Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment
Abstract
:1. Introduction
2. Methods
2.1. Process Description and Simulation
2.1.1. Membrane Process
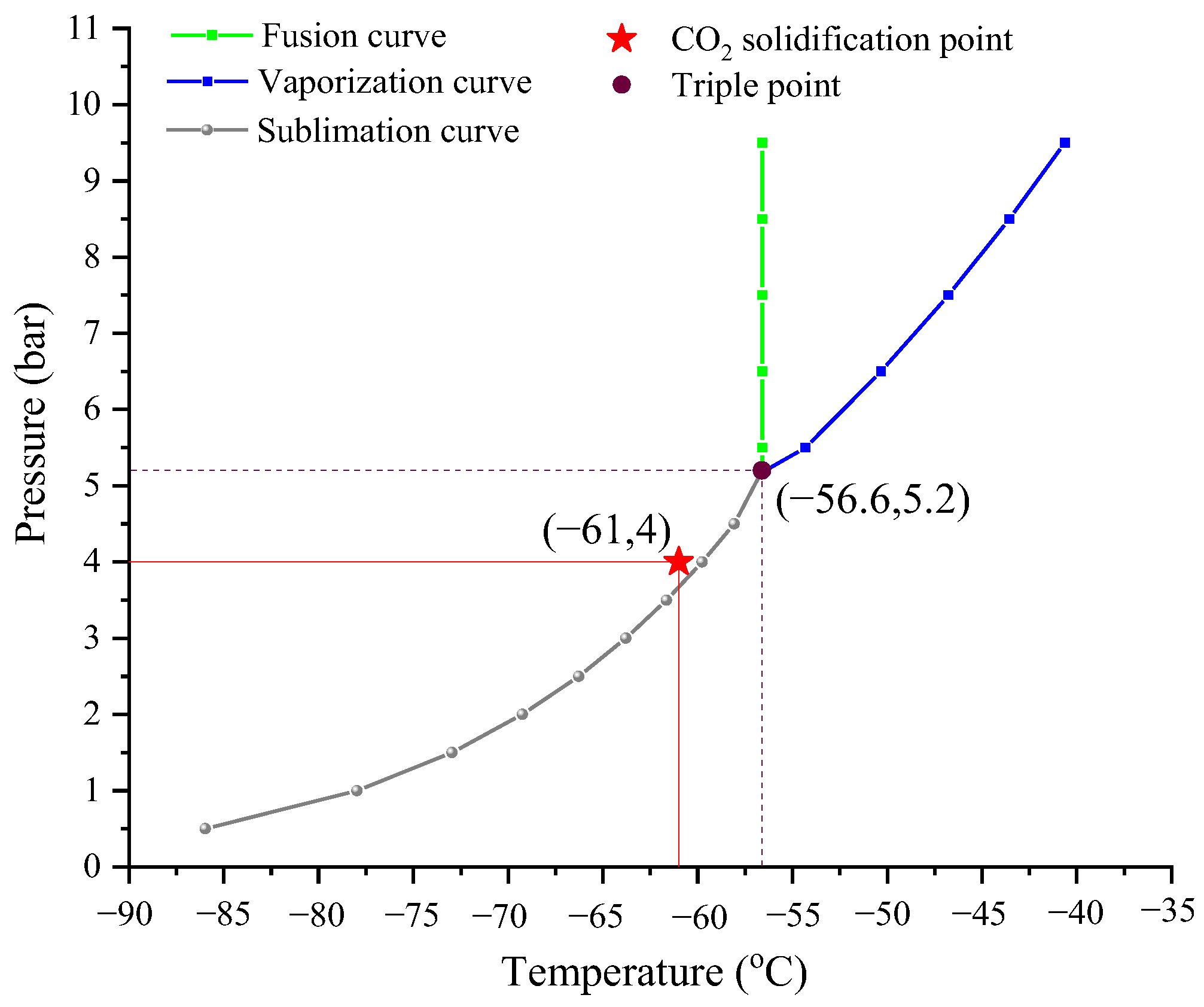
2.1.2. CO2 Solidification Process
2.1.3. Process Simulation
- No experimental data are available on the amount of CO2 that solidifies under the operating conditions; a 100% solidification rate is assumed.
