Enhanced Bacterial Cellulose Production Using Hempseed Meal: Optimal Conditions and Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Bacterial Cellulose Production
2.2.1. Culture Media Preparation
2.2.2. Culture Conditions
2.3. Analytical Methods of Culture Media During Fermentation
2.3.1. Sugar Analysis
2.3.2. Organic Acid Analysis
2.3.3. Total Phenolic and Flavonoid Content Analysis
2.3.4. pH and Total Soluble Solid Content (TSS) Analysis
2.4. Characterizations of Bacterial Cellulose
2.4.1. Mechanical Properties
2.4.2. Fourier Transform Infrared Spectroscopy (FT-IR)
2.4.3. X-Ray Diffraction (XRD)
2.4.4. Scanning Electron Microscopy (SEM)
2.4.5. Thermogravimetric Analysis (TGA)
2.4.6. Analysis of Bacterial Cellulose Functional Properties
2.5. Statistical Analysis
3. Results
3.1. The Bacterial Cellulose Production
3.1.1. Effects of pH, Organic Acids, and Phenolic Compounds on Bacterial Cellulose Production
3.1.2. The Change of Carbon Source During BC Fermentation
3.2. Bacterial Cellulose Characteristics
3.2.1. Morphology and Mechanical Properties
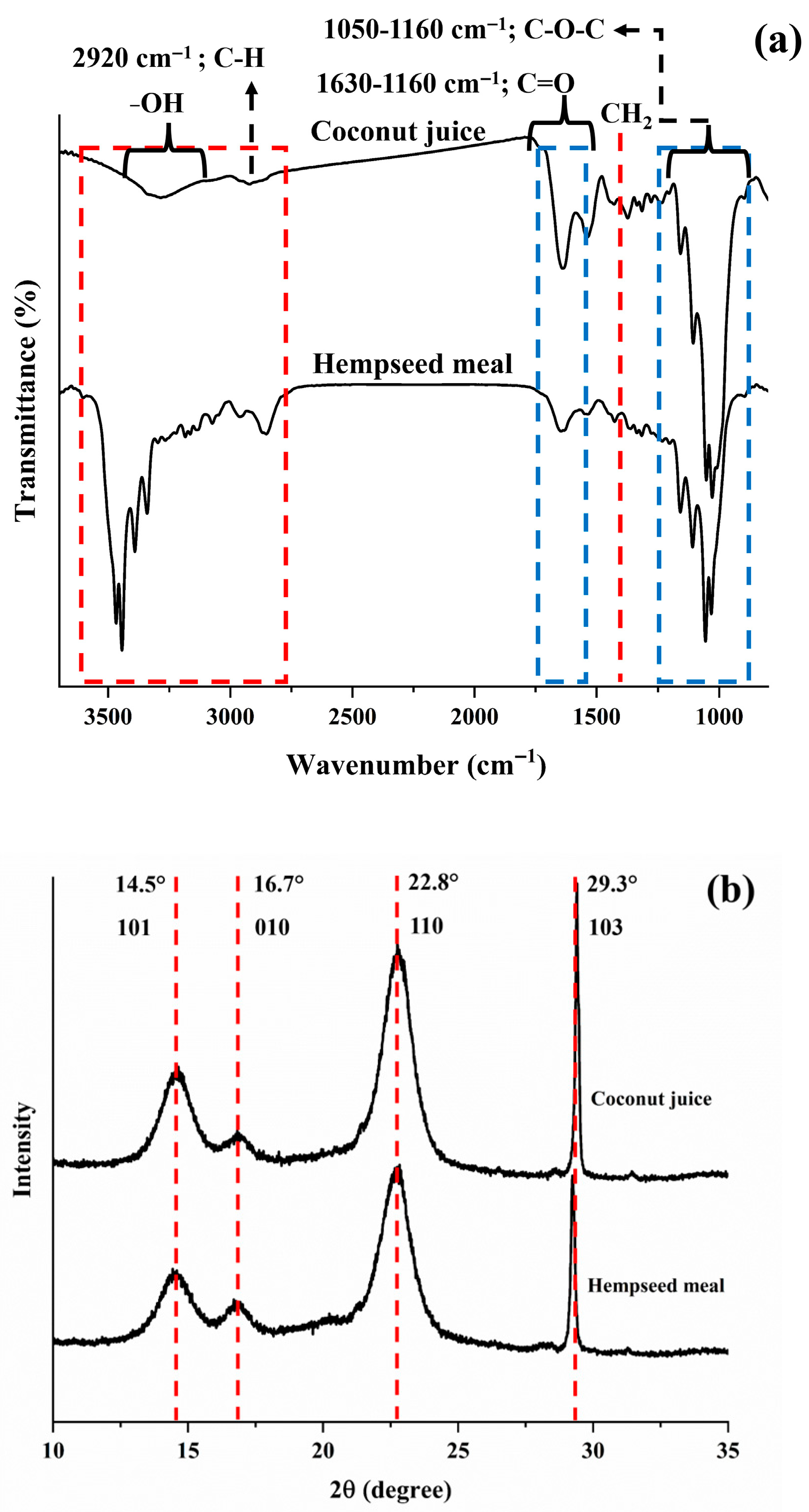
3.2.2. FT-IR and XRD Analysis
3.2.3. Functional and Thermal Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Montero, L.; Ballesteros-Vivas, D.; Gonzalez-Barrios, A.F.; Sánchez-Camargo, A.d.P. Hemp seeds: Nutritional value, associated bioactivities and the potential food applications in the Colombian context. Front. Nutr. 2023, 9, 1039180. [Google Scholar] [CrossRef]
- Singhania, R.R.; Patel, A.K.; Tseng, Y.-S.; Kumar, V.; Chen, C.-W.; Haldar, D.; Saini, J.K.; Dong, C.-D. Developments in bioprocess for bacterial cellulose production. Bioresour. Technol. 2022, 344, 126343. [Google Scholar] [CrossRef]
- Wang, Q.; Nnanna, P.C.; Shen, F.; Huang, M.; Tian, D.; Hu, J.; Zeng, Y.; Yang, G.; Deng, S. Full utilization of sweet sorghum for bacterial cellulose production: A concept of material crop. Ind. Crops Prod. 2021, 162, 113256. [Google Scholar] [CrossRef]
- Gao, H.; Sun, Q.; Han, Z.; Li, J.; Liao, B.; Hu, L.; Huang, J.; Zou, C.; Jia, C.; Huang, J.; et al. Comparison of bacterial nanocellulose produced by different strains under static and agitated culture conditions. Carbohydr. Polym. 2020, 227, 115323. [Google Scholar] [CrossRef] [PubMed]
