Status of Brain Imaging in Gastroparesis
Abstract
1. Introduction
2. Background
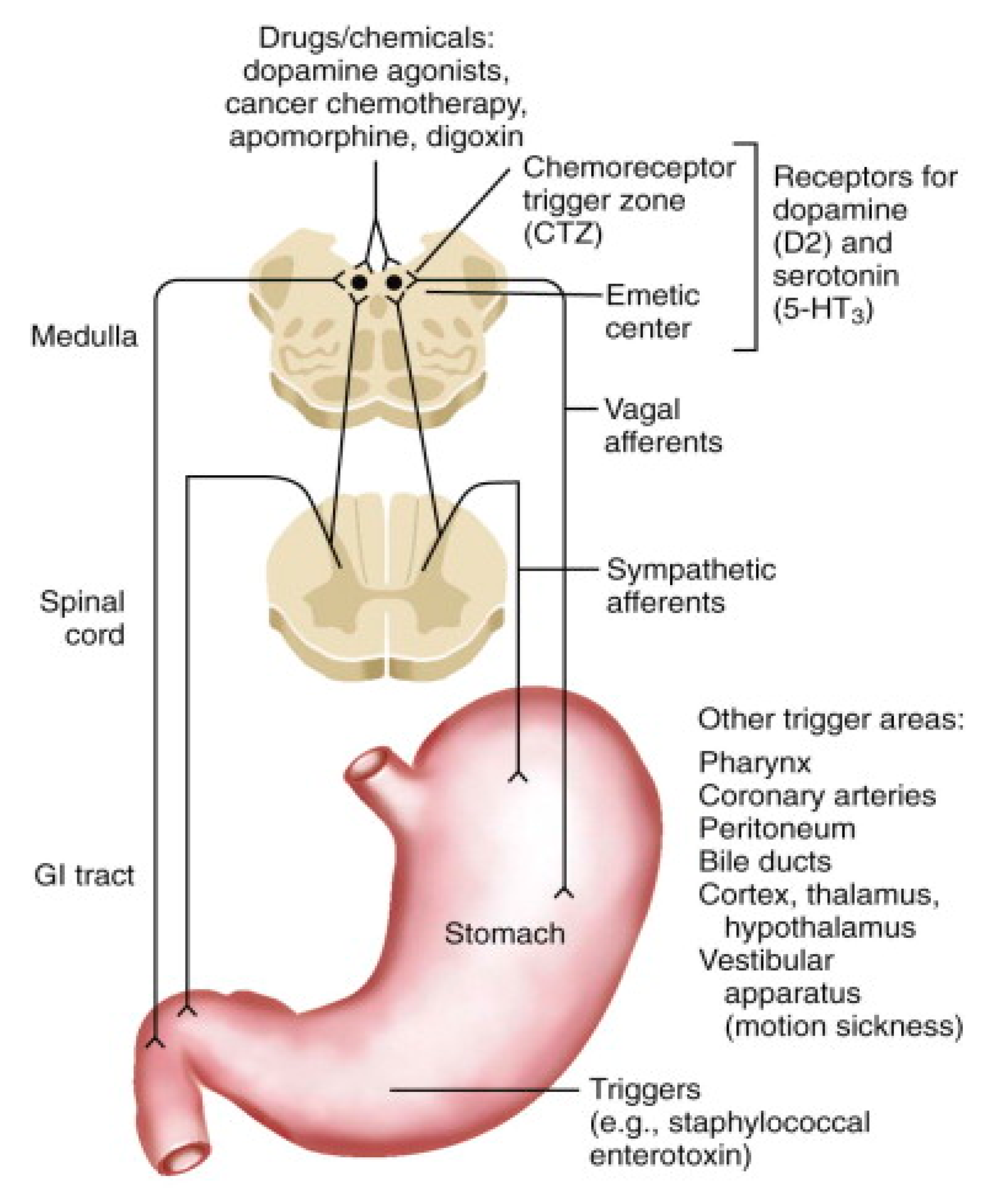
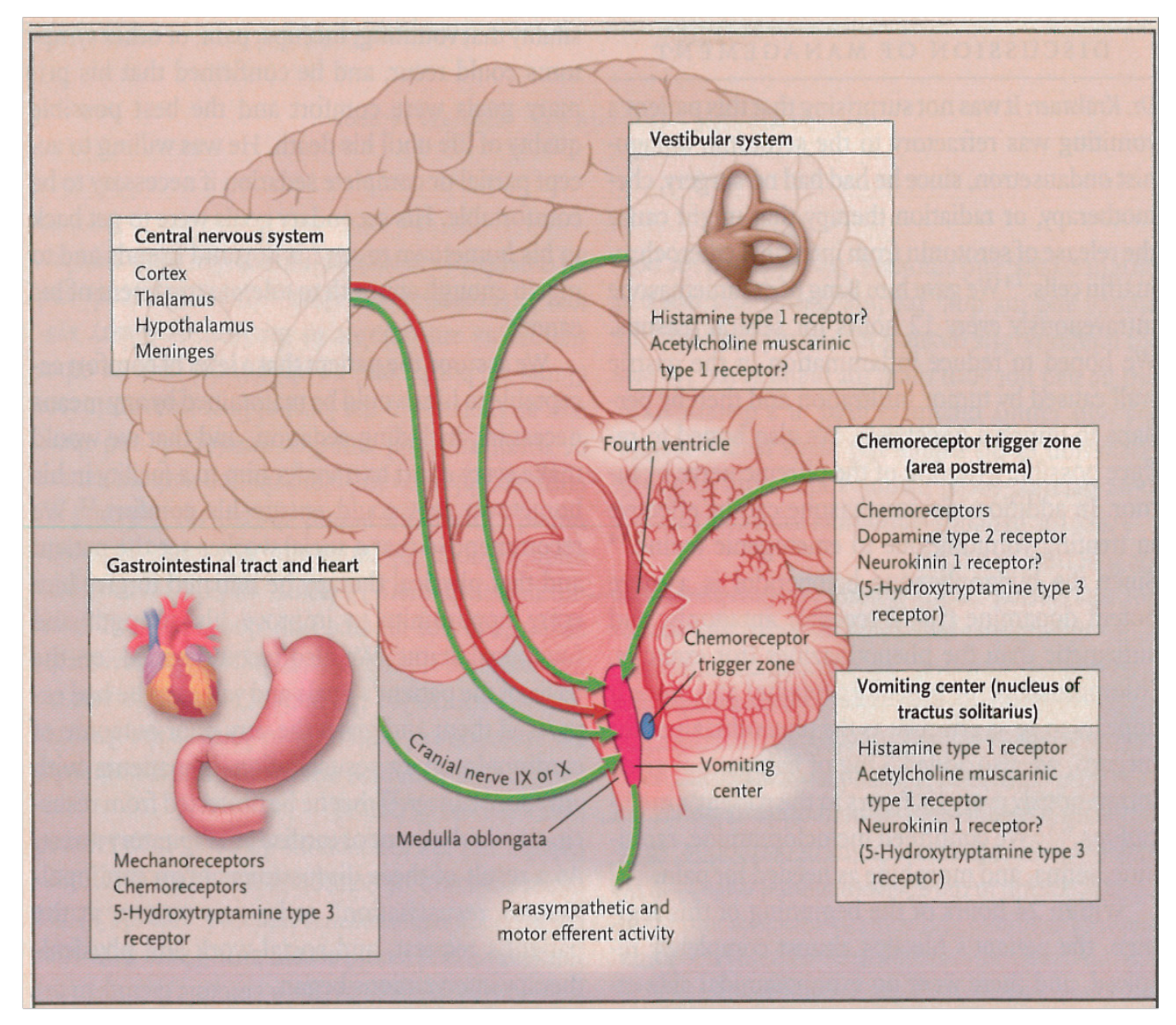
3. Physiology of Nausea and Vomiting
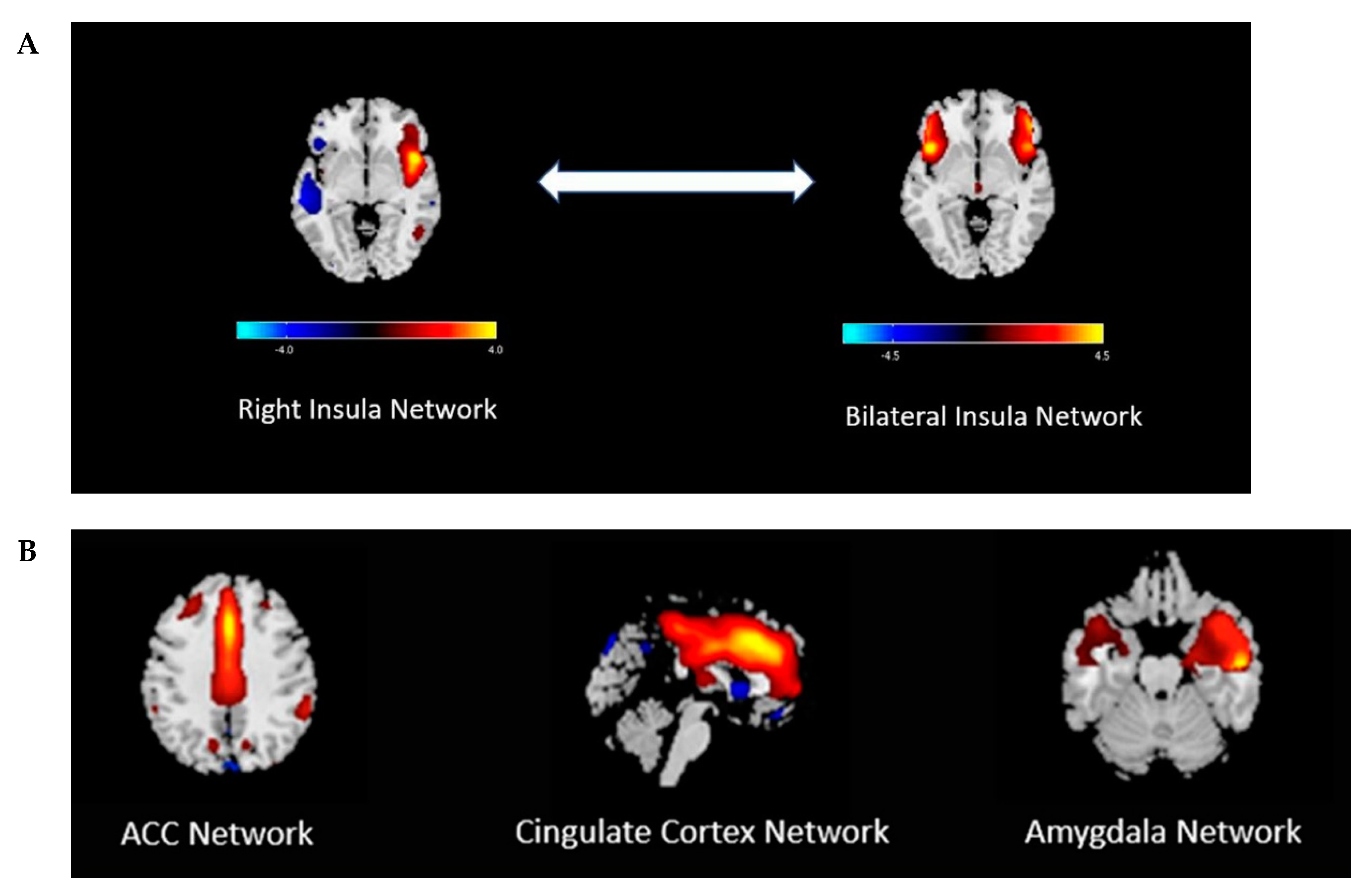
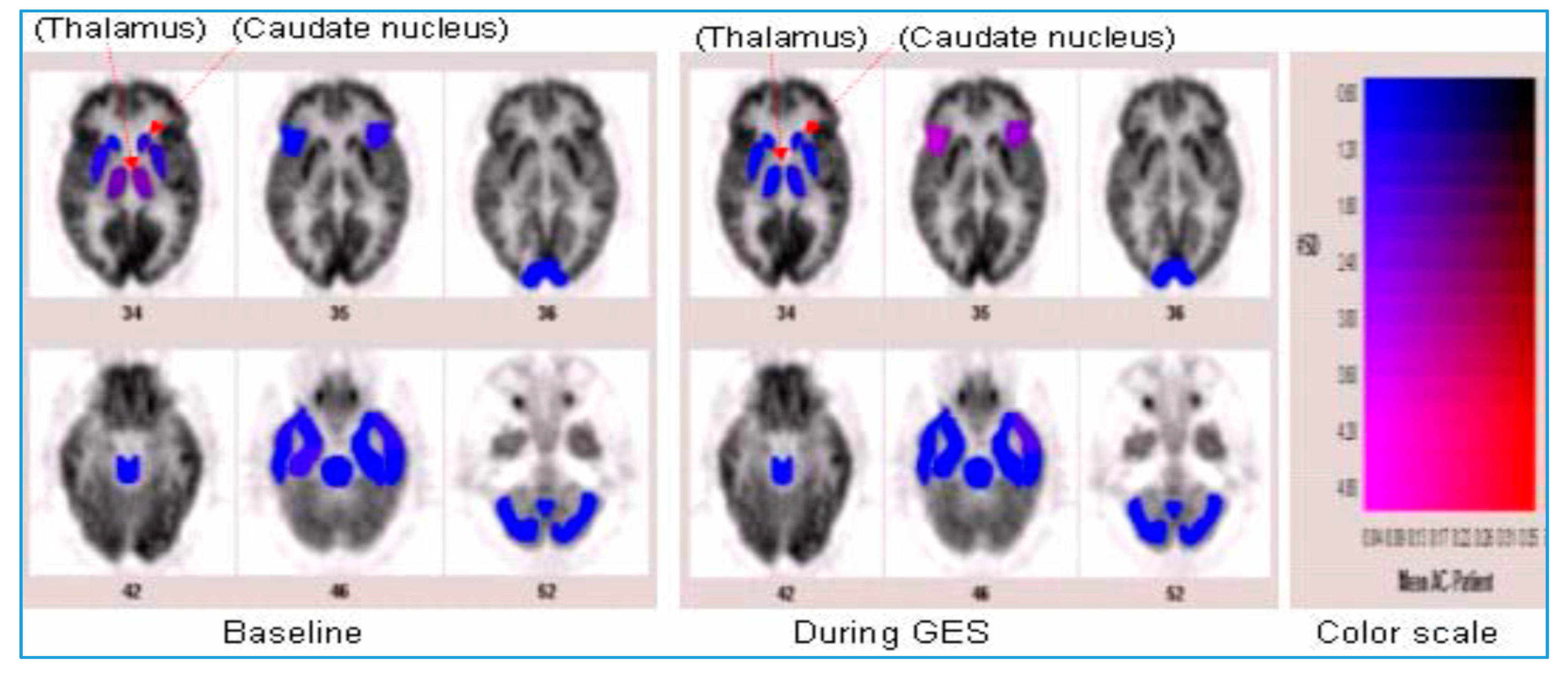
4. Discussion and Review of the Literature
5. Conclusion and Future Directions
Funding
Conflicts of Interest
References
- Parkman, H.P.; Hasler, W.L.; Fisher, R.S. American Gastroenterological Association medical position statement: Diagnosis and treatment of gastroparesis. Gastroenterology 2004, 127, 1589–1591. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M.; Hasler, W.L.; Parkman, H.P. AGA Technical Review on Nausea and Vomiting. Gastroenterology 2001, 120. [Google Scholar] [CrossRef] [PubMed]
- Hornby, P.J. Central neurocircuitry associated with emesis. Am. J. Med. 2001, 111 (Suppl. 8A), 106s–112s. [Google Scholar] [CrossRef]
- Ellingsen, D.M.; Garcia, R.G.; Lee, J.; Lin, R.L.; Kim, J.; Thurler, A.H.; Castel, S.; Dimisko, L.; Rosen, B.R.; Hadjikhani, N.; et al. Cyclic Vomiting Syndrome is characterized by altered functional brain connectivity of the insular cortex: A cross-comparison with migraine and healthy adults. Neurogastroenterol. Motil. 2017, 29. [Google Scholar] [CrossRef]
- Namin, F.; Patel, J.; Lin, Z.; Foran, P.; Dusing, R.; Mccallum, R. Recognizing abnormal patterns on PET brain images in adults patients with the cyclic vomiting syndrome: 52. Neurogastroenterol. Motil. 2006, 18, 681. [Google Scholar]
- Oki, J.; Miyamoto, A.; Takahashi, S.; Itoh, J.; Sakata, Y.; Okuno, A. Cyclic vomiting and elevation of creatine kinase associated with bitemporal hypoperfusion and EEG abnormalities: A migraine equivalent? Brain Dev. 1998, 20, 186–189. [Google Scholar] [CrossRef]
- Kim, J.; Napadow, V.; Kuo, B.; Barbieri, R. A combined HRV-fMRI approach to assess cortical control of cardiovagal modulation by motion sickness. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; Volume 2011, pp. 2825–2828. [Google Scholar] [CrossRef]
- Toschi, N.; Kim, J.; Sclocco, R.; Duggento, A.; Barbieri, R.; Kuo, B.; Napadow, V. Motion sickness increases functional connectivity between visual motion and nausea-associated brain regions. Auton. Neurosci. 2017, 202, 108–113. [Google Scholar] [CrossRef]
- Mayer, E.A.; Berman, S.; Suyenobu, B.; Labus, J.; Mandelkern, M.A.; Naliboff, B.D.; Chang, L. Differences in brain responses to visceral pain between patients with irritable bowel syndrome and ulcerative colitis. Pain 2005, 115, 398–409. [Google Scholar] [CrossRef]