- The compressors in the refrigeration cycle are operated with a compression ratio of ≤3.0.
- The pressure drop across all the coolers and heat exchangers is considered to be negligible.
- The efficiencies of the compressors, pumps, and expanders are maintained at 75%.
- The minimum internal approach temperature (MITA) for the multi-stream heat exchanger is maintained at 1.0–2.0 °C.
- The heat loss is considered to be negligible.
2.2. Process Optimization
2.3. Energy Analysis
2.3.1. Design Variable Analysis
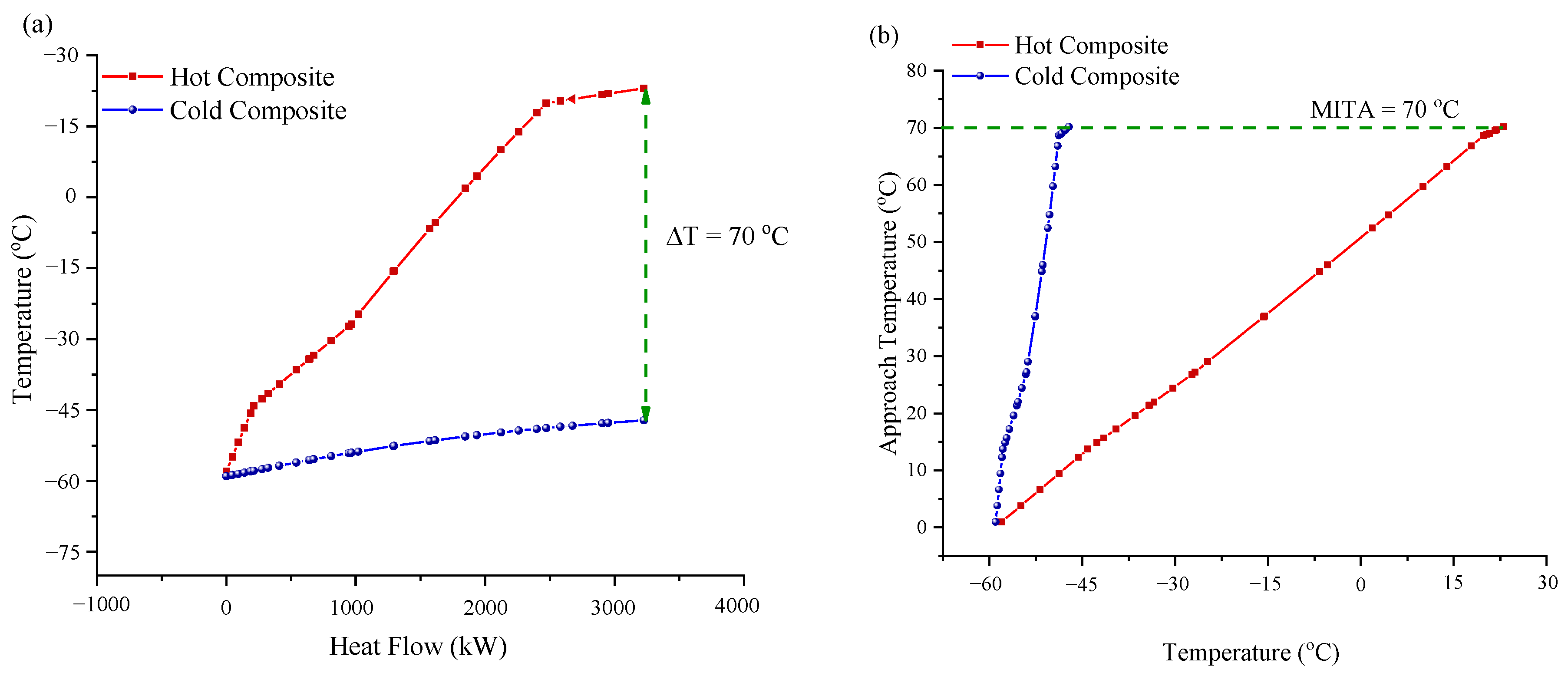