- Pääkkönen, T.; Spiliopoulos, P.; Nonappa, N.; Kontturi, K.S.; Penttilä, P.; Viljanen, M.; Svedström, K.; Kontturi, E. Sustainable High Yield Route to Cellulose Nanocrystals from Bacterial Cellulose. ACS Sustain. Chem. Eng. 2019, 7, 14384–14388. [Google Scholar] [CrossRef]
- Esa, F.; Tasirin, S.M.; Rahman, N.A. Overview of Bacterial Cellulose Production and Application. Agric. Agric. Sci. Procedia 2014, 2, 113–119. [Google Scholar] [CrossRef]
- Hur, D.H.; Choi, W.S.; Kim, T.Y.; Lee, S.Y.; Park, J.H.; Jeong, K.J. Enhanced Production of Bacterial Cellulose in Komagataeibacter xylinus via Tuning of Biosynthesis Genes with Synthetic RBS. J. Microbiol. Biotechnol. 2020, 30, 1430–1435. [Google Scholar] [CrossRef]
- Cakar, F.; Ozer, I.; Aytekin, A.; Sahin, F. Improvement production of bacterial cellulose by semi-continuous process in molasses medium. Carbohydr. Polym. 2014, 106, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Gama, F. Bacterial Nanocellulose, From Biotechnology to Bio-Economy; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Przygrodzka, K.; Charęza, M.; Banaszek, A.; Zielińska, B.; Ekiert, E.; Drozd, R. Bacterial Cellulose Production by Komagateibacter xylinus with the Use of Enzyme-Degraded Oligo- and Polysaccharides as the Substrates. Appl. Sci. 2022, 12, 12673. [Google Scholar] [CrossRef]
- Gullo, M.; La China, S.; Falcone, P.M.; Giudici, P. Biotechnological production of cellulose by acetic acid bacteria: Current state and perspectives. Appl. Microbiol. Biotechnol. 2018, 102, 6885–6898. [Google Scholar] [CrossRef]
- Kongruang, S. Bacterial cellulose production by Acetobacter xylinum strains from agricultural waste products. Appl. Biochem. Biotechnol. 2008, 148, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tavakoli, J.; Tang, Y. Bacterial cellulose production, properties and applications with different culture methods—A review. Carbohydr. Polym. 2019, 219, 63–76. [Google Scholar] [CrossRef]
- Mohammadkazemi, F.; Azin, M.; Ashori, A. Production of bacterial cellulose using different carbon sources and culture media. Carbohydr. Polym. 2015, 117, 518–523. [Google Scholar] [CrossRef]
- Kaur, P.; Singh, S.; Ghoshal, G.; Ramamurthy, P.; Parihar, P.; Singh, J.; Singh, A. Valorization of Agri-Food Industry Waste for the Production of Microbial Pigments: An Eco-Friendly Approach; Springer: Berlin/Heidelberg, Germany, 2022; pp. 137–167. [Google Scholar]
