Water Availability Associated with Coinoculation with Growth-Promoting Rhizobacteria in Cowpea
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Design
2.2. Irrigation System
2.3. Variables Analyzed
2.4. Statistical Analysis
3. Results and Discussion
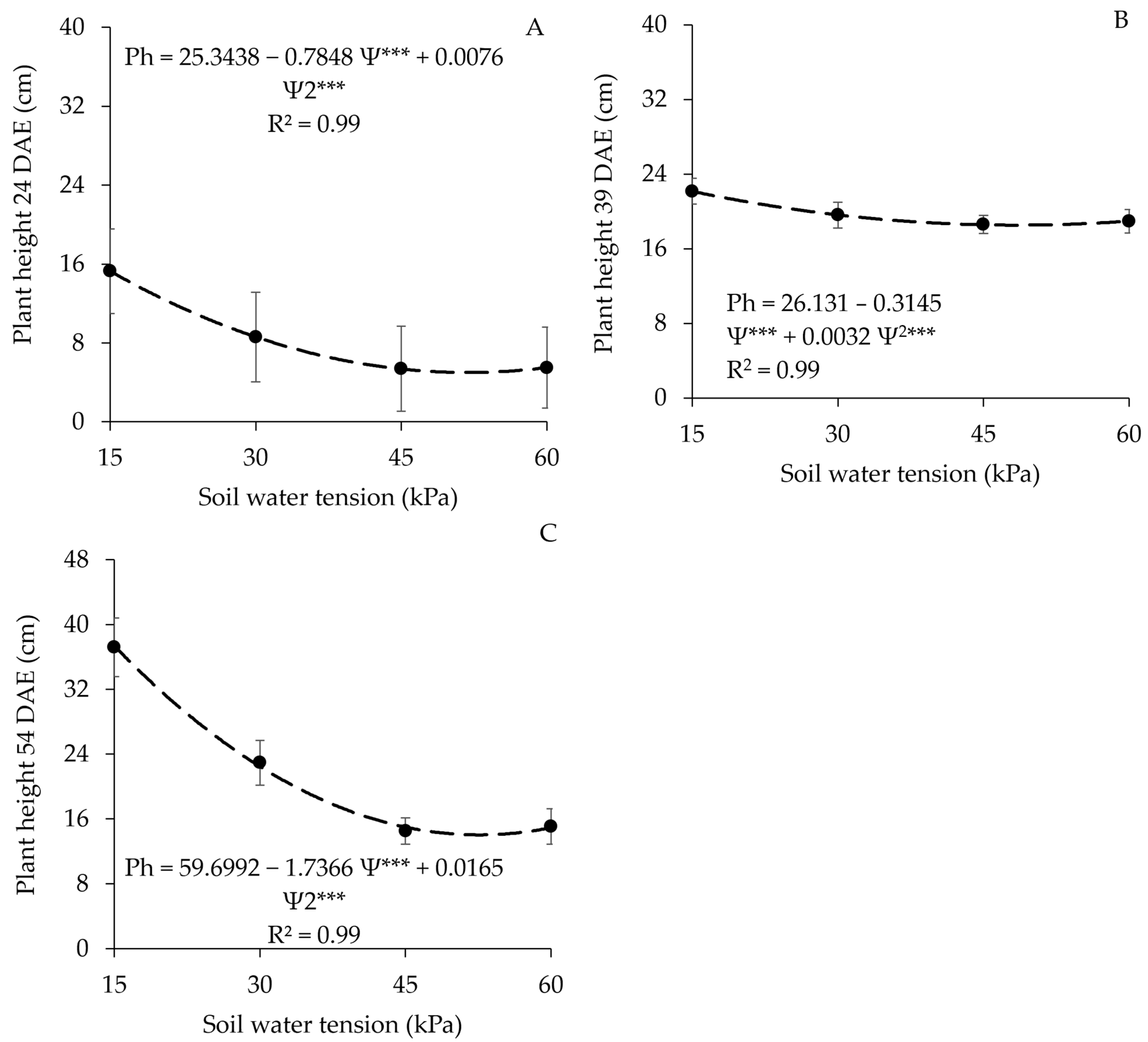
3.1. Plant Height
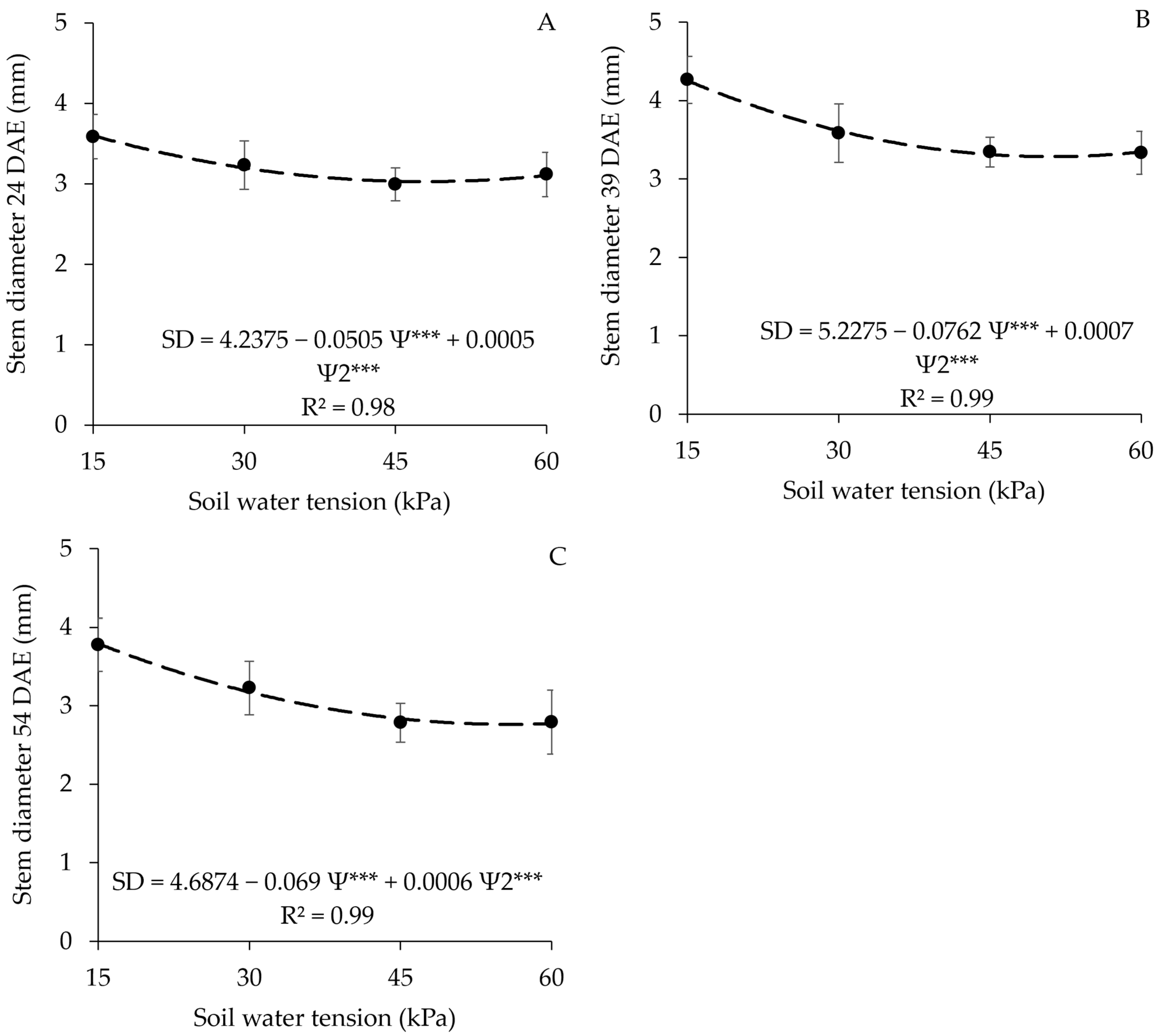
3.2. Stem Diameter
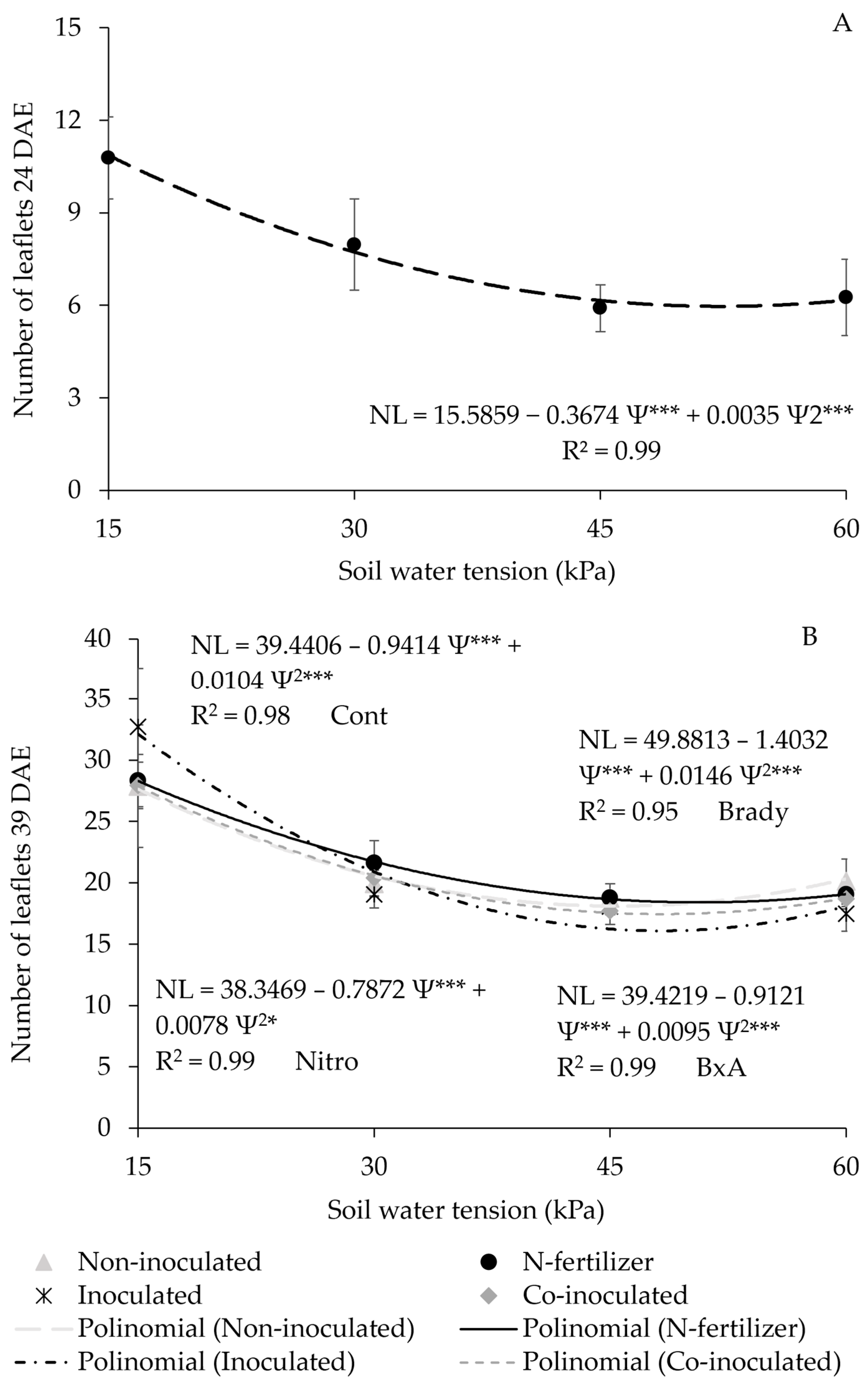
3.3. Number of Leaflets
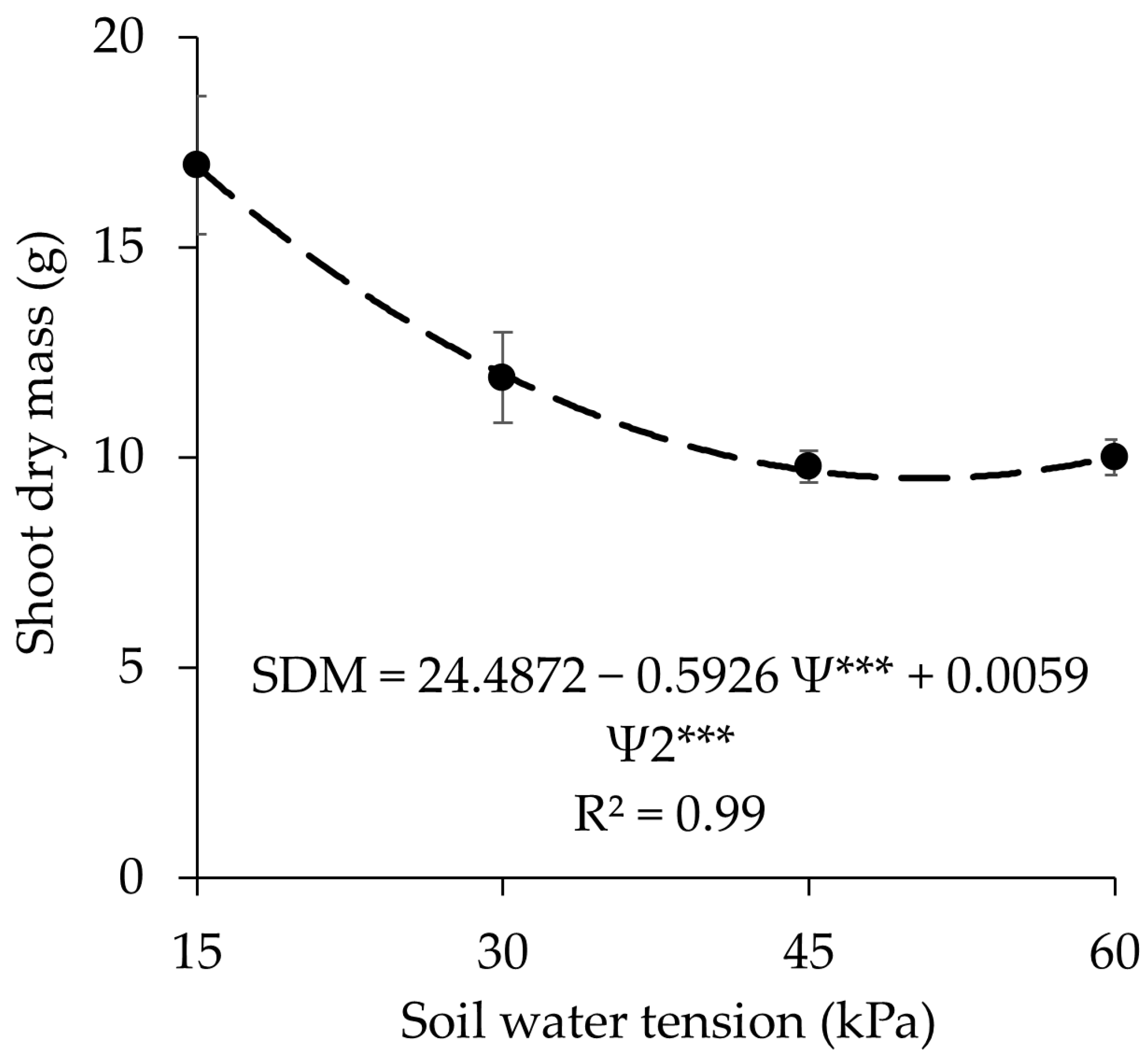
3.4. Shoot Dry Mass
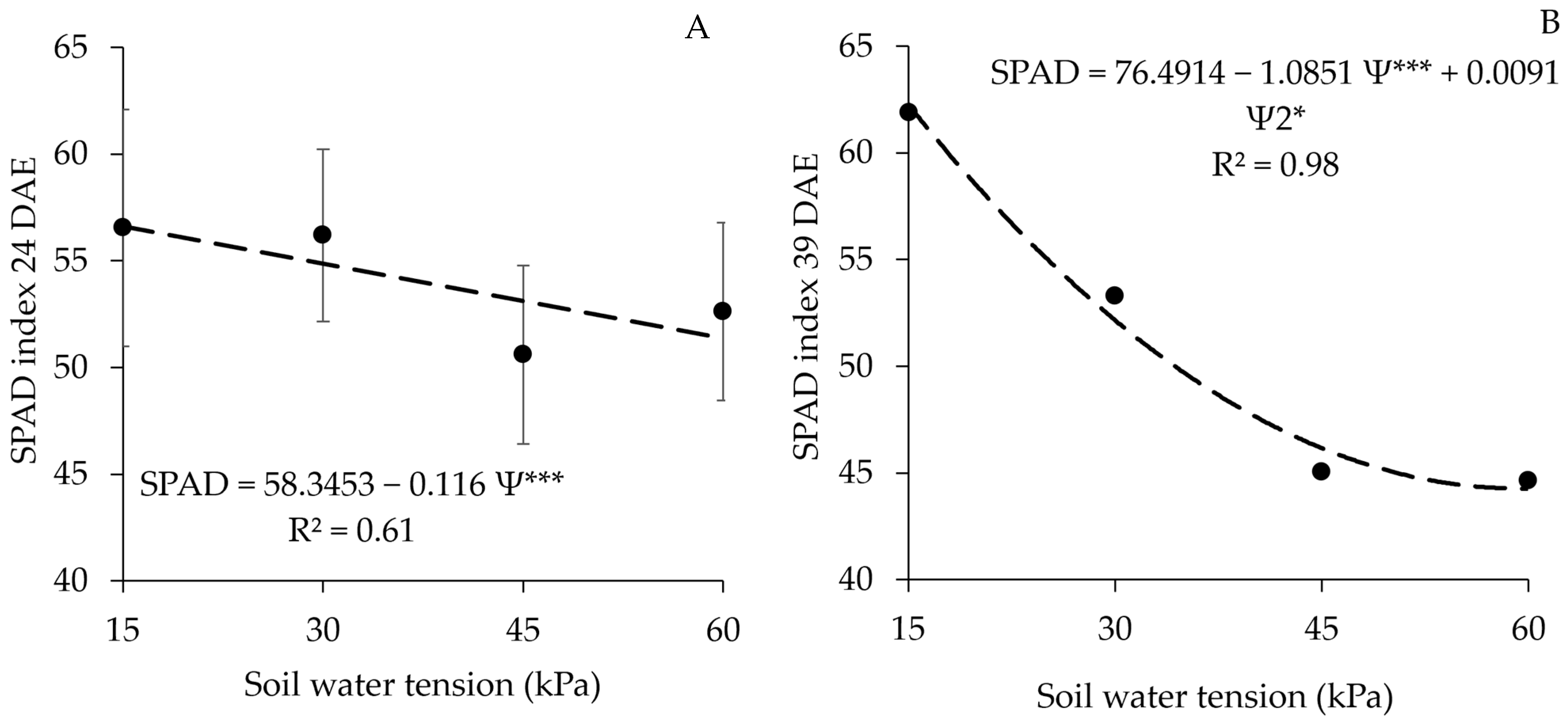
3.5. SPAD Index
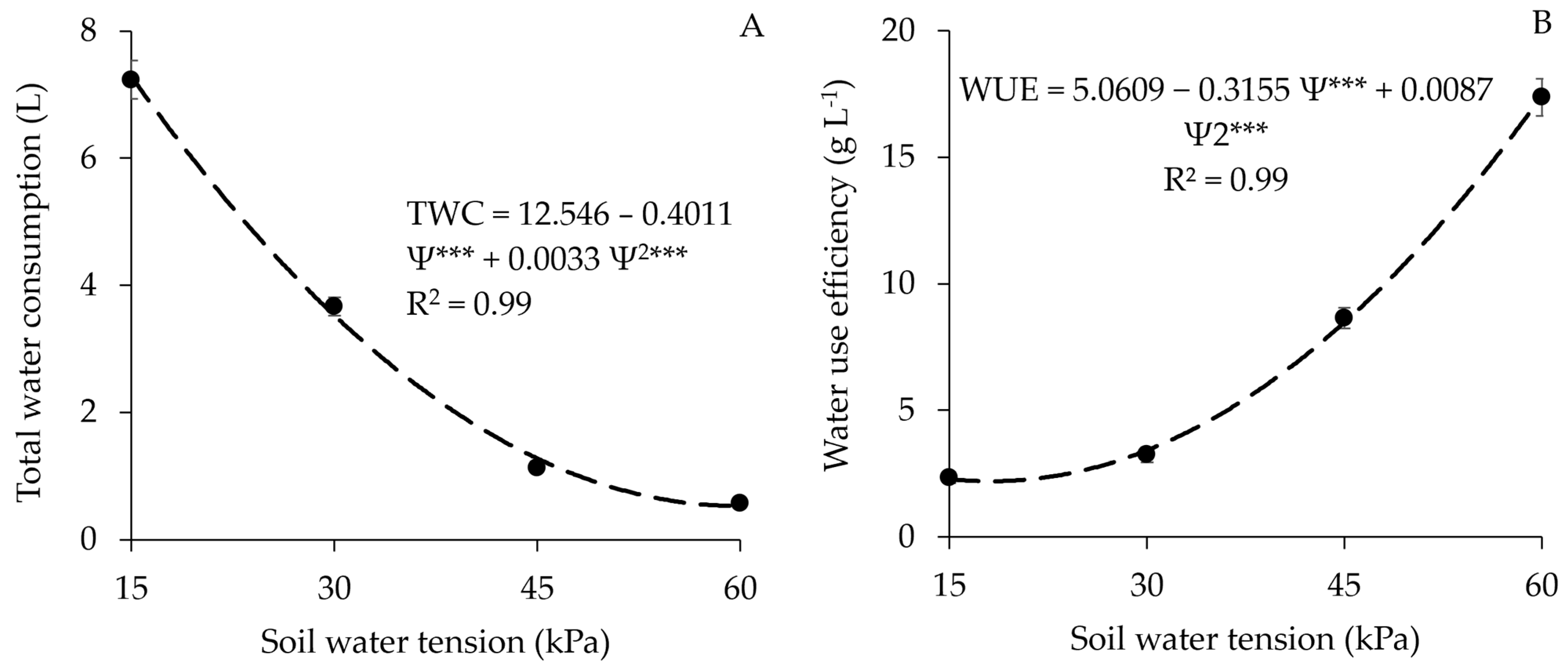
3.6. Total Water Consumption and Water Use Efficiency
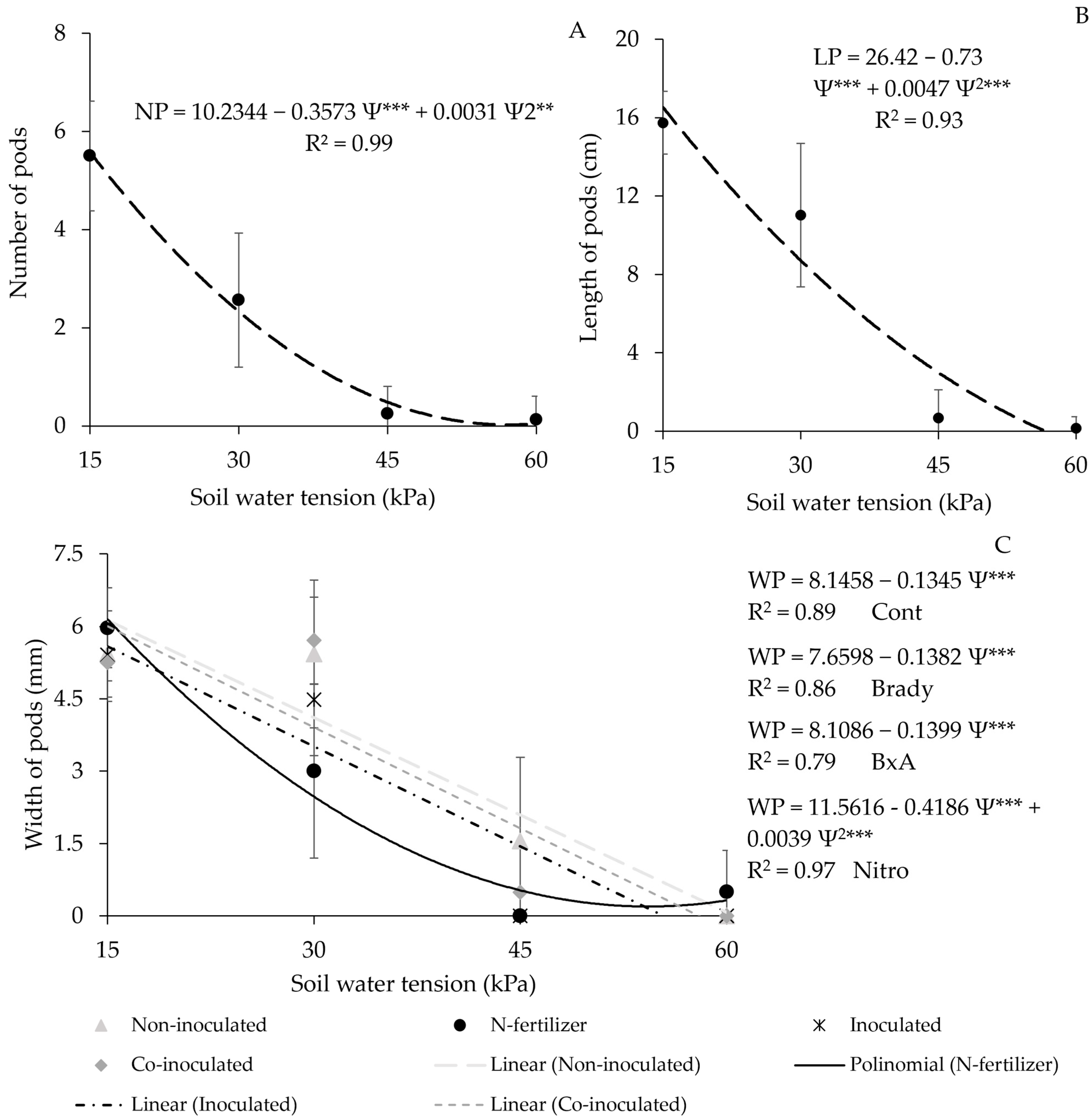
3.7. Number, Length, and Width of Pods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CONAB—National Supply Company. Monitoring the Brazilian Grain Harvest—v.1, n.1 (2013-)—Brasília: Conab. 