Leptin Signaling in the Hypothalamus: Cellular Insights and Therapeutic Perspectives in Obesity
Abstract
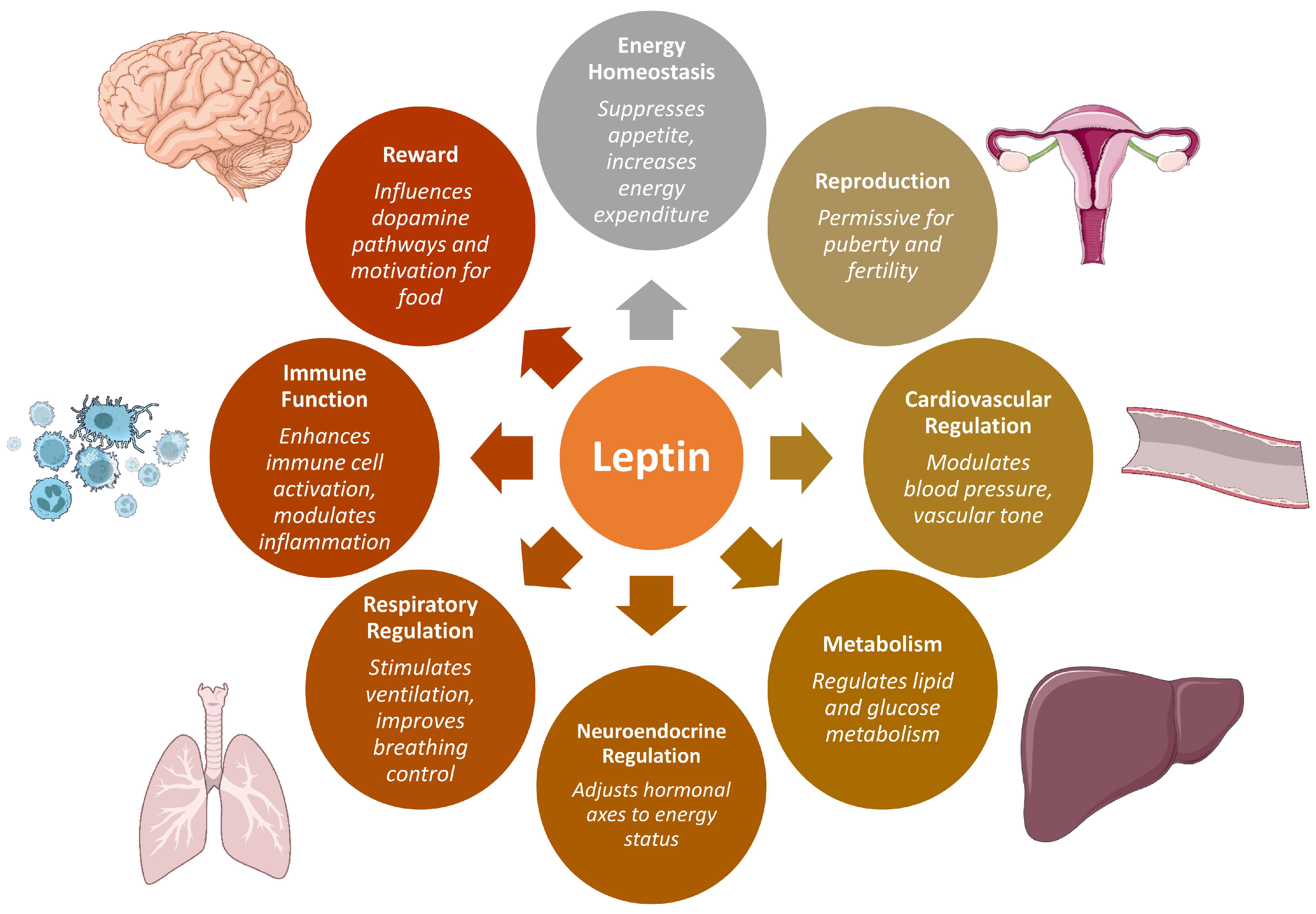
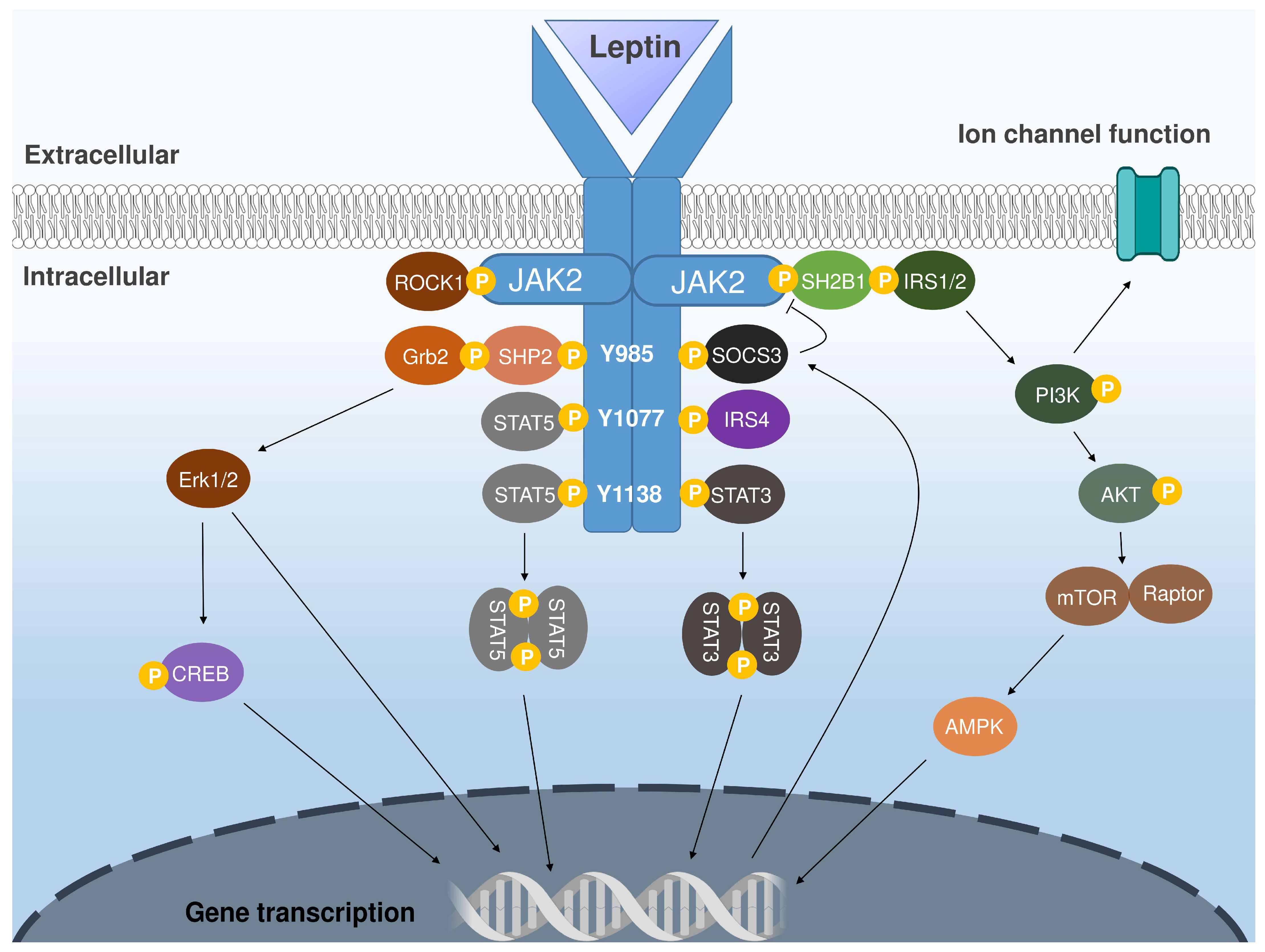
1. Introduction
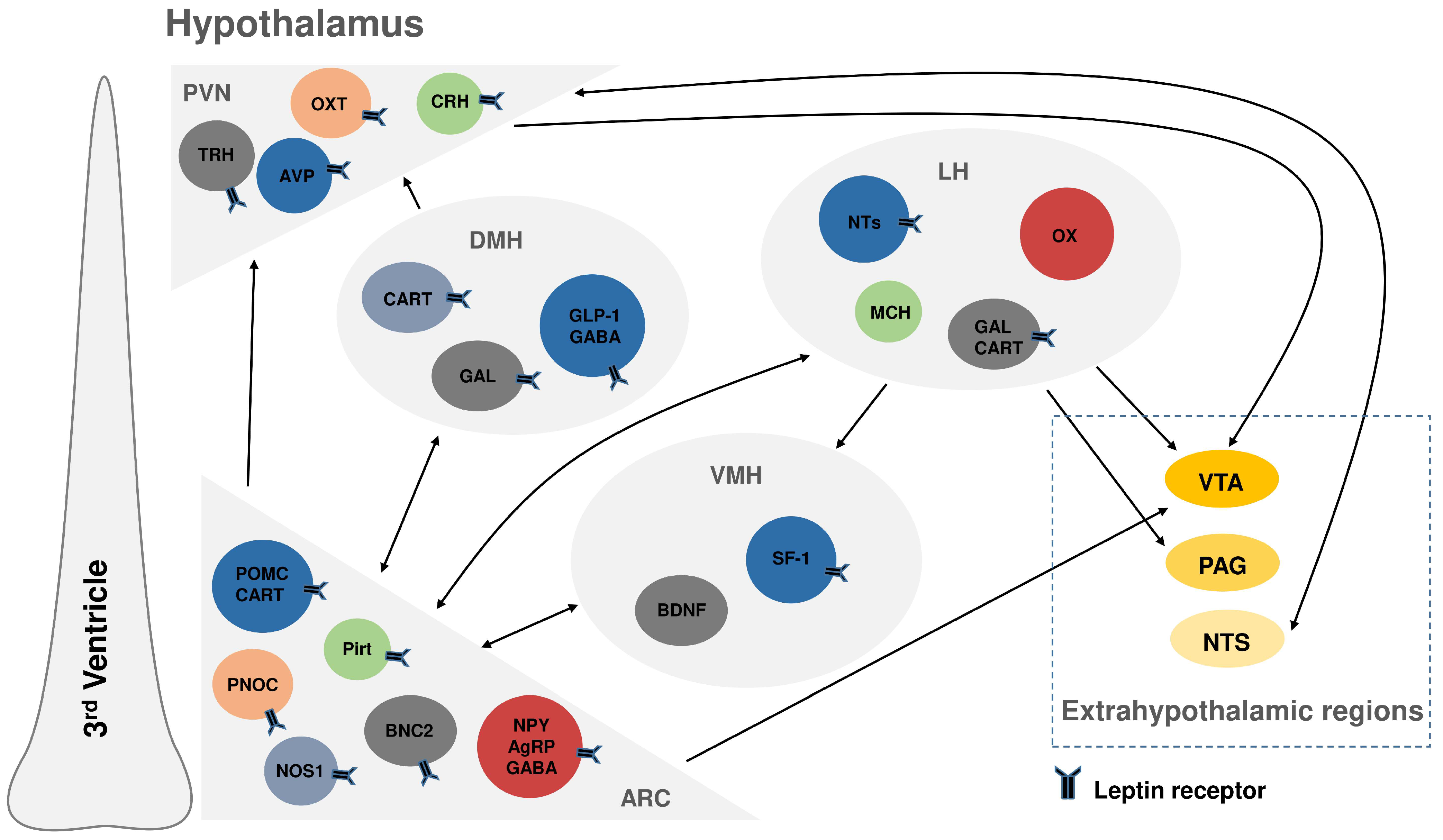
2. The Arcuate Nucleus as a Leptin-Sensing Hub: An Emerging Player in Appetite and Energy Balance Regulation
3. Leptin Action in the Ventromedial Hypothalamus: Synergistic Hormonal Signaling and Epigenetic Regulation
| Modulator | Site of Action | Mechanism | Reference(s) |
|---|---|---|---|
| Angiopoietin-like growth factor (AGF) | ARC (POMC neurons) | A peripheral promoter of energy expenditure that counteracts obesity. Leptin induces AGF expression via STAT3 phosphorylation. | [29] |
| Zinc-α2-glycoprotein | ARC (POMC neurons) | Increases energy expenditure, improves leptin and insulin sensitivity, alleviates hepatic steatosis, and promotes adipose browning. | [30] |
| Spexin | ARC (POMC neurons) | Leptin induces spexin expression, and inhibition of hypothalamic spexin blocks leptin’s effects on food intake, body weight, and Pomc expression. | [31] |
| Ras-related protein 1 (Rap1) | ARC (POMC neurons) | POMC-specific Rap1 deletion enhances leptin sensitivity, improves glucose homeostasis, and reduces weight gain under high-fat diet. | [32] |
| Mammalian target of rapamycin (mTOR) | ARC (POMC neurons) | mTOR signaling in POMC neurons drives leptin resistance in diet-induced obesity. Inhibition with rapamycin restores leptin sensitivity, reduces fat mass, and improves metabolism. | [33] |
| Growth factor receptor-bound protein 10 (Grb10) | ARC (POMC/AgRP neurons) | Overexpression of Grb10 reduces food intake and body weight by enhancing leptin’s excitatory effect on POMC neurons and inhibitory effect on AgRP neurons, while its deletion promotes obesity. | [34] |
| Spliced X-box binding protein 1 | ARC (AgRP neurons) | Prevents diet-induced obesity and enhances leptin sensitivity. | [35] |
| Jumonji D3 (JMJD3), a histone lysine demethylase containing a Jmjc domain | VMH neurons | Enhances leptin signaling by demethylating H3K27me3 at the LepRb promoter, increasing LepRb expression. Overexpression of JMJD3 in obese mice reduces body weight, fat mass, food intake, and glucose levels. | [42] |
4. The Dorsomedial Hypothalamus: Integrating Leptin’s Roles in Metabolism, Circadian Rhythms, and Respiratory Control
5. The Lateral Hypothalamus: Leptin’s Regulation of Feeding Motivation and Reward Pathways
| Hypothalamic Region | Neuronal Population | Functions | Reference(s) |
|---|---|---|---|
| ARC | LepRb PNOC neurons | Mediate leptin’s effects on feeding by regulating NPY; LepRb loss causes obesity, restoration reduces body weight. | [25] |
| LepRb BNC2 neurons | Suppress appetite by inhibiting AgRP neurons; LepRb loss causes obesity, highlighting their key role in appetite control. | [26] | |
| LepRb Pirt neurons | Regulate reproduction and suppress energy expenditure; LepRb loss impairs female fertility but not feeding. | [27,28] | |
