In Silico Modeling of the Interaction of NBD Steroids with Insect Steroid-Binding Protein SPC-2 †
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ciufo, L.F.; Murray, P.A.; Thompson, A.; Rigden, D.J.; Rees, H.H. Characterisation of a Desmosterol Reductase Involved in Phytosterol Dealkylation in the Silkworm, Bombyx mori. PLoS ONE 2011, 6, e21316. [Google Scholar] [CrossRef] [PubMed]
- Yoshiyama-Yanagawa, T.; Enya, S.; Shimada-Niwa, Y.; Yaguchi, S.; Haramoto, Y.; Matsuya, T.; Shiomi, K.; Sasakura, Y.; Takahashi, S.; Asashima, M.; et al. The Conserved Rieske Oxygenase DAF-36/Neverland Is a Novel Cholesterol-metabolizing Enzyme. J. Biol. Chem. 2011, 29, 25756–25762. [Google Scholar] [CrossRef] [PubMed]
- Satoko, H.-S.; Takashi, S. Cell surface control of the layer specific targeting in the Drosophila visual system. Genes Genet. Syst. 2014, 1, 9–15. [Google Scholar] [CrossRef]
- Ma, H.; Ma, Y.; Liu, X.; Dyer, D.H.; Xu, P.; Liu, K.; Lan, Q.; Hong, H.; Peng, J.; Peng, R. NMR structure and function of Helicoverpa armigera sterol carrier protein-2, an important insecticidal target from the cotton bollworm. Sci. Rep. 2015, 5, 18186. [Google Scholar] [CrossRef] [PubMed]
- Minsik, K.; Vilena, W.; Que, L. Identification of mosquito sterol carrier protein-2 inhibitors. Nat. Lib. Med. 2005, 46, 650–657. [Google Scholar] [CrossRef]
- Faletrov, Y.V.; Pozniak, H.I.; Yakovets, P.S.; Frolova, N.S.; Shkumatov, V.M. New lipophilic conjugates of fluorescent NBD-piperazine: Synthesis, in silico interactions with lipid bilayer and cytochromes P450. Proc. Natl. Acad. Sci. Belarus Chem. Ser. 2022, 58, 62–67. [Google Scholar] [CrossRef]