- Becker, J.B.; Berkley, K.J.; Geary, N.; Hampson, E.; Herman, J.P.; Young, E. Sex Differences in the Brain: From Genes to Behavior; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Naliboff, B.D.; Berman, S.; Chang, L.; Derbyshire, S.W.G.; Suyenobu, B.; Vogt, B.A.; Mandelkern, M.; Mayer, E.A. Sex-related Differences in IBS Patients: Central Processing of Visceral Stimuli. Gastroenterology 2003, 124. [Google Scholar] [CrossRef]
- Berman, S.; Munakata, J.; Naliboff, B.D.; Chang, L.; Mandelkern, M.; Silverman, D.; Kovalik, E.; Mayer, E.A. Gender Differences in Regional Brain Response to Visceral Pressure in IBS Patients. Eur. J. Pain (Lond. Engl.) 2000, 4. [Google Scholar] [CrossRef]
- Abell, T.L.; Adams, K.A.; Boles, R.G.; Bousvaros, A.; Chong, S.K.; Fleisher, D.R.; Hasler, W.L.; Hyman, P.E.; Issenman, R.M.; Li, B.U.; et al. Cyclic vomiting syndrome in adults. Neurogastroenterol. Motil. 2008, 20, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Parkman, H.P.; Hasler, W.L.; Fisher, R.S. American Gastroenterological Association technical review on the diagnosis and treatment of gastroparesis. Gastroenterology 2004, 127, 1592–1622. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, P.; Gianrico, F. Diabetic Gastroparesis: What We Have Learned and Had to Unlearn in the Past 5 Years. Gut 2010, 59. [Google Scholar] [CrossRef] [PubMed]
- Moshiree, B.; Potter, M.; Talley, N.J. Epidemiology and Pathophysiology of Gastroparesis. Gastrointest. Endosc. Clin. N. Am. 2019, 29, 1–14. [Google Scholar] [CrossRef]
- Parkman, H.P. Gastroparesis. In Functional and GI Motility Disorders; Karger Publishers: Basel, Switzerland, 2014; Volume 33, pp. 25–37. [Google Scholar]
- Wang, Y.R.; Fisher, R.S.; Parkman, H.P. Gastroparesis-related hospitalizations in the United States: Trends, characteristics, and outcomes, 1995–2004. Am. J. Gastroenterol. 2008, 103, 313–322. [Google Scholar] [CrossRef]
- Snodgrass, P.; Sandoval, H.; Calhoun, V.D.; Ramos-Duran, L.; Song, G.; Sun, Y.; Alvarado, B.; Bashashati, M.; Sarosiek, I.; McCallum, R.W. Central Nervous System Mechanisms of Nausea in Gastroparesis: An fMRI-Based Case-Control Study. Dig. Dis. Sci. 2019. [Google Scholar] [CrossRef]
- Stapleton, J.; Wo, J.M. Current Treatment of Nausea and Vomiting Associated with Gastroparesis: Antiemetics, Prokinetics, Tricyclics. Gastrointest. Endosc. Clin. N. Am. 2009, 19, 57–72. [Google Scholar] [CrossRef]
