Ceratozamia schiblii (Zamiaceae): A New Cycad Species from the Eastern Mountains of Oaxaca, Mexico
Abstract
:1. Introduction
2. Material and Methods
3. Results
New Species Description
| 1a. Plants with erect cone | |
| 2a: Plants with emerging orange leaves……………………………………………………………………………………………… | C. aurantiaca |
| 2b: Plants with green, reddish, yellowish, brown, or caramel emerging leaves | |
| 3a: Plants with visible veins on leaflets | |
| 4a: Plants with emerging yellowish-brown leaves……………………………………………………………………… | C. subroseophylla |
| 4b: Plants with emerging green leaves | |
| 5a: Leaflets membranaceous………………………………………………………………………………………… | C. leptoceras |
| 5b: Leaflets papyraceous……………………………………………………………………………………………………… | C. oliversacksii |
| 3b: Plants with inconspicuous veins on leaflets | |
| 6a: Plants with emerging reddish-brown leaves……………………………………………………………………………………… | C. robusta |
| 6b: Plants with newly emerging caramel colored leaves………………………………………………………………………………… | C. dominguezii |
| 1b. Plants with pendent cone | |
| 7a: Plants with emerging leaves of pea green color……………………………………………………………………………………… | C. mixeorum |
| 7b: Plants with emerging leaves of brown color…………………………………………………………………………………………… | C. schiblii |
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Whitelock, L.M. The Cycads; Timber Press: Portland, OR, USA, 2002; p. 532. [Google Scholar]
- Vovides, A.P.; Pérez-Farrera, M.A.; González, D.; Avendaño, S. Relationships and Phytogeography in Ceratozamia (Zamiaceae). In Cycad Classification: Concepts and Recommendations; Walters, T., Osborne, R., Eds.; CABI Publishing: Wallingford, UK, 2004; pp. 109–125. [Google Scholar]
- Pérez-Farrera, M.A.; Vovides, A.P.; López-Mendoza, S.; Hernández-Sandoval, L.; Martínez, M. Estimation of genetic variation in closely related cycad species in Ceratozamia (Zamiaceae, Cycadales) using RAPDs markers. Rev. Biol. Trop. 2017, 65, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Mankga, L.T.; Yessoufou, K. Factors driving the global decline of cycad diversity. AoB Plants 2017, 9, plx022. [Google Scholar] [CrossRef] [PubMed]
- Calonje, M.; Stevenson, D.W.; Osborne, R. The World List of Cycads, Online Edition. 2022. Available online: http://www.cycadlist.org (accessed on 20 April 2022).
- Gutiérrez-Ortega, J.S.; Pérez-Farrera, M.A.; Vovides, A.P.; Chávez-Cortázar, A.; López, S.; Santos-Hernández, N.G.; Ruíz-Roblero, S.K. Ceratozamia sanchezae (Zamiaceae): A new cycad species from Chiapas Highlands (Mexico). Phytotaxa 2021, 500, 201–216. [Google Scholar] [CrossRef]
- Pérez-Farrera, M.A.; Vovides, A.P.; Hernández-Sandoval, L.; González, D.; Martínez, M. A morphometric analysis of the Ceratozamia norstogii Stevenson complex (Zamiaceae). In Cycad Classification: Concepts and Recommendations; Walters, T., Osborne, R., Eds.; CABI Publishing: Wallingford, UK, 2004; pp. 127–136. [Google Scholar]
- Pérez-Farrera, M.A.; Gutiérrez-Ortega, J.S.; Haynes, J.L.; Chemnick, J.; Salas-Morales, S.H.; Calonje, M.; Vovides, A.P. Ceratozamia aurantiaca (Zamiaceae): A new cycad species from the northern rainforests of Oaxaca, Mexico. Taxonomy 2021, 1, 243–255. [Google Scholar] [CrossRef]
- Pérez-Farrera, M.A.; Gutiérrez-Ortega, J.S.; Vovides, A.P.; Calonje, M.; Díaz-Jiménez, P. Ceratozamia dominguezii (Zamiaceae): A new cycad species from southeastern Mexico. Taxonomy 2021, 1, 345–359. [Google Scholar] [CrossRef]
- Martínez-Domínguez, L.; Nicolalde-Morejón, F.; Lorea-Hernández, F.G.; Vergara-Silva, F.; Stevenson, D.W. A novelty in Ceratozamia (Zamiaceae, Cycadales) from the Sierra Madre del Sur, Mexico: Biogeographic and morphological patterns, DNA barcoding and phenology. PhytoKeys 2020, 156, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Domínguez, L.; Nicolalde-Morejón, F.; Vergara-Silva, F.; Stevenson, D.W. Ceratozamia oliversacksii (Zamiaceae), a new species of gymnosperm from western Oaxaca, Mexico. Kew Bull. 2022, 77, 211–219. [Google Scholar] [CrossRef]
- QGIS Geographic Information System. QGIS Association. Available online: http://www.qgis.org (accessed on 20 April 2022).