2.3.2. Composite Curve Analysis
2.4. Exergy Analysis
2.5. Economic Analysis
3. Results and Discussion: Process Analysis
3.1. Energy Analysis
3.1.1. Design Variables Analysis
3.1.2. Composite Curve (CC) Analysis
3.2. Exergy Analysis
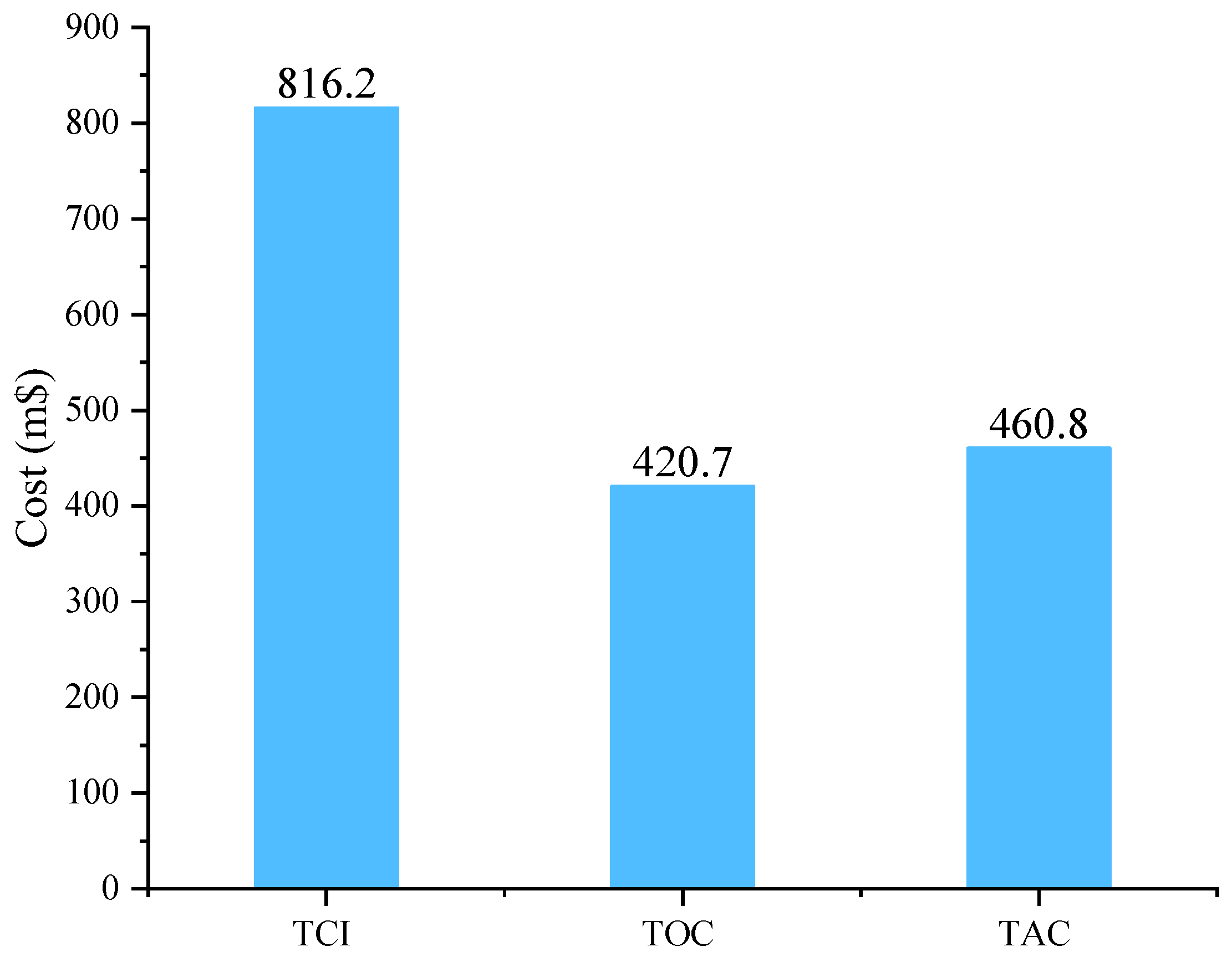
3.3. Economic Analysis
3.4. Comparison with Conceptual Studies
4. Conclusions and Future Work
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IEA. CO2 Emissions in 2022. Available online: https://www.iea.org/reports/co2-emissions-in-2022 (accessed on 14 June 2023).