- Parkman, H.P.; Camilleri, M.; Farrugia, G.; McCallum, R.W.; Bharucha, A.E.; Mayer, E.A.; Tack, J.F.; Spiller, R.; Horowitz, M.; Vinik, A.I.; et al. Gastroparesis and Functional Dyspepsia: Excerpts from the AGA/ANMS Meeting. Neurogastroenterol. Motil. 2010, 22, 113–133. [Google Scholar] [CrossRef]
- Mayer, E.A.; Aziz, Q.; Coen, S.; Kern, M.; Labus, J.S.; Lane, R.; Kuo, B.; Naliboff, B.; Tracey, I. Brain Imaging Approaches to the Study of Functional GI Disorders: A Rome Working Team Report. Neurogastroenterol. Motil. 2009, 21. [Google Scholar] [CrossRef]
- Malagelada, J.-R.; Malagelada, C. Nausea and Vomiting. In Sleisenger and Fordtran’s Gastrointestinal and Liver Disease, 10th ed.; Feldman, M., Friedman, L.S., Brandt, L.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; Chapter 15; pp. 207–220. [Google Scholar]
- Krakauer, E.L.; Zhu, A.X.; Bounds, B.C.; Sahani, D.; McDonald, K.R.; Brachtel, E.F. Case 6-2005: A 58-Year-Old Man with Esophageal Cancer and Nausea, Vomiting, and Intractable Hiccups. New Engl. J. Med. 2005, 352, 817–825. [Google Scholar] [CrossRef]
- Arthurs, O.J.; Boniface, S. How Well Do We Understand the Neural Origins of the fMRI BOLD Signal? Trends Neurosci. 2002, 25. [Google Scholar] [CrossRef]
- Logothetis, N.K. What We Can Do and What We Cannot Do With fMRI. Nature 2008, 453. [Google Scholar] [CrossRef] [PubMed]
- Macapinlac, H.A. Positron Emission Tomography of the Brain. Neuroimaging Clin. N. Am. 2006, 16. [Google Scholar] [CrossRef]
- Sharma, R.; Aboagye, E. Development of Radiotracers for Oncology—The Interface with Pharmacology. Br. J. Pharmacol. 2011, 163. [Google Scholar] [CrossRef]
- Napadow, V.; Sheehan, J.D.; Kim, J.; Lacount, L.T.; Park, K.; Kaptchuk, T.J.; Rosen, B.R.; Kuo, B. The brain circuitry underlying the temporal evolution of nausea in humans. Cereb. Cortex 2013, 23, 806–813. [Google Scholar] [CrossRef]
- Kuo, B.; Singh, P. Nausea and Vomiting Related to the Central Nervous System Diseases; Springer: Cham, Switzerland, 2017; pp. 109–118. [Google Scholar]
- Balaban, D.H.; Chen, J.; Lin, Z.; Tribble, C.G.; McCallum, R.W. Median arcuate ligament syndrome: A possible cause of idiopathic gastroparesis. Am. J. Gastroenterol. 1997, 92, 519–523. [Google Scholar]
- Frøkjaer, J.B.; Andersen, L.W.; Brock, C.; Simrén, M.; Ljungberg, M.; Søfteland, E.; Dimcevski, G.; Yavarian, Y.; Gregersen, H.; Drewes, A.M. Altered Brain Microstructure Assessed by Diffusion Tensor Imaging in Patients With Diabetes and Gastrointestinal Symptoms. Diabetes Care 2013, 36. [Google Scholar] [CrossRef]
