Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Retrospective Cohort Study
2.2. Statistical Analysis
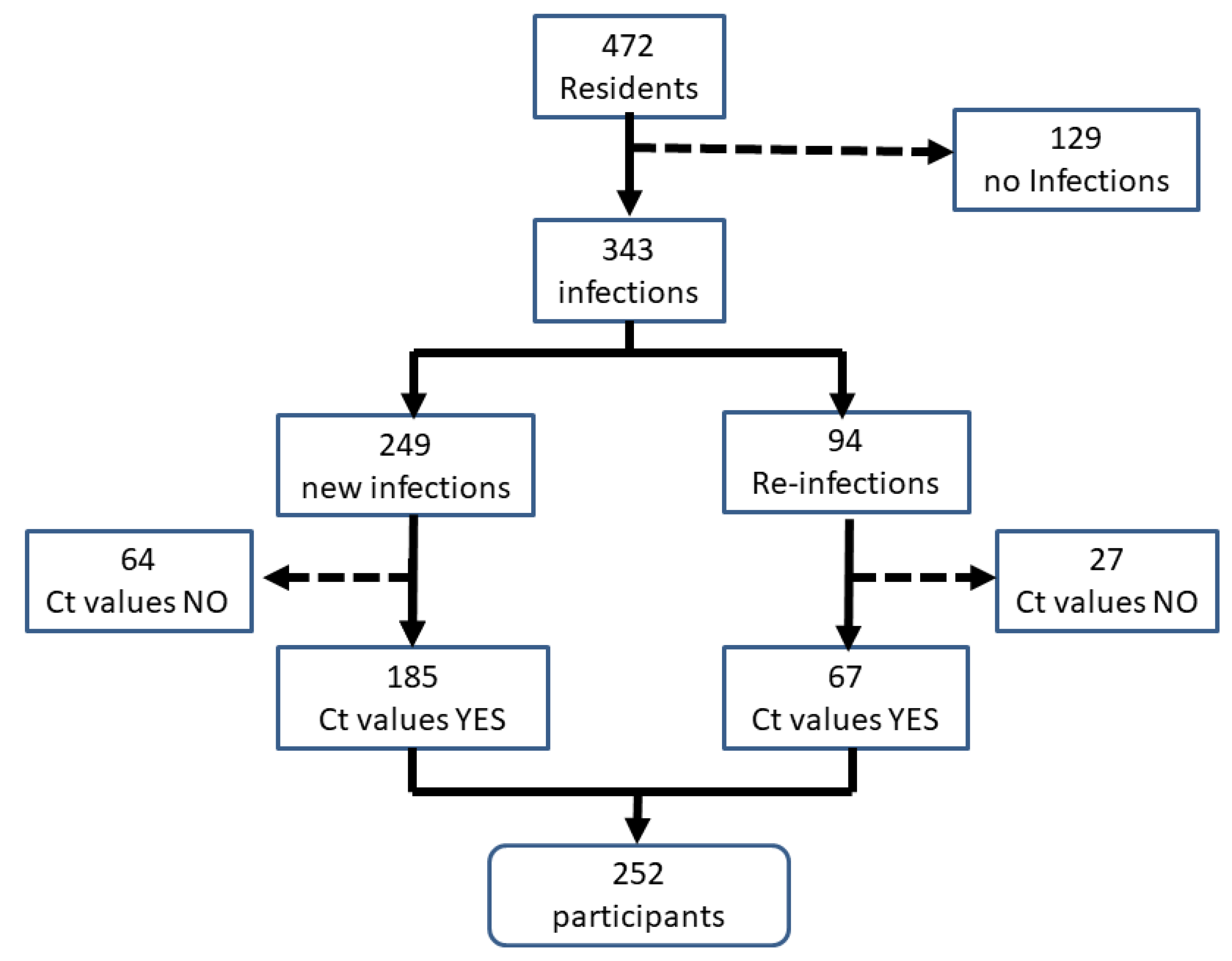
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rao, S.N.; Manissero, D.; Steele, V.R.; Pareja, J. A systematic review of the clinical utility of cycle threshold values in the context COVID-19. Infect. Dis. Ther. 2020, 9, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Aranha, C.; Patel, V.; Bhor, V.; Gogoi, D. Cycle threshold values in RT-PCR to determine dynamics of SARS-CoV-2 viral load: An approach to reduce the isolation period for COVID-19 patients. J. Med. Virol. 2021, 93, 6794–6797. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Yuen, A.; Rhoades, J.; Barnes, D.; Zakowski, P.; Megna, D.J.; Catarino, P.; Zaffiri, L.; Rampolla, R. Real-time transcription polymerase chain reaction cycle threshold values as criteria for utilization of incidental COVID-19 positive lung donors. J. Heart Lung Transpl. 2022, 42, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Hansel, D.E.; Curlin, M.E.; Townes, J.M.; Messer, W.B.; Fan, G.; Qin, X. Bimodal distribution pattern associated with the PCR cycle threshold (Ct) and implications in COVID-19 infections. Sci. Rep. 2022, 12, 14544. [Google Scholar] [CrossRef]
- Lopera, T.J.; Alzate-Ángel, J.C.; Díaz, F.J.; Rugeles, M.T.; Aguilar-Jiménez, W. The usefulness of antigen testing in predicting contagiousness in COVID-19. Microbiol. Spectr. 2022, 10, e0196221. [Google Scholar] [CrossRef]
- Abu-Raddad, L.J.; Chemaitelly, H.; Ayoub, H.H.; Tang, P.; Coyle, P.; Hasan, M.R.; Yassine, H.M.; Benslimane, F.M.; Al-Khatib, H.A.; Al-Kanaani, Z.; et al. Relative infectiousness of SARS-CoV-2 vaccine breakthrough infections, reinfections, and primary infections. Nat. Commun. 2022, 13, 532. [Google Scholar] [CrossRef]
- Phillips, M.C.; Quintero, D.; Wald-Dickler, N.; Holtom, P.; Butler-Wu, S.M. SARS-CoV-2 cycle threshold (Ct) values predict future COVID-19 cases. J. Clin. Virol. 2022, 150–151, 105153. [Google Scholar] [CrossRef]
- Maier, H.E.; Plazaola, M.; Lopez, R.; Sanchez, N.; Saborio, S.; Ojeda, S.; Barilla, C.; Kuan, G.; Balmaseda, A.; Gordon, A. SARS-CoV-2 infection-induced immunity and the duration of viral shedding: Results from a Nicaraguan household cohort study. Influenza Other Respir. Viruses 2023, 17, e13074. [Google Scholar] [CrossRef]
- Townsley, H.; Gahir, J.; Russell, T.W.; Greenwood, D.; Carr, E.J.; Dyke, M.; Adams, L.; Miah, M.; Clayton, B.; Smith, C.; et al. COVID-19 in non-hospitalised adults caused by either SARS-CoV-2 sub-variants Omicron BA.1, BA.2, BA.4/5 or Delta associates with similar illness duration, symptom severity and viral kinetics, irrespective of vaccination history. PLoS ONE 2024, 19, e0294897. [Google Scholar] [CrossRef]
- Kimball, A.; Hatfield, K.M.; Arons, M.; James, A.; Taylor, J.; Spicer, K.; Bardossy, A.C.; Oakley, L.P.; Tanwar, S.; Chisty, Z.; et al. Asymptomatic and presymptomatic SARS-CoV-2 infections in residents of a long-term care skilled nursing facility—King County, Washington, March 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 377–381. [Google Scholar] [CrossRef]