- Gourinchas, P.-O. Global Economic Growth Slows Amid Gloomy and More Uncertain Outlook 2023. Available online: https://www.imf.org/en/Blogs/Articles/2022/07/26/blog-weo-update-july-2022 (accessed on 14 June 2023).
- Qyyum, M.A.; Dickson, R.; Shah, S.F.A.; Niaz, H.; Khan, A.; Liu, J.J.; Lee, M. Availability, versatility, and viability of feedstocks for hydrogen production: Product space perspective. Renew. Sustain. Energy Rev. 2021, 145, 110843. [Google Scholar] [CrossRef]
- Osman, A.I.; Mehta, N.; Elgarahy, A.M.; Hefny, M.; Al-Hinai, A.; Al-Muhtaseb, A.H. Hydrogen production, storage, utilisation and environmental impacts: A review. Environ. Chem. Lett. 2021, 20, 153–188. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Hydrogen Program Plan 2020. Available online: www.hydrogen.energy.gov/pdfs/hydrogen-program-plan-2020.pdf (accessed on 14 June 2023).
- Stenina, I.; Yaroslavtsev, A. Modern Technologies of Hydrogen Production. Process 2022, 11, 56. [Google Scholar] [CrossRef]
- Ishaq, H.; Dincer, I.; Crawford, C. A review on hydrogen production and utilization: Challenges and opportunities. Int. J. Hydrog. Energy 2021, 47, 26238–26264. [Google Scholar] [CrossRef]
- Younas, M.; Shafique, S.; Hafeez, A.; Javed, F.; Rehman, F. An Overview of Hydrogen Production: Current Status, Potential, and Challenges. Fuel 2022, 316, 123317. [Google Scholar] [CrossRef]
- Du, Z.; Liu, C.; Zhai, J.; Guo, X.; Xiong, Y.; Su, W.; He, G. A review of hydrogen purification technologies for fuel cell vehicles. Catalysts 2021, 11, 393. [Google Scholar] [CrossRef]
- Wickham, D.; Hawkes, A.; Jalil-Vega, F. Hydrogen supply chain optimisation for the transport sector—Focus on hydrogen purity and purification requirements. Appl. Energy 2022, 305, 117740. [Google Scholar] [CrossRef]
- Genovese, M.; Cigolotti, V.; Jannelli, E.; Fragiacomo, P. Current standards and configurations for the permitting and operation of hydrogen refueling stations. Int. J. Hydrog. Energy 2023, 48, 19357–19371. [Google Scholar] [CrossRef]
- Vermaak, L.; Neomagus, H.W.J.P.; Bessarabov, D.G. Recent advances in membrane-based electrochemical hydrogen separation: A review. Membranes 2021, 11, 127. [Google Scholar] [CrossRef]
- Schorer, L.; Schmitz, S.; Weber, A. Membrane based purification of hydrogen system (MEMPHYS). Int. J. Hydrog. Energy 2019, 44, 12708–12714. [Google Scholar] [CrossRef]
- Lei, L.; Bai, L.; Lindbråthen, A.; Pan, F.; Zhang, X.; He, X. Carbon membranes for CO2 removal: Status and perspectives from materials to processes. Chem. Eng. J. 2020, 401, 126084. [Google Scholar] [CrossRef]
- Amosova, O.L.; Malykh, O.V.; Teplyakov, V.V. Integrated membrane/PSA systems for hydrogen recovery from gas mixtures. Desalin Water Treat. 2010, 14, 119–126. [Google Scholar] [CrossRef]
- Li, B.; He, G.; Jiang, X.; Dai, Y.; Ruan, X. Pressure swing adsorption/membrane hybrid processes for hydrogen purification with a high recovery. Front. Chem. Sci. Eng. 2016, 10, 255–264. [Google Scholar] [CrossRef]
- Ohs, B.; Falkenberg, M.; Wessling, M. Optimizing hybrid membrane-pressure swing adsorption processes for biogenic hydrogen recovery. Chem. Eng. J. 2019, 364, 452–461. [Google Scholar] [CrossRef]
- Lin, L.; Tian, Y.; Su, W.; Luo, Y.; Chen, C.; Jiang, L. Techno-economic analysis and comprehensive optimization of an on-site hydrogen refuelling station system using ammonia: Hybrid hydrogen purification with both high H2 purity and high recovery. Sustain. Energy Fuels 2020, 4, 3006–3017. [Google Scholar] [CrossRef]
