The Management of Resistant Hypercalcaemia Secondary to Cancer of Unknown Primary and Presenting with Pancreatitis
Abstract
:1. Introduction
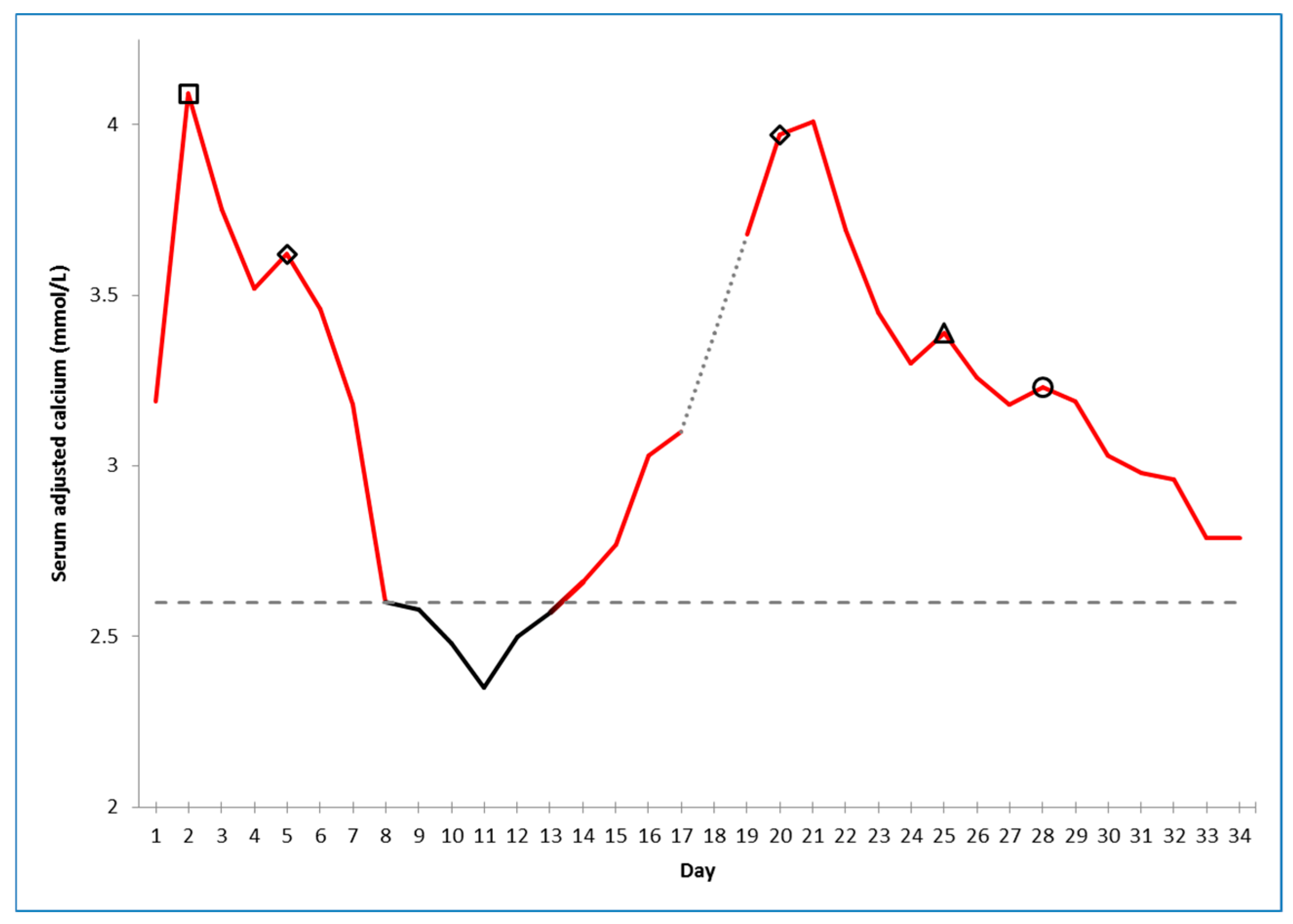
2. Case Presentation Section
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Minisola, S.; Pepe, J.; Piemonte, S.; Cipriani, C. The diagnosis and management of hypercalcaemia. BMJ 2015, 350, h2723. [Google Scholar] [CrossRef] [PubMed]
- Žofková, I. Hypercalcemia. Pathophysiological aspects. Physiol. Res. 2016, 65, 1–10. [Google Scholar] [PubMed]
- Pavlidis, N.; Pentheroudakis, G. Cancer of unknown primary site. Lancet 2012, 379, 1428–1435. [Google Scholar] [CrossRef]
- Krishna, S.; Lakhtakia, S.; Modi, K.D.; Kota, S.; Modi, K.; Kota, S.K.; Modi, K.D. Metabolic pancreatitis: Etiopathogenesis and management. J. Endocrinol. Metab. 2013, 17, 799–805. [Google Scholar]
- Jick, S.; Li, L.; Gastanaga, V.M.; Liede, A. Prevalence of hypercalcemia of malignancy among cancer patients in the UK: Analysis of the Clinical Practice Research Datalink database. Cancer Epidemiol. 2015, 39, 901–907. [Google Scholar] [PubMed]
- Lamy, O.; Jenzer-Closuit, A.; Burckhardt, P.; Jenzer-Closuit, A. Hypercalcaemia of malignancy: An undiagnosed and undertreated disease. J. Int. Med. 2001, 250, 73–79. [Google Scholar]
- Goldner, W. Cancer-Related Hypercalcemia. J. Oncol. Pract. 2016, 12, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Donovan, P.J.; Achong, N.; Griffin, K.; Galligan, J.; Pretorius, C.J.; McLeod, D.S.A. PTHrP-Mediated Hypercalcemia: Causes and Survival in 138 Patients. J. Clin. Endocrinol. Metab. 2015, 100, 2024–2029. [Google Scholar] [CrossRef] [PubMed]
- Mirrakhimov, A.E. Hypercalcemia of Malignancy: An Update on Pathogenesis and Management. Am. J. Med. Sci. 2015, 7, 483–493. [Google Scholar] [CrossRef]
- Vakiti, A.; Mewawalla, P. Malignancy-Related Hypercalcemia; StatPearls Publishing: Florida, 2018. [Google Scholar]
- Maxwell, J.E.; Howe, J.R. Imaging in neuroendocrine tumors: An update for the clinician. Int. J. Endocr. Oncol. 2015, 2, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Mojtahedi, A.; Thamake, S.; Tworowska, I.; Ranganathan, D.; Delpassand, E.S. The value of 68Ga-DOTATATE PET/CT in diagnosis and management of neuroendocrine tumors compared to current FDA approved imaging modalities: A review of literature. Am. J. Nucl. Med. Mol. 2014, 4, 426–434. [Google Scholar]