- Arons, M.M.; Hatfield, K.M.; Reddy, S.C.; Kimball, A.; James, A.; Jacobs, J.R.; Taylor, J.; Spicer, K.; Bardossy, A.C.; Oakley, L.P.; et al. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N. Engl. J. Med. 2020, 382, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Paap, K.C.; van Loon, A.M.; Koene, F.M.; van Buul, L.W.; Jurriaans, S.; Smalbrugge, M.; de Jong, M.D.; Hertogh, C.M.P.M. Clinical evaluation of single-swab sampling for rapid COVID-19 detection in outbreak settings in Dutch nursing homes. Eur. Geriatr. Med. 2022, 13, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.Y.; Jian, M.J.; Chang, C.K.; Lin, J.C.; Yeh, K.M.; Chen, C.W.; Hsieh, S.S.; Hung, K.S.; Tang, S.H.; Perng, C.L.; et al. Emergency SARS-CoV-2 variants of concern: Novel multiplex real-time RT-PCR assay for rapid detection and surveillance. Microbiol. Spectr. 2022, 10, e0251321. [Google Scholar] [CrossRef]
- Tso, C.F.; Garikipati, A.; Green-Saxena, A.; Mao, Q.; Das, R. Correlation of population SARS-CoV-2 cycle threshold values to local disease dynamics: Exploratory observational study. JMIR Public Health Surveill. 2021, 7, e28265. [Google Scholar] [CrossRef]
- Harrison, R.E.; Hamada, A.; Haswell, N.; Groves, A.; Vihta, K.D.; Cella, K.; Garner, S.; Walker, A.S.; Seale, A.C. Cycle threshold values as indication of increasing SARS-CoV-2 new variants, England, 2020–2022. Emerg. Infect. Dis. 2023, 29, 2024–2031. [Google Scholar] [CrossRef] [PubMed]
- Konetzka, R.T.; White, E.M.; Pralea, A.; Grabowski, D.C.; Mor, V. A systematic review of long-term care facility characteristics associated with COVID-19 outcomes. J. Am. Geriatr. Soc. 2021, 69, 2766–2777. [Google Scholar] [CrossRef]
- Arnedo-Pena, A.; Romeu-Garcia, M.A.; Gascó-Laborda, J.C.; Meseguer-Ferrer, N.; Safont-Adsuara, L.; Prades-Vila, L.; Flores-Medina, M.; Rusen, V.; Tirado-Balaguer, M.D.; Sabater-Vidal, S.; et al. Incidence, mortality, and risk factors of COVID-19 in nursing homes. Epidemiologia 2022, 3, 179–190. [Google Scholar] [CrossRef]
- Singanayagam, A.; Patel, M.; Charlett, A.; Lopez Bernal, J.; Saliba, V.; Ellis, J.; Ladhani, S.; Zambon, M.; Gopal, R. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Euro Surveill. 2020, 25, 2001483. [Google Scholar] [CrossRef]
- Girón Pérez, D.A.; Fonseca-Agüero, A.; Toledo-Ibarra, G.A.; Gomez-Valdivia, J.J.; Díaz-Resendiz, K.J.G.; Benitez-Trinidad, A.B.; Razura-Carmona, F.F.; Navidad-Murrieta, M.S.; Covantes-Rosales, C.E.; Giron-Pérez, M.I. Post-COVID-19 syndrome in outpatients and its association with viral load. Int. J. Environ. Res. Public Health 2022, 19, 15145. [Google Scholar] [CrossRef]
- Eyre, D.W.; Taylor, D.; Purver, M.; Chapman, D.; Fowler, T.; Pouwels, K.B.; Walker, A.S.; Peto, T.E.A. Effect of COVID-19 vaccination on transmission of Alpha and Delta variants. N. Engl. J. Med. 2022, 386, 744–756. [Google Scholar] [CrossRef]