- Ma, B.; Deng, C.; Chen, H.; Zhu, M.; Yang, M.; Feng, X. Hybrid Separation Process of Refinery Off-gas toward Near-Zero Hydrogen Emission: Conceptual Design and Techno-economic Analysis. Ind. Eng. Chem. Res. 2020, 59, 8715–8727. [Google Scholar] [CrossRef]
- Terrien, P.; Lockwood, F.; Granados, L.; Morel, T. CO2 capture from H2 plants: Implementation for, EOR. Energy Procedia 2014, 63, 7861–7866. [Google Scholar] [CrossRef] [Green Version]
- Pichot, D.; Granados, L.; Morel, T.; Schuller, A.; Dubettier, R.; Lockwood, F. Start-up of Port-Jérôme CRYOCAPTM Plant: Optimized Cryogenic CO2 Capture from H2 Plants. Energy Procedia 2017, 114, 2682–2689. [Google Scholar] [CrossRef]
- Van Acht, S.C.J.; Laycock, C.; Carr, S.J.W.; Maddy, J.; Guwy, A.J.; Lloyd, G.; Raymakers, L.F.J.M. Simulation of integrated novel PSA/EHP/C process for high-pressure hydrogen recovery from Coke Oven Gas. Int. J. Hydrog. Energy 2020, 45, 15196–15212. [Google Scholar] [CrossRef]
- Scholz, M. Membrane-Cryogenic Hybrid Processes. In Encyclopedia of Membranes; Drioli, E., Giorno, L., Eds.; Springer: Berlin/Heidelberg, Garmany, 2015. [Google Scholar] [CrossRef]
- Agrawal, R.; Auvil, S.R.; DiMartino, S.P.; Choe, J.S.; Hopkins, J.A. Membrane/cryogenic hybrid processes for hydrogen purification. Gas. Sep. Purif. 1988, 2, 9–15. [Google Scholar] [CrossRef]
- Baker, R.; Wijmans, J.; Kaschemekat, J. The design of membrane vapor–gas separation systems. J. Memb. Sci. 1998, 151, 55–62. [Google Scholar] [CrossRef]
- Baker, R.W.; Lokhandwala, K.A.; Pinnau, I. Ethylene/Nitrogen Separation Process. USOO5879431A, 9 March 1999. [Google Scholar]
- Lin, H.; He, Z.; Sun, Z.; Kniep, J.; Ng, A.; Baker, R.W.; Merkel, T.C. CO2-selective membranes for hydrogen production and CO2 capture—Part II: Techno-economic analysis. J. Memb. Sci. 2015, 493, 794–806. [Google Scholar] [CrossRef] [Green Version]
- Liao, Z.; Hu, Y.; Wang, J.; Yang, Y.; You, F. Systematic Design and Optimization of a Membrane–Cryogenic Hybrid System for CO 2 Capture. ACS Sustain. Chem. Eng. 2019, 7, 17186–17197. [Google Scholar] [CrossRef]
- Liao, Z.; Hu, Y.; Tu, G.; Sun, J.; Jiang, B.; Wang, J.; Yang, Y. Optimal design of hybrid cryogenic flash and membrane system. Chem. Eng. Sci. 2018, 179, 13–31. [Google Scholar] [CrossRef]
- Liu, B.; Yang, X.; Chiang, P.C.; Wang, T. Energy Consumption Analysis of Cryogenic-membrane Hybrid Process for CO2 Capture from CO2-EOR Extraction Gas. Aerosol Air Qual. Res. 2020, 20, 820–832. [Google Scholar] [CrossRef]
- Wankat, P.C.; Kostroski, K.P. Hybrid membrane-cryogenic distillation air separation process for oxygen production. Sep. Sci. Technol. 2011, 46, 1539–1545. [Google Scholar] [CrossRef]
- Burdyny, T.; Struchtrup, H. Hybrid membrane/cryogenic separation of oxygen from air for use in the oxy-fuel process. Energy 2010, 35, 1884–1897. [Google Scholar] [CrossRef]
- Park, J.; Kim, K.; Shin, J.-W.; Park, Y.-K. Analysis of Multistage Membrane and Distillation Hybrid Processes for Propylene/Propane Separation. Chem. Eng. Trans. 2019, 74, 871–876. [Google Scholar] [CrossRef]
- Xu, G.; Li, L.; Yang, Y.; Tian, L.; Liu, T.; Zhang, K. A novel CO2 cryogenic liquefaction and separation system. Energy 2012, 42, 522–529. [Google Scholar] [CrossRef]
