Preliminary Results: The Impact of Smartphone Use and Short-Wavelength Light during the Evening on Circadian Rhythm, Sleep and Alertness
Abstract
1. Introduction
2. Results
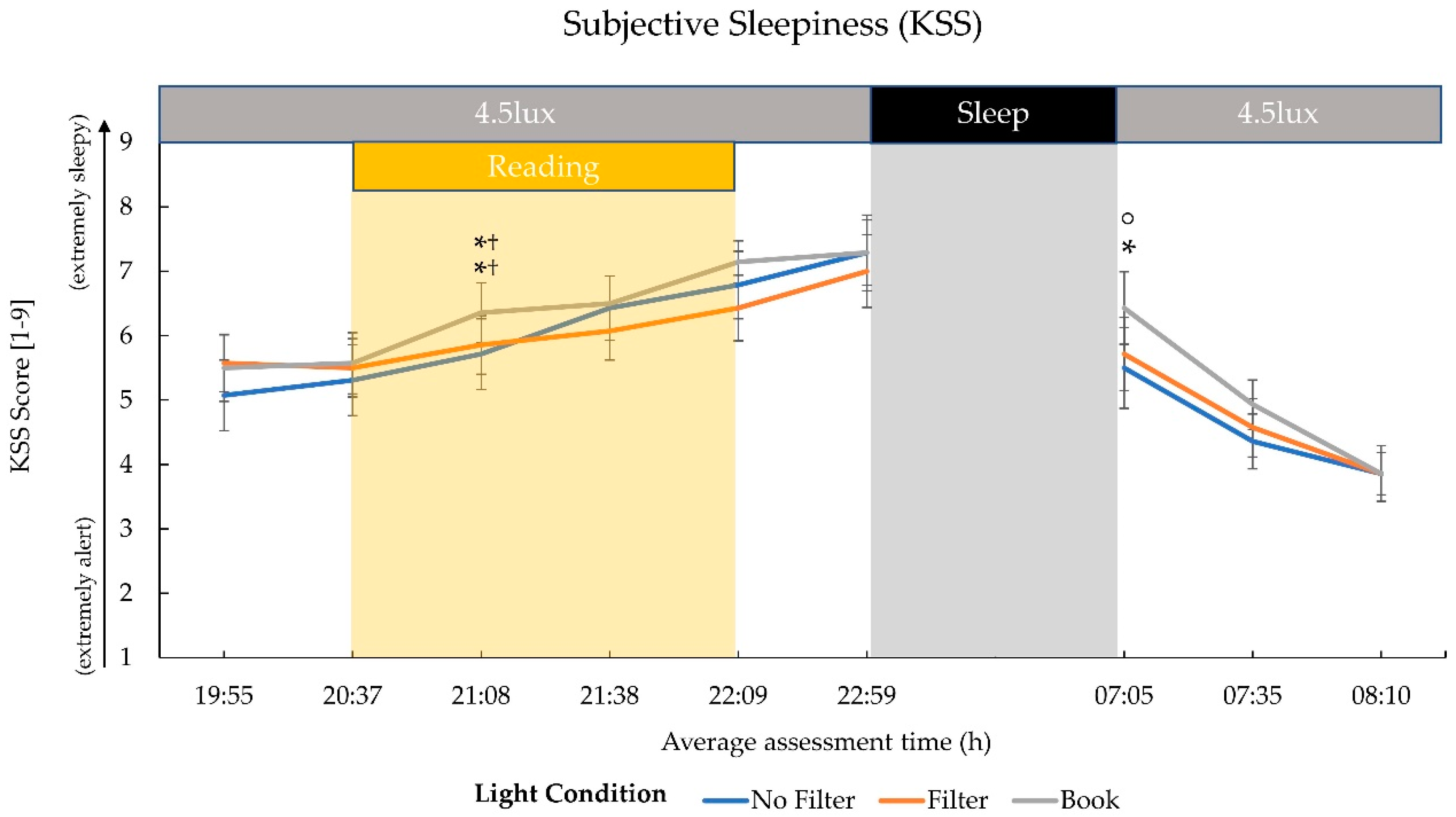
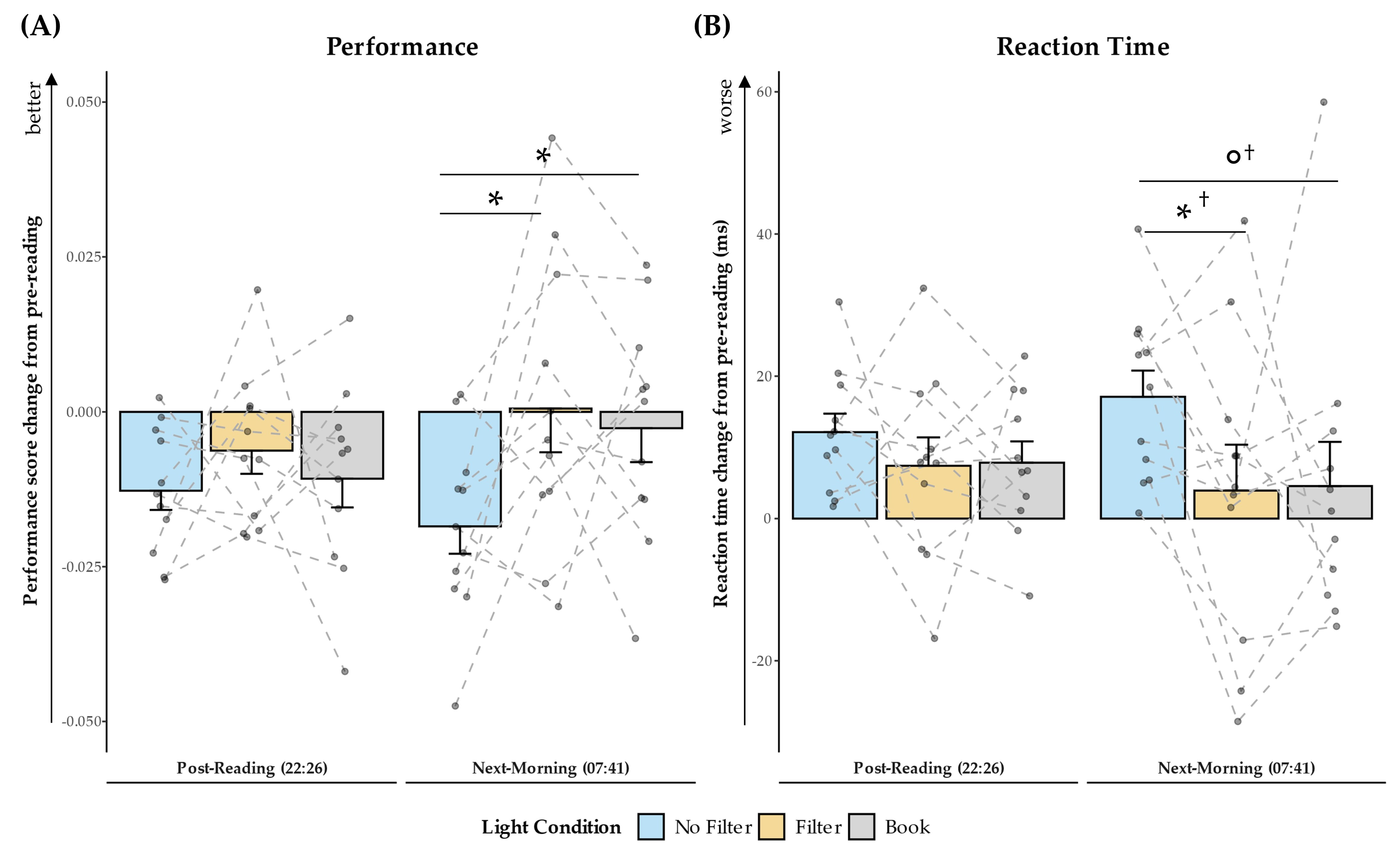
2.1. Subjective Sleepiness
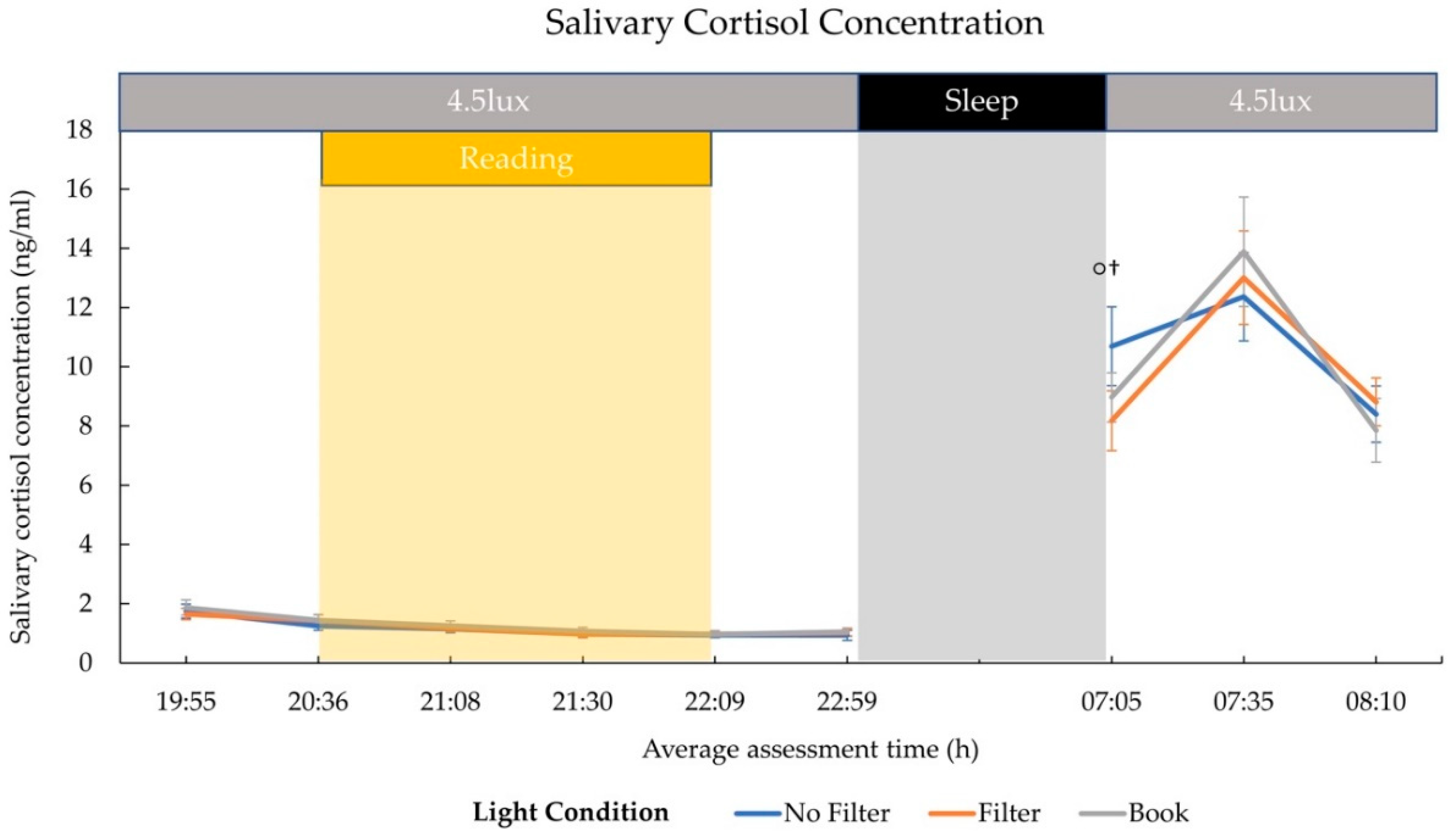
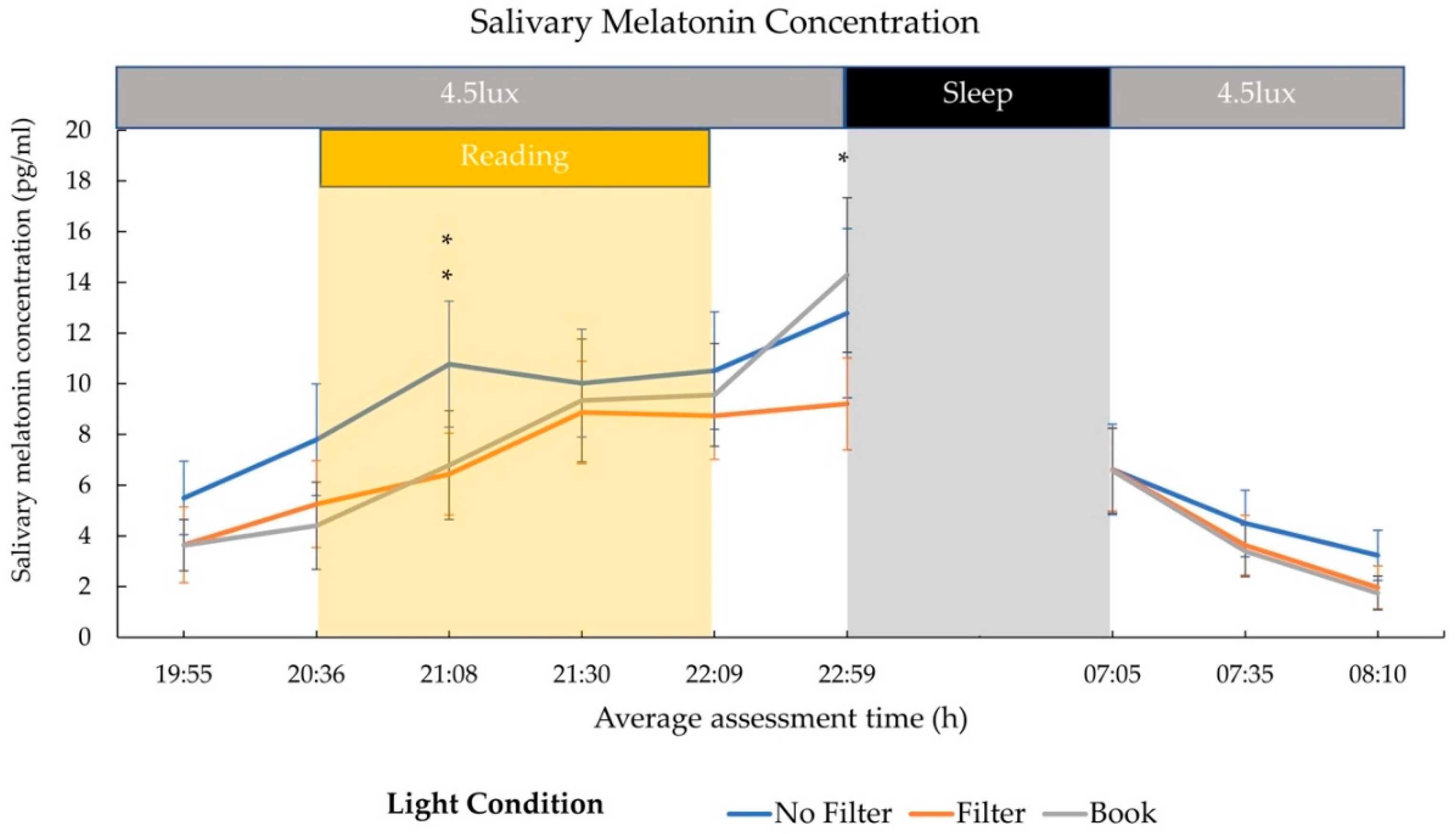
2.2. Cortisol and Melatonin
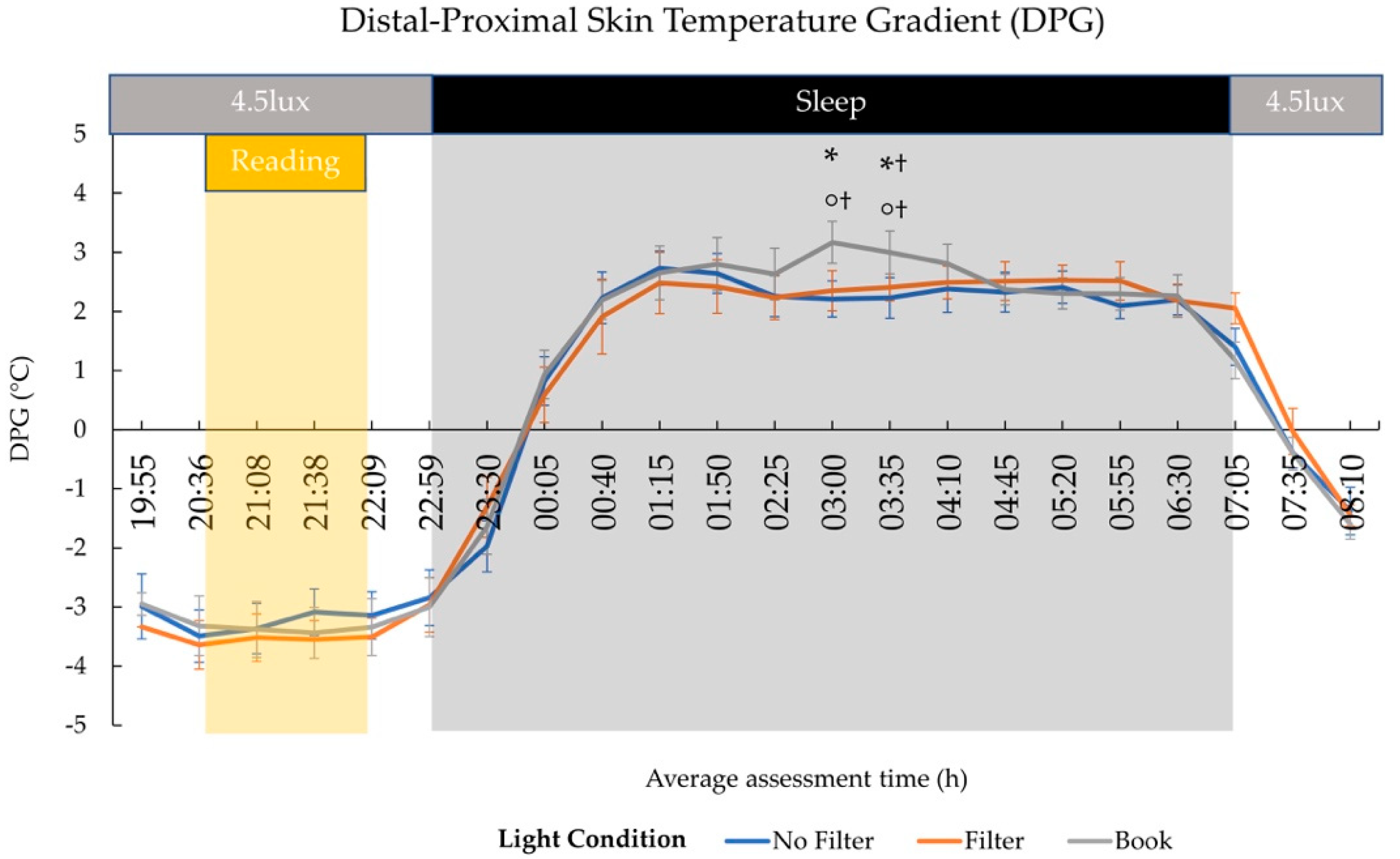
2.3. Body Temperature
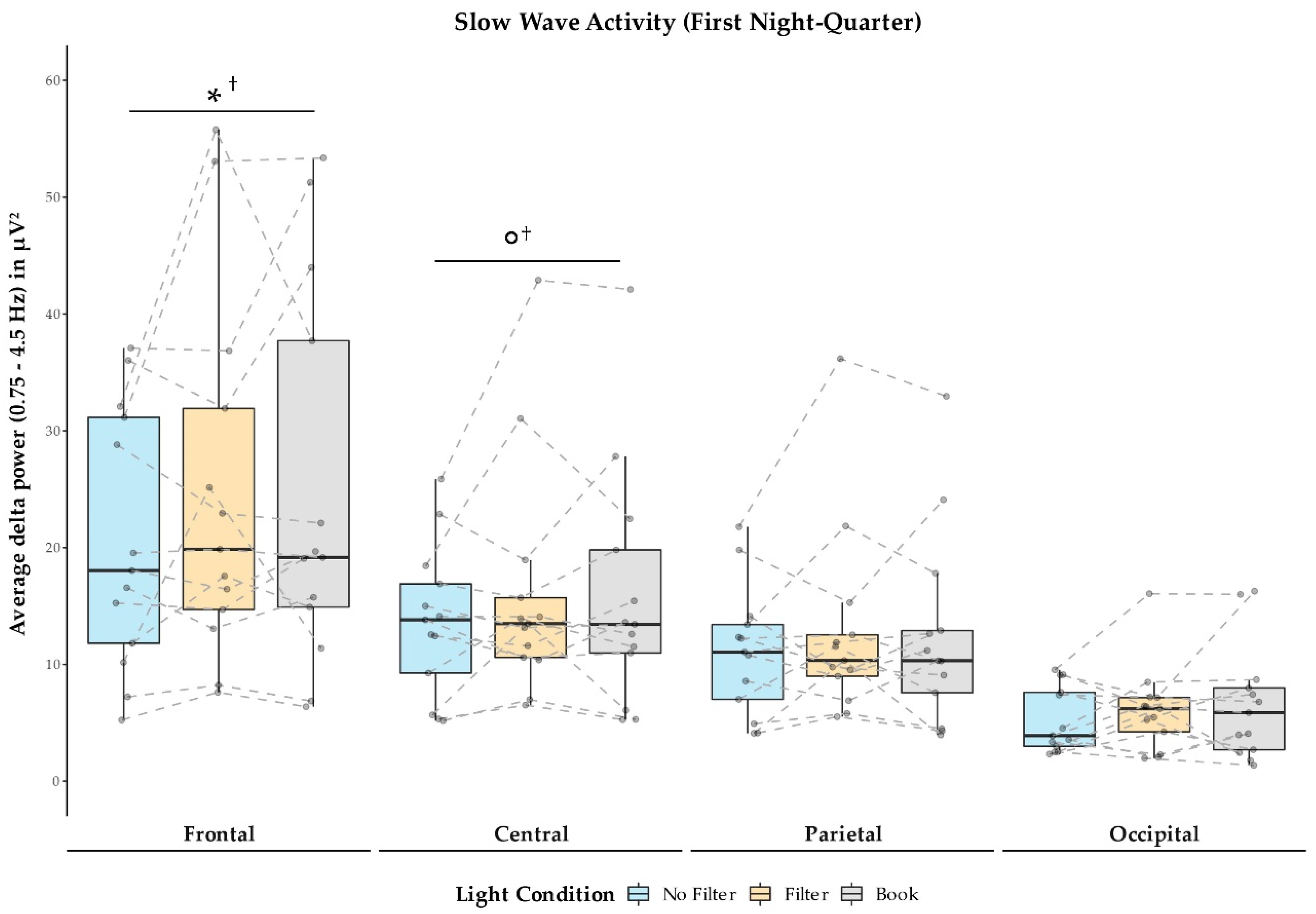
2.4. Sleep Measures
2.5. Objective Alertness
3. Discussion
3.1. Effects on Evening Sleepiness
3.2. Hormonal Effects
3.3. Effects on Sleep and Next-Morning Alertness
3.4. Limitations and Future Directions
4. Materials and Methods
4.1. Participants
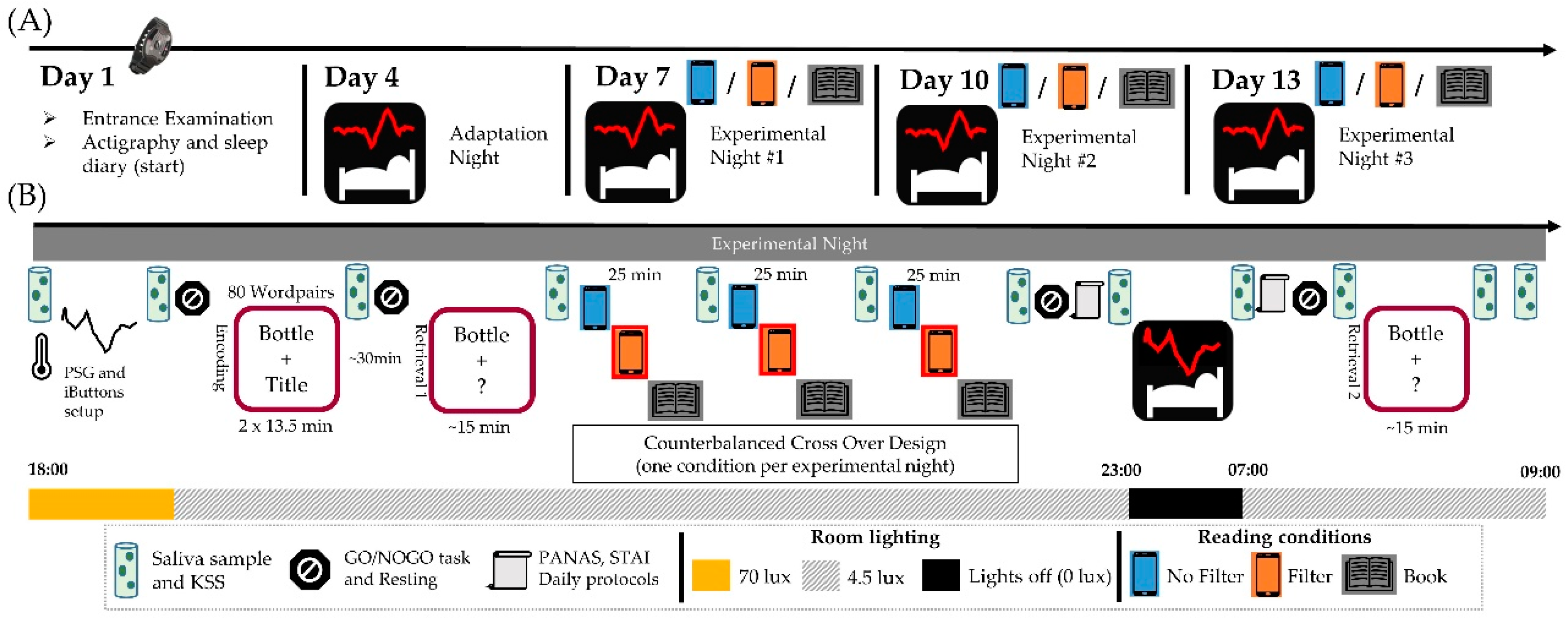
4.2. Protocol
4.3. Light Exposure (Reading Session)
4.4. Polysomnography
4.5. Subjective Sleepiness
4.6. Temperature
4.7. Melatonin and Cortisol
4.8. Alertness
4.9. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| CAR | Cortisol awakening response |
| CBT | Core body temperature |
| DPG | Distal proximal gradient |
| ipRGC | Intrinsically photosensitive retinal ganglion cell |
| IQR | Interquartile range |
| KSS | Karolinska Sleepiness Scale |
| PSG | Polysomnography |
| SCN | Suprachiasmatic nucleus |
| SWA | Slow wave activity |
| SWS | Slow wave sleep |
References
- Lemola, S.; Perkinson-Gloor, N.; Brand, S.; Dewald-Kaufmann, J.F.; Grob, A. Adolescents’ electronic media use at night, sleep disturbance, and depressive symptoms in the smartphone age. J. Youth Adolesc. 2014, 44, 405–418. [Google Scholar] [CrossRef]
- Rideout, V.J.; Foehr, U.G.; Roberts, D.F. Generation M2: Media in the Lives of 8 to 18 Year-Olds; Henry J. Kaiser Family Foundation: San Francisco, CA, USA, 2010. [Google Scholar]
- Pimputkar, S.; Speck, J.S.; Denbaars, S.P.; Nakamura, S. Prospects for LED lighting. Nat. Photonics 2009. [Google Scholar] [CrossRef]
- Gringras, P.; Middleton, B.; Skene, D.J.; Revell, V.L. Bigger, brighter, bluer-better? Current light-emitting devices—Adverse sleep properties and preventative strategies. Front. Public Health 2015, 3, 233. [Google Scholar] [CrossRef] [PubMed]
- Cajochen, C.; Frey, S.; Anders, D.; Späti, J.; Bues, M.; Pross, A.; Mager, R.; Wirz-Justice, A.; Stefani, O. Evening exposure to a light-emitting diodes (LED)-backlit computer screen affects circadian physiology and cognitive performance. J. Appl. Physiol. 2011, 110, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Münch, M.; Kobialka, S.; Steiner, R.; Oelhafen, P.; Wirz-Justice, A.; Cajochen, C. Wavelength-dependent effects of evening light exposure on sleep architecture and sleep EEG power density in men. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Vandewalle, G.; Maquet, P.; Dijk, D.J. Light as a modulator of cognitive brain function. Trends Cogn. Sci. 2009, 13, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Chellappa, S.L.; Steiner, R.; Blattner, P.; Oelhafen, P.; Götz, T.; Cajochen, C. Non-visual effects of light on melatonin, alertness and cognitive performance: Can blue-enriched light keep us alert? PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Chinoy, E.D.; Duffy, J.F.; Czeisler, C.A. Unrestricted evening use of light-emitting tablet computers delays self-selected bedtime and disrupts circadian timing and alertness. Physiol. Rep. 2018, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.A.; Hilaire, M.A.S.; Lockley, S.W. The effects of spectral tuning of evening ambient light on melatonin suppression, alertness and sleep. Physiol. Behav. 2017, 177, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.-M.; Aeschbach, D.; Duffy, J.F.; Czeisler, C.A. Evening use of light-emitting eReaders negatively affects sleep, circadian timing, and next-morning alertness. Proc. Natl. Acad. Sci. USA 2015, 112, 1232–1237. [Google Scholar] [CrossRef]
- Yang, M.; Ma, N.; Zhu, Y.; Su, Y.C.; Chen, Q.; Hsiao, F.C.; Ji, Y.; Yang, C.M.; Zhou, G. The acute effects of intermittent light exposure in the evening on alertness and subsequent sleep architecture. Int. J. Environ. Res. Public Health. 2018, 15, 524. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Allebrandt, K.V.; Merrow, M.; Vetter, C. Social jetlag and obesity. Curr. Biol. 2012, 22, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Van der Lely, S.; Frey, S.; Garbazza, C.; Wirz-Justice, A.; Jenni, O.G.; Steiner, R.; Wolf, S.; Cajochen, C.; Bromundt, V.; Schmidt, C. Blue blocker glasses as a countermeasure for alerting effects of evening light-emitting diode screen exposure in male teenagers. J. Adolesc. Heal. 2015, 56, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Provencio, I.; Rodriguez, I.R.; Jiang, G.; Hayes, W.P.; Moreira, E.F.; Rollag, M.D. A novel human opsin in the inner retina. J. Neurosci. 2000, 20, 600–605. [Google Scholar] [CrossRef]
- Bailes, H.J.; Lucas, R.J. Melanopsin and inner retinal photoreception. Cell. Mol. Life Sci. 2010, 67, 99–111. [Google Scholar] [CrossRef]
- Blume, C.; Garbazza, C.; Spitschan, M. Effects of light on human circadian rhythms, sleep and mood. Somnologie 2019, 23, 147–156. [Google Scholar] [CrossRef]
- Hannibal, J.; Christiansen, A.T.; Heegaard, S.; Fahrenkrug, J.; Kiilgaard, J.F. Melanopsin expressing human retinal ganglion cells: Subtypes, distribution, and intraretinal connectivity. J. Comp. Neurol. 2017, 525, 1934–1961. [Google Scholar] [CrossRef]
- Hellhammer, D.H.; Wüst, S.; Kudielka, B.M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology 2009, 34, 163–171. [Google Scholar] [CrossRef]
- Kern, S.; Oakes, T.R.; Stone, C.K.; McAuliff, E.; Kirschbaum, C.; Davidson, R.J. Glucose metabolic changes in the prefrontal cortex are associated with HPA axis response to a psychosocial stressor. Psychoneuroendocrinology 2008, 33, 517–529. [Google Scholar] [CrossRef]