- McEllistrem, M.C.; Clancy, C.J.; Buehrle, D.J.; Lucas, A.; Decker, B.K. Single dose of an mRNA Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) vaccine is associated with lower nasopharyngeal viral load among nursing home residents with asymptomatic coronavirus disease 2019 (COVID-19). Clin. Infect. Dis. 2021, 73, e1365–e1367. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Kalita, D.; Panda, P.K. Cycle threshold values versus reverse transcription-polymerase chain reaction positivity in COVID-19 de-isolation. Indian J. Med. Microbiol. 2021, 39, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Wünsch, K.; Anastasiou, O.E.; Alt, M.; Brochhagen, L.; Cherneha, M.; Thümmler, L.; van Baal, L.; Madel, R.J.; Lindemann, M.; Taube, C.; et al. COVID-19 in elderly, immunocompromised or diabetic patients-From immune monitoring to clinical management in the hospital. Viruses 2022, 14, 746. [Google Scholar] [CrossRef] [PubMed]
- McKay, S.L.; Tobolowsky, F.A.; Moritz, E.D.; Hatfield, K.M.; Bhatnagar, A.; LaVoie, S.P.; Jackson, D.A.; Lecy, K.D.; Bryant-Genevier, J.; Campbell, D.; et al. Performance evaluation of serial SARS-CoV-2 rapid antigen testing during a nursing home outbreak. Ann. Intern. Med. 2021, 174, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Leitão, I.C.; Calil, P.T.; Galliez, R.M.; Moreira, F.R.R.; Mariani, D.; Castiñeiras, A.C.P.; da Silva, G.P.D.; Maia, R.A.; Corrêa, I.A.; Monteiro, F.L.L.; et al. Prolonged SARS-CoV-2 positivity in immunocompetent patients: Virus isolation, genomic integrity, and transmission risk. Microbiol. Spectr. 2021, 9, e0085521. [Google Scholar] [CrossRef]
- Shoaib, N.; Noureen, N.; Faisal, A.; Zaheer, M.; Imran, M.; Ahsan, A.; Munir, R.; Zaidi, N. Factors associated with cycle threshold values (Ct-values) of SARS-CoV2-rRT-PCR. Mol. Biol. Rep. 2022, 49, 4101–4106. [Google Scholar] [CrossRef]
- Rhoads, D.D.; Pinsky, B.A. The truth about SARS-CoV-2 cycle threshold values is rarely pure and never simple. Clin. Chem. 2021, 68, 16–18. [Google Scholar] [CrossRef]
- Fan, G.; Jin, Y.; Wang, Q.; Yue, Y. Assessing the comparability of cycle threshold values derived from five external quality assessment rounds for Omicron nucleic acid testing. Virol. J. 2023, 20, 119. [Google Scholar] [CrossRef]
- Yip, C.C.; Sridhar, S.; Cheng, A.K.; Leung, K.H.; Choi, G.K.; Chen, J.H.; Poon, R.W.; Chan, K.H.; Wu, A.K.; Chan, H.S.; et al. Evaluation of the commercially available LightMix® Modular E-gene kit using clinical and proficiency testing specimens for SARS-CoV-2 detection. J. Clin. Virol. 2020, 129, 104476. [Google Scholar] [CrossRef]
- Ministerio de Sanidad. Estrategia de Detección Precoz, Vigilancia y Control de COVID-19. Centro de Coordinación de Alertas y Emergencias Sanitarias. Ministry of Health. Strategy for Early Detection Surveillance and Control of COVID-19. Health Alerts and Emergencies Coordination Center. Actualizado 22 de Diciembre de 2021. Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/COVID19_Estrategia_vigilancia_y_control_e_indicadores.pdf (accessed on 28 June 2024).