2013. Available online: https://www.conab.gov.br (accessed on 26 August 2025).
- Qaseem, M.F.; Qureshi, R.; Shaheen, H.; Kousar, R.; Röder, M.S. Genome-wide association mapping in bread wheat subjected to independent and combined high temperature and drought stress. PLoS ONE 2018, 13, e0199121. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Sarkar, S.; Das, S.; Dutta, S.; Choudhury, M.R.; Giri, A.; Bera, B.; Bag, K.; Mukherjee, B.; Banerjee, K.; et al. Water scarcity: A global hindrance to sustainable development and agricultural production–A critical review of the impacts and adaptation strategies. Camb. Prism. Water 2025, 3, e4. [Google Scholar] [CrossRef]
- Hungria, M.; Nogueira, M.A.; Araújo, R.S. Coinoculation of soybeans and common beans with rhizobia and azospirilla: Strategies to improve sustainability. Biol. Fertil. Soils 2013, 49, 791–801. [Google Scholar] [CrossRef]
- Bulegon, L.G.; Rampim, L.; Klein, J.; Kestring, D.; Guimarães, V.F.; Battistus, A.G.; Inagaki, A.M. Components of production and yield of soybean inoculated with Bradyrhizobium and Azospirillum. Terra Latinoam. 2016, 34, 169–176. [Google Scholar]
- Vanneste, S.; Pei, Y.; Friml, J. Mechanisms of auxin action in plant growth and development. Nat. Rev. Mol. Cell Biol. 2025, 26, 648–666. [Google Scholar] [CrossRef]
- Silva, A.F.; Carvalho, M.A.C.; Schoninger, E.L.; Monteiro, S.; Caione, G.; Santos, P.A. Doses of inoculant and nitrogen at soybean sowing in a first crop area. Biosci. J. 2011, 27, 404–412. [Google Scholar]