- Revicki, D.A.; Rentz, A.M.; Dubois, D.; Kahrilas, P.; Stanghellini, V.; Talley, N.J.; Tack, J. Development and validation of a patient-assessed gastroparesis symptom severity measure: The Gastroparesis Cardinal Symptom Index. Aliment. Pharmacol. Ther. 2003, 18, 141–150. [Google Scholar] [CrossRef]
- Sarosiek, I.; Song, G.; Sun, Y.; Sandoval, H.; Sands, S.; Chen, J.; McCallum, R.W. Central and Peripheral Effects of Transcutaneous Acupuncture Treatment for Nausea in Patients with Diabetic Gastroparesis. J. Neurogastroenterol. Motil. 2017, 23, 245–253. [Google Scholar] [CrossRef]
- Ueda, K.; Okamoto, Y.; Okada, G.; Yamashita, H.; Hori, T.; Yamawaki, S. Brain Activity During Expectancy of Emotional Stimuli: An fMRI Study. Neuroreport 2003, 14. [Google Scholar] [CrossRef]
- McCallum, R.W.; Lin, Z.; Forster, J.; Roeser, K.; Hou, Q.; Sarosiek, I. Gastric electrical stimulation improves outcomes of patients with gastroparesis for up to 10 years. Clin. Gastroenterol. Hepatol. 2011, 9, 314–319. [Google Scholar] [CrossRef]
- Abell, T.; Lou, J.; Tabbaa, M.; Batista, O.; Malinowski, S.; Al-Juburi, A. Gastric electrical stimulation for gastroparesis improves nutritional parameters at short, intermediate, and long-term follow-up. JPEN J. Parenter. Enteral Nutr. 2003, 27, 277–281. [Google Scholar] [CrossRef]
- Lin, Z.; McElhinney, C.; Sarosiek, I.; Forster, J.; McCallum, R. Chronic gastric electrical stimulation for gastroparesis reduces the use of prokinetic and/or antiemetic medications and the need for hospitalizations. Dig. Dis. Sci. 2005, 50, 1328–1334. [Google Scholar] [CrossRef]
- Lin, Z.; Sarosiek, I.; Forster, J.; McCallum, R.W. Symptom responses, long-term outcomes and adverse events beyond 3 years of high-frequency gastric electrical stimulation for gastroparesis. Neurogastroenterol. Motil. 2006, 18, 18–27. [Google Scholar] [CrossRef]
- Lin, Z.; Forster, J.; Sarosiek, I.; McCallum, R.W. Treatment of gastroparesis with electrical stimulation. Dig. Dis. Sci. 2003, 48, 837–848. [Google Scholar] [CrossRef]
- Abell, T.; McCallum, R.; Hocking, M.; Koch, K.; Abrahamsson, H.; Leblanc, I.; Lindberg, G.; Konturek, J.; Nowak, T.; Quigley, E.M.; et al. Gastric electrical stimulation for medically refractory gastroparesis. Gastroenterology 2003, 125, 421–428. [Google Scholar] [CrossRef]