- Hijmans, R.J.; Guarino, L.; Mathur, P. Diva-Gis Version 7.5 Manual. University of California. Available online: https://www.diva-gis.org (accessed on 21 April 2022).
- Hammer, Ø.; Harper, D.A.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Thiers, B. Index Herbariorum: A Global Directory of Public Herbaria and Associated Staff. New York Botanical Garden’s Virtual Herbarium. 2020. Available online: http://sweetgum.nybg.org/science/ih/ (accessed on 3 June 2022).
- Rzedowski, J. Vegetación de México, 1ra. Edición Digital; Comisión Nacional Para el Conocimiento y Uso de la Biodiversidad: Mexico City, Mexico, 2006; p. 504. [Google Scholar]
- Wendt, T. Las selvas de Uxpanapa, Veracruz-Oaxaca, México: Evidencia de refugios florísticos cenozoicos. An. Inst. Biol. UNAM Ser. Bot. 1989, 58, 29–54. [Google Scholar]
- Gutiérrez-Ortega, J.S.; Salinas-Rodríguez, M.M.; Ito, T.; Pérez-Farrera, M.A.; Vovides, A.P.; Martínez, J.F.; Molina-Freaner, F.; Hernández-López, A.; Kawaguchi, L.; Nagano, A.J.; et al. Niche conservatism promotes speciation in cycads: The case of Dioon merolae (Zamiaceae) in Mexico. New Phytol. 2020, 227, 1872–1884. [Google Scholar] [CrossRef] [PubMed]