- Pruessner, J.C.; Wolf, O.T.; Hellhammer, D.H.; Buske-Kirschbaum, A.; von Auer, K.; Jobst, S.; Kaspers, F.; Kirschbaum, C. Free cortisol levels after awakening: A reliable biological marker for the assessment of adrenocortical activity. Life Sci. 1997. [Google Scholar] [CrossRef]
- Wilhelm, I.; Born, J.; Kudielka, B.M.; Schlotz, W.; Wüst, S. Is the cortisol awakening rise a response to awakening? Psychoneuroendocrinology 2007, 32, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Federenko, I.; Wüst, S.; Hellhammer, D.H.; Dechoux, R.; Kumsta, R.; Kirschbaum, C. Free cortisol awakening responses are influenced by awakening time. Psychoneuroendocrinology 2004, 29, 174–184. [Google Scholar] [CrossRef]
- Devine, J.K.; Wolf, J.M. Determinants of cortisol awakening responses to naps and nighttime sleep. Psychoneuroendocrinology 2016, 63, 128–134. [Google Scholar] [CrossRef]
- Schmidt, C.; Xhrouet, M.; Hamacher, M.; Delloye, E.; LeGoff, C.; Cavalier, E.; Collette, F.; Vandewalle, G. Light exposure via a head-mounted device suppresses melatonin and improves vigilant attention without affecting cortisol and comfort. PsyCh J. 2018, 7, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Leproult, R.; Colecchia, E.F.; L’Hermite-Balériaux, M.; van Cauter, E. Transition from dim to bright light in the morning induces an immediate elevation of cortisol levels. J. Clin. Endocrinol. Metab. 2001, 86, 151–157. [Google Scholar] [CrossRef]
- Cajochen, C.; Kräuchi, K.; Wirz-Justice, A. Role of melatonin in the regulation of human circadian rhythms and sleep. J. Neuroendocrinol. 2003, 15, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Dubocovich, M.L.; Rivera-Bermudez, M.A.; Gerdin, M.J.; Masana, M.I. Molecular pharmacology, regulation and function of mammalian melatonin receptors. Front. Biosci. 2003, 8, 1093–1108. [Google Scholar] [CrossRef]
- Cajochen, C.; Münch, M.; Kobialka, S.; Kräuchi, K.; Steiner, R.; Oelhafen, P.; Orgül, S.; Wirz-Justice, A. High sensitivity of human melatonin, alertness, thermoregulation, and heart rate to short wavelength light. J. Clin. Endocrinol. Metab. 2005, 90, 1311–1316. [Google Scholar] [CrossRef]
- Kräuchi, K.; Cajochen, C.; Werth, E.; Wirz-Justice, A. Functional link between distal vasodilation and sleep-onset latency? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 278, 741–748. [Google Scholar] [CrossRef]
- Kräuchi, K.; Cajochen, C.; Wirz-Justice, A. Waking up properly: Is there a role of thermoregulation in sleep inertia? J. Sleep Res. 2004, 13, 121–127. [Google Scholar] [CrossRef]
- Kräuchi, K.; Cajochen, C.; Werth, E.; Wirz-Justice, A. Warm feet promote the rapid onset of sleep. Nature 1999, 401, 36–37. [Google Scholar] [CrossRef] [PubMed]
- Dijk, D.J.; Cajochen, C. Melatonin and the circadian regulation of sleep initiation, consolidation, structure, and the sleep EEG. J. Biol. Rhythms 1997, 12, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Wehr, T.; Aeschbach, D.; Duncan, W.C., Jr. Evidence for a biological dawn and dusk in the human circadian timing system. J. Appl. Phys. 2001, 353, 937–951. [Google Scholar] [CrossRef] [PubMed]