- Wukovits, W.; Chudzicki, M.; Makaruk, A.; Friedl, A. Simulation Study on the Applicability and Performance of Conventional and Reverse-Selective Membranes for Upgrading of H2/CO2 Mixtures via Gas-Permeation. Chem. Eng. Trans. 2012, 29, 1171–1176. [Google Scholar] [CrossRef]
- Riaz, A.; Chaniago, Y.D.; Hussain, A.; Andika, R.; Kim, G.; Lim, H.; Lee, M. Thermodynamic, economic, and emissions assessment of integrated power to methanol concept with membrane-based biogas up-gradation and plasma electrolysis. J. Clean. Prod. 2022, 363, 132367. [Google Scholar] [CrossRef]
- Harlacher, T.; Melin, T.; Wessling, M. Techno-economic Analysis of Membrane-Based Argon Recovery in a Silicon Carbide Process. Ind. Eng. Chem. Res. 2013, 52, 10460–10466. [Google Scholar] [CrossRef]
- Huang, W.; Jiang, X.; He, G.; Ruan, X.; Chen, B.; Nizamani, A.K.; Li, X.; Wu, X.; Xiao, W. A Novel Process of H2/CO2 Membrane Separation of Shifted Syngas Coupled with Gasoil Hydrogenation. Processes 2020, 8, 590. [Google Scholar] [CrossRef]
- Naquash, A.; Haider, J.; Qyyum, M.A.; Islam, M.; Min, S.; Lee, S.; Lim, H.; Lee, M. Hydrogen enrichment by CO2 anti-sublimation integrated with triple mixed refrigerant-based liquid hydrogen production process. J. Clean. Prod. 2022, 341, 130745. [Google Scholar] [CrossRef]
- Naquash, A.; Qyyum, M.A.; Haider, J.; Lim, H.; Lee, M. Renewable LNG production: Biogas upgrading through CO2 solidification integrated with single-loop mixed refrigerant biomethane liquefaction process. Energy Convers. Manag. 2021, 243, 114363. [Google Scholar] [CrossRef]
- Yurata, T.; Lei, H.; Tang, L.; Lu, M.; Patel, J.; Lim, S.; Piumsomboon, P.; Chalermsinsuwan, B.; Li, C. Feasibility and sustainability analyses of carbon dioxide—Hydrogen separation via de-sublimation process in comparison with other processes. Int. J. Hydrog. Energy 2019, 44, 23120–23134. [Google Scholar] [CrossRef]
- Peng, D.Y.; Robinson, D.B. A New Two-Constant Equation of State. Ind. Eng. Chem. Fundam. 1976, 15, 59–64. [Google Scholar] [CrossRef]
- Lopez-Echeverry, J.S.; Reif-Acherman, S.; Araujo-Lopez, E. Peng-Robinson equation of state: 40 years through cubics. Fluid. Phase Equilib. 2017, 447, 39–71. [Google Scholar] [CrossRef]
- Aasadnia, M.; Mehrpooya, M. Large-scale liquid hydrogen production methods and approaches: A review. Appl. Energy 2018, 212, 57–83. [Google Scholar] [CrossRef]
- Qyyum, M.A.; Riaz, A.; Naquash, A.; Haider, J.; Qadeer, K.; Nawaz, A.; Lee, H.; Lee, M. 100% saturated liquid hydrogen production: Mixed-refrigerant cascaded process with two-stage ortho-to-para hydrogen conversion. Energy Convers. Manag. 2021, 246, 114659. [Google Scholar] [CrossRef]
- Naquash, A.; Qyyum, M.A.; Chaniago, Y.D.; Riaz, A.; Yehia, F.; Lim, H.; Lee, M. Separation and purification of syngas-derived hydrogen: A comparative evaluation of membrane- and cryogenic-assisted approaches. Chemosphere 2023, 313, 137420. [Google Scholar] [CrossRef] [PubMed]
- Richard, T. Analysis, Synthesis, and Design of Chemical Processes; Prentice Hall: London, UK, 2012. [Google Scholar]
- Ahmad, F.; Lau, K.K.; Shariff, A.M.; Murshid, G. Process simulation and optimal design of membrane separation system for CO2 capture from natural gas. Comput. Chem. Eng. 2012, 36, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Xe.com. Euros to USD n.d. Available online: https://www.xe.com/ (accessed on 13 June 2023).