| VMH | LepRb neurons | Enable CCK-induced satiety, especially on high-fat diets, integrating central leptin and peripheral CCK signals for energy regulation. | [40] |
| LepRb neurons | Require intact PACAP signaling to mediate leptin’s effects on satiety, metabolism, and associated molecular responses. | [41] | |
| DMH | LepRb Glp1r neurons | Essential for regulating food intake and body weight, mediating the anorexigenic effects of both leptin and GLP-1R agonists. | [47,48] |
| LepRb neurons | Coordinate feeding-related circadian rhythms, linking leptin signaling to metabolic timing. | [49,50] | |
| LepRb neurons | Crucial for central respiratory control, promoting ventilation and maintaining airway patency. | [54,55,56] | |
| LH | LepRb GABA neurons | Regulate motivated behavior rather than baseline feeding. Their activation enhances goal-directed actions, while inhibition disrupts Pavlovian appetitive learning and attenuates reward-specific behaviors. | [65,66,67] |
6. Conclusions and Future Directions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| α-MSH | α-melanocyte-stimulating hormone |
| AGF | Angiopoietin-like growth factor |
| AMPK | AMP-activated protein kinase |
| ARC | Arcuate nucleus |
| AZGP1 | Zinc-α2-glycoprotein |
| BDNF | Brain-derived neurotrophic factor |
| CCK | Cholecystokinin |
| CNS | Central nervous system |
| CREB | cAMP response element-binding protein |
| DMH | Dorsomedial hypothalamus |
| ELT | Early life trauma |
| ER | Endoplasmic reticulum |
| ERK1/2 | Extracellular signal-regulated kinases 1 and 2 |
| GABA | Gamma-aminobutyric acid |
| GFP | Green fluorescent protein |
| GLP-1 | Glucagon-like peptide-1 |
| GLP-1R | Glucagon-like peptide-1 receptor |
| Grb10 | Growth factor receptor-bound protein 10 |
| HFD | High-fat diet |
| JAK2 | Janus kinase 2 |
| LH | Lateral hypothalamus |
| LepR/LepRb | Leptin receptor/Leptin receptor long isoform |
| MAPK | Mitogen-activated protein kinase |
| MC3R | Melanocortin-3 receptor |
| MC4R | Melanocortin-4 receptor |
| MCH | Melanin-concentrating hormone |
| mTOR | Mammalian target of rapamycin |
| NREM | Non-rapid eye movement (sleep) |
| NPY | Neuropeptide Y |
| NTS | Nucleus of the solitary tract |
| PACAP | Pituitary adenylate cyclase-activating polypeptide |
| PI3K | Phosphoinositide 3-kinase |
| Pirt | Phosphoinositide-interacting regulator of TRP |
| PNOC | Prepronociceptin |
| POMC | Proopiomelanocortin |
| Rap1 | Ras-related protein 1 |
| REM | Rapid eye movement (sleep) |
| SOCS3 | Suppressor of cytokine signaling 3 |
| STAT3 | Signal transducer and activator of transcription 3 |
| VMH | Ventromedial hypothalamus |
| vlPAG | Ventrolateral periaqueductal gray |
| VTA | Ventral tegmental area |
| Xbp1s | Spliced X-box binding protein 1 |
References
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Mantzoros, C.S.; Magkos, F.; Brinkoetter, M.; Sienkiewicz, E.; Dardeno, T.A.; Kim, S.Y.; Hamnvik, O.P.; Koniaris, A. Leptin in human physiology and pathophysiology. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E567–E584. [Google Scholar] [CrossRef]