- European Centre for Disease Prevention and Control Reinfection with SARS-CoV-2: Implementation of a Surveillance Case Definition within the EU/EEA. 2021. Available online: https://www.ecdc.europa.eu/en/publications-data/reinfection-sars-cov-2-implementation-surveillance-case-definition-within-eueea (accessed on 20 June 2022).
- Khorraminejad-Shirazi, M.; Nabavizadeh, S.S.; Aminnia, S.; Ahmadifar, M.; Borazjani, R.; Sadeghi, E.; Izadpanah, S.; Heidari Esfahani, M.; Mokhtari, M.; Monabati, A. Association of demographic, clinical, and vaccination characteristics with COVID-19 viral load assessed by qRT-PCR. Arch. Iran Med. 2023, 26, 688–694. [Google Scholar] [CrossRef]
- Dimcheff, D.E.; Blair, C.N.; Zhu, Y.; Chappell, J.D.; Gaglani, M.; McNeal, T.; Ghamande, S.; Steingrub, J.S.; Shapiro, N.I.; Duggal, A.; et al. Total and subgenomic RNA viral load in patients infected with SARS-CoV-2 Alpha, Delta, and Omicron variants. J. Infect. Dis. 2023, 228, 235–244. [Google Scholar] [CrossRef]
- Ravindran, S.; Gubbay, J.B.; Cronin, K.; Sullivan, A.; Zygmunt, A.; Johnson, K.; Buchan, S.A.; Parpia, A.S. Association between cycle threshold value and vaccination status among severe acute respiratory syndrome coronavirus 2 Omicron variant cases in Ontario, Canada, in December 2021. Open Forum Infect. Dis. 2023, 10, ofad282. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Liu, Y.K.; Sun, Q.D.; Du, Z.; Fang, Y.Q.; Guo, F.; Wang, Y.B.; He, Y.; Cen, Y.; Zeng, F. Clinical characteristics and risk factors for a prolonged length of stay of patients with asymptomatic and mild COVID-19 during the wave of Omicron from Shanghai, China. BMC Infect. Dis. 2022, 22, 947. [Google Scholar] [CrossRef] [PubMed]
- Textor, J.; van der Zander, B.; Gilthorpe, M.S.; Liskiewicz, M.; Ellison, G.T. Robust causal inference using directed acyclic graphs: The R package ‘dagitty’. Int. J. Epidemiol. 2016, 45, 1887–1894. [Google Scholar] [CrossRef]
- Meyer, E.D.; Sandfort, M.; Bender, J.; Matysiak-Klose, D.; Dörre, A.; Bojara, G.; Beyrer, K.; Hellenbrand, W. BNT162b2 vaccination reduced infections and transmission in a COVID-19 outbreak in a nursing home in Germany, 2021. Influenza Other Respir. Viruses 2023, 17, e13051. [Google Scholar] [CrossRef]
- Admon, A.J.; Wander, P.L.; Iwashyna, T.J.; Ioannou, G.N.; Boyko, E.J.; Hynes, D.M.; Bowling, C.B.; Bohnert, A.S.B.; O’Hare, A.M.; Smith, V.A.; et al. Consensus elements for observational research on COVID-19-related long-term outcomes. Medicine 2022, 101, e31248. [Google Scholar] [CrossRef]
- Martínek, J.; Tomášková, H.; Janošek, J.; Zelená, H.; Kloudová, A.; Mrázek, J.; Ježo, E.; Král, V.; Pohořská, J.; Šturcová, H.; et al. Immune response 5–7 months after vaccination against SARS-CoV-2 in elderly nursing home residents in the Czech Republic: Comparison of three vaccines. Viruses 2022, 14, 1086. [Google Scholar] [CrossRef]
- Duncombe, T.R.; Garrod, M.; Wang, X.; Ng, J.; Lee, E.; Short, K.; Tan, K. Risk factors associated with severe acute respiratory coronavirus virus 2 (SARS-CoV-2) transmission, outbreak duration, and mortality in acute-care settings. Infect. Control Hosp. Epidemiol. 2023, 44, 1643–1649. [Google Scholar] [CrossRef]
- American Association for Clinical Chemistry. Recommendation for Reporting SARS-CoV-2 Cycle Threshold (CT) Values. Available online: https://www.myadlm.org/Media/Press-Release-Archive/2021/07-Jul/AACC-Recommends-Against-Using-Coronavirus-Test-Ct-Values-to-Guide-COVID19-Patient-Care (accessed on 1 March 2023).
- Quiroz-Ruiz, H.R.; Chimoy-Effio, P.J.; Vértiz-Osores, J.J.; Bazán-Mayra, J.E. Correlación entre el umbral de ciclo de la RPC en tiempo real y la clasificación clínica de la COVID-19. Rev. Chilena Infectol. 2022, 39, 35–44. [Google Scholar] [CrossRef]
- Ley 14/1986, de 25 de Abril, General de Sanidad (Law General of Health). Available online: https://www.boe.es/eli/es/l/1986/04/25/14/con (accessed on 2 August 2021). (In Spanish).
