Dicamba Injury on Soybean Assessed Visually and with Spectral Vegetation Index
Abstract
1. Introduction
2. Materials and Methods
2.1. Treatments
2.2. Evaluations
2.3. Statistical Analysis
3. Results and Discussion
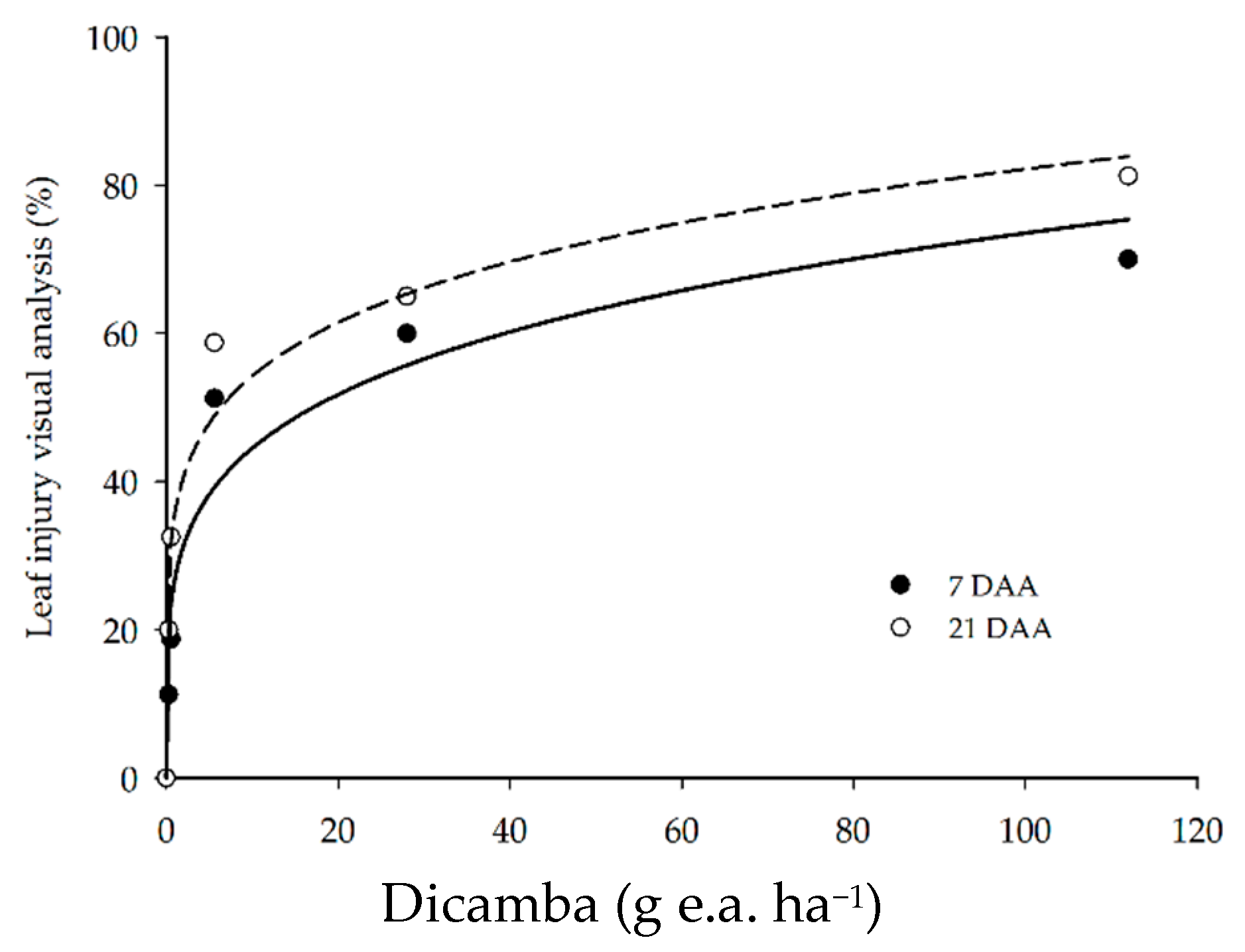
3.1. Leaf Injury Visual Analysis
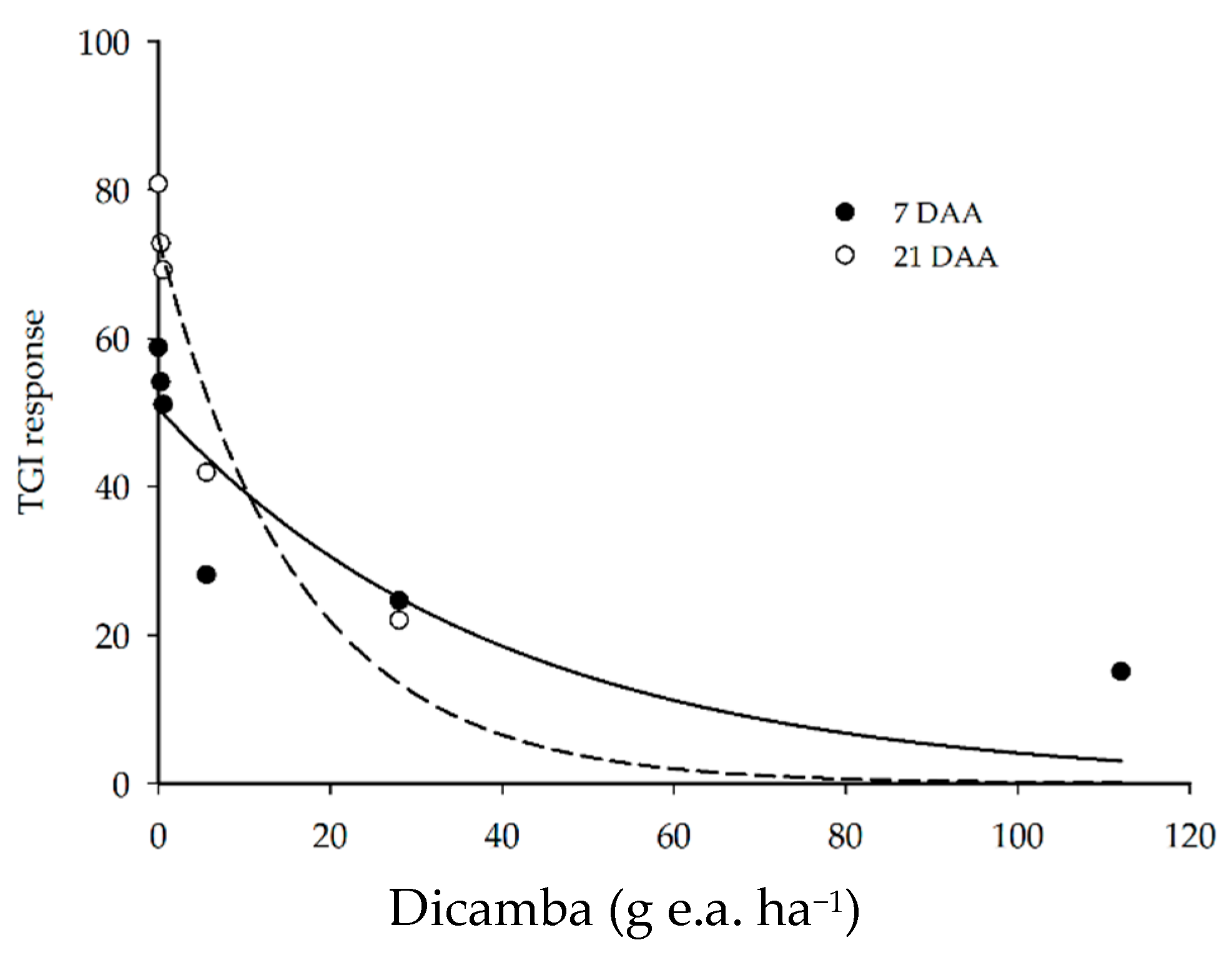
3.2. Spectral Vegetation Responses
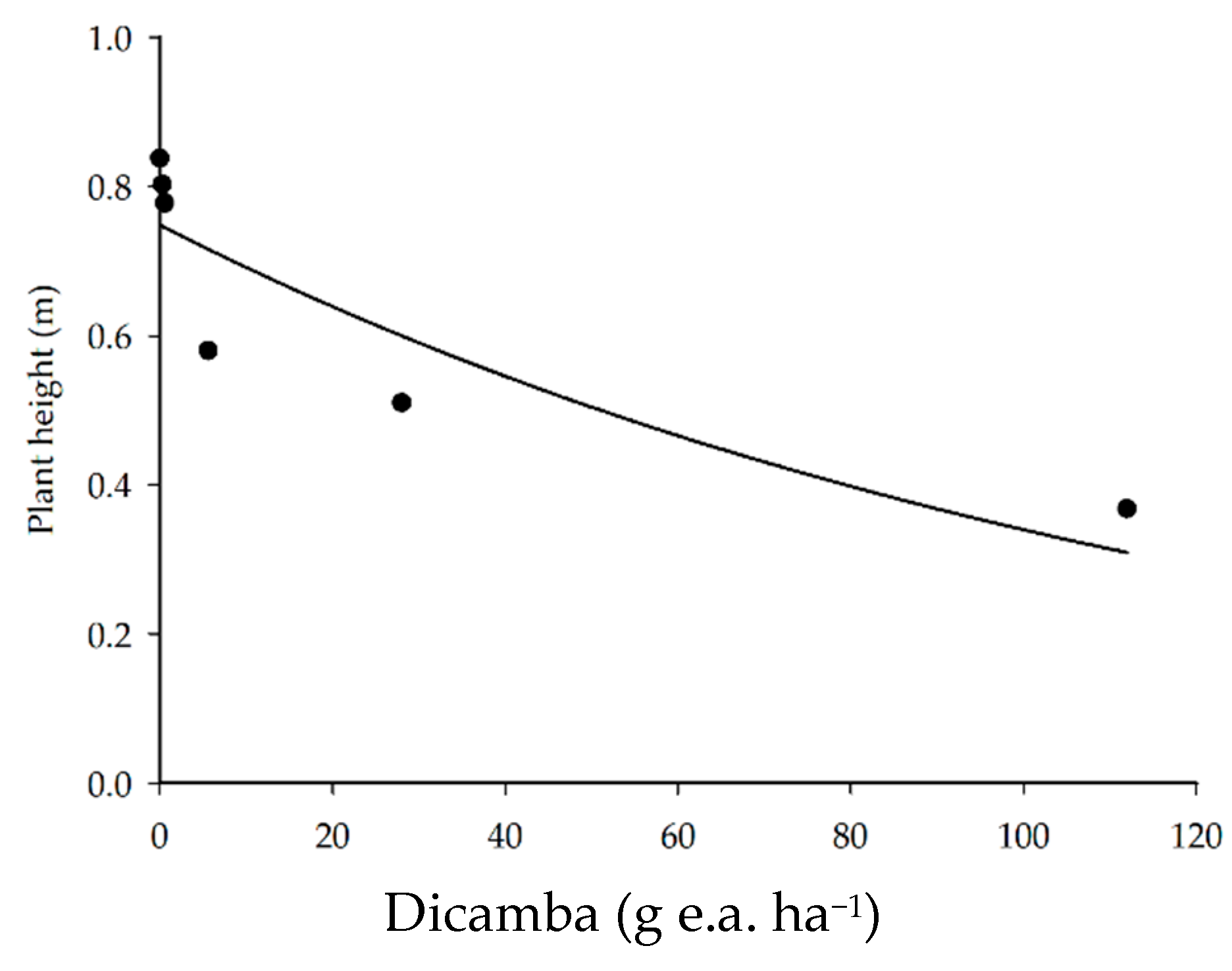
3.3. Plant Height
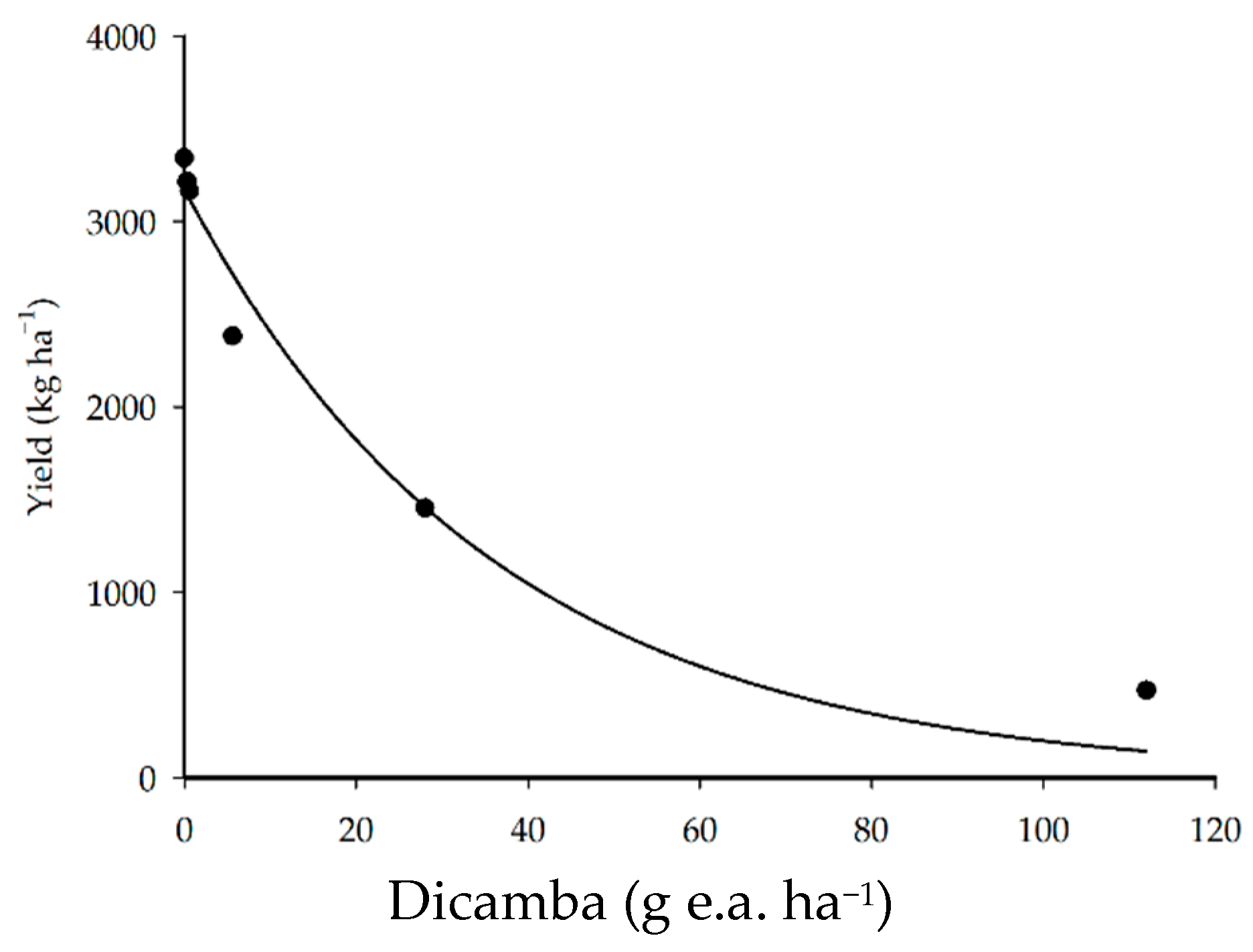
3.4. Crop Yield
3.5. Correlations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Behrens, M.R.; Mutlu, N.; Chakraborty, S.; Dumitru, R.; Jiang, W.Z.; Lavallee, B.J.; Herman, P.L.; Clemente, T.E.; Weeks, D.P. Dicamba resistance: Enlarging and preserving biotechnology-based weed management strategies. Science 2007, 316, 1185–1188. [Google Scholar] [CrossRef] [PubMed]
- Byker, H.P.; Soltani, N.; Robinson, D.; Tardif, F.J.; Lawton, M.B.; Sikkema, P.H. Control of glyphosate-resistant horseweed (conyza canadensis) with dicamba applied preplant and postemergence in dicamba-resistant soybean. Weed Technol. 2013, 27, 492–496. [Google Scholar] [CrossRef]