- Chemnick, J.; Gregory, T.J.; Salas-Morales, S. Ceratozamia mixeorum (Zamiaceae), a new species from Oaxaca, Mexico with comments on distribution, habitat, and species relationships. Phytologia 1997, 83, 47–52. [Google Scholar]
- González, D.; Vovides, V.P. Low intralineage divergence in Ceratozamia (Zamiaceae) detected with nuclear ribosomal DNA ITS and chloroplast DNA trnL-F non-coding region. Syst. Bot. 2002, 27, 654–661. [Google Scholar]
- Vovides, A.P.; Pérez-Farrera, M.A.; Gutiérrez-Ortega, J.S.; Avendaño, S.; Medina-Villarreal, A.; González-Astorga, J.; Galicia, S. A revision of the Ceratozamia miqueliana (Zamiaceae) species complex based on analyses of leaflet anatomical characters. Flora 2020, 270, 151649. [Google Scholar] [CrossRef]
- Medina-Villarreal, A.; González-Astorga, J.; de Los Monteros, A.E. Evolution of Ceratozamia cycads: A proximate-ultimate approach. Mol. Phylogenet. Evol. 2019, 139, 106530. [Google Scholar] [CrossRef] [PubMed]
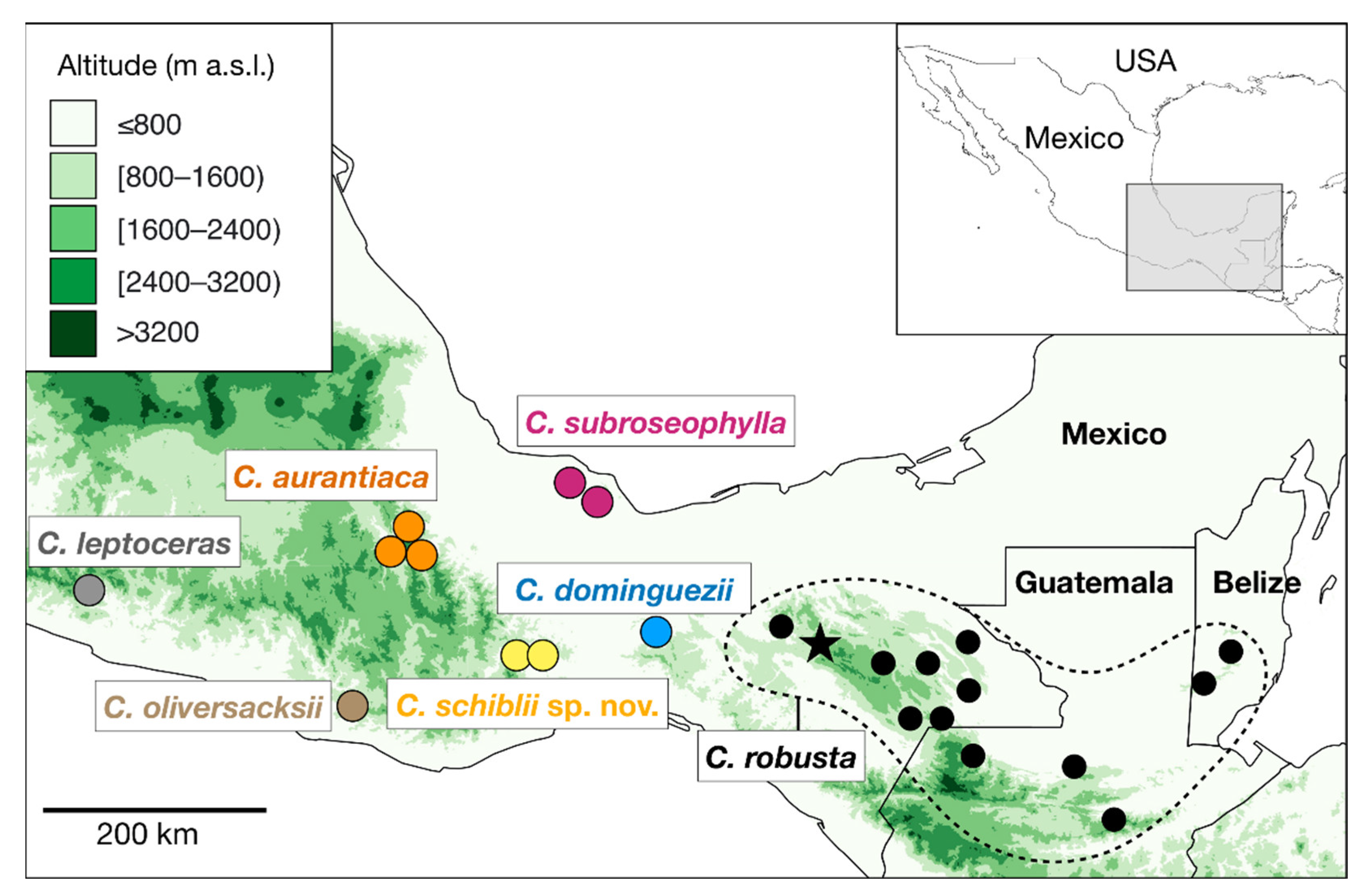
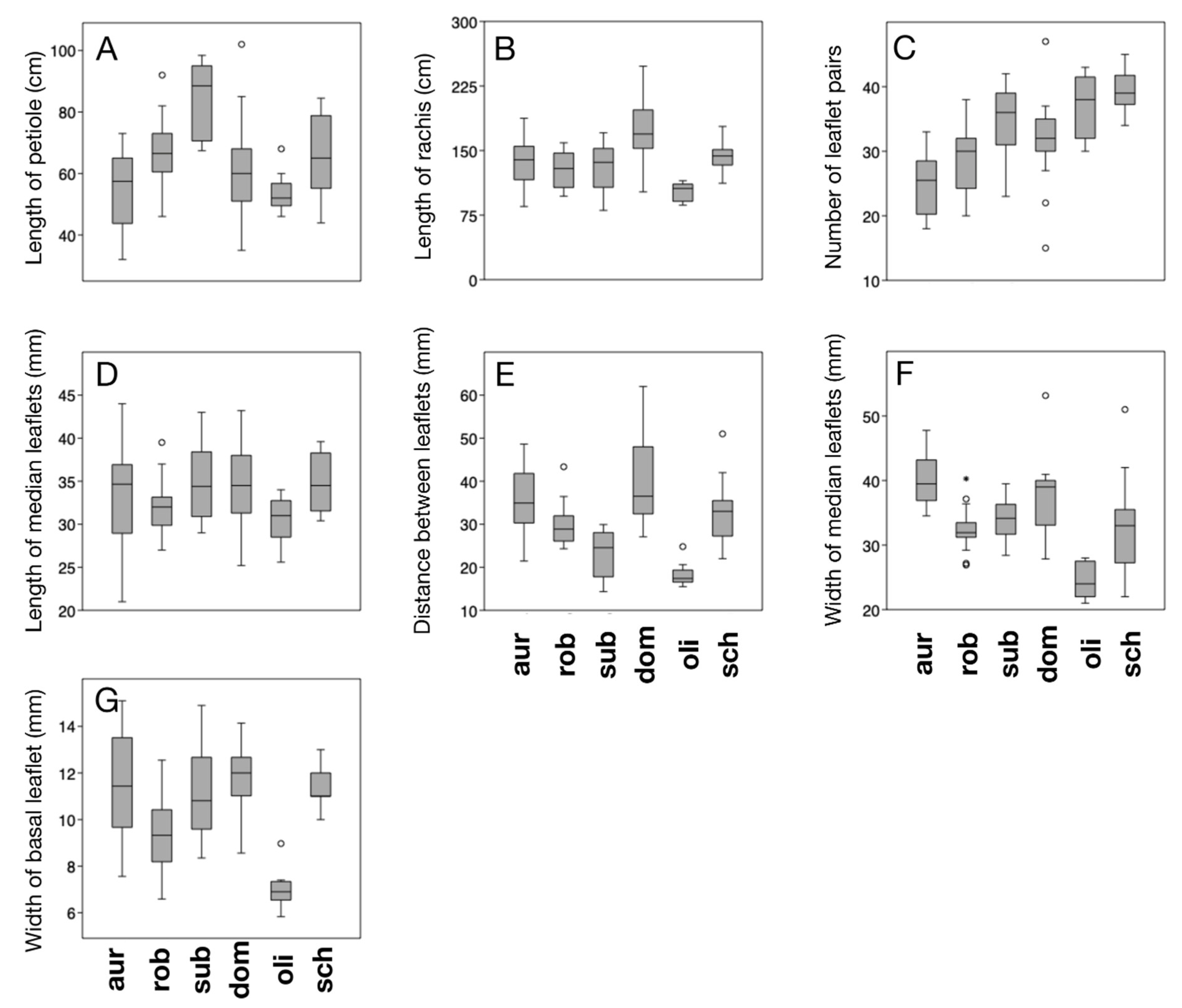
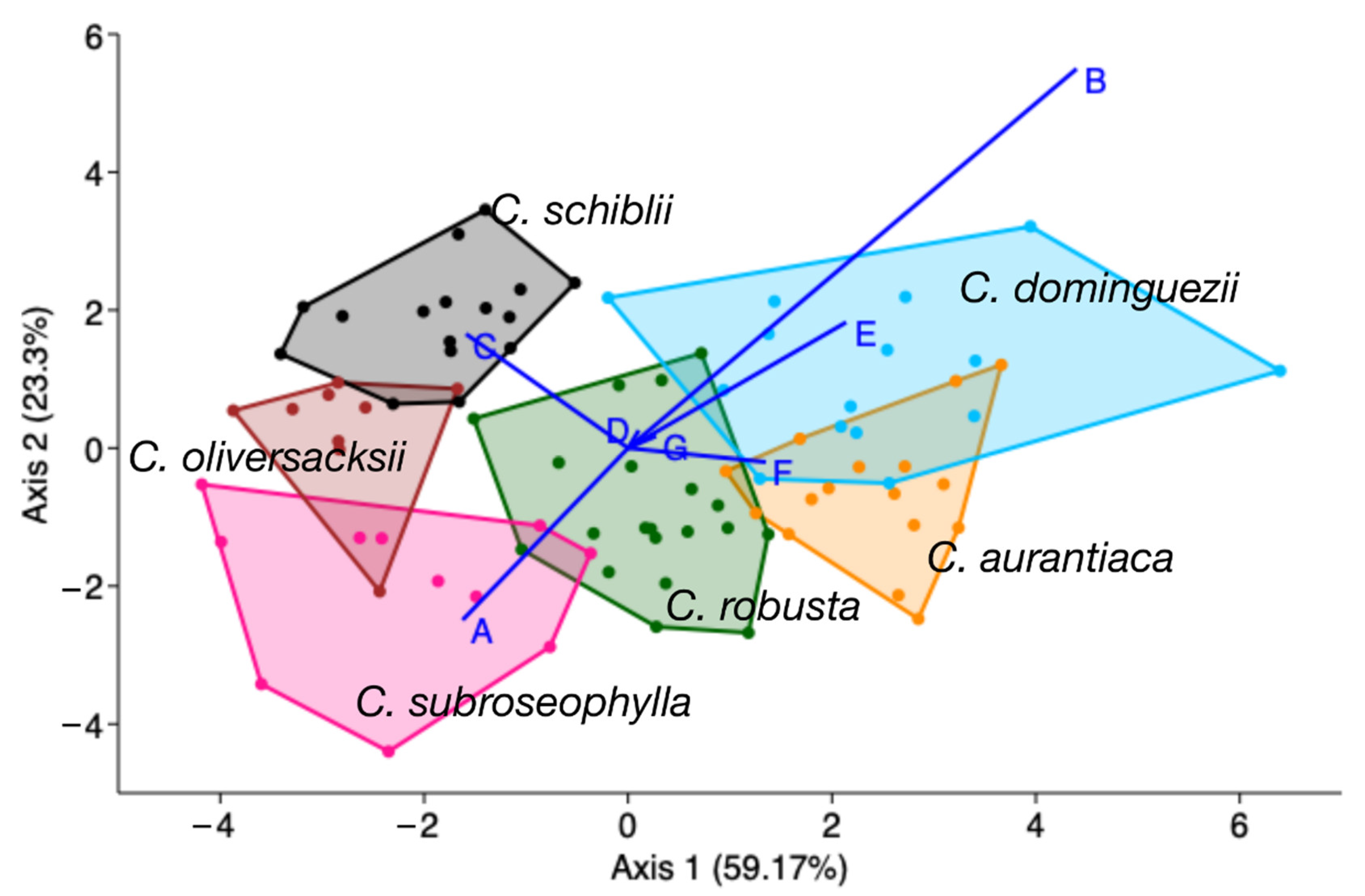




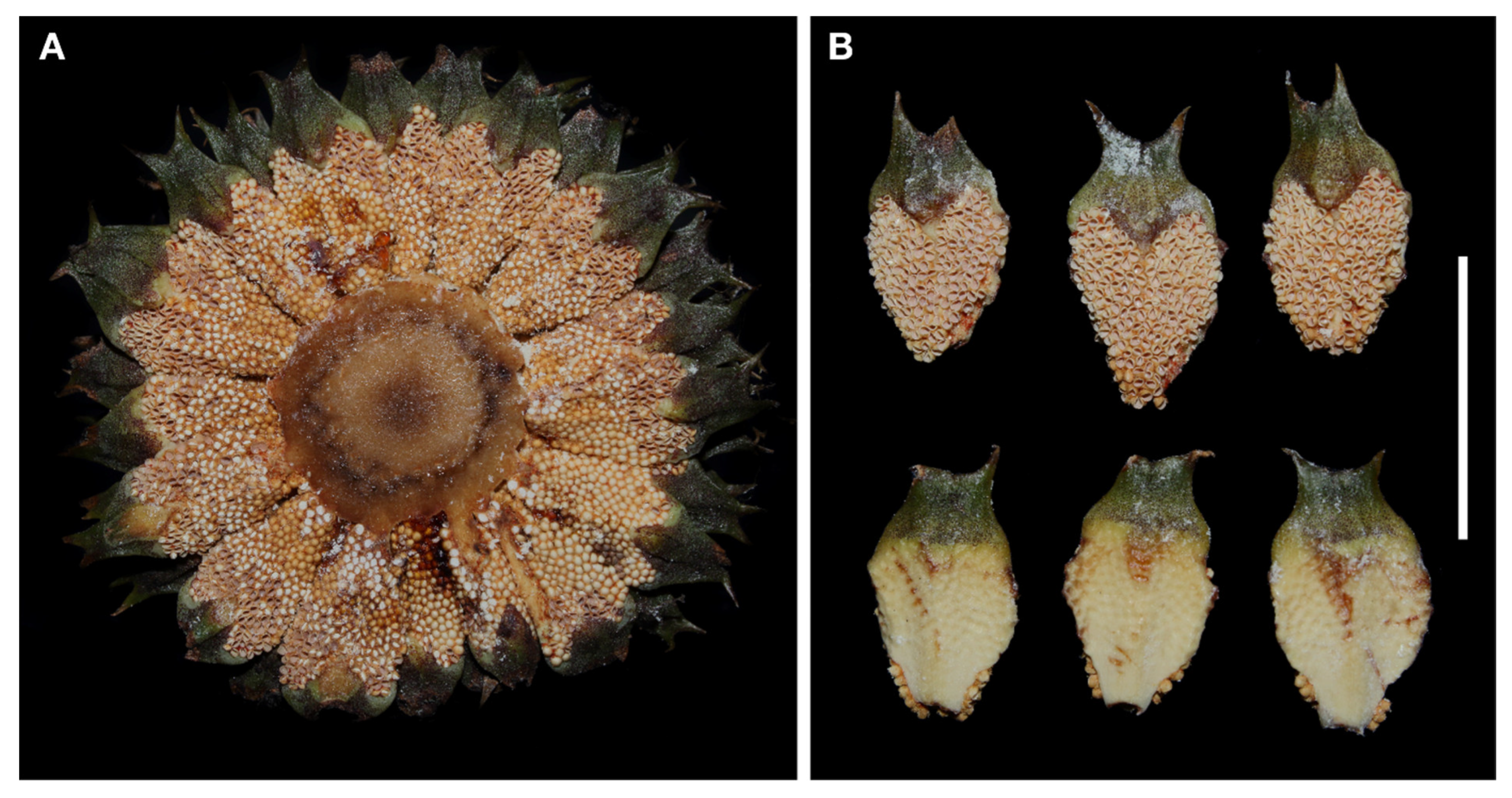
| Species | Key | Elevation (m a.s.l.) | Locality | n |