- Ley 16/2003, de 28 de Mayo, de Cohesión y Calidad del Sistema Nacional de Salud. (Law of Cohesion and Quality of the National System of Health). Available online: https://www.boe.es/eli/es/l/2003/05/28/16 (accessed on 2 August 2021). (In Spanish).
- Ley 33/2011, de 4 de Octubre, General de Salud Pública (Law General of Public Health). Available online: https://www.boe.es/eli/es/l/2011/10/04/33/con (accessed on 2 August 2021). (In Spanish).
- Ministry of Health Agreed in the Interterritorial Council of the National Health System. Early Response Plan in a COVID-19 Pandemic Control Scenario. 2020. Available online: https://www.sanidad.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov/documentos/COVID19_Plan_de_respuesta_temprana_escenario_control.pdf (accessed on 2 August 2021). (In Spanish)
- Bobrovitz, N.; Ware, H.; Ma, X.; Li, Z.; Hosseini, R.; Cao, C.; Selemon, A.; Whelan, M.; Premji, Z.; Issa, H.; et al. Protective effectiveness of previous SARS-CoV-2 infection and hybrid immunity against the omicron variant and severe disease: A systematic review and meta-regression. Lancet Infect. Dis. 2023, 23, 556–567. [Google Scholar] [CrossRef]
- Gascó-Laborda, J.C.; Gil-Fortuño, M.; Ortiz-Rambla, J.; Meseguer-Ferrer, N.; Pérez-Olaso, Ó.; Lluch-Bacas, L.; Moya-Malo, R.; Moliner-Urdiales, D.; Bellido-Blasco, J.B. COVID-19 outbreak in a properly vaccinated nursing home. Influence of hybrid immunity on viral load, risk of infection and risk of disease progression. Rev. Esp. Salud. Publica 2024, 98, e202405036. [Google Scholar] [PubMed]
- Mauriz, E.; Fernández-Vázquez, J.P.; Díez-Flecha, C.; Reguero-Celada, S.; Fernández-Villa, T.; Fernández-Somoano, A.; Caylà, J.A.; Lozano-García, J.A.; Vázquez-Casares, A.M.; Martín-Sánchez, V. Impact of a COVID-19 outbreak in an elderly care home after primary vaccination. Vaccines 2023, 11, 1382. [Google Scholar] [CrossRef] [PubMed]
- Henriques, H.R.; Sousa, D.; Faria, J.; Pinto, J.; Costa, A.; Henriques, M.A.; Durão, M.C. Learning from the COVID-19 outbreaks in long-term care facilities: A systematic review. BMC Geriatr. 2023, 23, 618. [Google Scholar] [CrossRef]
- Meyers, E.; Deschepper, E.; Duysburgh, E.; De Rop, L.; De Burghgraeve, T.; Van Ngoc, P.; Digregorio, M.; Delogne, S.; Coen, A.; De Clercq, N.; et al. Declining prevalence of SARS-CoV-2 antibodies among vaccinated nursing home residents and staff six months after the primary BNT162b2 vaccination campaign in Belgium: A prospective cohort study. Viruses 2022, 14, 2361. [Google Scholar] [CrossRef]
- Moyet, J.; Helle, F.; Bourdenet, G.; Joseph, C.; Gubler, B.; Deschasse, G.; Defouilloy, I.; Slovenski, T.; François, C.; Liabeuf, S.; et al. Kinetics of SARS-CoV-2-neutralising antibodies of residents of long-term care facilities. J. Nutr. Health Aging 2022, 26, 57–63. [Google Scholar] [CrossRef]
- Petersen, J.; Jhala, D. COVID-19 cycle threshold/cycle number testing at a community living center. Fed. Pract. 2022, 39, 254–260. [Google Scholar] [CrossRef]
- Lennon, N.J.; Bhattacharyya, R.P.; Mina, M.J.; Rehm, H.L.; Hung, D.T.; Smole, S.; Woolley, A.; Lander, E.S.; Gabriel, S.B. Cross-Sectional assessment of SARS-CoV-2 viral load by symptom status in Massachusetts congregate living facilities. J. Infect. Dis. 2021, 224, 1658–1663. [Google Scholar] [CrossRef]