- Goyal, R.K.; Mattoo, A.K.; Schmidt, M.A. Rhizobial–host interactions and symbiotic nitrogen fixation in legume crops toward agriculture sustainability. Front. Microbiol. 2021, 12, 669404. [Google Scholar] [CrossRef]
- Tien, T.M.; Gaskins, M.H.; Hubbell, D.H. Plant growth substances produced by Azospirillum brasilense and their effect on the growth of pearl millet (Pennisetum americanum L.). Appl. Environ. Microbiol. 1979, 37, 1016–1024. [Google Scholar] [CrossRef]
- Döbereiner, J.; Pedrosa, F.O. Nitrogen-Fixing Bacteria in Nonleguminous Crop Plants; Science Tech; Springer: Madison, WI, USA, 1987; pp. 1–155. [Google Scholar]
- Coniglio, A.; Larama, G.; Nievas, S.; Cale, N.L.; Mora, V.; Torres, D.; Lopez, G.; Donadio, F.; Rodriguez, B.; Marin, A.; et al. Auxin-mediated modulation of the maize rhizosphere microbiome: Insights from inoculation with Azospirillum and treatment with indole-3-acetic acid. J. Soil Sci. Plant Nutr. 2024, 24, 6906–6921. [Google Scholar] [CrossRef]
