Concomitant Rare KRAS and BRAF Mutations in Lung Adenocarcinoma: A Case Report
Abstract
1. Introduction
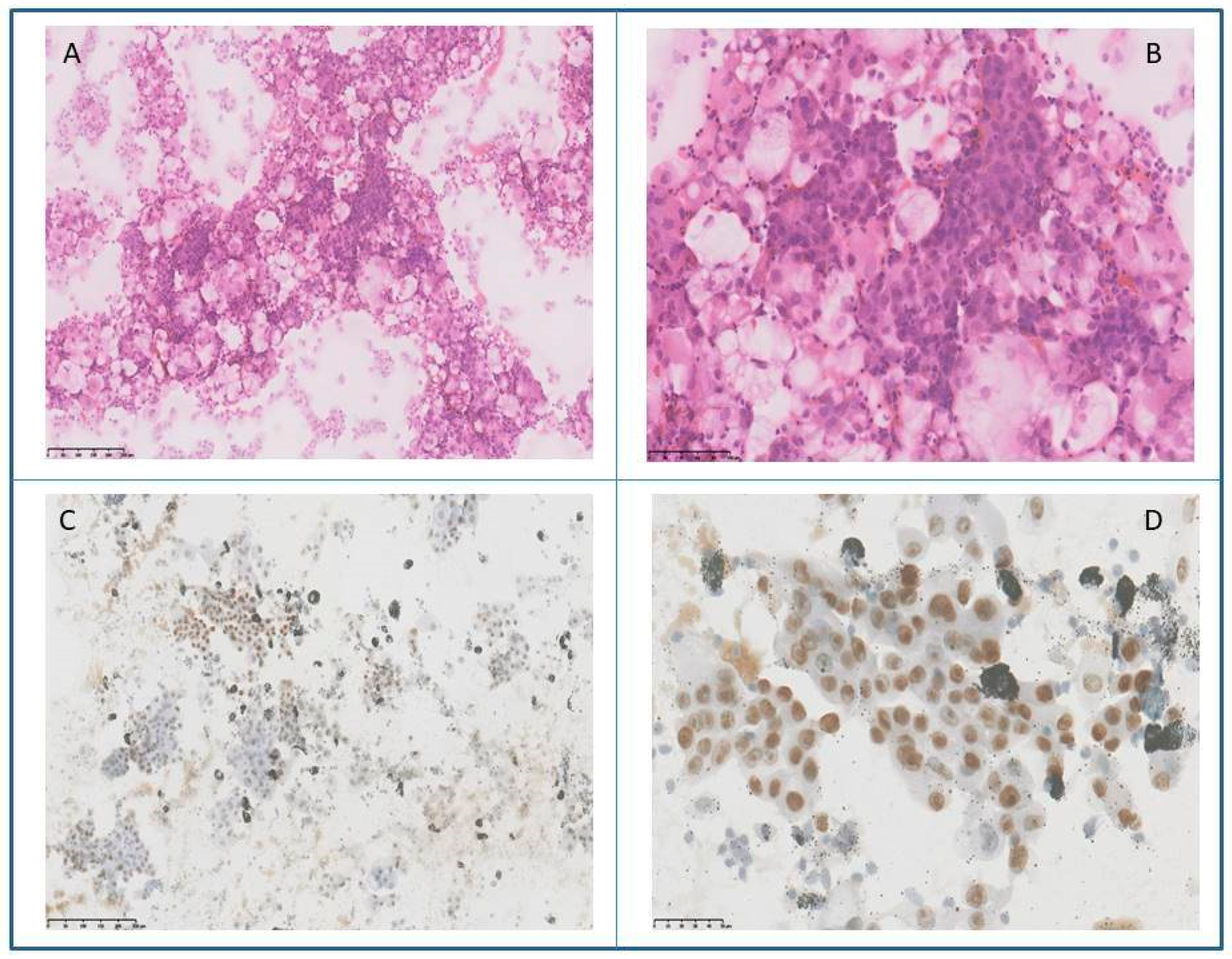
2. Case Report
3. Ethical Statement
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Malapelle, U.; Bellevicine, C.; De Luca, C.; Salatiello, M.; De Stefano, A.; Rocco, D.; de Rosa, N.; Vitiello, F.; Russo, S.; Pepe, F.; et al. EGFR mutations detected on cytology samples by a centralized laboratory reliably predict response to gefitinib in non-small cell lung carcinoma patients. Cancer Cytopathol. 2013, 121, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, N.I.; Cagle, P.T.; Aisner, D.L.; Arcila, M.E.; Beasley, M.B.; Bernicker, E.H.; Colasacco, C.; Dacic, S.; Hirsch, F.R.; Kerr, K.; et al. Updated Molecular Testing Guideline for the Selection of Lung Cancer Patients for Treatment With Targeted Tyrosine Kinase Inhibitors: Guideline From the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. Arch. Pathol. Lab. Med. 2018, 142, 321–346. [Google Scholar] [PubMed]
- Kalemkerian, G.P.; Narula, N.; Kennedy, E.B.; Biermann, W.A.; Donington, J.; Leighl, N.B.; Lew, M.; Pantelas, J.; Ramalingam, S.S.; Reck, M.; et al. Molecular Testing Guideline for the Selection of Patients With Lung Cancer for Treatment With Targeted Tyrosine Kinase Inhibitors: American Society of Clinical Oncology Endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 911–919. [Google Scholar] [PubMed]
- Ettinger, D.S.; Aisner, D.L.; Wood, D.E.; Akerley, W.; Bauman, J.; Chang, J.Y.; Chirieac, L.R.; D’Amico, T.A.; Dilling, T.J.; Dobelbower, M.; et al. NCCN Guidelines Insights: Non-Small Cell Lung Cancer, Version 5.2018. J. Natl. Compr. Cancer Netw. 2018, 16, 807–821. [Google Scholar] [CrossRef] [PubMed]
- AMG 510 First to Inhibit “Undruggable” KRAS. Cancer Discov. 2019, 9, 988–989.
- Vigliar, E.; Malapelle, U.; de Luca, C.; Bellevicine, C.; Troncone, G. Challenges and opportunities of next-generation sequencing: A cytopathologist’s perspective. Cytopathology 2015, 26, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Malapelle, U.; Mayo de-Las-Casas, C.; Rocco, D.; Garzon, M.; Pisapia, P.; Jordana-Ariza, N.; Russo, M.; Sgariglia, R.; De Luca, C.; Pepe, F.; et al. Development of a gene panel for next-generation sequencing of clinically relevant mutations in cell-free DNA from cancer patients. Br. J. Cancer. 2017, 116, 802–810. [Google Scholar] [CrossRef]
- Pepe, F.; De Luca, C.; Smeraglio, R.; Pisapia, P.; Sgariglia, R.; Nacchio, M.; Russo, M.; Serra, N.; Rocco, D.; Battiloro, C.; et al. Performance analysis of SiRe next-generation sequencing panel in diagnostic setting: Focus on NSCLC routine samples. J. Clin. Pathol. 2019, 72, 38–45. [Google Scholar] [CrossRef]