- Prolo, P.; Wong, M.L.; Licinio, J. Leptin. Int. J. Biochem. Cell Biol. 1998, 30, 1285–1290. [Google Scholar] [CrossRef]
- Harvey, J. Novel Leptin-Based Therapeutic Strategies to Limit Synaptic Dysfunction in Alzheimer’s Disease. Int. J. Mol. Sci. 2024, 25, 7352. [Google Scholar] [CrossRef]
- Yupanqui-Lozno, H.; Bastarrachea, R.A.; Yupanqui-Velazco, M.E.; Alvarez-Jaramillo, M.; Medina-Méndez, E.; Giraldo-Peña, A.P.; Arias-Serrano, A.; Torres-Forero, C.; Garcia-Ordoñez, A.M.; Mastronardi, C.A.; et al. Congenital Leptin Deficiency and Leptin Gene Missense Mutation Found in Two Colombian Sisters with Severe Obesity. Genes 2019, 10, 342. [Google Scholar] [CrossRef]
- Hristov, M.; Lazarov, L. Inhibition of nitric oxide synthase or cystathionine gamma-lyase abolishes leptin-induced fever in male rats. J. Therm. Biol. 2023, 112, 103443. [Google Scholar] [CrossRef] [PubMed]
- van Swieten, M.M.; Pandit, R.; Adan, R.A.; van der Plasse, G. The neuroanatomical function of leptin in the hypothalamus. J. Chem. Neuroanat. 2014, 61–62, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Wauman, J.; Zabeau, L.; Tavernier, J. The Leptin Receptor Complex: Heavier Than Expected? Front. Endocrinol. 2017, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Boyle, A.M.; Holland, O.J.; Hryciw, D.H. Link Between Umbilical Cord Blood Adipokines and Early Childhood Health. Endocrines 2025, 6, 13. [Google Scholar] [CrossRef]
- Münzberg, H.; Morrison, C.D. Structure, production and signaling of leptin. Metab. Clin. Exp. 2015, 64, 13–23. [Google Scholar] [CrossRef]
- Coppari, R.; Bjørbæk, C. Leptin revisited: Its mechanism of action and potential for treating diabetes. Nat. Rev. Drug Discov. 2012, 11, 692–708. [Google Scholar] [CrossRef]
- Myers, M.G.; Cowley, M.A.; Münzberg, H. Mechanisms of leptin action and leptin resistance. Annu. Rev. Physiol. 2008, 70, 537–556. [Google Scholar] [CrossRef]
- Cui, H.; López, M.; Rahmouni, K. The cellular and molecular bases of leptin and ghrelin resistance in obesity. Nat. Rev. Endocrinol. 2017, 13, 338–351. [Google Scholar] [CrossRef]
- Hristov, M.; Landzhov, B.; Yakimova, K. Cafeteria diet-induced obesity reduces leptin-stimulated NADPH-diaphorase reactivity in the hypothalamic arcuate nucleus of rats. Acta Histochem. 2020, 122, 151616. [Google Scholar] [CrossRef]
- Hristov, M.; Landzhov, B.; Nikolov, R.; Yakimova, K. Central, but not systemic, thermoregulatory effects of leptin are impaired in rats with obesity: Interactions with GABAB agonist and antagonist. Amino Acids 2019, 51, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.H.; Proenca, R.; Montez, J.M.; Carroll, K.M.; Darvishzadeh, J.G.; Lee, J.I.; Friedman, J.M. Abnormal splicing of the leptin receptor in diabetic mice. Nature 1996, 379, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.C.; Lord, R.A.; Anderson, G.M. Multiple Leptin Signalling Pathways in the Control of Metabolism and Fertility: A Means to Different Ends? Int. J. Mol. Sci. 2021, 22, 9210. [Google Scholar] [CrossRef] [PubMed]