- Paap, K.C.; van Loon, A.M.; van Rijs, S.M.; Helmich, E.; Buurman, B.M.; Smalbrugge, M.; Hertogh, C.M.P.M. Symptom- and prevention-based testing of COVID-19 in nursing home residents: A retrospective cohort study. Gerontol. Geriatr. Med. 2021, 7, 23337214211055338. [Google Scholar] [CrossRef]
- Wilson, W.W.; Hatfield, K.M.; Tressler, S.; Bicking Kinsey, C.; Parra, G.; Zell, R.; Denson, A.; Williams, C.; Spicer, K.B.; Kamal-Ahmed, I.; et al. Characteristics of nursing home residents and healthcare personnel with repeated severe acute respiratory coronavirus virus 2 (SARS-CoV-2) tests positive ≥90 days after initial infection: Four US jurisdictions, July 2020–March 2021. Infect. Control Hosp. Epidemiol. 2023, 44, 809–812. [Google Scholar] [CrossRef]
- Shrotri, M.; Krutikov, M.; Palmer, T.; Giddings, R.; Azmi, B.; Subbarao, S.; Fuller, C.; Irwin-Singer, A.; Davies, D.; Tut, G.; et al. Vaccine effectiveness of the first dose of ChAdOx1 nCoV-19 and BNT162b2 against SARS-CoV-2 infection in residents of long-term care facilities in England (VIVALDI): A prospective cohort study. Lancet Infect. Dis. 2021, 21, 1529–1538. [Google Scholar] [CrossRef]
- Johnson, B.N.; Cooper, L.; Beck, J.J.; Finnicum, C.T.; Davis, C.M.; Van Asselt, A.J.; Kallsen, N.; Silvernail, C.; Viet, S.; Long, K.; et al. More than results: The clinical and research relationship in the evolving detection and surveillance of SARS-CoV-2. S. D. Med. 2023, 76, 248–256. [Google Scholar] [PubMed]
- Shoaib, N.; Iqbal, A.; Shah, F.A.; Zainab, W.; Qasim, M.; Zerqoon, N.; Naseem, M.O.; Munir, R.; Zaidi, N. Population-level median cycle threshold (Ct) values for asymptomatic COVID-19 cases can predict the trajectory of future cases. PLoS ONE 2023, 18, e0281899. [Google Scholar] [CrossRef] [PubMed]
- Dehesh, P.; Baradaran, H.R.; Eshrati, B.; Motevalian, S.A.; Salehi, M.; Donyavi, T. The relationship between population-level SARS-CoV-2 cycle threshold values and trend of COVID-19 infection: Longitudinal study. JMIR Public Health Surveill. 2022, 8, e36424. [Google Scholar] [CrossRef]
- Boogaerts, H.L.F.; Smits, P.; Hans, G.; Bouly, L.; Coeck, E.; Vandamme, S.; Jansens, H.; Goossens, H.; Matheeussen, V. Laboratory analysis of two Delta SARS-CoV-2 variant outbreaks in the Port of Antwerp. Acta. Clin. Belg. 2022, 77, 925–932. [Google Scholar] [CrossRef]
- Nyaruaba, R.; Mwaliko, C.; Dobnik, D.; Neužil, P.; Amoth, P.; Mwau, M.; Yu, J.; Yang, H.; Wei, H. Digital PCR Applications in the SARS-CoV-2/COVID-19 era: A roadmap for future outbreaks. Clin. Microbiol. Rev. 2022, 35, e0016821. [Google Scholar] [CrossRef]
- Miranda, R.L.; Guterres, A.; de Azeredo Lima, C.H.; Filho, P.N.; Gadelha, M.R. Misinterpretation of viral load in COVID-19 clinical outcomes. Virus Res. 2021, 296, 198340. [Google Scholar] [CrossRef]
- Han, M.S.; Byun, J.H.; Cho, Y.; Rim, J.H. RT-PCR for SARS-CoV-2: Quantitative versus qualitative. Lancet Infect. Dis. 2021, 21, 165. [Google Scholar] [CrossRef]
- Infectious Disease Society of America and Association for Molecular Pathology Joint Statement on the Use of SARS-CoV-2 PCR Cycle Threshold (Ct) Values for Clinical Decision-Making. Available online: https://www.idsociety.org/globalassets/idsa/public-health/covid-19/idsa-amp-statement.pdf (accessed on 1 March 2024).