- Pisapia, P.; Pepe, F.; Smeraglio, R.; Russo, M.; Rocco, D.; Sgariglia, R.; Nacchio, M.; De Luca, C.; Vigliar, E.; Bellevicine, C.; et al. Cell free DNA analysis by SiRe(®) next generation sequencing panel in non small cell lung cancer patients: Focus on basal setting. J. Thorac. Dis. 2017, 9 (Suppl. 13), S1383–S1390. [Google Scholar] [CrossRef]
- Pisapia, P.; Rocco, D.; Pepe, F.; De Luca, C.; Battiloro, C.; Smeraglio, R.; Cieri, M.; Bellevicine, C.; Troncone, G.; Malapelle, U. EGFR exon 19 deletion switch and development of p.L792Q mutation as a new resistance mechanism to osimertinib: A case report and literature review. Transl. Cancer Res. 2019, 8 (Suppl. 1), S64–S69. [Google Scholar] [CrossRef]
- Clery, E.; Pisapia, P.; Migliatico, I.; Pepe, F.; De Luca, C.; Russo, M.; De Rosa, F.; Smeraglia, F.; Insabato, L.; Vigliar, E.; et al. Cytology meets next generation sequencing and liquid biopsy: A case of lung adenocarcinoma presenting as metastasis to the phalanx. Diagn. Cytopathol. 2020, 48, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Malapelle, U.; Pepe, F.; Pisapia, P.; Sgariglia, R.; Nacchio, M.; De Luca, C.; Lacalamita, R.; Tommasi, S.; Pinto, R.; Palomba, G.; et al. Harmonization of Next-Generation Sequencing Procedure in Italian Laboratories: A Multi-Institutional Evaluation of the SiRe® Panel. Front Oncol. 2020, 10, 236. [Google Scholar] [CrossRef] [PubMed]
- Weyn, C.; Van Raemdonck, S.; Dendooven, R.; Maes, V.; Zwaenepoel, K.; Lambin, S.; Pauwels, P. Clinical performance evaluation of a sensitive, rapid low-throughput test for KRAS mutation analysis using formalin-fixed, paraffin-embedded tissue samples. BMC Cancer 2017, 17, 139. [Google Scholar] [CrossRef] [PubMed]
- Downward, J. Targeting RAS signalling pathways in cancer therapy. Nat. Rev. Cancer 2003, 3, 11–22. [Google Scholar] [CrossRef]
- Passiglia, F.; Malapelle, U.; Del Re, M.; Righi, L.; Pagni, F.; Furlan, D.; Danesi, R.; Troncone, G.; Novello, S. KRAS inhibition in non-small cell lung cancer: Past failures, new findings and upcoming challenges. Eur. J. Cancer 2020, 137, 57–68. [Google Scholar] [CrossRef]
- Dueling KRASG12C Inhibitors Achieve Responses. Cancer Discov. 2020, 10, 10. [CrossRef]
- AACR Project GENIE Consortium. AACR Project GENIE: Powering Precision Medicine through an International Consortium. Cancer Discov. 2017, 7, 818–831. [Google Scholar] [CrossRef]
- Janakiraman, M.; Vakiani, E.; Zeng, Z.; Pratilas, C.A.; Taylor, B.S.; Chitale, D.; Halilovic, E.; Wilson, M.; Huberman, K.; Ricarte Filho, J.C.; et al. Genomic and biological characterization of exon 4 KRAS mutations in human cancer. Cancer Res. 2010, 70, 5901–5911. [Google Scholar] [CrossRef] [PubMed]