- Lomize, A.L.; Pogozheva, I.D.; Mosberg, H.I. Anisotropic solvent model of the lipid bilayer. 2. Energetics of insertion of small molecules, peptides, and proteins in membranes. J. Chem. Inform. Mod. 2011, 51, 930–946. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Faletrov, Y.V.; Gilep, K.A.; Falchevskaya, A.S.; Horetski, M.S.; Panada, J.V.; Andrievskaya, E.V.; Rudaya, E.V.; Frolova, N.S.; Brzostek, A.; Plocinska, R.; et al. In silico modeling of izoniazid steroid conjugates interactions with cytochromes P450 of mycobacteria and their bioconversion in vitro by the cells. Biomed. Khimiya 2020, 66, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Faletrov, Y.V.; Staravoitava, V.A.; Dudko, A.R.; Shkumatov, V.M. Application of docking-based inverse high throughput virtual screening to found phytochemical covalent inhibitors of SARS-CoV-2 main protease, NSP12 and NSP16. Res. Square 2022, preprint. [Google Scholar] [CrossRef]
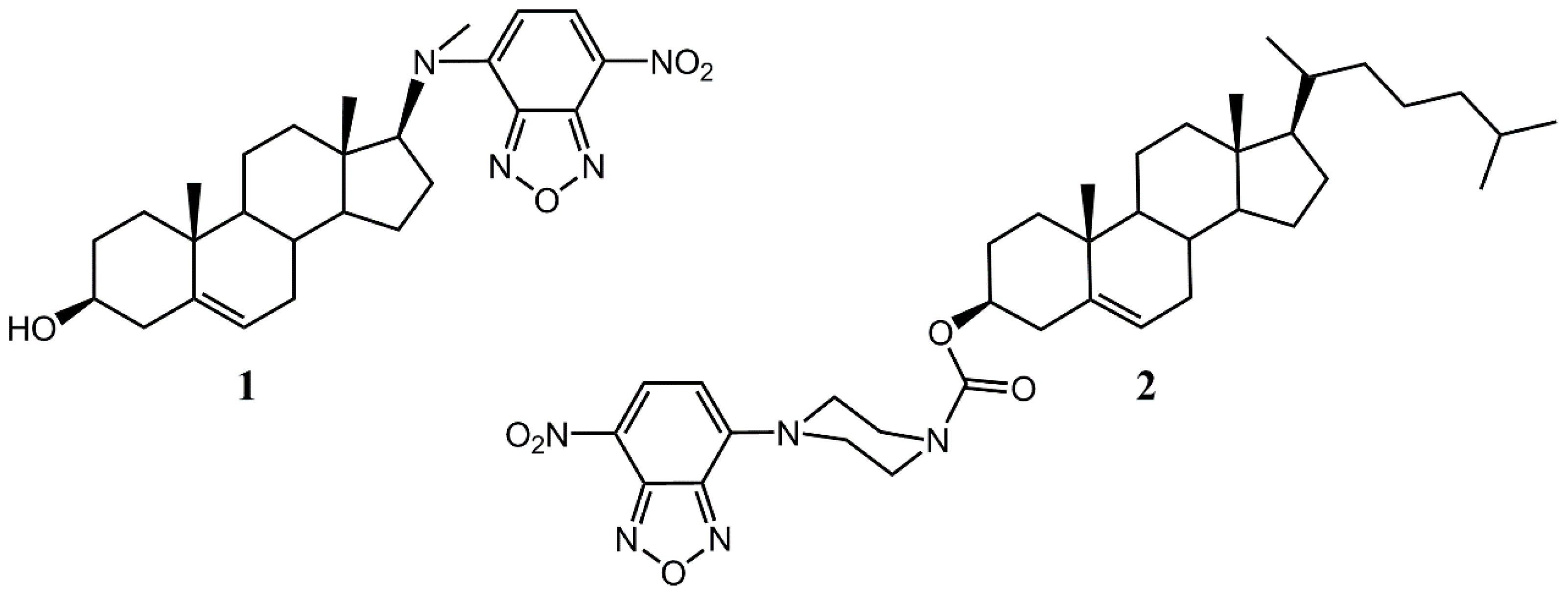
| Compound | Free Energy of Binding (DOPC), kcal/mol | Log of Perm. Coeff.-BLM | Log of Perm. Coeff-BBB | Log of Perm. Coeff–CACO2 |
|---|---|---|---|---|
| 1 | −6.45 | 1.85 | −2.24 | −3.01 |
| 2 | −10.20 | 2.24 | −2.10 | −2.91 |
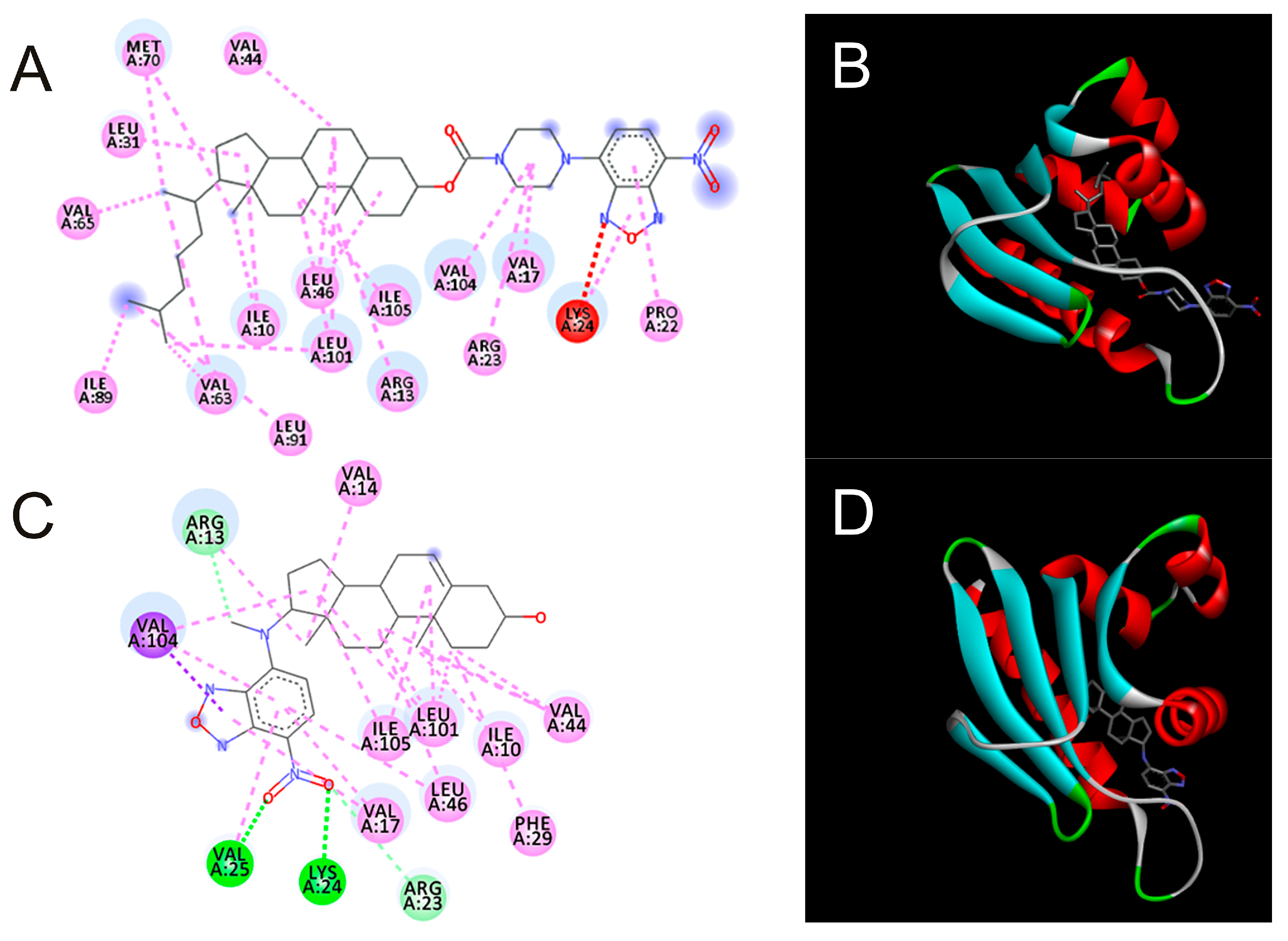
| PDB Code | Ligand Code | Ebind | Some Amino Acid Residues within 0.4 nm of Ligand Atoms | Position of NBD |
|---|---|---|---|---|
| 2KSH | CCFNBDpip | −9.0 | phe100, lys82, glu97, val96, ile99, val92 | OUT |
| 2KSH | Cholesterol | −7.5 | lys82, phe100, val96, glu97, ile99, ala81 | OUT |
