, B.; Tsao, S.W.; Chan, K.W.; Ludwig, D.L.; Novosyadlyy, R.; Li, Y.Y.; He, Q.Y.; Cheung, A.L. Id1-induced IGF-II and its autocrine/endocrine promotion of esophageal cancer progression and chemoresistance--implications for IGF-II and IGF-IR-targeted therapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 2651–2662. [Google Scholar] [CrossRef] [Green Version]
- Chan, D.; Zhou, Y.; Chui, C.H.; Lam, K.H.; Law, S.; Chan, A.S.; Li, X.; Lam, A.K.; Tang, J.C.O. Expression of Insulin-Like Growth Factor Binding Protein-5 (IGFBP5) Reverses Cisplatin-Resistance in Esophageal Carcinoma. Cells 2018, 7, 143. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Pan, T.; Zhong, X.; Cheng, C. Resistance to cetuximab in EGFR-overexpressing esophageal squamous cell carcinoma xenografts due to FGFR2 amplification and overexpression. J. Pharmacol. Sci. 2014, 126, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Saito, S.; Morishima, K.; Ui, T.; Hoshino, H.; Matsubara, D.; Ishikawa, S.; Aburatani, H.; Fukayama, M.; Hosoya, Y.; Sata, N.J.B.c. The role of HGF/MET and FGF/FGFR in fibroblast-derived growth stimulation and lapatinib-resistance of esophageal squamous cell carcinoma. BMC Cancer 2015, 15, 82. [Google Scholar] [CrossRef] [Green Version]
- Janjigian, Y.Y.; Sanchez-Vega, F.; Jonsson, P.; Chatila, W.K.; Hechtman, J.F.; Ku, G.Y.; Riches, J.C.; Tuvy, Y.; Kundra, R.; Bouvier, N.; et al. Genetic Predictors of Response to Systemic Therapy in Esophagogastric Cancer. Cancer Discov. 2018, 8, 49–58. [Google Scholar] [CrossRef] [Green Version]
- Allegra, A.; Cancemi, G.; Mirabile, G.; Tonacci, A.; Musolino, C.; Gangemi, S. Circulating Tumour Cells, Cell Free DNA and Tumour-Educated Platelets as Reliable Prognostic and Management Biomarkers for the Liquid Biopsy in Multiple Myeloma. Cancers 2022, 14, 4136. [Google Scholar] [CrossRef]
- Iacob, R.; Mandea, M.; Iacob, S.; Pietrosanu, C.; Paul, D.; Hainarosie, R.; Gheorghe, C. Liquid Biopsy in Squamous Cell Carcinoma of the Esophagus and of the Head and Neck. Front. Med. 2022, 9, 827297. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Wang, J.; Yang, B.; Zhu, J.; Wang, Y.; Jiao, J.; Zhu, K.; Zhang, M.; Zhai, L.; Gong, T. Urinary protein biomarker panel predicts esophageal squamous carcinoma from control cases and other tumors. Esophagus 2022, 19, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Goto, M.; Liu, M.J.E. Chemokines and their receptors as biomarkers in esophageal cancer. Esophagus 2020, 17, 113–121. [Google Scholar] [CrossRef]
- Luo, H.; Li, H.; Hu, Z.; Wu, H.; Liu, C.; Li, Y.; Zhang, X.; Lin, P.; Hou, Q.; Ding, G.; et al. Noninvasive diagnosis and monitoring of mutations by deep sequencing of circulating tumor DNA in esophageal squamous cell carcinoma. Biochem. Biophys. Res. Commun. 2016, 471, 596–602. [Google Scholar] [CrossRef]
- Ueda, M.; Iguchi, T.; Masuda, T.; Nakahara, Y.; Hirata, H.; Uchi, R.; Niida, A.; Momose, K.; Sakimura, S.; Chiba, K.; et al. Somatic mutations in plasma cell-free DNA are diagnostic markers for esophageal squamous cell carcinoma recurrence. Oncotarget 2016, 7, 62280–62291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsieh, C.C.; Hsu, H.S.; Chang, S.C.; Chen, Y.J. Circulating Cell-Free DNA Levels Could Predict Oncological Outcomes of Patients Undergoing Esophagectomy for Esophageal Squamous Cell Carcinoma. Int. J. Mol. Sci. 2016, 17, 2131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, W.; Han, Y.; Zhao, X.; Duan, L.; Zhou, W.; Wang, X.; Shi, G.; Che, Y.; Zhang, Y.; Liu, J.; et al. Advances in prognostic biomarkers for esophageal cancer. Expert Rev. Mol. Diagn. 2019, 19, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Peng, J.; Zhu, W.; Tao, G.; Song, Y.; Zhou, X.; Wang, W. Combined downregulation of microRNA-133a and microRNA-133b predicts chemosensitivity of patients with esophageal squamous cell carcinoma undergoing paclitaxel-based chemotherapy. Med. Oncol. 2014, 31, 263. [Google Scholar] [CrossRef]
- Zhou, Y.; Hong, L. Prediction value of miR-483 and miR-214 in prognosis and multidrug resistance of esophageal squamous cell carcinoma. Genet. Test. Mol. Biomark. 2013, 17, 470–474. [Google Scholar] [CrossRef] [Green Version]
- Ju, C.; He, J.; Wang, C.; Sheng, J.; Jia, J.; Du, D.; Li, H.; Zhou, M.; He, F. Current advances and future perspectives on the functional roles and clinical implications of circular RNAs in esophageal squamous cell carcinoma: More influential than expected. Biomark. Res. 2022, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Yu, Z.; Yang, H.; Lin, Y. Increased MALAT1 expression predicts poor prognosis in esophageal cancer patients. Biomed. Pharmacother. 2016, 83, 8–13. [Google Scholar] [CrossRef] [PubMed]