- Egan, J.F.; Mortensen, D.A. Quantifying vapor drift of dicamba herbicides applied to soybean. Environ. Toxicol. Chem. 2012, 31, 1023–1031. [Google Scholar] [CrossRef]
- Osipitan, O.A.; Scott, J.E.; Knezevic, S.Z. Glyphosate-resistant soybean response to micro-rates of three dicamba-based herbicides. Agrosyst. Geosci. Environ. 2019, 2. [Google Scholar] [CrossRef]
- Andersen, S.M.; Clay, S.A.; Wrage, L.J.; Matthees, D. Soybean foliage residues of dicamba and 2,4-d and correlation to application rates and yield. Agron. J. 2004, 96, 750–760. [Google Scholar] [CrossRef]
- Foster, M.R.; Griffin, J.L. Changes in soybean yield components in response to dicamba. Agrosyst. Geosci. Environ. 2019, 2. [Google Scholar] [CrossRef]
- Foster, M.R.; Griffin, J.L.; Copes, J.T.; Blouin, D.C. Development of a model to predict soybean yield loss from dicamba exposure. Weed Technol. 2019, 33, 287–295. [Google Scholar] [CrossRef]
- Kniss, A.R. Soybean response to dicamba: A meta-analysis. Weed Technol. 2018, 32, 507–512. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, Y.; Reddy, K.N.; Wang, B. Assessing crop damage from dicamba on non-dicamba-tolerant soybean by hyperspectral imaging through machine learning. Pest Manag. Sci. 2019, 75, 3260–3272. [Google Scholar] [CrossRef]
- Solomom, C.B.; Bradley, K.W. Influence of application timings and sublethal rates of synthetic auxin herbicides on soybean. Weed Tech. 2014, 28, 454–464. [Google Scholar] [CrossRef]
- Everitt, J.D.; Keeling, J.W. Cotton growth and yield response to simulated 2,4-D and dicamba drift. Weed Tech. 2009, 23, 503–506. [Google Scholar] [CrossRef]
- Hunt, E.R.; Daughtry, C.S.T. What good are unmanned aircraft systems for agricultural remote sensing and precision agriculture? Int. J. Remote Sens. 2018, 39, 5345–5376. [Google Scholar] [CrossRef]