|---|---|---|---|---|
| C. aurantiaca | aur | 480 | El Faro, Sierra Norte, Oaxaca (type population) | 16 |
| C. robusta | rob | 1200 | Cañón del Sumidero, Chiapas (neotype population) | 20 |
| C. subroseophylla | sub | 500 | Santiago Tuxtla, Veracruz (type population) | 11 |
| C. dominguezii | dom | 130 | Uxpanapa, Veracruz (type population) | 15 |
| C. oliversacksii | oli | 840 | San Gabriel Mixtepec, Oaxaca | 9 |
| C. schiblii | sch | 1290 | Santiago Lachiguiri, Oaxaca (type population) | 16 |
| Trait | C. aurantiaca | C. robusta | C. subroseophylla | C. dominguezii | C. oliversacksii | C. leptoceras | C. schiblii sp. nov. |
|---|---|---|---|---|---|---|---|
| Trunk | Erect to decumbent | Erect | Erect to decumbent | Erect to decumbent | Erect | Erect to decumbent | Erect to decumbent |
| Color of emerging leaves | Orange-brown to orange | Reddish-brown | Yellowish-brown | Caramel color | Green | Green to copper-green | Brown |
| Crown of leaves | Open | Closed | Open | Open | Open | Open | Open |
| Texture of leaflets | Coriaceous | Papyraceous | Papyraceous | Papyraceous | Papyraceous | Membranaceous | Coriaceous |