- Lazaridis, G.; Pentheroudakis, G.; Fountzilas, G.; Pavlidis, N. Liver metastases from cancer of unknown primary (CUPL): A retrospective analysis of presentation, management and prognosis in 49 patients and systematic review of the literature. Treat. Rev. 2008, 34, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Frick, T.W. The role of calcium in acute pancreatitis. Surgery 2012, 152, S157–S163. [Google Scholar] [CrossRef] [PubMed]
- Frick, T.W.; Mithöfer, K.; Castillo, C.F.-D.; Rattner, D.W.; Warshaw, A.L. Hypercalcemia causes acute pancreatitis by pancreatic secretory block, intracellular zymogen accumulation, and acinar cell injury. Am. J. Surg. 1995, 169, 167–172. [Google Scholar] [CrossRef]
- Rossi, R.E.; Naik, K.; Navalkissoor, S.; Imber, C.; O’Beirne, J.; Toumpanakis, C.; Caplin, M.E. Case report of multimodality treatment for metastatic parathyroid hormone-related peptide-secreting pancreatic neuroendocrine tumour. Tumori J. 2014, 100, e153–e156. [Google Scholar] [CrossRef]
- Milanesi, A.; Yu, R.; Wolin, E.M. Humoral hypercalcemia of malignancy caused by parathyroid hormone-related peptide-secreting neuroendocrine tumors: Report of six cases. Pancreatology 2013, 13, 324–326. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, J.; Lauer, J.; Gunatilake, R.; Knudtson, E. Multiple Myeloma Presenting as Hypercalcemic Pancreatitis During Pregnancy. Obstet. Gynecol. 2014, 124, 461–463. [Google Scholar] [CrossRef] [PubMed]
- Popescu, M.; Popov, V.; Popescu, G.; Dobrea, C.; Sandu, A.; Grigorean, V.T.; Strâmbu, V.; Pleşea, I.E. Acute pancreatitis: The onset digestive manifestation, in a patient with adult T-cell leukemia/lymphoma. Roman. J. Morphol. Embryol. 2012, 53, 847–850. [Google Scholar]
- Çelik, İ.; Apras, S.; Kars, A.; Barista, I. Persistent Hypercalcemia Leading to Acute Pancreatitis in a Patient with Relapsed Myeloma and Renal Failure. Nephron 1998, 79, 109–110. [Google Scholar] [CrossRef]
- Senba, M.; Senba, M.I. Acute pancreatitis associated with hypercalcaemia in adult T-cell leukaemia-lymphoma. J. Gastroenterol. Hepatol. 1996, 11, 180–182. [Google Scholar] [CrossRef]
- Ono, Y.; Kimura, T.; Nakano, I.; Furukawa, M.; Ito, T.; Sakamoto, S.; Konomi, K.; Nawata, H. Acute pancreatitis induced by hypercalcaemia associated with adult T-cell leukaemia: A case report. J. Gastroenterol. Hepatol. 1996, 11, 193–195. [Google Scholar] [CrossRef]