- Forster, J.; Sarosiek, I.; Delcore, R.; Lin, Z.; Raju, G.S.; McCallum, R.W. Gastric pacing is a new surgical treatment for gastroparesis. Am. J. Surg. 2001, 182, 676–681. [Google Scholar] [CrossRef]
- Qin, C.; Sun, Y.; Chen, J.D.Z.; Foreman, R.D. Gastric Electrical Stimulation Modulates Neuronal Activity in Nucleus Tractus Solitarii in Rats. Auton. Neurosci. Basic Clin. 2005, 119. [Google Scholar] [CrossRef]
- Tang, M.; Zhang, J.; Chen, J.D.Z. Central Mechanisms of Gastric Electrical Stimulation Involving Neurons in the Paraventricular Nucleus of the Hypothalamus in Rats. Obes. Surg. 2006, 16. [Google Scholar] [CrossRef]
- Qin, C.; Li, J.; Tang, K. The Paraventricular Nucleus of the Hypothalamus: Development, Function, and Human Diseases. Endocrinology 2018, 159, 3458–3472. [Google Scholar] [CrossRef]
- McCallum, R.W.; Dusing, R.W.; Sarosiek, I.; Cocjin, J.; Forster, J.; Lin, Z. Mechanisms of high-frequency electrical stimulation of the stomach in gastroparetic patients. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2006, 1, 5400–5403. [Google Scholar] [CrossRef]
- McCallum, R.W.; Dusing, R.W.; Sarosiek, I.; Cocjin, J.; Forster, J.; Lin, Z. Mechanisms of symptomatic improvement after gastric electrical stimulation in gastroparetic patients. Neurogastroenterol. Motil. 2010, 22, e150–e161. [Google Scholar] [CrossRef]
- Collins, Z.; Dusing, R.; McCallum, R.; McMillin, C.; Lin, Z.; Forster, J. Comparison of positron emission tomography (PET) imaged cerebral activity in gastroparetic patients before and during gastric electrical stimulation (GES). J. Nucl. Med. 2006, 47, 306. [Google Scholar]
- Abell, T.L.; Van Cutsem, E.; Abrahamsson, H.; Huizinga, J.D.; Konturek, J.W.; Galmiche, J.P.; VoelIer, G.; Filez, L.; Everts, B.; Waterfall, W.E.; et al. Gastric electrical stimulation in intractable symptomatic gastroparesis. Digestion 2002, 66, 204–212. [Google Scholar] [CrossRef]
- Ducrotte, P.; Coffin, B.; Bonaz, B.; Fontaine, S.; Bruley Des Varannes, S.; Zerbib, F.; Caiazzo, R.; Grimaud, J.C.; Mion, F.; Hadjadj, S.; et al. Gastric Electrical Stimulation Reduces Refractory Vomiting in a Randomized Crossover Trial. Gastroenterology 2020, 158, 506–514. [Google Scholar] [CrossRef]
- McCallum, R.W.; Snape, W.; Brody, F.; Wo, J.; Parkman, H.P.; Nowak, T. Gastric electrical stimulation with Enterra therapy improves symptoms from diabetic gastroparesis in a prospective study. Clin. Gastroenterol. Hepatol. 2010, 8, 947–954. [Google Scholar] [CrossRef]