| Drugs Name | Targeting Pathway | Mode of Action | Findings | Dose and Dosing Frequency | Side Effects | References |
|---|---|---|---|---|---|---|
| Cetuximab | EGFR | Anti EGFR monoclonal antibody | Effective in combination with chemotherapy; increased survival rate in resectable ESCC patients | 400 mg/m2 over 120 min (day 1), followed by weekly dose of 250 mg/m2 over 60 min for 14 weeks. | Hypomagnesemia | [29,80] |
| Nimotuzumab | EGFR | Anti-EGFR monoclonal antibody | Nimotuzumab + radiotherapy = used in phase I clinical trial; Stopped G2 phase of ESCC cell cycle | Nimotuzumab (200 mg/weekly), diluted in 250 mL of 0.9% sodium chloride, for 5 weeks. | Esophagitis, pneumonitis, leukopenia, gastrointestinal reaction, thrombocytopenia, radiothermitis and fever | [37,81,82] |
| Gefitinib | EGFR | Anti-EGFR-RTK inhibitor | Gefitinib + 5-FU = increased survival rate of advance ESCC patients | 250 mg/day | Dry skin, Itching, rash, acne, mouth sores, and weakness, diarrhea skin toxicity, fatigue | [42,83] |
| Icotinib | EGFR | Anti-EGFR-RTK inhibitor | 150 mg, orally three times daily | Rash and diarrhea | [44,84,85] | |
| Transtuzumab | HER2 | Monoclonal anti-HER2 antibody | Used in combination with cisplatin + fluoropyrimidine (either capecitabine or 5-fluorouracil) for patient with HER2-positive metastatic gastric or GE adenocarcinoma | 200 mg/m2 to 1000 mg/m2 | Fever and chills, cough, and headache | [49,86,87] |
| Lapatinib | Dual RTK inhibitor. Bind to ATP binding site and inhibit kinase activity | Lapatinib + paclitaxel = suppressed cell proliferation and decreased ESCC cell migration, invasion | 1250 mg per day | Face redness, dizziness, headache, shortness of breath, and anxiety | [57,88,89] | |
| Ramucirumab | VEGF | Monoclonal antibody, block binding of VEGF to VEGFR | Ramucirumab + paclitaxel = used in 2nd line treatment of advanced GEJ adenocarcinoma | 8 mg/kg | Hypertension, thromboembolism, rash, diarrhea, and myelosuppression | [90,91,92] |
| Endastar | VEGF | Suppress the signaling of VEGFR and inhibit endothelial growth and migration | Endastar in combination with chemotherapy decreased tumor weight | Nausea, vomiting, fever, etc. | [93,94] | |
| Sorafenib | VEGF | Inhibit VEGFR2 | Reduced development of EAC and GEJ in phase II clinical trial | 400 mg orally twice a day for 21 days | fatigue, weakness, redness of the skin, hair loss, itching, dry or peeling skin, and a lack of appetite, etc. | [95,96,97] |
| Apatinib | VEGF | Inhibit RTK-VEGFR2 receptor | Had an anti-esophageal-cancer effect | A starting dosage of 250 mg once every day. After a week, if the first dose was well tolerated, apatinib dosage was increased to 500 mg. | Diarrhea, nausea, vomiting, dry skin etc. | [98,99,100] |
| Anlotinib | VEGF | Inhibit RTK-VEGFR2/3 receptor | Increased disease control rate in pretreated advanced ESCC patients | 10 mg orally/day | thrombocytopenia and neutropenia, hypercholesterolemia, dermal toxicity hypertriglyceridemia | [101,102,103,104] |
| Rilotumumab | HGF-c-MET | Inhibit interaction of HGF with c-MET | Rilotumumab could not effectively treat the patients. | 5.4–6.4/kg | Nausea, vomiting, fever etc. | [3,75] |
| Obinutuzumab | HGF-c-MET | Block MET from binding to HGF | Obinutuzumab could not improve the patient survival rate | Decrease in the number of WBC and platelets cause infection and bleeding. Fever; tiredness and weakness, headache; hair loss. | [3] | |
| Rapamycin and Everolimus | m-TOR | Inhibit m-TOR | Decreased cell proliferation and growth | 10 mg/day or 50 mg/week | Stomatitis, rash, tiredness, hyperglycemia, hyperlipidemia, etc. | [105,106,107] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panda, P.; Verma, H.K.; Bhaskar, L.V.K.S. Precision Medicine Revolutionizing Esophageal Cancer Treatment: Surmounting Hurdles and Enhancing Therapeutic Efficacy through Targeted Drug Therapies. Onco 2023, 3, 127-146. https://doi.org/10.3390/onco3030010
Panda P, Verma HK, Bhaskar LVKS. Precision Medicine Revolutionizing Esophageal Cancer Treatment: Surmounting Hurdles and Enhancing Therapeutic Efficacy through Targeted Drug Therapies. Onco. 2023; 3(3):127-146. https://doi.org/10.3390/onco3030010
Chicago/Turabian StylePanda, Poojarani, Henu Kumar Verma, and Lakkakula V. K. S. Bhaskar. 2023. "Precision Medicine Revolutionizing Esophageal Cancer Treatment: Surmounting Hurdles and Enhancing Therapeutic Efficacy through Targeted Drug Therapies" Onco 3, no. 3: 127-146. https://doi.org/10.3390/onco3030010
APA StylePanda, P., Verma, H. K., & Bhaskar, L. V. K. S. (2023). Precision Medicine Revolutionizing Esophageal Cancer Treatment: Surmounting Hurdles and Enhancing Therapeutic Efficacy through Targeted Drug Therapies. Onco, 3(3), 127-146. https://doi.org/10.3390/onco3030010