- Radočaj, O.; Dimić, E.; Tsao, R. Effects of hemp (Cannabis sativa L.) seed oil press-cake and decaffeinated green tea leaves (Camellia sinensis) on functional characteristics of gluten-free crackers. J. Food Sci. 2014, 79, C318–C325. [Google Scholar] [CrossRef]
- Kauffmann, A.C.; Castro, V.S. Phenolic Compounds in Bacterial Inactivation: A Perspective from Brazil. Antibiotics 2023, 12, 645. [Google Scholar] [CrossRef] [PubMed]
- Mollica, D.; Thanthithum, C.; Chaves Lopez, C.; Prakitchaiwattana, C. Bioconversion of mangosteen pericarp extract juice with bacterial cellulose production and probiotic integration: A promising approach for functional food supplement development. Food Biosci. 2024, 61, 104549. [Google Scholar] [CrossRef]
- Masaoka, S.; Ohe, T.; Sakota, N. Production of cellulose from glucose by Acetobacter xylinum. J. Ferment. Bioeng. 1993, 75, 18–22. [Google Scholar] [CrossRef]
- Izquierdo-Llopart, A.; Carretero, A.; Saurina, J. Organic Acid Profiling by Liquid Chromatography for the Characterization of Base Vines and Sparkling Wines. Food Anal. Methods 2020, 13, 1852–1866. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Ligor, M.; Kornysova, O.; Maruska, A.; Buszewski, B. Determination of Flavonoids in Tea and Rooibos Extracts by TLC and HPLC. JPC-J. Planar Chromatogr.-Mod. TLC 2008, 21, 355–360. [Google Scholar] [CrossRef]
- Montenegro-Silva, P.; Ellis, T.; Dourado, F.; Gama, M.; Domingues, L. Enhanced bacterial cellulose production in Komagataeibacter sucrofermentans: Impact of different PQQ-dependent dehydrogenase knockouts and ethanol supplementation. Biotechnol. Biofuels Bioprod. 2024, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Song, Z.; Hao, Y.; Hu, X.; Lin, X.; Liu, S.; Li, C. Effect of co-culture of Komagataeibacter nataicola and selected Lactobacillus fermentum on the production and characterization of bacterial cellulose. LWT 2023, 173, 114224. [Google Scholar] [CrossRef]
- Arserim-Uçar, D.K.; Korel, F.; Liu, L.; Yam, K.L. Characterization of bacterial cellulose nanocrystals: Effect of acid treatments and neutralization. Food Chem. 2021, 336, 127597. [Google Scholar] [CrossRef]
- Nie, W.; Zheng, X.; Feng, W.; Liu, Y.; Li, Y.; Liang, X. Characterization of bacterial cellulose produced by Acetobacter pasteurianus MGC-N8819 utilizing lotus rhizome. LWT 2022, 165, 113763. [Google Scholar] [CrossRef]