- Michaelis, S.; Schneider, C.; Schnedl, W.J.; Baranyi, A.; Enko, D. Comparison of cycle-threshold-values between two commercial SARS-CoV-2 PCR assays. EXCLI J. 2023, 22, 397–399. [Google Scholar]
- Dahdouh, E.; Lázaro-Perona, F.; Romero-Gómez, M.P.; Mingorance, J.; García-Rodriguez, J. Ct values from SARS-CoV-2 diagnostic PCR assays should not be used as direct estimates of viral load. J. Infect. 2021, 82, 414–451. [Google Scholar] [CrossRef]
- Ingberg, E.; Ahlstrand, E.; Cajander, P.; Löf, E.; Sundqvist, M.; Wegener, M.; Lidén, M.; Cajander, S. RT-PCR cycle threshold value in combination with visual scoring of chest computed tomography at hospital admission predicts outcome in COVID-19. Infect. Dis. 2022, 54, 431–440. [Google Scholar] [CrossRef]
- Markewitz, R.; Dargvainiene, J.; Junker, R.; Wandinger, K.P. Cycle threshold of SARS-CoV-2 RT-PCR as a driver of retesting. Sci. Rep. 2024, 14, 2423. [Google Scholar] [CrossRef]
- Buchta, C.; Görzer, I.; Chiba, P.; Camp, J.V.; Holzmann, H.; Puchhammer-Stöckl, E.; Mayerhofer, M.; Müller, M.M.; Aberle, S.W. Variability of cycle threshold values in an external quality assessment scheme for detection of the SARS-CoV-2 virus genome by RT-PCR. Clin. Chem. Lab. Med. 2020, 59, 987–994. [Google Scholar] [CrossRef]
| Variables | Center 1 n (%) | Center 2 n (%) | Center 3 n (%) | Center 4 n (%) | Total n (%) |
|---|---|---|---|---|---|
| Date of reporting (month/year) | August 2021 | March 2022 | January 2022 | February 2022 | |
| COVID-19 Variant outbreak | Delta B.1.617.2 | Omicron | Omicron | Omicron | |
| Total residents | 125 | 130 | 111 | 106 | 472 |
| Age (years) mean SD 1 | 84.4 ± 8.3 | 82.0 ± 11.0 | 83.8 ± 8.9 | 82.4 ± 10.8 | 83.1 ± 9.8 |
| Female | 90 (72%) | 92 (70.8%) | 74 (66.7%) | 62 (58.5%) | 318 (67.4%) |
| Male | 35 (28%) | 38 (29.2%) | 37 (33.3%) | 44 (41.5%) | 154 (32.6%) |
| Elapsed time from former SARS-CoV-2 outbreak in days | 339.9 | 433.1 | 350.5 | 634.9 | 391.8 2 |
| SARS-CoV-2 Vaccine | 122 (97.6%) | 130 (100%) | 111 (100%) | 104 (98.1%) | 467 (98.9%) |
| Elapsed time from the last vaccine doses; mean SD in days | 177 ± 28.4 | 154 ± 73.6 | 108 ± 36.2 | 154 ± 22.2 | 157 ± 50.1 |
| Total SARS-CoV-2 infections | 57 (45.6%) | 125 (96.2%) | 94 (84.7%) | 67 (63.2%) | 343 (72.7%) |
| Incidence attack rate (%) 3 | 45.6% | 96.2% | 84.7% | 63.2% | 72.7% |
| New SARS-CoV-2 infections | 50 (40%) | 63 (48.5%) | 93 (83.8%) | 43 (40.6%) | 249 (52.8%) |
| SARS-CoV-2 reinfections | 7 (5.6%) | 62 (47.7%) | 1 (0.90%) | 24 (22.6%) | 94 (19.9%) |
| Reinfections attack rate (%) 3 | 5.6% | 47.7% | 0.90% | 22.6% | 19.9% |
| Mortality rate | 6 (4.8%) | 7 (5.4%) | 6 (5.4%) | 0 | 19 (4.0%) |
| Fatality rate | 10.6% | 17.9% | 1.1% | 0 | 5.5% |
| Variables | Centre 1 n (%) | Centre 2 n (%) | Centre 3 n (%) | Centre 4 n (%) | Total n (%) |
|---|---|---|---|---|---|
| Residents with PCR-Ct | 38 | 90 | 92 | 32 | 252 |
| Age | 85.0 ± 6.7 | 81.9 ± 11.6 | 84.7 ± 8.4 | 80.4 ± 12.0 | 83.2 ± 10.0 |
| Female | 32 (84.2) | 66 (73.3) | 66 (71.7) | 20 (62.5) | 184 (73.0) |
| Male | 6 (15.8) | 24 (26.7) | 26 (28.3) | 12 (37.5) | 68 (27.0) |
| First infection with PCR-Ct | 33 (86.8) | 42 (46.7) | 91 (98.9) | 19 (59.4) | 185 (73.4) |
| Reinfections with PCR-Ct | 5 (13.2) | 48 (53.3) | 1 (1.1) | 13 (40.6) | 67 (26.6) |
| Reinfections without PCR-Ct | 2 (5.3) | 14 (15.6) | 0 | 11 (34.4) | 27 (10.7) |
| Cycle threshold (median range) | 23.3 (17–38) | 33.5 (11–39) | 27 (17–38) | 29 (15–37) | 29 (11–39) |
| Cycle thresholds’ mean ± SD | 25.2 ± 6.2 | 30.9 ± 7.0 | 27.2 ± 5.7 | 28.4 ± 6.6 | 28.4 ± 6.7 |
| Ct SARS-CoV-2 reinfections | 36.4 ± 3.0 | 32.5 ± 4.9 | 26.0 | 28.1 ± 5.7 | 31.9 ± 5.4 |