- Ito, T.; Kimura, T.; Yamashita, S.; Abe, Y.; Haji, M.; Nawata, H. Acute Pancreatitis Associated with Hypercalcemia in a Patient with Multiple Myeloma. Pancreas 1992, 7, 396–398. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-G.; Huang, T.-Y.; Hsieh, T.-Y.; Chen, P.-J. Acute pancreatitis due to hypercalcemia related to bone metastasis from breast cancer. QJM Int. J. Med. 2011, 106, 367–369. [Google Scholar] [CrossRef]
- Hussain, A.; Adnan, A.; EL-Hasani, S. Small Cell Carcinoma of the Lung Presented as Acute Pancreatitis. Case Report and Review of the Literature. JOP J. Pancreas 2012, 13, 702–704. [Google Scholar] [CrossRef]
- Kanno, K.; Hikichi, T.; Saito, K.; Watanabe, K.; Takagi, T.; Shibukawa, G.; Wakatsuki, T.; Imamura, H.; Takahashi, Y.; Sato, A.; et al. A case of esophageal small cell carcinoma associated with hypercalcemia causing severe acute pancreatitis. Fukushima J. Med Sci. 2007, 53, 51–60. [Google Scholar] [CrossRef]
- Wymenga, A.; Van Der Werf, T.; Van Der Graaf, W.; Tulleken, J.; Zijlstra, J.; Ligtenberg, J. Lessons from an unusual case: Malignancy associated hypercalcemia, pancreatitis and respiratory failure due to ARDS. Neth. J. Med. 1999, 54, 27–30. [Google Scholar] [CrossRef]
- Hsieh, T.L.; Chen, J.J.; Chien, C.S.; Chiou, S.S.; Tai, D.I.; Lee, C.N.; Kuo, C.H.; Chiu, K.W.; Chuah, S.K.; Hu, T.H. Small cell lung cancer with liver and bone metastasis associated with hypercalcemia and acute pancreatitis—A case report. Changgeng Yi Xue Za Zhi 1995, 18, 190–193. [Google Scholar] [PubMed]
- Dhar, S.; Kokroo, T.; Grossman, C.E. Metastatic Neuroendocrine Tumor of Unknown Primary Presenting as Acute Pancreatitis. Am. J. Med. Sci. 2008, 335, 304–306. [Google Scholar] [CrossRef]
- Munro, A.; Brown, M.; Niblock, P.; Steele, R.; Carey, F. Do Multidisciplinary Team (MDT) processes influence survival in patients with colorectal cancer? A population-based experience. BMC Cancer 2015, 15, 313. [Google Scholar] [CrossRef]
- Bydder, S.; Nowak, A.; Marion, K.; Phillips, M.; Atun, R. The impact of case discussion at a multidisciplinary team meeting on the treatment and survival of patients with inoperable non-small cell lung cancer. Int. Med. J. 2009, 39, 838–841. [Google Scholar] [CrossRef] [PubMed]
- Patkar, V.; Acosta, D.; Davidson, T.; Jones, A.; Fox, J.; Keshtgar, M. Cancer Multidisciplinary Team Meetings: Evidence, Challenges, and the Role of Clinical Decision Support Technology. Int. J. Breast Cancer 2011, 2011, 1–7. [Google Scholar] [CrossRef]