- Narh, C.; Frimpong, C.; Mensah, A.; Wei, Q. Rice bran, an alternative nitrogen source for Acetobacter xylinum bacterial cellulose synthesis. Bioresources 2018, 13, 4346–4363. [Google Scholar] [CrossRef]
- Li, J.; Fu, J.; Ma, Y.; He, Y.; Fu, R.; Qayum, A.; Jiang, Z.; Wang, L. Low temperature extrusion promotes transglutaminase cross-linking of whey protein isolate and enhances its emulsifying properties and water holding capacity. Food Hydrocoll. 2022, 125, 107410. [Google Scholar] [CrossRef]
- Ashaolu, T.J.; Zhao, G. Fabricating a Pickering Stabilizer from Okara Dietary Fibre Particulates by Conjugating with Soy Protein Isolate via Maillard Reaction. Foods 2020, 9, 143. [Google Scholar] [CrossRef]
- Chawla, P.; Bajaj, I.; Survase, S.; Singhal, R. Microbial Cellulose: Fermentative Production and Applications. Food Technol. Biotechnol. 2009, 47, 107–124. [Google Scholar]
- Akil, M.; Biak, D.; Zainuddin, H. Production of Bacteria Cellulose from Hydrolysate of Banana, Sago, and Potato Wastes. Open Biotechnol. J. 2025, 19, e18740707372946. [Google Scholar] [CrossRef]
- Keshk, S.M.A.S. Vitamin C enhances bacterial cellulose production in Gluconacetobacter xylinus. Carbohydr. Polym. 2014, 99, 98–100. [Google Scholar] [CrossRef]
- Raghavendran, V.; Asare, E.; Roy, I. Chapter Three—Bacterial cellulose: Biosynthesis, production, and applications. In Advances in Microbial Physiology; Poole, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 77, pp. 89–138. [Google Scholar]
- He, F.; Yang, H.; Zeng, L.; Hu, H.; Hu, C. Production and characterization of bacterial cellulose obtained by Gluconacetobacter xylinus utilizing the by-products from Baijiu production. Bioprocess Biosyst. Eng. 2020, 43, 927–936. [Google Scholar] [CrossRef]
- Zhao, H.; Xia, J.; Wang, J.; Yan, X.; Wang, C.; Lei, T.; Xian, M.; Zhang, H. Production of bacterial cellulose using polysaccharide fermentation wastewater as inexpensive nutrient sources. Biotechnol. Biotechnol. Equip. 2018, 32, 350–356. [Google Scholar] [CrossRef]
- Liu, M.; Li, S.; Xie, Y.; Jia, S.; Hou, Y.; Zou, Y.; Zhong, C. Enhanced bacterial cellulose production by Gluconacetobacter xylinus via expression of Vitreoscilla hemoglobin and oxygen tension regulation. Appl. Microbiol. Biotechnol. 2018, 102, 1155–1165. [Google Scholar] [CrossRef]