- Atzberger, C. Advances in remote sensing of agriculture: Context description, existing operational monitoring systems and major information needs. Remote Sens. 2013, 5, 949–981. [Google Scholar] [CrossRef]
- Henry, W.B.; Shaw, D.R.; Reddy, K.R.; Bruce, L.M.; Tamhankar, H.D. Remote sensing to detect herbicide drift on crops. Weed Technol. 2004, 18, 358–368. [Google Scholar] [CrossRef]
- Xue, J.; Su, B. Significant remote sensing vegetation indices: A review of developments and applications. J. Sens. 2017, 2017, 1353691. [Google Scholar] [CrossRef]
- Duddu, H.S.N.; Johnson, E.N.; Willenborg, C.J.; Shirtliffe, S.J. High-throughput UAV image-based method is more precise than manual rating of herbicide tolerance. Plant Phenomics 2019, 2019, 6036453. [Google Scholar] [CrossRef]
- Huang, Y.; Yuan, L.; Reddy, K.N.; Zhang, J. In-situ plant hyperspectral sensing for early detection of soybean injury from dicamba. Biosyst. Eng. 2016, 149, 51–59. [Google Scholar] [CrossRef]
- Freire-Silva, J.; Paz, Y.E.; Lima-Silva, P.P.; Pereira, J.A.S.; Candeias, A.L.B. Índices de vegetação do sensoriamento remoto para processamento de imagens na faixa do visível (RGB). J. Hyperspectr. Remote Sens. 2019, 9, 228–239. [Google Scholar] [CrossRef]
- Hunt, E.R.; Doraiswamy, P.C.; McMurtrey, J.E.; Daughtry, C.S.; Perry, E.M.; Akhmedov, B. A visible band index for remote sensing leaf chlorophyll content at the canopy scale. Int. J. Appl. Earth Obs. Geoinf. 2013, 21, 103–112. [Google Scholar] [CrossRef]
- Netto, A.F.A.; Martins, R.N.; Souza, G.S.A.; Santos, F.F.L.; Rosas, J.T.F. Evaluation of a low-cost camera for agricultural applications. J. Exp. Agric. Int. 2019, 32, 1–9. [Google Scholar] [CrossRef]
- Hunt, E.R.; Daughtry, C.S.T.; Eitel, J.U.; Long, D.S. Remote sensing leaf chlorophyll content using a visible band index. Agron. J. 2011, 103, 1090–1099. [Google Scholar] [CrossRef]
- State of New South Wales. New South Wales Weed Control Handbook, NSW. 2018. Available online: https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0017/123317/weed-control-handbook.pdf (accessed on 26 April 2021).