- Cajochen, C.; Dijk, D.; Borbeiy, A.A. Dynamics of EEG slow-wave activity and core body temperature in human sleep after exposure to bright light. Sleep 1992, 15, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Aritake-Okada, S.; Tanabe, K.; Mochizuki, Y.; Ochiai, R.; Hibi, M.; Kozuma, K.; Katsuragi, Y.; Ganeko, M.; Takeda, N.; Uchida, S. Diurnal repeated exercise promotes slow-wave activity and fast-sigma power during sleep with increase in body temperature: A human crossover trial. J. Appl. Physiol. 2019, 127. [Google Scholar] [CrossRef] [PubMed]
- Chellappa, S.L.; Steiner, R.; Oelhafen, P.; Lang, D.; Götz, T.; Krebs, J.; Cajochen, C. Acute exposure to evening blue-enriched light impacts on human sleep. J. Sleep Res. 2013, 22, 573–580. [Google Scholar] [CrossRef]
- Grønli, J.; Byrkjedal, I.K.; Bjorvatn, B.; Nødtvedt, O.; Hamre, B.; Pallesen, S. Reading from an iPad or from a book in bed: The impact on human sleep. A randomized controlled crossover trial. Sleep Med. 2016, 21, 86–92. [Google Scholar] [CrossRef]
- Prayag, A.; Münch, M.; Aeschbach, D.; Chellappa, S.L.; Gronfier, C. Light modulation of human clocks, wake, and sleep. Clocks Sleep 2019, 1, 17. [Google Scholar] [CrossRef]
- Carter, B.; Rees, P.; Hale, L.; Bhattacharjee, D.; Paradkar, M.S. Association between portable screen-based media device access or use and sleep outcomes a systematic review and meta-analysis. JAMA Pediatr. 2016, 170, 1202–1208. [Google Scholar] [CrossRef]
- Green, A.; Cohen-Zion, M.; Haim, A.; Dagan, Y. Evening light exposure to computer screens disrupts human sleep, biological rhythms, and attention abilities. Chronobiol. Int. 2017, 34. [Google Scholar] [CrossRef]
- Knufinke, M.; Fittkau-Koch, L.; Møst, E.I.S.; Kompier, M.A.J.; Nieuwenhuys, A. Restricting short-wavelength light in the evening to improve sleep in recreational athletes—A pilot study. Eur. J. Sport Sci. 2019, 19, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.Y.; Kim, K.; Fava, M.; Mischoulon, D.; Papakostas, G.I.; Kim, M.J.; Kim, D.J.; Chang, K.A.J.; Oh, Y.; Yu, B.H.; et al. Effects of smartphone use with and without blue light at night in healthy adults: A randomized, double-blind, cross-over, placebo-controlled comparison. J. Psychiatr. Res. 2017, 87, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, R.; Alighanbari, N.; Zamanian, Z. The effects of screen light filtering software on cognitive performance and sleep among night workers. Health Promot. Perspect. 2019, 9, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, S.A.R.; Parhoodeh, S.; Hosseini, M.A.; Arabi, H.; Malakooti, H.; Nematollahi, S.; Mortazavi, G.; Darvish, L.; Mortazavi, S.M.J. Blocking short-wavelength component of the visible light emitted by smartphones’ screens improves human sleep quality. J. Biomed. Phys. Eng. 2018, 8, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Heath, M.; Sutherland, C.; Bartel, K.; Gradisar, M.; Williamson, P.; Lovato, N.; Micic, G. Does one hour of bright or short-wavelength filtered tablet screenlight have a meaningful effect on adolescents’ pre-bedtime alertness, sleep, and daytime functioning? Chronobiol. Int. 2014, 31, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Nagare, R.; Plitnick, B.; Figueiro, M.G. Does the iPad night shift mode reduce melatonin suppression? Light. Res. Technol. 2019, 51, 373–383. [Google Scholar] [CrossRef]