- Digel, I.; Akimbekov, N.; Rogachev, E.; Pogorelova, N. Bacterial cellulose produced by Medusomyces gisevii on glucose and sucrose: Biosynthesis and structural properties. Cellulose 2023, 30, 11439–11453. [Google Scholar] [CrossRef]
- Li, Z.; Chen, S.Q.; Cao, X.; Li, L.; Zhu, J.; Yu, H. Effect of pH Buffer and Carbon Metabolism on the Yield and Mechanical Properties of Bacterial Cellulose Produced by Komagataeibacter hansenii ATCC 53582. J. Microbiol. Biotechnol. 2021, 31, 429–438. [Google Scholar] [CrossRef]
- Shah, N.; Ul-Islam, M.; Khattak, W.A.; Park, J.K. Overview of bacterial cellulose composites: A multipurpose advanced material. Carbohydr. Polym. 2013, 98, 1585–1598. [Google Scholar] [CrossRef]
- White, C.E.; Gavina, J.M.A.; Morton, R.; Britz-McKibbin, P.; Finan, T.M. Control of hydroxyproline catabolism in Sinorhizobium meliloti. Mol. Microbiol. 2012, 85, 1133–1147. [Google Scholar] [CrossRef] [PubMed]
- Deppenmeier, U.; Ehrenreich, A. Physiology of acetic acid bacteria in light of the genome sequence of Gluconobacter oxydans. J. Mol. Microbiol. Biotechnol. 2009, 16, 69–80. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Xie, Z.; Zhang, H.; Liebl, W.; Toyama, H.; Chen, F. Oxidative Fermentation of Acetic Acid Bacteria and Its Products. Front. Microbiol. 2022, 13, 879246. [Google Scholar] [CrossRef] [PubMed]
- de Souza, E.R.P.; Braz, M.; Castro, R.N.; Pereira, M.D.; Riger, C.J. Influence of microbial fermentation on the antioxidant activity of phenolic substances in Saccharomyces cerevisiae. J. Appl. Microbiol. 2023, 134, lxad148. [Google Scholar] [CrossRef]
- Enderle, J.D. Chapter 8—Biochemical Reactions and Enzyme Kinetics. In Introduction to Biomedical Engineering, 3rd ed.; Enderle, J.D., Bronzino, J.D., Eds.; Academic Press: Boston, MA, USA, 2012; pp. 447–508. [Google Scholar]
- Ahmad, M.; Wolberg, A.; Kahwaji, C.I. Biochemistry, Electron Transport Chain; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Tzin, V.; Galili, G.; Aharoni, A. Shikimate Pathway and Aromatic Amino Acid Biosynthesis. eLS 2012, 8, 32. [Google Scholar] [CrossRef]
- Zhan, J. Biosynthesis of bacterial aromatic polyketides. Curr. Top. Med. Chem. 2009, 9, 1598–1610. [Google Scholar] [CrossRef] [PubMed]
- Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef]
- Lou, H.; Hu, L.; Lu, H.; Wei, T.; Chen, Q. Metabolic Engineering of Microbial Cell Factories for Biosynthesis of Flavonoids: A Review. Molecules 2021, 26, 4522. [Google Scholar] [CrossRef]