- Poulin, E.J.; Bera, A.K.; Lu, J.; Lin, Y.J.; Strasser, S.D.; Paulo, J.A.; Huang, T.Q.; Morales, C.; Yan, W.; Cook, J.; et al. Tissue-Specific Oncogenic Activity of KRASA146T. Cancer Discov. 2019, 9, 738–755. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lv, Q.; Xu, Y.; Cai, Z.; Zheng, J.; Cheng, X.; Dai, Y.; Jänne, P.A.; Ambrogio, C.; Köhler, J. An integrative pharmacogenomics analysis identifies therapeutic targets in KRAS-mutant lung cancer. EBioMedicine 2019, 49, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Ruan, X.; Sun, Y.; Wang, W.; Ye, J.; Zhang, D.; Gong, Z.; Yang, M. Multiplexed molecular profiling of lung cancer with malignant pleural effusion using next generation sequencing in Chinese patients. Oncol. Lett. 2020, 19, 3495–3505. [Google Scholar] [CrossRef]
- Leonetti, A.; Facchinetti, F.; Rossi, G.; Minari, R.; Conti, A.; Friboulet, L.; Tiseo, M.; Planchard, D. BRAF in non-small cell lung cancer (NSCLC): Pickaxing another brick in the wall. Cancer Treat. Rev. 2018, 66, 82–94. [Google Scholar] [CrossRef]
- Pisapia, P.; Pepe, F.; Malapelle, U.; Troncone, G. BRAF Mutations in Lung Cancer. Acta Cytol. 2019, 63, 247–250. [Google Scholar] [CrossRef]
- Salimian, K.J.; Fazeli, R.; Zheng, G.; Ettinger, D.; Maleki, Z. V600E BRAF versus Non-V600E BRAF Mutated Lung Adenocarcinomas: Cytomorphology, Histology, Coexistence of Other Driver Mutations and Patient Characteristics. Acta Cytol. 2018, 62, 79–84. [Google Scholar] [CrossRef]
- Noeparast, A.; Teugels, E.; Giron, P.; Verschelden, G.; De Brakeleer, S.; Decoster, L.; De Grève, J. Non-V600 BRAF mutations recurrently found in lung cancer predict sensitivity to the combination of Trametinib and Dabrafenib. Oncotarget 2016, 8, 60094–60108. [Google Scholar] [CrossRef]
- Planchard, D.; Besse, B.; Groen, H.J.M.; Souquet, P.J.; Quoix, E.; Baik, C.S.; Barlesi, F.; Kim, T.M.; Mazieres, J.; Novello, S.; et al. Dabrafenib plus trametinib in patients with previously treated BRAF(V600E)-mutant metastatic non-small cell lung cancer: An open-label, multicentre phase 2 trial. Lancet Oncol. 2016, 17, 984–993. [Google Scholar] [CrossRef]
- Planchard, D.; Smit, E.F.; Groen, H.J.M.; Mazieres, J.; Besse, B.; Helland, Å.; Giannone, V.; D’Amelio AMJr Zhang, P.; Mookerjee, B.; Johnson, B.E. Dabrafenib plus trametinib in patients with previously untreated BRAFV600E-mutant metastatic non-small-cell lung cancer: An open-label, phase 2 trial. Lancet Oncol. 2017, 18, 1307–1316. [Google Scholar] [CrossRef]