- Åkerstedt, T.; Gillberg, M. Subjective and objective sleepiness in the active individual. Int. J. Neurosci. 1990, 52, 29–37. [Google Scholar] [CrossRef]
- Lockley, S.W.; Evans, E.E.; Scheer, F.A.J.L.; Brainard, G.C.; Czeisler, C.A.; Aeschbach, D. Short-wavelength sensitivity for the direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans. Sleep 2006, 29, 161–168. [Google Scholar] [CrossRef]
- Yoshimura, M.; Kitazawa, M.; Maeda, Y.; Mimura, M.; Tsubota, K.; Kishimoto, T. Smartphone viewing distance and sleep: An experimental study utilizing motion capture technology. Nat. Sci. Sleep 2017, 9, 59–65. [Google Scholar] [CrossRef]
- Zeitzer, J.M.; Najjar, R.P.; Wang, C.-A.; Kass, M. Impact of blue-depleted white light on pupil dynamics, melatonin suppression and subjective alertness following real-world light exposure. Sleep Sci. Pract. 2018, 2. [Google Scholar] [CrossRef]
- Kräuchi, K.; Cajochen, C.; Pache, M.; Flammer, J.; Wirz-Justice, A. Thermoregulatory effects of melatonin in relation to sleepiness. Chronobiol. Int. 2006, 23, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Wirz-Justice, A. Circadian clues to sleep onset mechanisms. Neuropsychopharmacology 2001, 25, 92–96. [Google Scholar] [CrossRef]
- Kräuchi, K.; Deboer, T. The interrelationship between sleep regulation and thermoregulation. Front. Biosci. 2010, 15, 604–625. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Cajochen, C.; Danilenko, K.V.; Wirz-Justice, A. The hypothermic effect of late evening melatonin does not block the phase delay induced by concurrent bright light in human subjects. Neurosci. Lett. 1997, 232, 57–61. [Google Scholar] [CrossRef]
- Borbély, A.A. A two process model of sleep regulation. Hum. Neurobiol. 1982, 1, 195–204. [Google Scholar]
- Tobler, I.; Borbély, A.A. Sleep EEG in the rat as a function of prior waking. Electroencephalogr. Clin. Neurophysiol. 1986, 64, 74–76. [Google Scholar] [CrossRef]
- Chung, S.J.; An, H.; Suh, S. What do people do before going to bed? A study of bedtime procrastination using time use surveys. Sleep 2020, 43. [Google Scholar] [CrossRef]
- Robb, M.B. The New Normal: Parents, Teens, Screens, and Sleep in the United States; Common Sense Media: San Francisco, CA, USA, 2019. [Google Scholar]
- Chida, Y.; Steptoe, A. Cortisol awakening response and psychosocial factors: A systematic review and meta-analysis. Biol. Psychol. 2009, 80, 265–278. [Google Scholar] [CrossRef]
- Higuchi, S.; Motohashi, Y.; Liu, Y.; Maeda, A. Effects of playing a computer game using a bright display on presleep physiological variables, sleep latency, slow wave sleep and REM sleep. J. Sleep Res. 2005, 14, 267–273. [Google Scholar] [CrossRef]