| Veins on abaxial surface of leaflets | Not visible | Not visible | Visible | Not visible | Not visible | Visible | Not visible |
| Apex of seed cone | Mucronate | Acuminate | Mucronate | Acuminate | Acuminate | Acute | Mucronate |
| Habit of mature seed cones | Erect | Erect | Erect | Erect | Erect | Erect | Pendulous |
| Peduncle of seed cone | Short | Short | Short | Short | Short | Short | Long |
| Key | Trait | F | p |
|---|---|---|---|
| A | Length of petiole | 9.12 | <0.0001 |
| B | Length of rachis | 10.58 | <0.0001 |
| C | Number of leaflet pairs | 17.43 | <0.0001 |
| D | Length of median leaflets | 2.052 | 0.08 |
| E | Distance between median leaflets | 15.13 | <0.0001 |
| F | Width of median leaflets | 14.05 | <0.0001 |
| G | Width of basal leaflets | 14.39 | <0.0001 |
| Trait | sch vs. aur | sch vs. rob | sch vs. sub | sch vs. dom | sch vs. oli | |
|---|---|---|---|---|---|---|
| A | Length of petiole | 5.49 * | 0.27 ns | 14.83 *** | 0.76 ns | 6.46 * |
| B | Length of rachis | 0.88 ns | 6.42 * | 1.55 ns | 9.77 ** | 49.16 *** |
| C | Number of leaflet pairs | 112.9 *** | 58.84 *** | 8.61 * | 17.1 *** | 2.32 ns |