- Villaruz, L.C.; Socinski, M.A.; Abberbock, S.; Berry, L.D.; Johnson, B.E.; Kwiatkowski, D.J.; Iafrate, A.J.; Varella-Garcia, M.; Franklin, W.A.; Camidge, D.R.; et al. Clinicopathologic features and outcomes of patients with lung adenocarcinomas harboring BRAF mutations in the Lung Cancer Mutation Consortium. Cancer 2015, 121, 448–456. [Google Scholar] [CrossRef]
- Illei, P.B.; Belchis, D.; Tseng, L.H.; Nguyen, D.; De Marchi, F.; Haley, L.; Riel, S.; Beierl, K.; Zheng, G.; Brahmer, J.R.; et al. Clinical mutational profiling of 1006 lung cancers by next generation sequencing. Oncotarget 2017, 8, 96684–96696. [Google Scholar] [CrossRef]
- Gautschi, O.; Milia, J.; Cabarrou, B.; Bluthgen, M.V.; Besse, B.; Smit, E.F.; Wolf, J.; Peters, S.; Früh, M.; Koeberle, D.; et al. Targeted Therapy for Patients with BRAF-Mutant Lung Cancer: Results from the European EURAF Cohort. J. Thorac. Oncol. 2015, 10, 1451–1457. [Google Scholar] [CrossRef]
- Sullivan, R.J.; Infante, J.R.; Janku, F.; Wong, D.J.L.; Sosman, J.A.; Keedy, V.; Patel, M.R.; Shapiro, G.I.; Mier, J.W.; Tolcher, A.W.; et al. First-in-Class ERK1/2 Inhibitor Ulixertinib (BVD-523) in Patients with MAPK Mutant Advanced Solid Tumors: Results of a Phase I Dose-Escalation and Expansion Study. Cancer Discov. 2018, 8, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R.J.; Hollebecque, A.; Flaherty, K.T.; Shapiro, G.I.; Rodon Ahnert, J.; Millward, M.J.; Zhang, W.; Gao, L.; Sykes, A.; Willard, M.D.; et al. A Phase I Study of LY3009120, a Pan-RAF Inhibitor, in Patients with Advanced or Metastatic Cancer. Mol. Cancer Ther. 2020, 19, 460–467. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iaccarino, A.; Pisapia, P.; De Felice, M.; Pepe, F.; Gragnano, G.; De Luca, C.; Ianniello, G.; Malapelle, U. Concomitant Rare KRAS and BRAF Mutations in Lung Adenocarcinoma: A Case Report. J. Mol. Pathol. 2020, 1, 36-42. https://doi.org/10.3390/jmp1010006
Iaccarino A, Pisapia P, De Felice M, Pepe F, Gragnano G, De Luca C, Ianniello G, Malapelle U. Concomitant Rare KRAS and BRAF Mutations in Lung Adenocarcinoma: A Case Report. Journal of Molecular Pathology. 2020; 1(1):36-42. https://doi.org/10.3390/jmp1010006
Chicago/Turabian StyleIaccarino, Antonino, Pasquale Pisapia, Marco De Felice, Francesco Pepe, Gianluca Gragnano, Caterina De Luca, Giovanni Ianniello, and Umberto Malapelle. 2020. "Concomitant Rare KRAS and BRAF Mutations in Lung Adenocarcinoma: A Case Report" Journal of Molecular Pathology 1, no. 1: 36-42. https://doi.org/10.3390/jmp1010006
APA StyleIaccarino, A., Pisapia, P., De Felice, M., Pepe, F., Gragnano, G., De Luca, C., Ianniello, G., & Malapelle, U. (2020). Concomitant Rare KRAS and BRAF Mutations in Lung Adenocarcinoma: A Case Report. Journal of Molecular Pathology, 1(1), 36-42. https://doi.org/10.3390/jmp1010006