- King, D.L.; Gradisar, M.; Drummond, A.; Lovato, N.; Wessel, J.; Micic, G.; Douglas, P.; Delfabbro, P. The impact of prolonged violent video-gaming on adolescent sleep: An experimental study. J. Sleep Res. 2013, 22, 137–143. [Google Scholar] [CrossRef]
- Figueiro, M.G.; Sahin, L.; Wood, B.; Plitnick, B. Light at night and measures of alertness and performance: Implications for shift workers. Biol. Res. Nurs. 2016, 18, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Kayaba, M.; Iwayama, K.; Ogata, H.; Seya, Y.; Kiyono, K.; Satoh, M.; Tokuyama, K. The effect of nocturnal blue light exposure from light-emitting diodes on wakefulness and energy metabolism the following morning. Environ. Health Prev. Med. 2014, 19, 354–361. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rahman, S.A.; Flynn-Evans, E.E.; Aeschbach, D.; Brainard, G.C.; Czeisler, C.A.; Lockley, S.W. Diurnal spectral sensitivity of the acute alerting effects of light. Sleep 2014, 37, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Souman, J.L.; Tinga, A.M.; te Pas, S.F.; van Ee, R.; Vlaskamp, B.N.S. Acute alerting effects of light: A systematic literature review. Behav. Brain Res. 2018, 337, 228–239. [Google Scholar] [CrossRef]
- Chellappa, S.L. Individual differences in light sensitivity affect sleep and circadian rhythms. Sleep 2020, 214. [Google Scholar] [CrossRef]
- Campbell, S.S.; Dawson, D. Aging young sleep: A test of the phase advance hypothesis of sleep disturbance in the elderly. J. Sleep Res. 1992, 1, 205–210. [Google Scholar] [CrossRef]
- Carrier, J.; Dumont, M. Sleep propensity and sleep architecture after bright light exposure at three different times of day. J. Sleep Res. 1995, 4, 202–211. [Google Scholar] [CrossRef]
- Thorne, H.C.; Jones, K.H.; Peters, S.P.; Archer, S.N.; Dijk, D.J. Daily and seasonal variation in the spectral composition of light exposure in humans. Chronobiol. Int. 2009, 26, 854–866. [Google Scholar] [CrossRef]
- Adamsson, M.; Laike, T.; Morita, T. Annual variation in daily light exposure and circadian change of melatonin and cortisol concentrations at a northern latitude with large seasonal differences in photoperiod length. J. Physiol. Anthropol. 2017, 36, 1–15. [Google Scholar] [CrossRef]
- Loughran, S.P.; McKenzie, R.J.; Jackson, M.L.; Howard, M.E.; Croft, R.J. Individual differences in the effects of mobile phone exposure on human sleep: Rethinking the problem. Bioelectromagnetics 2012, 33, 86–93. [Google Scholar] [CrossRef]
- Lowden, A.; Nagai, R.; Åkerstedt, T.; Mild, K.H.; Hillert, L. Effects of evening exposure to electromagnetic fields emitted by 3G mobile phones on health and night sleep EEG architecture. J. Sleep Res. 2019, 28. [Google Scholar] [CrossRef] [PubMed]