| Feed Conditions | Values |
|---|---|
| Vapor/Phase Fraction | 1.00 |
| Temperature (°C) | 35.00 |
| Pressure (bar) | 5.00 |
| Mass Flow (kg/s) | 100.00 |
| H2 (mol%) | 0.20 |
| Carbon Dioxide (mol%) | 0.80 |
| Refrigerants Conditions | Values |
|---|---|
| C2 (mol%) | 25.33 |
| C3 (mol%) | 74.67 |
| Suction Pressure (bar) | 1.18 |
| Discharge Pressure (bar) | 14.40 |
| Temperature (°C) | 23.02 |
| Stream ID | CO2 | H2 | C2 | C3 |
|---|---|---|---|---|
| Feed | 0.8000 | 0.2000 | 0 | 0 |
| CO2-m | 0.9908 | 0.0092 | 0 | 0 |
| H2-m | 0.9996 | 0.0004 | 0 | 0 |
| 14 | 0 | 0 | 0.2533 | 0.7467 |
| H2 | 0 | 1.000 | 0 | 0 |
| sCO2 | 1.000 | 0 | 0 | 0 |
| Equipment | Exergy Destruction Equations |
|---|---|
| Compressors | |
| Expanders | |
| Pumps | |
| Phase separators | |
| Heat exchangers | |
| Cryogenic heat exchanger | |
| Cold chamber | |
| Membranes |
| Section | Variables | Values |
|---|---|---|
| Membrane Separation | High pressure (bar) | 10.0 |
| Low pressure (bar) | 0.10 | |
| Mem-1 area (m2) | 9000 | |
| Mem-2 area (m2) | 500 | |
| Mem-3 area (m2) | 1000 | |
| CO2 purity (%) | 99.08 | |
| CO2 recovery (%) | 98.13 | |
| De-sublimation cycle | C2 (kg/s) | 2.305 |
| C3 (kg/s) | 9.962 | |
| Discharge Pressure (bar) | 14.40 | |
| Suction Pressure (bar) | 1.177 | |
| Desublimation temperature (°C) | −61 | |
| Desublimation pressure (bar) | 4.00 | |
| CHX-1 MITA (°C) | 1.00 | |
| H2 purity (%) | 99.96 | |
| H2 recovery (%) | 95.9 |
| Membrane section SEC (kWh/kg) | 2.26 |
| Desublimation section SEC (kWh/kg) | 0.11 |
| Net SEC (kWh/kg) | 2.37 |
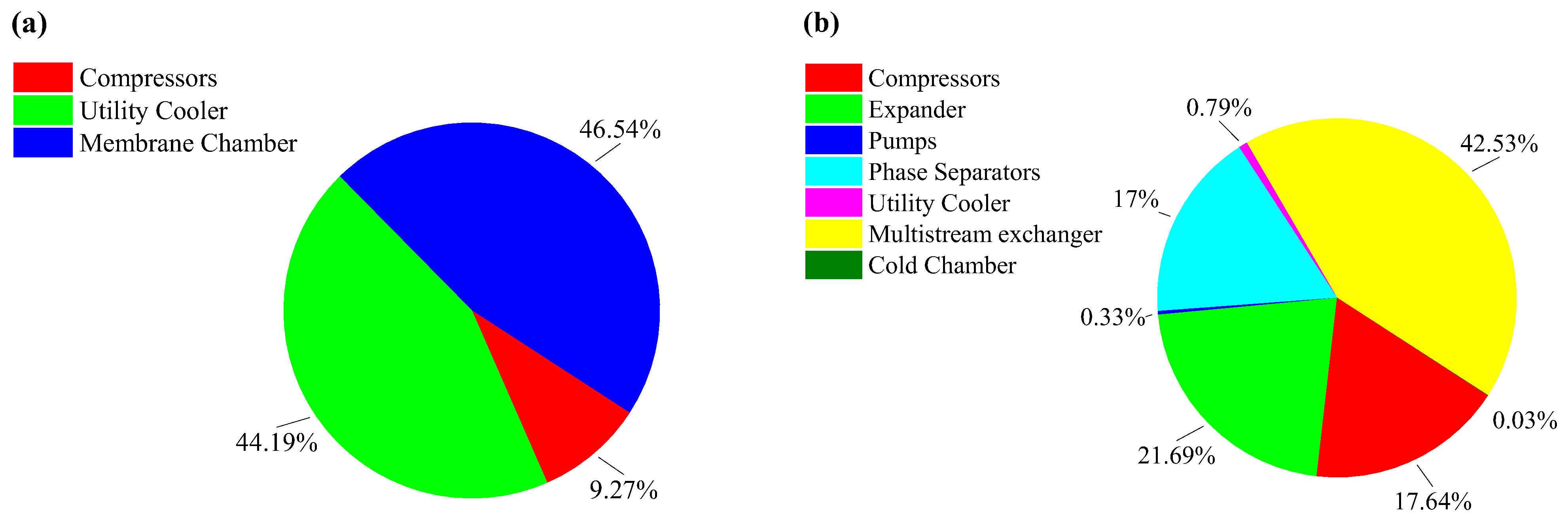
| Section | Equipment | Exergy Destruction (kW) |
|---|---|---|
| Membrane section | Compressors | 77,274.1 |
| Coolers | 368,514.5 | |