- McCallum, R.W.; Sarosiek, I.; Parkman, H.; Snape, W.; Brody, F.; Wo, J.; Nowak, T. Gastric electrical stimulation with Enterra therapy improves symptoms of idiopathic gastroparesis. Neurogastroenterol. Motil. 2013, 25. [Google Scholar] [CrossRef]
- Lin, Z.; Hou, Q.; Sarosiek, I.; Forster, J.; McCallum, R. Association between changes in symptoms and gastric emptying in gastroparetic patients treated with gastric electrical stimulation. Neurogastroenterol. Motil. 2008, 20, 464–470. [Google Scholar] [CrossRef]
- Brody, F.; Vaziri, K.; Saddler, A.; Ali, A.; Drenon, E.; Hanna, B.; Akin, E.; Gonzalez, F.; Soffer, E. Gastric electrical stimulation for gastroparesis. J. Am. Coll. Surg. 2008, 207, 533–538. [Google Scholar] [CrossRef]
- Maranki, J.L.; Lytes, V.; Meilahn, J.E.; Harbison, S.; Friedenberg, F.K.; Fisher, R.S.; Parkman, H.P. Predictive factors for clinical improvement with Enterra gastric electric stimulation treatment for refractory gastroparesis. Dig. Dis. Sci. 2008, 53, 2072–2078. [Google Scholar] [CrossRef]
- Gourcerol, G.; Chaput, U.; LeBlanc, I.; Gallas, S.; Michot, F.; Leroi, A.M.; Ducrotte, P. Gastric electrical stimulation in intractable nausea and vomiting: Assessment of predictive factors of favorable outcomes. J. Am. Coll. Surg. 2009, 209, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Stanghellini, V.; Chan, F.K.L.; Hasler, W.L.; Malagelada, J.R.; Suzuki, H.; Tack, J.; Talley, N.J. Gastroduodenal Disorders. Gastroenterology 2016, 150, 1380–1392. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Caenepeel, P.; Fischler, B.; Piessevaux, H.; Janssens, J. Symptoms associated with hypersensitivity to gastric distention in functional dyspepsia. Gastroenterology 2001, 121, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Caenepeel, P.; Corsetti, M.; Janssens, J. Role of tension receptors in dyspeptic patients with hypersensitivity to gastric distention. Gastroenterology 2004, 127, 1058–1066. [Google Scholar] [CrossRef]
- Notivol, R.; Coffin, B.; Azpiroz, F.; Mearin, F.; Serra, J.; Malagelada, J.-R. Gastric tone determines the sensitivity of the stomach to distention. Gastroenterology 1995, 108, 330–336. [Google Scholar] [CrossRef]
- Van Oudenhove, L.; Vandenberghe, J.; Dupont, P.; Geeraerts, B.; Vos, R.; Dirix, S.; Bormans, G.; Vanderghinste, D.; Van Laere, K.; Demyttenaere, K. Abnormal regional brain activity during rest and (anticipated) gastric distension in functional dyspepsia and the role of anxiety: A H215O-PET study. Am. J. Gastroenterol. 2010, 105, 913–924. [Google Scholar] [CrossRef]