- Nair, A.; Sarma, S.J. The impact of carbon and nitrogen catabolite repression in microorganisms. Microbiol. Res. 2021, 251, 126831. [Google Scholar] [CrossRef]
- Nadeem, H.; Rashid, M.H.; Siddique, M.H.; Azeem, F.; Muzammil, S.; Javed, M.R.; Ali, M.A.; Rasul, I.; Riaz, M. Microbial invertases: A review on kinetics, thermodynamics, physiochemical properties. Process Biochem. 2015, 50, 1202–1210. [Google Scholar] [CrossRef]
- Kierans, S.; Taylor, C. Glycolysis: A multifaceted metabolic pathway and signaling hub. J. Biol. Chem. 2024, 300, 107906. [Google Scholar] [CrossRef]
- Nocek, B.; Stein, A.J.; Jedrzejczak, R.; Cuff, M.E.; Li, H.; Volkart, L.; Joachimiak, A. Structural studies of ROK fructokinase YdhR from Bacillus subtilis: Insights into substrate binding and fructose specificity. J. Mol. Biol. 2011, 406, 325–342. [Google Scholar] [CrossRef]
- van Heeswijk, W.C.; Westerhoff, H.V.; Boogerd, F.C. Nitrogen assimilation in Escherichia coli: Putting molecular data into a systems perspective. Microbiol. Mol. Biol. Rev. 2013, 77, 628–695. [Google Scholar] [CrossRef] [PubMed]
- Vigentini, I.; Fabrizio, V.; Dellacà, F.; Rossi, S.; Azario, I.; Mondin, C.; Benaglia, M.; Foschino, R. Set-Up of Bacterial Cellulose Production From the Genus Komagataeibacter and Its Use in a Gluten-Free Bakery Product as a Case Study. Front. Microbiol. 2019, 10, 1953. [Google Scholar] [CrossRef]
- Hebeish, A.; Higazy, A.; El-Shafei, A.; Sharaf, S. Synthesis of carboxymethyl cellulose (CMC) and starch-based hybrids and their applications in flocculation and sizing. Carbohydr. Polym. 2010, 79, 60–69. [Google Scholar] [CrossRef]
- Fan, S.; Zhang, Z.; Duncan, G.J.; Morris, A.; Scobbie, L.; Henderson, D.; Morrice, P.; Russell, W.R.; Duncan, S.H.; Neacsu, M. Bioprocessing of Hempseed (Cannabis sativa L.) Food By-Products Increased Nutrient and Phytochemical In Vitro Bioavailability during Digestion and Microbial Fermentation. Appl. Sci. 2023, 13, 5781. [Google Scholar] [CrossRef]
- Torres, N.; Tobón-Cornejo, S.; Velazquez-Villegas, L.A.; Noriega, L.G.; Alemán-Escondrillas, G.; Tovar, A.R. Amino Acid Catabolism: An Overlooked Area of Metabolism. Nutrients 2023, 15, 3378. [Google Scholar] [CrossRef] [PubMed]