| D | Length of median leaflets | 0.45 ns | 6.55 * | 0.00 ns | 0.01 ns | 11.27 * |
| E | Distance between median leaflets | 1.16 ns | 2.47 ns | 14.49 *** | 4.77 * | 32.33 *** |
| F | Width of median leaflets | 11.78 ** | 0.09 ns | 0.24 ns | 3.20 ns | 10.82 ** |
| G | Width of basal leaflets | 0.09 ns | 23.51 *** | 0 ns | 1.13 ns | 140.4 *** |
| aur | rob | sub | dom | oli | sch | Total | |
|---|---|---|---|---|---|---|---|
| aur | 13 | 1 | 0 | 2 | 0 | 0 | 16 |
| rob | 1 | 18 | 0 | 1 | 0 | 0 | 20 |
| sub | 0 | 0 | 11 | 0 | 0 | 0 | 11 |
| dom | 2 | 1 | 0 | 11 | 0 | 1 | 15 |
| oli | 0 | 0 | 0 | 0 | 9 | 0 | 9 |
| sch | 0 | 0 | 0 | 0 | 0 | 16 | 16 |
| Total | 16 | 20 | 11 | 14 | 9 | 17 | 87 |
| Aur | rob | sub | dom | oli | sch | |
|---|---|---|---|---|---|---|
| aur | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| rob | 7.8582 | <0.001 | <0.001 | <0.001 | <0.001 | |
| sub | 26.485 | 11.609 | <0.001 | 0.001 | <0.001 | |
| dom | 6.2325 | 9.7395 | 32.998 | <0.001 | <0.001 | |
| oli | 32.383 | 12.87 | 16.229 | 33.386 | <0.001 | |