| 2KSH | DAMNBD | −7.4 | lys82, phe100, pro104, val96, met85, ile99 | IN |
| 3BDQ | CCFNBDpip | −11.9 | ile105, leu101, ile10, met70, val65, ile73 | OUT |
| 3BDQ | DAMNBD | −11.7 | arg23, pro22, val17, leu101, ile105, leu31 | IN |
| 3BDQ | Cholesterol | −9.7 | leu101, arg13, val104, val63, ile105, ile10 | IN |
| 3BKS | Cholesterol | −9.6 | phe48, lys120, lys116, phe117, leu113, val19 | OUT |
| 3BKS | DAMNBD | −8.7 | lys120, val19, arg24, ser25, phe26, lys23 | OUT |
| NVLD | DAMNBD | −11.9 | gly210, gln308, ile206, pro207, gln286 | - |
| NVLD | CCFNBDpip | −8.6 | asn26, tyr191, thr67, tyr325, pro24, gln65 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pozniak, H.; Stoliarchuk, A.; Faletrov, Y.; Shkumatov, V. In Silico Modeling of the Interaction of NBD Steroids with Insect Steroid-Binding Protein SPC-2. Chem. Proc. 2022, 12, 86. https://doi.org/10.3390/ecsoc-26-13712
Pozniak H, Stoliarchuk A, Faletrov Y, Shkumatov V. In Silico Modeling of the Interaction of NBD Steroids with Insect Steroid-Binding Protein SPC-2. Chemistry Proceedings. 2022; 12(1):86. https://doi.org/10.3390/ecsoc-26-13712
Chicago/Turabian StylePozniak, Hleb, Anna Stoliarchuk, Yaroslav Faletrov, and Vladimir Shkumatov. 2022. "In Silico Modeling of the Interaction of NBD Steroids with Insect Steroid-Binding Protein SPC-2" Chemistry Proceedings 12, no. 1: 86. https://doi.org/10.3390/ecsoc-26-13712
APA StylePozniak, H., Stoliarchuk, A., Faletrov, Y., & Shkumatov, V. (2022). In Silico Modeling of the Interaction of NBD Steroids with Insect Steroid-Binding Protein SPC-2. Chemistry Proceedings, 12(1), 86. https://doi.org/10.3390/ecsoc-26-13712