- Karabulut, G.; Feng, H.; Yemis, O. Physicochemical and Antioxidant Properties of Industrial Hemp Seed Protein Isolate Treated by High-Intensity Ultrasound. Plant Foods Hum. Nutr. 2022, 77, 3. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yuan, H.; Wang, H.; Yu, H. Coproduction of bacterial cellulose and pear vinegar by fermentation of pear peel and pomace. Bioprocess Biosyst. Eng. 2021, 44, 2231–2244. [Google Scholar] [CrossRef]
- Oliveira, R.; Vieira, J.; Barud, H.; Assunção, R.; Filho, G.; Ribeiro, S.; Messadeqq, Y. Synthesis and Characterization of Methylcellulose Produced from Bacterial Cellulose under Heterogeneous Condition. J. Braz. Chem. Soc. 2015, 26, 1861–1870. [Google Scholar] [CrossRef]
- Sharma, A.; Thakre, S.; Kumaraswamy, G. Microstructural differences between Viscose and Lyocell revealed by in-situ studies of wet and dry fibers. Cellulose 2020, 27, 1195–1206. [Google Scholar] [CrossRef]
- Dasari, M.; Rajasekaran, P.R.; Iyer, R.; Kohli, P. Calligraphic solar cells: Acknowledging paper and pencil. J. Mater. Res. 2016, 31, 2578–2589. [Google Scholar] [CrossRef]
- Pang, J.; Mehandzhiyski, A.; Zozoulenko, I. A computational study of cellulose regeneration: Coarse-grained molecular dynamics simulations. Carbohydr. Polym. 2023, 313, 120853. [Google Scholar] [CrossRef]
- Imai, M.; Horikawa, Y.; Kiyoto, S.; Imai, T.; Sugiyama, J. Structural changes in sugarcane bagasse cellulose caused by enzymatic hydrolysis. J. Wood Sci. 2020, 66, 11. [Google Scholar] [CrossRef]
- Amorim, L.; Li, L.; Gomes, A.; Fangueiro, R.; Gouveia, I. Sustainable bacterial cellulose production by low cost feedstock: Evaluation of apple and tea by-products as alternative sources of nutrients. Cellulose 2023, 30, 5589–5606. [Google Scholar] [CrossRef]
- Islam, M.U.; Ullah, M.W.; Khan, S.; Shah, N.; Park, J.K. Strategies for cost-effective and enhanced production of bacterial cellulose. Int. J. Biol. Macromol. 2017, 102, 1166–1173. [Google Scholar] [CrossRef]
- Ul-Islam, M.; Khan, T.; Park, J.K. Water holding and release properties of bacterial cellulose obtained by in situ and ex situ modification. Carbohydr. Polym. 2012, 88, 596–603. [Google Scholar] [CrossRef]
- Lin, S.-P.; Huang, Y.-H.; Hsu, K.-D.; Lai, Y.-J.; Chen, Y.-K.; Cheng, K.-C. Isolation and identification of cellulose-producing strain Komagataeibacter intermedius from fermented fruit juice. Carbohydr. Polym. 2016, 151, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Ciolacu, D.; Ciolacu, F.; Popa, V. Amorphous cellulose—Structure and characterization. Cellul. Chem. Technol. 2011, 45, 13–21. [Google Scholar]