| Causes | Conditions |
|---|---|
| Parathyroid hormone mediated | Sporadic (adenoma, hyperplasia, or carcinoma); Familial (multiple endocrine neoplasia 1, 2a, or 4, hyperparathyroidism jaw tumour syndrome, familial isolated hyperparathyroidism, familial hypocalciuria hypercalcaemia); Ectopic parathyroid hormone in malignancy (rare); “Tertiary” hyperparathyroidism. |
| Malignancy | Humoral hypercalcaemia of malignancy (parathyroid hormone related protein); Local osteolysis (cytokines, chemokines, parathyroid hormone related protein); Ectopic parathyroid hormone in malignancy (rare); Calcitriol related hypercalcaemia. |
| Vitamin D related | Granulomatous disease (for example, sarcoidosis, tuberculosis, berylliosis, coccidiodomycosis, histoplasmosis, leprosy, inflammatory bowel disease, foreign body granuloma); Vitamin D intoxication (vitamin D supplements, metabolites, or analogues). |
| Endocrine disorders | Thyrotoxicosis; Adrenal insufficiency; Pheochromocytoma; VIPoma (Verner–Morrison) syndrome |
| Drugs | Thiazide diuretics; Lithium; Milk–alkali syndrome (calcium and antacids); Vitamin A; Parathyroid hormone. |
| Other | Coexisting malignancy and primary hyperparathyroidism; Immobilisation; Acute renal failure; Chronic renal failure treated with calcium and calcitriol or vitamin D analogues; Renal transplant. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
George, J.; Reed, E.; Patel, K.; Gardner-Thorpe, J. The Management of Resistant Hypercalcaemia Secondary to Cancer of Unknown Primary and Presenting with Pancreatitis. Reports 2019, 2, 13. https://doi.org/10.3390/reports2020013
George J, Reed E, Patel K, Gardner-Thorpe J. The Management of Resistant Hypercalcaemia Secondary to Cancer of Unknown Primary and Presenting with Pancreatitis. Reports. 2019; 2(2):13. https://doi.org/10.3390/reports2020013
Chicago/Turabian StyleGeorge, Jayan, Emily Reed, Kuunal Patel, and James Gardner-Thorpe. 2019. "The Management of Resistant Hypercalcaemia Secondary to Cancer of Unknown Primary and Presenting with Pancreatitis" Reports 2, no. 2: 13. https://doi.org/10.3390/reports2020013
APA StyleGeorge, J., Reed, E., Patel, K., & Gardner-Thorpe, J. (2019). The Management of Resistant Hypercalcaemia Secondary to Cancer of Unknown Primary and Presenting with Pancreatitis. Reports, 2(2), 13. https://doi.org/10.3390/reports2020013