- Mier, W.; Mier, D. Advantages in functional imaging of the brain. Front. Hum. Neurosci. 2015, 9, 249. [Google Scholar] [CrossRef]
- Vandenberghe, J.; Dupont, P.; Van Oudenhove, L.; Bormans, G.; Demyttenaere, K.; Fischler, B.; Geeraerts, B.; Janssens, J.; Tack, J. Regional cerebral blood flow during gastric balloon distention in functional dyspepsia. Gastroenterology 2007, 132, 1684–1693. [Google Scholar] [CrossRef]
- Tack, J.; Bisschops, R.; Sarnelli, G. Pathophysiology and treatment of functional dyspepsia. Gastroenterology 2004, 127, 1239–1255. [Google Scholar] [CrossRef]
- Van Oudenhove, L.; Kindt, S.; Vos, R.; Coulie, B.; Tack, J. Influence of buspirone on gastric sensorimotor function in man. Aliment. Pharmacol. Ther. 2008, 28, 1326–1333. [Google Scholar] [CrossRef]
- Dinan, T.; Mahmud, N.; Rathore, O.; Thakore, J.; Scott, L.; Carr, E.; Naesdal, J.; O’Morain, C.; Keeling, P. A double-blind placebo-controlled study of buspirone-stimulated prolactin release in non-ulcer dyspepsia—are central serotoninergic responses enhanced? Aliment. Pharmacol. Ther. 2001, 15, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Sawhney, M.S.; Prakash, C.; Lustman, P.J.; Clouse, R.E. Tricyclic antidepressants for chronic vomiting in diabetic patients. Dig. Dis. Sci. 2007, 52, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Clouse, R.E. Antidepressants for functional gastrointestinal syndromes. Dig. Dis. Sci. 1994, 39, 2352–2363. [Google Scholar] [CrossRef] [PubMed]
- Loldrup, D.; Langemark, M.; Hansen, H.J.; Olesen, J.; Bech, P. Clomipramine and mianserin in chronic idiopathic pain syndrome. Psychopharmacology 1989, 99, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mertz, H.; Fass, R.; Kodner, A.; Yan-Go, F.; Fullerton, S.; Mayer, E. Effect of amitryptiline on symptoms, sleep, and visceral perception in patients with functional dyspepsia. Am. J. Gastroenterol. 1998, 93, 160–165. [Google Scholar] [CrossRef]
- Prakash, C.; Lustman, P.J.; Freedland, K.E.; Clouse, R.E. Tricyclic antidepressants for functional nausea and vomiting (clinical outcome in 37 patients). Dig. Dis. Sci. 1998, 43, 1951–1956. [Google Scholar] [CrossRef]
- Parkman, H.P.; Van Natta, M.L.; Abell, T.L.; McCallum, R.W.; Sarosiek, I.; Nguyen, L.; Snape, W.J.; Koch, K.L.; Hasler, W.L.; Farrugia, G.; et al. Effect of nortriptyline on symptoms of idiopathic gastroparesis: The NORIG randomized clinical trial. JAMA 2013, 310, 2640–2649. [Google Scholar] [CrossRef]