- Silva, E.R.; Zoz, J.; Oliveira, C.E.S.; Zuffo, A.M.; Steiner, F.; Zoz, T.; Vendruscolo, E.P. Can co-inoculation of Bradyrhizobium and Azospirillum alleviate adverse effects of drought stress on soybean (Glycine max L. Merrill.)? Arch. Microbiol. 2019, 201, 325–335. [Google Scholar] [CrossRef]
- Rondina, A.B.L.; Sanzovo, A.W.S.; Guiramães, G.S.; Wendling, J.R.; Nogueira, M.A.; Hungria, M. Changes in root morphological traits in soybean coinoculated with Bradyrhizibium spp. and Azospirillum brasilense or treated with A. brasilense exudates. Biol. Fertil. Soils 2020, 56, 537–549. [Google Scholar] [CrossRef]
- Guimarães, S.L.; Neves, L.C.R.; Bonfim-Silva, E.M.; Campos, D.T.S. Development of pigeon pea inoculated with Rhizobium isolated from cowpea trap host plants. Rev. Caatinga 2016, 29, 789–795. [Google Scholar] [CrossRef]
- Batista, E.R.; Guimarães, S.L.; Bonfim-Silva, E.M.; Souza, A.C.P. Combined inoculation of rhizobia on the cowpea development in the soil of Cerrado. Rev. Ciência Agron. 2017, 48, 745–755. [Google Scholar] [CrossRef]
- Guimarães, S.L.; Bonfim-Silva, E.M.; Moreira, J.C.F.; Silva, L.S.; Bosa, C.K.; Silva, T.J.A.; Koetz, M. Structural and productive characteristics of Cowpea (Vigna ungiculata) under different water availabilities. Afr. J. Agric. Res. 2018, 13, 2741–2747. [Google Scholar] [CrossRef]
- Van Raij, B. (Ed.) Soil Fertility and Fertilization; Ceres—Potafós: Piracicaba City, Brazil, 1991; Volume 1. [Google Scholar]
- Bonfim-Silva, E.M.; Costa, A.S.; José, J.V.; Ferraz, A.P.F.; Damasceno, A.P.A.B.; Silva, T.J.A. Correction of Acidity of a Brazilian Cerrado Oxisol with Limestone and Wood Ash on the Initial Growth of Cowpea. Agric. Sci. 2019, 10, 841–851. [Google Scholar] [CrossRef]
- Van Genuchten, M.T.A. A closed-form equation to predict the hydraulic conductivity of unsaturated soils. Soil Sci. Soc. Am. J. 1980, 44, 892–898. [Google Scholar] [CrossRef]
- Dourado Neto, D.; Jong Van Lier, Q.; Botrel, T.A.; Libardi, P.L. Program for the preparation of the soil water retention curve using the Genuchten model. Eng. Rural. 1990, 1, 92–102. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing, Version 3.6.1; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Ferreira, E.B.; Cavalcanti, P.P.; Nogueira, D.A. ExpDes: Experimental Designs (Portuguese), R Package Version 1.2.0. 2018. Available online: https://cran.r-project.org/web/packages/ExpDes/index.html (accessed on 22 April 2025).