- Behrens, R.; Lueschen, W.E. Dicamba volatility. Weed Sci. 1979, 27, 486–493. [Google Scholar] [CrossRef]
- Starý, K.; Jelínek, Z.; Kumhálová, J.; Chyba, J.; Balázová, K. Comparing RGB—based vegetation indices from UAV imageries to estimate hops canopy area. Agron. Res. 2020, 18, 2592–2601. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- McCown, S.; Barber, T.; Norsworthy, J.K. Response of non–dicamba-resistant soybean to dicamba as influenced by growth stage and herbicide rate. Weed Technol. 2018, 32, 513–519. [Google Scholar] [CrossRef]
- Schneider, T.; Rizzardi, M.A.; Rockenbach, A.P.; Peruzzo, S.T. Subdoses of dicamba herbicide on yield componentes in function of the soybean growth stage. J. Agric. Sci. 2019, 11, 407–415. [Google Scholar] [CrossRef]
- Costa, E.M.; Jakelaitis, A.; Zuchi, J.; Pereira, L.S.; Ventura, M.V.A.; Oliveira, G.S.; Sousa, G.D.; Silva, J.N. Simulated drift of dicamba and 2,4-D on soybeans: Effects of application dose and time. Biosci. J. 2020, 36, 857–864. [Google Scholar] [CrossRef]
- Weidenhamer, J.D.; Triplett, G.B.; Sobotka, F.E. Dicamba injury to soybean. Agron. J. 1989, 81, 637–643. [Google Scholar] [CrossRef]
- Hickman, M.V.; Everitt, J.H.; Escobar, D.E.; Richardson, A.J. Aerial photography and videography for detecting and mapping dicamba injury patterns. Weed Technol. 1991, 5, 700–706. [Google Scholar] [CrossRef]
- Huang, Y.; Reddy, K.N.; Thomson, S.J.; Yao, H. Assessment of soybean injury from glyphosate using airborne multispectral remote sensing. Pest Manag. Sci. 2015, 71, 545–552. [Google Scholar] [CrossRef]
- Huang, Y.; Thomson, S.J.; Molin, W.T.; Reddy, K.N.; Yao, H. Early detection of soybean plant injury from glyphosate by measuring chlorophyll reflectance and fluorescence. J. Agric. Sci. 2012, 4, 117–124. [Google Scholar] [CrossRef]
- Grossmann, K. Auxin herbicide action: Lifting the veil step by step. Plant Signal. Behav. 2007, 2, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, K. Auxin herbicides: Current status of mechanism and mode of action. Pest Manag. Sci. 2009, 66, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Peailillo, F.; Ortega-Farias, S.; Rivera, M.; Bardeen, M.; Moreno, M. Comparison of vegetation indices acquired from RGB and multispectral sensors placed on UAV. In Proceedings of the IEEE International Conference on Automation, Concepcion, Chile, 17–19 October 2018. [Google Scholar]
- Robinson, A.P.; Simpson, D.M.; Johnson, W.G. Response of glyphosate-tolerant soybean yield components to dicamba exposure. Weed Sci. 2013, 61, 526–536. [Google Scholar] [CrossRef]