- Baker, F.C.; Driver, H.S. Circadian rhythms, sleep, and the menstrual cycle. Sleep Med. 2007, 8, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Chellappa, S.L.; Steiner, R.; Oelhafen, P.; Cajochen, C. Sex differences in light sensitivity impact on brightness perception, vigilant attention and sleep in humans. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kessel, L.; Lundeman, J.H.; Herbst, K.; Andersen, T.V.; Larsen, M. Age-related changes in the transmission properties of the human lens and their relevance to circadian entrainment. J. Cataract Refract. Surg. 2010, 36, 308–312. [Google Scholar] [CrossRef]
- Kessel, L.; Siganos, G.; Jørgensen, T.; Larsen, M. Sleep disturbances are related to decreased transmission of blue light to the retina caused by lens yellowing. Sleep 2011, 34, 1215–1219. [Google Scholar] [CrossRef]
- Cajochen, C.; Münch, M.; Knoblauch, V.; Blatter, K.; Wirz-Justice, A. Age-related changes in the circadian and homeostatic regulation of human sleep. Chronobiol. Int. 2006, 23, 461–474. [Google Scholar] [CrossRef]
- Skeldon, A.C.; Derks, G.; Dijk, D.J. Modelling changes in sleep timing and duration across the lifespan: Changes in circadian rhythmicity or sleep homeostasis? Sleep Med. Rev. 2016, 28, 96–107. [Google Scholar] [CrossRef]
- Griefahn, B.; Künemund, C.; Bröde, P.; Mehnert, P. Zur validität der deutschen übersetzung des morningness-eveningness-questionnaires von Horne und Östberg. Somnologie 2001, 5, 71–80. [Google Scholar] [CrossRef]
- Saletu, B.; Wessely, P.; Grünberger, J.; Schultes, M. Erste klinische Erfahrungen mit einem neuen schlafanstoßenden Benzodiazepin. Neuropsychiatrie 1987, 1, 169–176. [Google Scholar]
- Donders, F.C. On the speed of mental processes. Acta Psychol. 1969, 30, 412–431. [Google Scholar] [CrossRef]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.E. State-Trait Anxiety Inventory Manual; Mind Garden, Inc.: Menlo Park, CA, USA, 1970. [Google Scholar]
- Schabus, M.; Gruber, G.; Parapatics, S.; Sauter, C.; Klösch, G.; Anderer, P.; Klimesch, W.; Saletu, B.; Zeitlhofer, J. Sleep spindles and their significance for declarative memory consolidation. Sleep 2004, 27, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Kling, M.-U. Die Känguru Chroniken: Ansichten eines Vorlauten Beuteltiers, 1st ed.; Ullstein: Berlin, Germany, 2009. [Google Scholar]
- Golluch, N. 555 Populäre Irrtümer: Warum Angela Merkel Eigentlich ein Wessi ist, man Eier Nicht Abschrecken Muss und Erdnüsse Keine Nüsse Sind; Riva: München, Germany, 2014. [Google Scholar]
- Wölk, J. 13 Gegen das Sommerloch: 13 Autoren—13 Geschichten—13 x Lesespaß; BookRix: Munich, Germany, 2015. [Google Scholar]
- Spitschan, M.; Stefani, O.; Blattner, P.; Gronfier, C.; Lockley, S.; Lucas, R. How to report light exposure in human chronobiology and sleep research experiments. Clocks Sleep 2019, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.; Quan, S.F. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Cohen, M.X. Analyzing Neural Time Series Data: Theory and Practice. Issues in Clinical and Cognitive Neuropsychology; The MIT Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Krauchi, K.; Wirz-Justice, A. Circadian rhythm of heat production, heart rate, and skin and core temperature under unmasking conditions in men. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1994. [Google Scholar] [CrossRef]