- Donato, J.; Frazão, R., Jr.; Elias, C.F. The PI3K signaling pathway mediates the biological effects of leptin. Arq. Bras. Endocrinol. E Metabol. 2010, 54, 591–602. [Google Scholar] [CrossRef]
- Hâkansson, M.L.; Brown, H.; Ghilardi, N.; Skoda, R.C.; Meister, B. Leptin receptor immunoreactivity in chemically defined target neurons of the hypothalamus. J Neurosci. 1998, 18, 559–572. [Google Scholar] [CrossRef]
- Scott, M.M.; Lachey, J.L.; Sternson, S.M.; Lee, C.E.; Elias, C.F.; Friedman, J.M.; Elmquist, J.K. Leptin targets in the mouse brain. J. Comp. Neurol. 2009, 514, 518–532. [Google Scholar] [CrossRef]
- Manfredi-Lozano, M.; Roa, J.; Ruiz-Pino, F.; Piet, R.; Garcia-Galiano, D.; Pineda, R.; Zamora, A.; Leon, S.; Sanchez-Garrido, M.A.; Romero-Ruiz, A.; et al. Defining a novel leptin-melanocortin-kisspeptin pathway involved in the metabolic control of puberty. Mol. Metab. 2016, 5, 844–857. [Google Scholar] [CrossRef]
- Lavoie, O.; Michael, N.J.; Caron, A. A critical update on the leptin-melanocortin system. J. Neurochem. 2023, 165, 467–486. [Google Scholar] [CrossRef]
- Perello, M.; Scott, M.M.; Sakata, I.; Lee, C.E.; Chuang, J.C.; Osborne-Lawrence, S.; Rovinsky, S.A.; Elmquist, J.K.; Zigman, J.M. Functional implications of limited leptin receptor and ghrelin receptor coexpression in the brain. J. Comp. Neurol. 2012, 520, 281–294. [Google Scholar] [CrossRef]
- Baquero, A.F.; de Solis, A.J.; Lindsley, S.R.; Kirigiti, M.A.; Smith, M.S.; Cowley, M.A.; Zeltser, L.M.; Grove, K.L. Developmental switch of leptin signaling in arcuate nucleus neurons. J. Neurosci. 2014, 34, 9982–9994. [Google Scholar] [CrossRef]
- Solheim, M.H.; Stroganov, S.; Chen, W.; Subagia, P.S.; Bauder, C.A.; Wnuk-Lipinski, D.; Del Río-Martín, A.; Sotelo-Hitschfeld, T.; Beddows, C.A.; Klemm, P.; et al. Hypothalamic PNOC/NPY neurons constitute mediators of leptin-controlled energy homeostasis. Cell 2025, 188, 3550–3566.e22. [Google Scholar] [CrossRef]
- Tan, H.L.; Yin, L.; Tan, Y.; Ivanov, J.; Plucinska, K.; Ilanges, A.; Herb, B.R.; Wang, P.; Kosse, C.; Cohen, P.; et al. Leptin-activated hypothalamic BNC2 neurons acutely suppress food intake. Nature 2024, 636, 198–205. [Google Scholar] [CrossRef]
- Santinga, J. The Role of Lepr/Pirt Neurons in the Control of Reproductive Function by Leptin. Bachelor’s Thesis in Neuroscience with Honors, University of Michigan, Ann Arbor, MI, USA, 30 March 2023. [Google Scholar]
- Onoharigho, R.; Campbell, J. A novel neuron population that selectively controls metabolism. Physiology 2025, 40 (Suppl. S1), 0789. [Google Scholar] [CrossRef]
- Jang, Y.; Heo, J.Y.; Lee, M.J.; Zhu, J.; Seo, C.; Go, D.H.; Yoon, S.K.; Yukari, D.; Oike, Y.; Sohn, J.-W.; et al. Angiopoietin-Like Growth Factor Involved in Leptin Signaling in the Hypothalamus. Int. J. Mol. Sci. 2021, 22, 3443. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Wu, Q.; Wang, H.; Liu, D.; Chen, C.; Zhu, Z.; Zheng, H.; Yang, G.; Li, L.; Yang, M. AZGP1 in POMC neurons modulates energy homeostasis and metabolism through leptin-mediated STAT3 phosphorylation. Nat. Commun. 2024, 15, 3377. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.; Kim, K.-K.; Lee, T.-H.; Kim, H.-R.; Park, B.-S.; Park, J.-W.; Jeong, J.-K.; Seong, J.-Y.; Lee, B.-J. Spexin Regulates Hypothalamic Leptin Action on Feeding Behavior. Biomolecules 2022, 12, 236. [Google Scholar] [CrossRef]