- Callegari-Jacques, S.M. Bioestatística: Princípios e Aplicações; Artmed: Porto Alegre, Brazil, 2009. [Google Scholar]

| Source of Variation | df | Visual Analysis | TGI | Plant Height | Yield | ||
|---|---|---|---|---|---|---|---|
| 7 DAA | 21 DAA | 7 DAA | 21 DAA | ||||
| FC Test (p Value) | |||||||
| Dicamba | 5 | 1116.35 | 146.61 | 29.38 | 111.13 | 62.92 | 70.66 |
| (<0.001) | (<0.001) | (<0.001) | (<0.001) | (<0.001) | (<0.001) | ||
| Block | 3 | ||||||
| Error | 15 | ||||||
| CV (%) | 4.91 | 11.78 | 17.42 | 14.15 | 7.38 | 11.80 | |
| SW | 0.2436 + | 0.3547 + | 0.8041 + | 0.7992 + | 0.8388 + | 0.0289 + | |
| O&M | 0.5613 + | 0.1863 + | 0.4307 + | 0.0867 + | 0.4286 + | 0.8870 + | |
| T | 0.4927 + | 0.1624 + | 0.9643 + | 0.9643 + | 0.9467 + | 0.8759 + | |
| Variable | Function | R2 | FC | p Value | |
|---|---|---|---|---|---|
| visual analysis | 7 DAA | ŷ = 26.9312 x0.2181 | 92.78 | 51.4210 | 0.0020 |
| 21 DAA | ŷ = 35.8384 x0.1803 | 96.23 | 102.1533 | 0.0005 | |
| TGI | 7 DAA | ŷ = 50.5499 e−0.0252x | 71.18 | 9.8811 | 0.0347 |
| 21 DAA | ŷ = 73.5128 e−0.0606x | 94.89 | 74.2232 | 0.0010 | |
| Plant height | ŷ = 0.7482 e−0.0079x | 76.46 | 12.9927 | 0.0227 | |
| Yield | ŷ = 3172.6639 e−0.0278x | 96.24 | 102.3146 | 0.0005 | |
| V.A. 7 DAA | V.A. 21 DAA | TGI 7 DAA | TGI 21 DAA | Plant Height | Yield | |
|---|---|---|---|---|---|---|
| V.A. 7 DAA | 1 | 0.975 * | −0.951 * | −0.941 * | −0.955 * | −0.911 * |
| V.A. 21 DAA | 1 | −0.918 * | −0.920 * | −0.936 * | −0.890 * | |
| TGI 7 DAA | 1 | 0.932 * | 0.950 * | 0.883 * | ||
| TGI 21 DAA | 1 | 0.972 * | 0.952 * | |||
| Plant height | 1 | 0.950 * | ||||
| Yield | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marques, M.G.; da Cunha, J.P.A.R.; Lemes, E.M. Dicamba Injury on Soybean Assessed Visually and with Spectral Vegetation Index. AgriEngineering 2021, 3, 240-250. https://doi.org/10.3390/agriengineering3020016
Marques MG, da Cunha JPAR, Lemes EM. Dicamba Injury on Soybean Assessed Visually and with Spectral Vegetation Index. AgriEngineering. 2021; 3(2):240-250. https://doi.org/10.3390/agriengineering3020016
Chicago/Turabian StyleMarques, Matheus Gregorio, João Paulo Arantes Rodrigues da Cunha, and Ernane Miranda Lemes. 2021. "Dicamba Injury on Soybean Assessed Visually and with Spectral Vegetation Index" AgriEngineering 3, no. 2: 240-250. https://doi.org/10.3390/agriengineering3020016
APA StyleMarques, M. G., da Cunha, J. P. A. R., & Lemes, E. M. (2021). Dicamba Injury on Soybean Assessed Visually and with Spectral Vegetation Index. AgriEngineering, 3(2), 240-250. https://doi.org/10.3390/agriengineering3020016