- Van Dongen, H.P.A.; Olofsen, E.; van Hartevelt, J.H.; Kruyt, E.W. Searching for biological rhythms: Peak detection in the periodogram of unequally spaced data. J. Biol. Rhythms. 1999, 14, 617–620. [Google Scholar] [CrossRef]
- Borchard, J.P.; Barry, R.J.; de Blasio, F.M. Sequential processing in an auditory equiprobable Go/NoGo task with variable interstimulus interval. Int. J. Psychophysiol. 2015, 97, 145–152. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics Using IBM SPSS Statistics, 4th ed.; SAGE Publications Inc.: Washington, DC, USA, 2013. [Google Scholar]

| No Filter | Filter | Book | χ2 | p | |
|---|---|---|---|---|---|
| TIB (min) | 480.50 (0.50) | 480.50 (1.00) | 480.50 (1.00) | 1.72 | 0.423 |
| TST (min) | 466.00 (34.00) | 468.00 (48.00) | 469.00 (11.75) | 2.46 | 0.292 |
| SEFF (%) | 96.98 (7.42) | 97.40 (9.98) | 97.51 (2.59) | 1.92 | 0.383 |
| SOL N2 (min) | 13.50 (6.75) | 11.00 (12.25) | 11.00 (6.75) | 0.28 | 0.869 |
| N1 (%) | 10.13 (7.24) | 11.42 (7.51) | 10.49 (7.55) | 2.46 | 0.292 |
| N2 (%) | 39.66 (4.33) | 38.35 (8.85) | 40.44 (10.04) | 3.23 | 0.199 |
| N3 (%) | 28.53 (9.88) | 31.65 (10.73) | 25.45 (9.50) | 1.92 | 0.383 |
| REM (%) | 19.25 (6.25) | 20.18 (4.78) | 20.90 (6.66) | 0.46 | 0.794 |
| WASO (min) | 12.00 (26.25) | 6.50 (41.50) | 7.00 (9.25) | 1.92 | 0.383 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Höhn, C.; Schmid, S.R.; Plamberger, C.P.; Bothe, K.; Angerer, M.; Gruber, G.; Pletzer, B.; Hoedlmoser, K. Preliminary Results: The Impact of Smartphone Use and Short-Wavelength Light during the Evening on Circadian Rhythm, Sleep and Alertness. Clocks & Sleep 2021, 3, 66-86. https://doi.org/10.3390/clockssleep3010005
Höhn C, Schmid SR, Plamberger CP, Bothe K, Angerer M, Gruber G, Pletzer B, Hoedlmoser K. Preliminary Results: The Impact of Smartphone Use and Short-Wavelength Light during the Evening on Circadian Rhythm, Sleep and Alertness. Clocks & Sleep. 2021; 3(1):66-86. https://doi.org/10.3390/clockssleep3010005
Chicago/Turabian StyleHöhn, Christopher, Sarah R. Schmid, Christina P. Plamberger, Kathrin Bothe, Monika Angerer, Georg Gruber, Belinda Pletzer, and Kerstin Hoedlmoser. 2021. "Preliminary Results: The Impact of Smartphone Use and Short-Wavelength Light during the Evening on Circadian Rhythm, Sleep and Alertness" Clocks & Sleep 3, no. 1: 66-86. https://doi.org/10.3390/clockssleep3010005
APA StyleHöhn, C., Schmid, S. R., Plamberger, C. P., Bothe, K., Angerer, M., Gruber, G., Pletzer, B., & Hoedlmoser, K. (2021). Preliminary Results: The Impact of Smartphone Use and Short-Wavelength Light during the Evening on Circadian Rhythm, Sleep and Alertness. Clocks & Sleep, 3(1), 66-86. https://doi.org/10.3390/clockssleep3010005