- Kaneko, K.; Lu, W.; Xu, Y.; Morozov, A.; Fukuda, M. The small GTPase Rap1 in POMC neurons regulates leptin actions and glucose metabolism. Mol. Metab. 2025, 95, 102117. [Google Scholar] [CrossRef]
- Tan, B.; Hedbacker, K.; Kelly, L.; Zhang, Z.; Moura-Assis, A.; Luo, J.D.; Rabinowitz, J.D.; Friedman, J.M. A cellular and molecular basis of leptin resistance. Cell Metab. 2025, 37, 723–741.e6. [Google Scholar] [CrossRef]
- Liu, H.; He, Y.; Bai, J.; Zhang, C.; Zhang, F.; Yang, Y.; Luo, H.; Yu, M.; Liu, H.; Tu, L.; et al. Hypothalamic Grb10 enhances leptin signalling and promotes weight loss. Nat. Metab. 2023, 5, 147–164. [Google Scholar] [CrossRef]
- Ajwani, J.; Hwang, E.; Portillo, B.; Lieu, L.; Wallace, B.; Kabahizi, A.; He, Z.; Dong, Y.; Grose, K.; Williams, K.W. Upregulation of Xbp1 in NPY/AgRP neurons reverses diet-induced obesity and ameliorates leptin and insulin resistance. Neuropeptides 2024, 108, 102461. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.B.S. Low-dose peripheral leptin infusion produces selective activation of ventromedial hypothalamic and hindbrain STAT3. American journal of physiology. Endocrinol. Metab. 2023, 325, E72–E82. [Google Scholar] [CrossRef]
- Ravussin, Y.; LeDuc, C.A.; Watanabe, K.; Mueller, B.; Skowronski, A.; Rosenbaum, M.; Leibel, R. Effects of chronic leptin infusion on subsequent body weight and composition in mice: Can body weight set point be reset? Mol. Metab. 2014, 3, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, H.; Zigman, J.M.; Ye, C.; Lee, C.E.; McGovern, R.A.; Tang, V.; Kenny, C.D.; Christiansen, L.M.; White, R.D.; Edelstein, E.A.; et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 2006, 49, 191–203. [Google Scholar] [CrossRef]
- Ameroso, D.; Meng, A.; Chen, S.; Felsted, J.; Dulla, C.G.; Rios, M. Astrocytic BDNF signaling within the ventromedial hypothalamus regulates energy homeostasis. Nat. Metab. 2022, 4, 627–643. [Google Scholar] [CrossRef]
- Ahn, W.; Latremouille, J.; Harris, R.B.S. Leptin receptor-expressing cells in the ventromedial nucleus of the hypothalamus contribute to enhanced CCK-induced satiety following central leptin injection. American journal of physiology. Endocrinol. Metab. 2022, 323, E267–E280. [Google Scholar] [CrossRef]
- Hurley, M.M.; Anderson, E.M.; Chen, C.; Maunze, B.; Hess, E.M.; Block, M.E.; Patel, N.; Cooper, Z.; McCoy, R.; Dabra, T.; et al. Acute Blockade of PACAP-Dependent Activity in the Ventromedial Nucleus of the Hypothalamus Disrupts Leptin-Induced Behavioral and Molecular Changes in Rats. Neuroendocrinology 2020, 110, 271–281. [Google Scholar] [CrossRef]
- Oladun, B.T.; Kim, M.H. Epigenetic Regulation of Leptin Signaling by Hypothalamic JMJD3 Mitigates High-Fat Diet-Induced Obesity in Mice. Curr. Dev. Nutr. 2024, 8 (Suppl. S2), 103638. [Google Scholar] [CrossRef]