| Membranes | 388,083.5 | |
| Desublimation section | Compressors | 287.4 |
| Expanders | 353.5 | |
| Pumps | 5.3 | |
| Vessels | 277.1 | |
| Coolers | 12.8 | |
| CHX-1 | 693.2 | |
| Cold box | 0.5 |
| Technology | Product Conditions | Energy Requirements | Cost | Ref. |
|---|---|---|---|---|
| Membrane–PSA | H2 purity: 99.98 % H2 recovery: 97% | N/A | N/A | [16] |
| Membrane–PSA | H2 purity: 99.0 % H2 recovery: 98% | Energy efficiency: 77.5% | H2 production cost: 0.6 €/kg (0.65 $/kg) * | [17] |
| Cryogenic–Membrane | H2 purity: 90.4% H2 recovery: 98% | Energy consumption: 36.6 MW | N/A | [27] |
| Cryogenic–Membrane | H2 purity: 90% H2 recovery: 98% | SEC: 0.546 GJ/ton CO2 | TAC: 40.9 m$/y | [28] |
| This study (Membrane–Cryogenic) | H2 purity: 99% H2 recovery: 96% | SEC: 2.37 kWh/kg | TAC: 460.8 m$/y |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naquash, A.; Riaz, A.; Yehia, F.; Chaniago, Y.D.; Lim, H.; Lee, M. Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment. Gases 2023, 3, 92-105. https://doi.org/10.3390/gases3030006
Naquash A, Riaz A, Yehia F, Chaniago YD, Lim H, Lee M. Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment. Gases. 2023; 3(3):92-105. https://doi.org/10.3390/gases3030006
Chicago/Turabian StyleNaquash, Ahmad, Amjad Riaz, Fatma Yehia, Yus Donald Chaniago, Hankwon Lim, and Moonyong Lee. 2023. "Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment" Gases 3, no. 3: 92-105. https://doi.org/10.3390/gases3030006
APA StyleNaquash, A., Riaz, A., Yehia, F., Chaniago, Y. D., Lim, H., & Lee, M. (2023). Hydrogen Purification through a Membrane–Cryogenic Integrated Process: A 3 E’s (Energy, Exergy, and Economic) Assessment. Gases, 3(3), 92-105. https://doi.org/10.3390/gases3030006