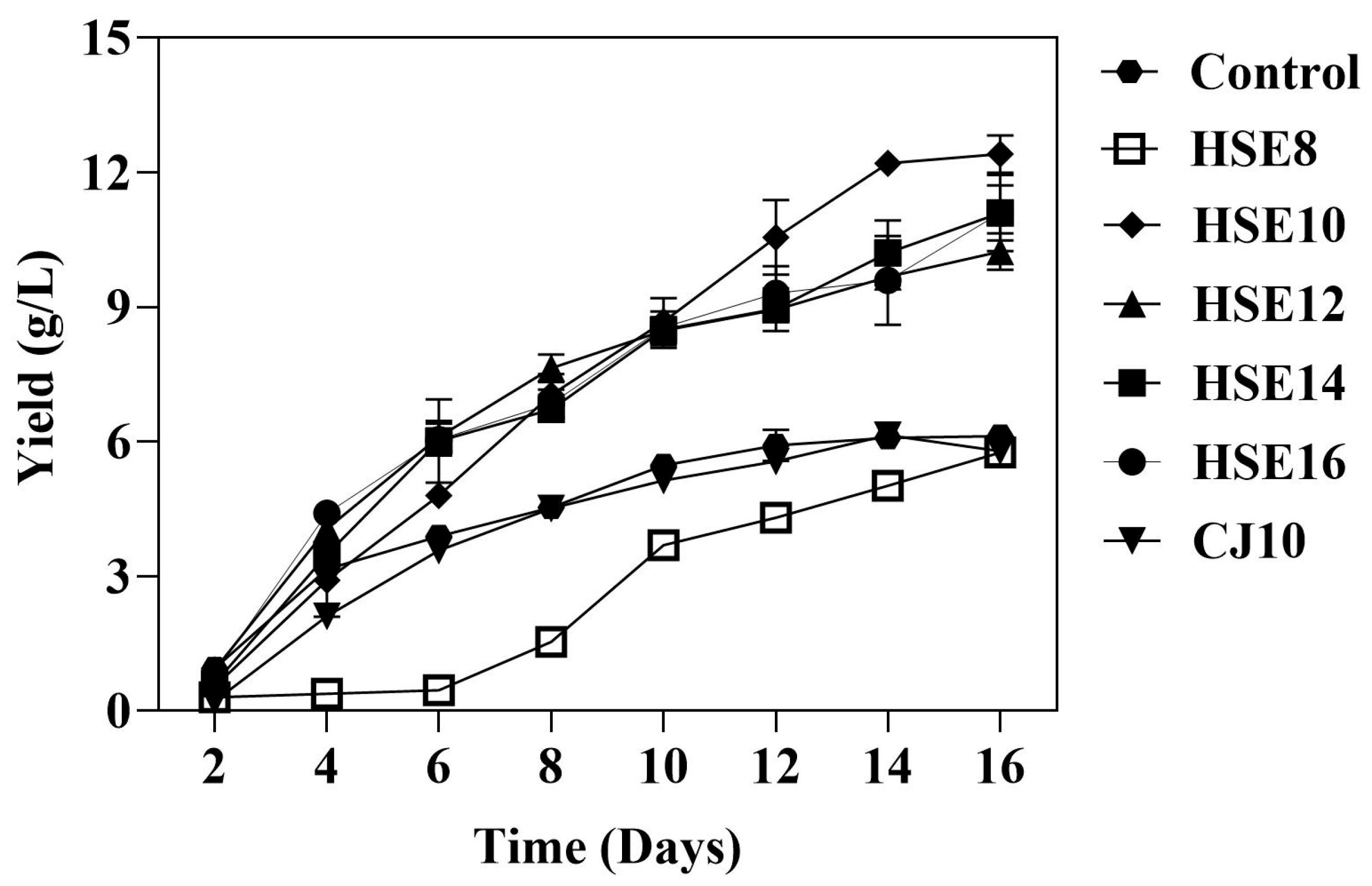
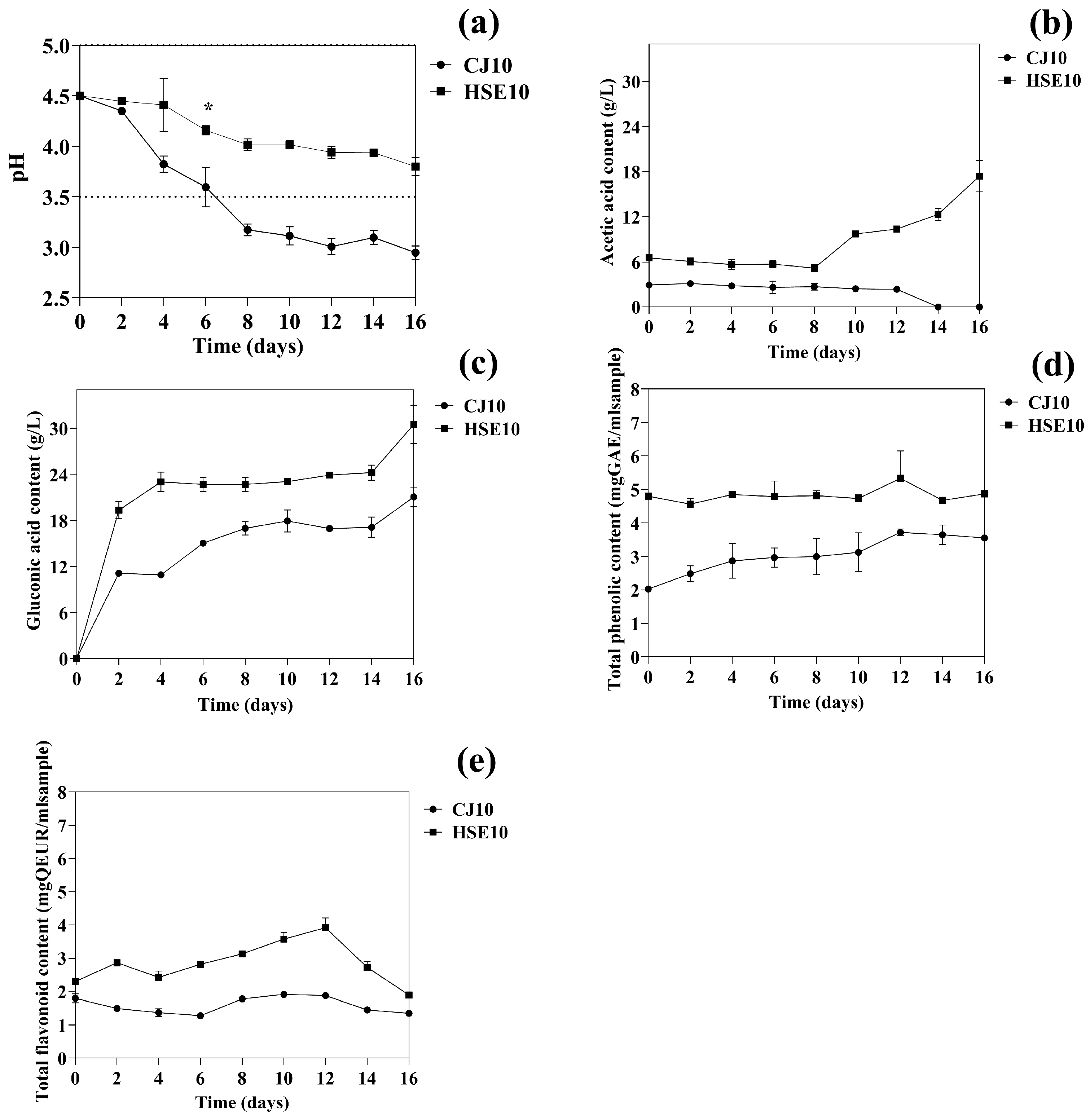
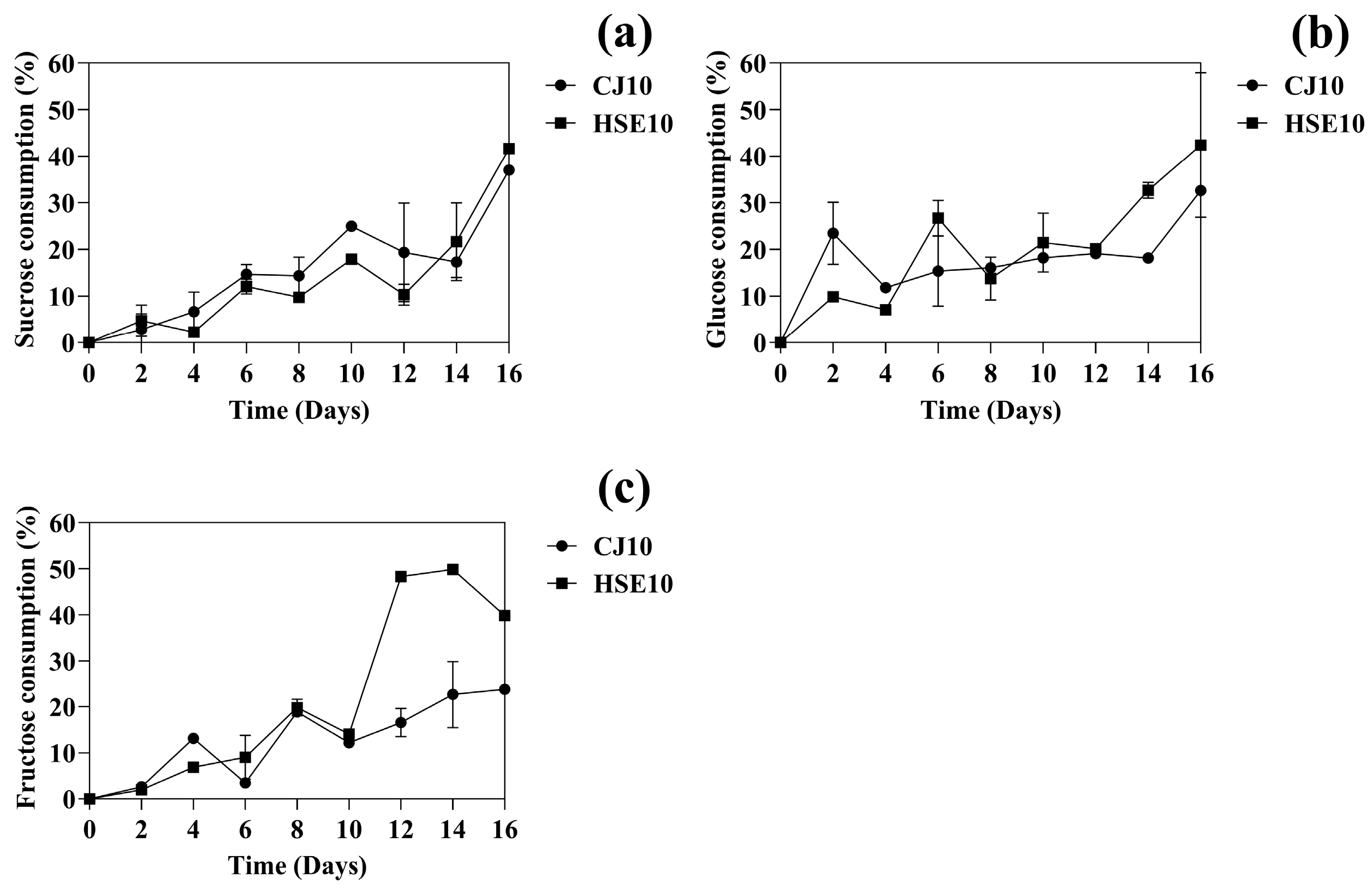
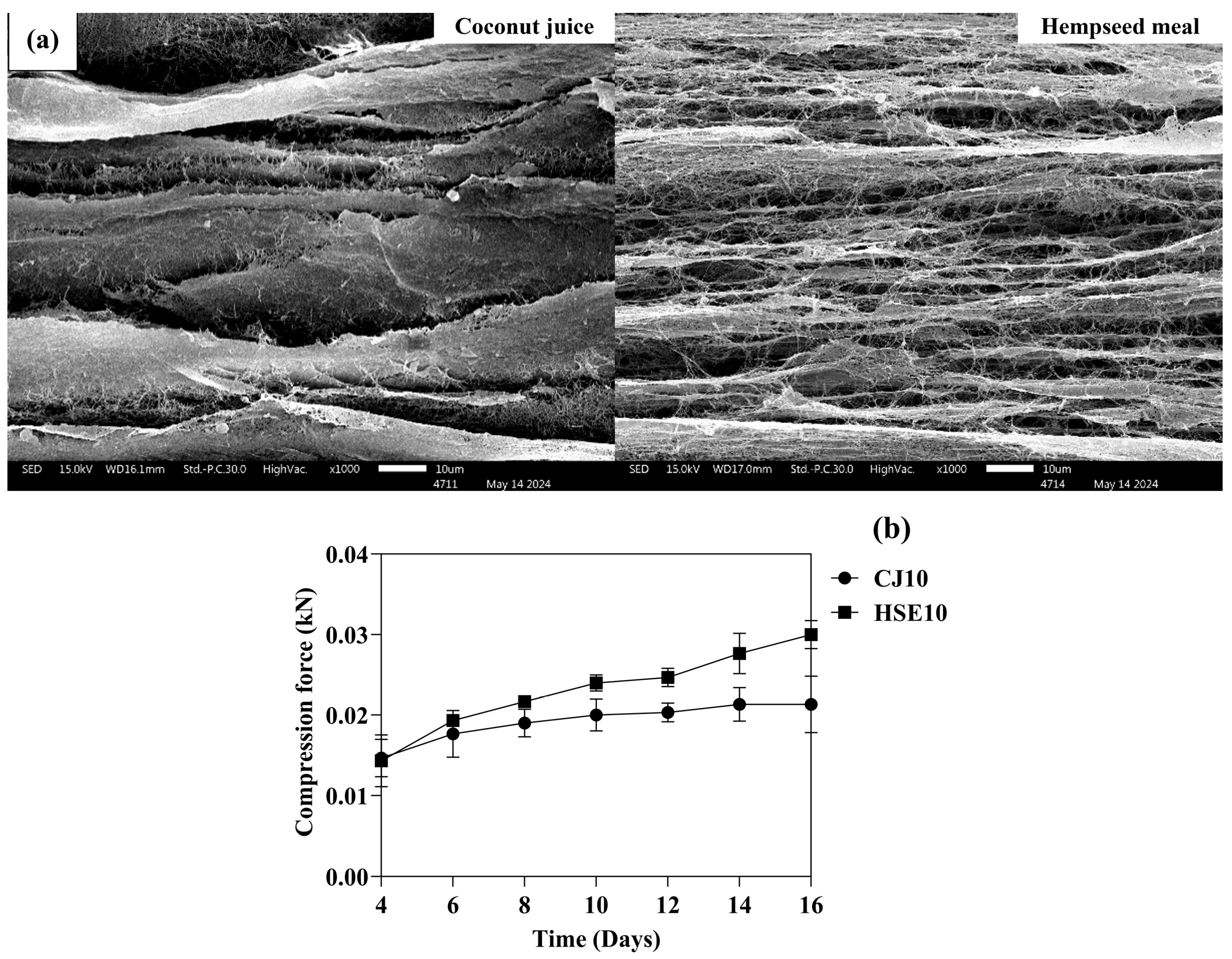
| Strain Used | Duration (Days) | Carbon Source | Nitrogen Source | BC Yield (g/L) | Reference |
|---|---|---|---|---|---|
| K. natacola | 14 | Hempseed meal | Yeast extract | 12.41 | This study |
| K. xylinus G29 | 7 | Reducing sugar, acetate and lactate | Peptone and yeast extract | 7.4 | [35] |
| K. xylinus BC-11 | 10 | Reducing sugars | Peptone and yeast extract | 1.2 | [36] |
| Gluconacetobacter xylinus | 14 | Glucose | Peptone and yeast extract | 4.3 | [37] |
| Acetobacter xylinum ATCC 23767 | 6 | Sweet sorghum | Peptone and yeast extract | 2.54 | [3] |
| Functional Properties | BCh | BCc |
|---|---|---|
| WHC (%, w/v) | 11.21 ± 0.31 a | 10.54 ± 0.45 a |
| OWC (%, w/v) | 2.71 ± 0.05 a | 3.86 ± 0.13 b |
| EA (%, v/v) | 34.33 ± 0.34 b | 29.19 ± 0.07 a |
| ES (%, v/v) | 39.11 ± 0.39 a | 38.36 ± 0.89 a |
| Degradation temperature (°C) | 358.58 ± 0.09 a | 366.41 ± 0.34 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orpool, S.; Kamthai, S.; Siriwoharn, T.; Khaw-on, P.; Deenu, A.; Naruenartwongsakul, S. Enhanced Bacterial Cellulose Production Using Hempseed Meal: Optimal Conditions and Properties. BioTech 2025, 14, 66. https://doi.org/10.3390/biotech14030066
Orpool S, Kamthai S, Siriwoharn T, Khaw-on P, Deenu A, Naruenartwongsakul S. Enhanced Bacterial Cellulose Production Using Hempseed Meal: Optimal Conditions and Properties. BioTech. 2025; 14(3):66. https://doi.org/10.3390/biotech14030066
Chicago/Turabian StyleOrpool, Sawichaya, Suthaphat Kamthai, Thanyaporn Siriwoharn, Patompong Khaw-on, Aree Deenu, and Srisuwan Naruenartwongsakul. 2025. "Enhanced Bacterial Cellulose Production Using Hempseed Meal: Optimal Conditions and Properties" BioTech 14, no. 3: 66. https://doi.org/10.3390/biotech14030066
APA StyleOrpool, S., Kamthai, S., Siriwoharn, T., Khaw-on, P., Deenu, A., & Naruenartwongsakul, S. (2025). Enhanced Bacterial Cellulose Production Using Hempseed Meal: Optimal Conditions and Properties. BioTech, 14(3), 66. https://doi.org/10.3390/biotech14030066