- Enriori, P.J.; Sinnayah, P.; Simonds, S.E.; Garcia Rudaz, C.; Cowley, M.A. Leptin action in the dorsomedial hypothalamus increases sympathetic tone to brown adipose tissue in spite of systemic leptin resistance. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 12189–12197. [Google Scholar] [CrossRef]
- Dodd, G.T.; Worth, A.A.; Nunn, N.; Korpal, A.K.; Bechtold, D.A.; Allison, M.B.; Myers, M.G.; Statnick, M.A., Jr.; Luckman, S.M. The thermogenic effect of leptin is dependent on a distinct population of prolactin-releasing peptide neurons in the dorsomedial hypothalamus. Cell Metab. 2014, 20, 639–649. [Google Scholar] [CrossRef]
- Zhang, Y.; Kerman, I.A.; Laque, A.; Nguyen, P.; Faouzi, M.; Louis, G.W.; Jones, J.C.; Rhodes, C.; Münzberg, H. Leptin-receptor-expressing neurons in the dorsomedial hypothalamus and median preoptic area regulate sympathetic brown adipose tissue circuits. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 1873–1884. [Google Scholar] [CrossRef]
- Francois, M.; Kaiser, L.; He, Y.; Xu, Y.; Salbaum, J.M.; Yu, S.; Morrison, C.D.; Berthoud, H.R.; Münzberg, H. Leptin receptor neurons in the dorsomedial hypothalamus require distinct neuronal subsets for thermogenesis and weight loss. Metab. Clin. Exp. 2025, 163, 156100. [Google Scholar] [CrossRef] [PubMed]
- Rupp, A.C.; Tomlinson, A.J.; Affinati, A.H.; Yacawych, W.T.; Duensing, A.M.; True, C.; Lindsley, S.R.; Kirigiti, M.A.; MacKenzie, A.; Polex-Wolf, J.; et al. Suppression of food intake by Glp1r/Lepr-coexpressing neurons prevents obesity in mouse models. J. Clin. Investig. 2023, 133, e157515. [Google Scholar] [CrossRef] [PubMed]
- Polex-Wolf, J.; Deibler, K.; Hogendorf, W.F.J.; Bau, S.; Glendorf, T.; Stidsen, C.E.; Tornøe, C.W.; Tiantang, D.; Lundh, S.; Pyke, C.; et al. Glp1r-Lepr coexpressing neurons modulate the suppression of food intake and body weight by a GLP-1/leptin dual agonist. Sci. Transl. Med. 2024, 16, eadk4908. [Google Scholar] [CrossRef]
- Faber, C.L.; Deem, J.D.; Phan, B.A.; Doan, T.P.; Ogimoto, K.; Mirzadeh, Z.; Schwartz, M.W.; Morton, G.J. Leptin receptor neurons in the dorsomedial hypothalamus regulate diurnal patterns of feeding, locomotion, and metabolism. eLife 2021, 10, e63671. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Godschall, E.; Brennan, C.D.; Zhang, Q.; Abraham-Fan, R.J.; Williams, S.P.; Güngül, T.B.; Onoharigho, R.; Buyukaksakal, A.; Salinas, R.; et al. Leptin receptor neurons in the dorsomedial hypothalamus input to the circadian feeding network. Sci. Adv. 2023, 9, eadh9570. [Google Scholar] [CrossRef]
- Yao, Q.; Pho, H.; Kirkness, J.; Ladenheim, E.E.; Bi, S.; Moran, T.H.; Fuller, D.D.; Schwartz, A.R.; Polotsky, V.Y. Localizing Effects of Leptin on Upper Airway and Respiratory Control during Sleep. Sleep 2016, 39, 1097–1106. [Google Scholar] [CrossRef]
- Berger, S.; Polotsky, V.Y. Leptin and Leptin Resistance in the Pathogenesis of Obstructive Sleep Apnea: A Possible Link to Oxidative Stress and Cardiovascular Complications. Oxidative Med. Cell. Longev. 2018, 2018, 5137947. [Google Scholar] [CrossRef] [PubMed]