| Ct first SARS-CoV-2 infections | 23.5 ± 4.5 | 29.0 ± 8.5 | 27.3 ± 5.7 | 28.6 ± 7.3 | 27.1 ± 6.6 |
| p-values | 0.000 | 0.016 | 0.876 | 0.820 | 0.000 |
| Variables | Ct ≥ 30 n = 123 (%) | Ct < 30 n = 129 (%) | Total | cRR | 95% CI | aRR | 95% CI | p-Value |
|---|---|---|---|---|---|---|---|---|
| Age 1 mean ± SD | 82.9 ± 10.1 | 83.5 ± 10.0 | - | 1.00 | 0.98–1.01 | 0.99 1 | 0.98–1.01 | 0.675 |
| Male 2 | 32 (47.1) | 36 (52.9) | 68 | 0.95 | 0.64–1.42 | 0.92 2 | 0.59–1.42 | 0.703 |
| Female | 91 (49.5) | 93 (50.5) | 184 | 1.00 | 1.00 | |||
| Elapse time from former SARS-CoV-2 outbreak 3 (days) mean ± SD | 427.9 ± 86.8 | 401.7 ± 97.3 | - | 1.00 | 0.99–1.00 | 1.00 3 | 1.00–1.01 | 0.031 |
| Nursing homes 4 | ||||||||
| Centre 1 | ||||||||
| SARS-CoV-2 Reinfections | 5 (100) | 0 (0) | 5 | 6.60 | 1.91–22.80 | 6.70 | 1.80–25.0 | 0.005 |
| First SARS-CoV-2 Infections | 5 (15.2) | 28 (84.8) | 33 | 1.00 | ||||
| Centre 2 | ||||||||
| SARS-CoV-2 Reinfections | 39 (81.3) | 9 (18.8) | 48 | 1.31 | 0.80–2.16 | 1.31 | 0.80–2.15 | 0.287 |
| First SARS-CoV-2 Infections | 26 (61.9) | 16 (38.1) | 42 | 1.00 | ||||
| Centre 3 | ||||||||
| SARS-CoV-2 Reinfections | 0 (0) | 1 (100) | 1 | 1.93 5 | 0.0–10.76 | 2.18 6 | 0.0–14.25 | 1.000 |
| First SARS-CoV-2 Infections | 33 (34.1) | 58 (63.7) | 91 | 1.00 | ||||
| Centre 4 | ||||||||
| SARS-CoV-2 Reinfections | 5 (38.5) | 8 (61.5) | 13 | 0.73 | 0.20–2.34 | 0.70 | 0.24–2.05 | 0.513 |
| First SARS-CoV-2 Infections | 10 (52.6) | 9 (47.4) | 19 | 1.00 | ||||
| Total nursing homes 7 | ||||||||
| SARS-CoV-2 Reinfections | 49 (73.1) | 18 (26.9) | 67 | 1.83 | 1.27–2.62 | 1.66 | 1.10–2.51 | 0.015 |
| First SARS-CoV-2 Infections | 74 (40.0) | 111 (60.0) | 185 | 1.00 | 1.00 |
| Yes n = 67 | No n = 185 | cOR | 95% CI | aOR | 95% | p-Value | |
|---|---|---|---|---|---|---|---|
| Cycle threshold 1 | n (%) | n (%) | |||||
| Minor (<18.83) | 1 (1.5) | 19 (10.3) | 1.00 | 1.00 | |||
| Medium (≥18.83–30.10) | 17 (25.4) | 99 (53.5) | 3.26 | 0.41–26.00 | 5.98 | 0.73–49.12 | 0.096 |
| High (>30.10) | 49 (73.1) | 67 (36.2) | 13.90 | 1.80–107.33 | 17.63 | 2.22–139.86 | 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gascó-Laborda, J.C.; Gil-Fortuño, M.; Tirado-Balaguer, M.D.; Meseguer-Ferrer, N.; Sabalza-Baztán, O.; Pérez-Olaso, Ó.; Gómez-Alfaro, I.; Poujois-Gisbert, S.; Hernández-Pérez, N.; Lluch-Bacas, L.; et al. Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study. Epidemiologia 2024, 5, 658-668. https://doi.org/10.3390/epidemiologia5040046
Gascó-Laborda JC, Gil-Fortuño M, Tirado-Balaguer MD, Meseguer-Ferrer N, Sabalza-Baztán O, Pérez-Olaso Ó, Gómez-Alfaro I, Poujois-Gisbert S, Hernández-Pérez N, Lluch-Bacas L, et al. Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study. Epidemiologia. 2024; 5(4):658-668. https://doi.org/10.3390/epidemiologia5040046
Chicago/Turabian StyleGascó-Laborda, Juan Carlos, Maria Gil-Fortuño, Maria Dolores Tirado-Balaguer, Noemi Meseguer-Ferrer, Oihana Sabalza-Baztán, Óscar Pérez-Olaso, Iris Gómez-Alfaro, Sandrine Poujois-Gisbert, Noelia Hernández-Pérez, Lledó Lluch-Bacas, and et al. 2024. "Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study" Epidemiologia 5, no. 4: 658-668. https://doi.org/10.3390/epidemiologia5040046
APA StyleGascó-Laborda, J. C., Gil-Fortuño, M., Tirado-Balaguer, M. D., Meseguer-Ferrer, N., Sabalza-Baztán, O., Pérez-Olaso, Ó., Gómez-Alfaro, I., Poujois-Gisbert, S., Hernández-Pérez, N., Lluch-Bacas, L., Rusen, V., Arnedo-Pena, A., & Bellido-Blasco, J. B. (2024). Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study. Epidemiologia, 5(4), 658-668. https://doi.org/10.3390/epidemiologia5040046