- Huang, W.; Jiang, S.-M.; Jia, L.; You, L.-Q.; Huang, Y.-X.; Gong, Y.-M.; Wang, G.-Q. Effect of amitriptyline on gastrointestinal function and brain-gut peptides: A double-blind trial. World J. Gastroenterol. 2013, 19, 4214. [Google Scholar] [CrossRef]
- Lawson, K. A brief review of the pharmacology of amitriptyline and clinical outcomes in treating fibromyalgia. Biomedicines 2017, 5, 24. [Google Scholar] [CrossRef]
- Ford, A.C.; Luthra, P.; Tack, J.; Boeckxstaens, G.E.; Moayyedi, P.; Talley, N.J. Efficacy of psychotropic drugs in functional dyspepsia: Systematic review and meta-analysis. Gut 2017, 66, 411–420. [Google Scholar] [CrossRef]
- Talley, N.J.; Locke, G.R.; Saito, Y.A.; Almazar, A.E.; Bouras, E.P.; Howden, C.W.; Lacy, B.E.; DiBaise, J.K.; Prather, C.M.; Abraham, B.P. Effect of amitriptyline and escitalopram on functional dyspepsia: A multicenter, randomized controlled study. Gastroenterology 2015, 149, 340–349. [Google Scholar] [CrossRef]
- Malamood, M.; Roberts, A.; Kataria, R.; Parkman, H.P.; Schey, R. Mirtazapine for symptom control in refractory gastroparesis. Drug Des. Dev. Ther. 2017, 11, 1035. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Janssen, P.; Masaoka, T.; Farré, R.; Van Oudenhove, L. Efficacy of Buspirone, a Fundus-Relaxing Drug, in Patients with Functional Dyspepsia. Clin. Gastroenterol. Hepatol. 2012, 10, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Yoon, S.S.; Kuo, B. Nausea: A review of pathophysiology and therapeutics. Ther. Adv. Gastroenterol. 2016, 9, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Naliboff, B.D.; Craig, A.D.B. Neuroimaging of the Brain-Gut Axis: From Basic Understanding to Treatment of Functional GI Disorders. Gastroenterology 2006, 131. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez, Z.; McCallum, R.W. Status of Brain Imaging in Gastroparesis. Gastrointest. Disord. 2020, 2, 58-70. https://doi.org/10.3390/gidisord2020006
Gonzalez Z, McCallum RW. Status of Brain Imaging in Gastroparesis. Gastrointestinal Disorders. 2020; 2(2):58-70. https://doi.org/10.3390/gidisord2020006
Chicago/Turabian StyleGonzalez, Zorisadday, and Richard W. McCallum. 2020. "Status of Brain Imaging in Gastroparesis" Gastrointestinal Disorders 2, no. 2: 58-70. https://doi.org/10.3390/gidisord2020006
APA StyleGonzalez, Z., & McCallum, R. W. (2020). Status of Brain Imaging in Gastroparesis. Gastrointestinal Disorders, 2(2), 58-70. https://doi.org/10.3390/gidisord2020006