- Phipps, P.R.; Starritt, E.; Caterson, I.; Grunstein, R.R. Association of serum leptin with hypoventilation in human obesity. Thorax 2002, 57, 75–76. [Google Scholar] [CrossRef] [PubMed]
- Pho, H.; Berger, S.; Freire, C.; Kim, L.J.; Shin, M.K.; Streeter, S.R.; Hosamane, N.; Cabassa, M.E.; Anokye-Danso, F.; Dergacheva, O.; et al. Leptin receptor expression in the dorsomedial hypothalamus stimulates breathing during NREM sleep in db/db mice. Sleep 2021, 44, zsab046. [Google Scholar] [CrossRef]
- Amorim, M.R.; Wang, X.; Pho, H.; Bevans-Fonti, S.; Anokye-Danso, F.; Escobar, J.; Dergacheva, O.; Branco, L.G.; Mendelowitz, D.; Polotsky, V.Y. Activation of Leptin-Receptor-Expressing Neurons in the Dorsomedial Hypothalamus Increases Hypercapcnic Ventilatory Response and Relieves Obstructive Sleep Apnea in Obese Mice. FASEB J. 2022, 36, R2522. [Google Scholar] [CrossRef]
- Amorim, M.R.; Wang, X.; Aung, O.; Bevans-Fonti, S.; Anokye-Danso, F.; Ribeiro, C.; Escobar, J.; Freire, C.; Pho, H.; Dergacheva, O.; et al. Leptin signaling in the dorsomedial hypothalamus couples breathing and metabolism in obesity. Cell Rep. 2023, 42, 113512. [Google Scholar] [CrossRef] [PubMed]
- Bonnavion, P.; Mickelsen, L.E.; Fujita, A.; de Lecea, L.; Jackson, A.C. Hubs and spokes of the lateral hypothalamus: Cell types, circuits and behaviour. J. Physiol. 2016, 594, 6443–6462. [Google Scholar] [CrossRef]
- Fakhoury, M.; Salman, I.; Najjar, W.; Merhej, G.; Lawand, N. The Lateral Hypothalamus: An Uncharted Territory for Processing Peripheral Neurogenic Inflammation. Front. Neurosci. 2020, 14, 101. [Google Scholar] [CrossRef]
- Goforth, P.B.; Leinninger, G.M.; Patterson, C.M.; Satin, L.S.; Myers, M.G., Jr. Leptin acts via lateral hypothalamic area neurotensin neurons to inhibit orexin neurons by multiple GABA-independent mechanisms. J. Neurosci. 2014, 34, 11405–11415. [Google Scholar] [CrossRef]
- Leinninger, G.M.; Opland, D.M.; Jo, Y.H.; Faouzi, M.; Christensen, L.; Cappellucci, L.A.; Rhodes, C.J.; Gnegy, M.E.; Becker, J.B.; Pothos, E.N.; et al. Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab. 2011, 14, 313–323. [Google Scholar] [CrossRef]
- Opland, D.; Sutton, A.; Woodworth, H.; Brown, J.; Bugescu, R.; Garcia, A.; Christensen, L.; Rhodes, C.; Myers, M.; Leinninger, G., Jr. Loss of neurotensin receptor-1 disrupts the control of the mesolimbic dopamine system by leptin and promotes hedonic feeding and obesity. Mol. Metab. 2013, 2, 423–434. [Google Scholar] [CrossRef]
- Laque, A.; Yu, S.; Qualls-Creekmore, E.; Gettys, S.; Schwartzenburg, C.; Bui, K.; Rhodes, C.; Berthoud, H.R.; Morrison, C.D.; Richards, B.K.; et al. Leptin modulates nutrient reward via inhibitory galanin action on orexin neurons. Mol. Metab. 2015, 4, 706–717. [Google Scholar] [CrossRef]
- Liu, J.J.; Bello, N.T.; Pang, Z.P. Presynaptic Regulation of Leptin in a Defined Lateral Hypothalamus-Ventral Tegmental Area Neurocircuitry Depends on Energy State. J. Neurosci. 2017, 37, 11854–11866. [Google Scholar] [CrossRef] [PubMed]