| sch | 25.085 | 14.141 | 16.102 | 20.021 | 10.575 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Farrera, M.A.; Gutiérrez-Ortega, J.S.; Gregory, T.J.; Chemnick, J.; Salas-Morales, S.; Calonje, M.; Díaz-Jiménez, P. Ceratozamia schiblii (Zamiaceae): A New Cycad Species from the Eastern Mountains of Oaxaca, Mexico. Taxonomy 2022, 2, 324-338. https://doi.org/10.3390/taxonomy2030025
Pérez-Farrera MA, Gutiérrez-Ortega JS, Gregory TJ, Chemnick J, Salas-Morales S, Calonje M, Díaz-Jiménez P. Ceratozamia schiblii (Zamiaceae): A New Cycad Species from the Eastern Mountains of Oaxaca, Mexico. Taxonomy. 2022; 2(3):324-338. https://doi.org/10.3390/taxonomy2030025
Chicago/Turabian StylePérez-Farrera, Miguel Angel, José Said Gutiérrez-Ortega, Timothy J. Gregory, Jeffrey Chemnick, Silvia Salas-Morales, Michael Calonje, and Pedro Díaz-Jiménez. 2022. "Ceratozamia schiblii (Zamiaceae): A New Cycad Species from the Eastern Mountains of Oaxaca, Mexico" Taxonomy 2, no. 3: 324-338. https://doi.org/10.3390/taxonomy2030025
APA StylePérez-Farrera, M. A., Gutiérrez-Ortega, J. S., Gregory, T. J., Chemnick, J., Salas-Morales, S., Calonje, M., & Díaz-Jiménez, P. (2022). Ceratozamia schiblii (Zamiaceae): A New Cycad Species from the Eastern Mountains of Oaxaca, Mexico. Taxonomy, 2(3), 324-338. https://doi.org/10.3390/taxonomy2030025