- Kakava-Georgiadou, N.; Drkelic, V.; Garner, K.M.; Luijendijk, M.C.M.; Basak, O.; Adan, R.A.H. Molecular profile and response to energy deficit of leptin-receptor neurons in the lateral hypothalamus. Sci. Rep. 2022, 12, 13374. [Google Scholar] [CrossRef]
- Schiffino, F.L.; Siemian, J.N.; Petrella, M.; Laing, B.T.; Sarsfield, S.; Borja, C.B.; Gajendiran, A.; Zuccoli, M.L.; Aponte, Y. Activation of a lateral hypothalamic-ventral tegmental circuit gates motivation. PLoS ONE 2019, 14, e0219522. [Google Scholar] [CrossRef]
- Siemian, J.N.; Arenivar, M.A.; Sarsfield, S.; Borja, C.B.; Russell, C.N.; Aponte, Y. Lateral hypothalamic LEPR neurons drive appetitive but not consummatory behaviors. Cell Rep. 2021, 36, 109615. [Google Scholar] [CrossRef]
- Lee, Y.H.; Kim, Y.B.; Kim, K.S.; Jang, M.; Song, H.Y.; Jung, S.H.; Ha, D.S.; Park, J.S.; Lee, J.; Kim, K.M.; et al. Lateral hypothalamic leptin receptor neurons drive hunger-gated food-seeking and consummatory behaviours in male mice. Nat. Commun. 2023, 14, 1486. [Google Scholar] [CrossRef]
- Guillaume, S.; Jaussent, I.; Maimoun, L.; Ryst, A.; Seneque, M.; Villain, L.; Hamroun, D.; Lefebvre, P.; Renard, E.; Courtet, P. Associations between adverse childhood experiences and clinical characteristics of eating disorders. Sci. Rep. 2016, 6, 35761. [Google Scholar] [CrossRef] [PubMed]
- Brustenghi, F.; Mezzetti, F.A.F.; Di Sarno, C.; Giulietti, C.; Moretti, P.; Tortorella, A. Eating Disorders: The Role of Childhood Trauma and the Emotion Dysregulation. Psychiatr. Danub. 2019, 31 (Suppl. S3), 509–511. [Google Scholar] [PubMed]
- Shin, S.; You, I.J.; Jeong, M.; Bae, Y.; Wang, X.Y.; Cawley, M.L.; Han, A.; Lim, B.K. Early adversity promotes binge-like eating habits by remodeling a leptin-responsive lateral hypothalamus-brainstem pathway. Nat. Neurosci. 2023, 26, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Hristov, M.; Landzhov, B.; Yakimova, K. Effect of leptin on nitrergic neurons in the lateral hypothalamic area and the supraoptic nucleus of rats. Biotech. Histochem. Off. Publ. Biol. Stain Comm. 2024, 99, 125–133. [Google Scholar] [CrossRef]
- Hristov, M.; Landzhov, B.; Yakimova, K. Increased NADPH-diaphorase reactivity in the hypothalamic paraventricular nucleus and tanycytes following systemic administration of leptin in rats. Acta Histochem. 2019, 121, 690–694. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hristov, M. Leptin Signaling in the Hypothalamus: Cellular Insights and Therapeutic Perspectives in Obesity. Endocrines 2025, 6, 42. https://doi.org/10.3390/endocrines6030042
Hristov M. Leptin Signaling in the Hypothalamus: Cellular Insights and Therapeutic Perspectives in Obesity. Endocrines. 2025; 6(3):42. https://doi.org/10.3390/endocrines6030042
Chicago/Turabian StyleHristov, Milen. 2025. "Leptin Signaling in the Hypothalamus: Cellular Insights and Therapeutic Perspectives in Obesity" Endocrines 6, no. 3: 42. https://doi.org/10.3390/endocrines6030042
APA StyleHristov, M. (2025). Leptin Signaling in the Hypothalamus: Cellular Insights and Therapeutic Perspectives in Obesity. Endocrines, 6(3), 42. https://doi.org/10.3390/endocrines6030042