- Wahab, A.; Abdi, G.; Saleem, M.H.; Ali, B.; Ullah, S.; Shah, W.; Mumtaz, S.; Yasin, G.; Muresan, C.C.; Marc, R.A. Plants’ Physio-Biochemical and Phyto-Hormonal Responses to Alleviate the Adverse Effects of Drought Stress: A Comprehensive Review. Plants 2022, 11, 1620. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Ansary, M.H.; Rahmani, H.A.; Ardakani, M.R.; Paknejad, F.; Habibi, D.; Mafakheri, S. Effect of Pseudomonas fluorescente on proline and phytohormonal status of maize (Zea mays L.) under water deficit stress. Ann. Biol. Res. 2012, 3, 1054–1062. [Google Scholar]
- Bray, E.A. Plant responses to water deficit. Trends Plant Sci. 1997, 2, 48–54. [Google Scholar] [CrossRef]
- Yu, Z.; Duan, X.; Luo, L.; Dai, S.; Ding, Z.; Xia, G. How plant hormones mediate salt stress responses. Trends Plant Sci. 2020, 25, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Brito, M.M.P.; Muraoka, T.; Silva, E.C. Soil nitrogen uptake, fertilizer and symbiotic fixation gait in cowpea (Vigna unguiculata (L.) WALP.) and common bean (Phaseolus vulgaris L.) determined using 15N. Braz. J. Soil Sci. 2009, 33, 895–905. [Google Scholar]
- Lopes, M.J.D.S.; Dias-Filho, M.B.; Gurgel, E.S.C. Successful plant growth-promoting microbes: Inoculation methods and abiotic factors. Front. Sustain. Food Syst. 2021, 5, 606454. [Google Scholar] [CrossRef]
- Vurukonda, S.S.K.P.; Fotopoulos, V.; Saeid, A. Production of a rich fertilizer base for plants from waste organic residues by microbial formulation technology. Microorganisms 2024, 12, 541. [Google Scholar] [CrossRef]
- Martínez-Viveros, O.; Jorquera, M.A.; Crowley, D.E.; Gajardo, G.; Mora, M.L. Mechanisms and practical considerations involved in plant growth promotion by rhizobacteria. J. Soil Sci. Plant Nutr. 2010, 10, 293–319. [Google Scholar] [CrossRef]
- Pál, M.; Tajti, J.; Szalai, G.; Peeva, V.; Végh, B.; Janda, T. Interaction of Polyamines, Abscisic Acid and Proline under Osmotic Stress in the Leaves of Wheat Plants. Sci. Rep. 2018, 8, 12839. [Google Scholar] [CrossRef]
- Dourado, L.G.A.; Bonfim-Silva, E.M.; da Silva, T.J.A.; Pinheiro, E.A.R.; Fenner, W. Effects of wood ash and soil water potential on the vegetative development of mung bean (‘Vigna radiata’ L.). Aust. J. Crop Sci. 2021, 15, 354–361. [Google Scholar] [CrossRef]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulatory mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef]
- García-Sánchez, F.; Syvertsen, J.P.; Gimeno, V.; Botía, P.; Perez-Perez, J.G. Responses to flooding and drought stress by two citrus rootstock seedlings with different water-use efficiency. Physiol. Plant. 2007, 130, 532–542. [Google Scholar] [CrossRef]
- Das, S.; KAR, R.K. Abscisic acid mediated differential growth responses of root and shoot of Vigna radiata (L.) Wilczek seedlings under water stress. Plant Physiol. Biochem. 2017, 123, 213–221. [Google Scholar] [CrossRef]
- Sun, Y.; Wu, M.; Xie, S.; Zang, J.; Wang, X.; Yang, Y.; Li, C.; Wang, J. Homogenization of bacterial plastisphere community in soil: A continental-scale microcosm study. ISME Commun. 2024, 4, 1. [Google Scholar] [CrossRef]
- Halsall, D.M.; Gibson, A.H. Nitrogenase activity of a range of diazotrophic bacteria on straw, straw breakdown products and related compounds. Soil Biol. Biochem. 1989, 21, 291–298. [Google Scholar] [CrossRef]
- Kasim, W.A.; Osman, M.E.; Omar, M.N.; Adb El-Daim, I.A.; Bejai, S.; Meijer, J. Control of drought stress in wheat using plant-growth-promoting bacteria. J. Plant Growth Regul. 2012, 32, 122–130. [Google Scholar] [CrossRef]
- Melo, H.F.; Souza, E.R.; Dourado, P.R.M.; Lins, C.M.T.; Santos, H.R.B.; Monteiro, D.R.; Paulino, M.K.S.S.; Almeida, B.G.; Santos, M.A. Comparison of water and osmotic potentials on Vigna unguiculata stress response. Rev. Bras. Ciência Solo 2020, 44, 1–16. [Google Scholar] [CrossRef]
- Paul, M.J.; Primavesi, L.F.; Jhurreea, D.; Zhang, Y. Trehalose metabolism and signaling. Annu. Rev. Plant Biol. 2008, 59, 417–441. [Google Scholar] [CrossRef]
- Naseem, H.; Bano, A. Role of plant growth-promoting rhizobacteria and their exopolysaccharide in drought tolerance in maize. J. Plant Interact. 2014, 9, 689–701. [Google Scholar] [CrossRef]
- Yadav, V.K.; Raghav, M.; Sharma, S.K.; Bhagat, N. Rhizobacteriome: Promising candidate for conferring drought tolerance in crops. J. Pure Appl. Microbiol. 2020, 14, 73–92. [Google Scholar] [CrossRef]
- Vardharajula, S.; Ali, S.Z.; Grover, M.; Reddy, G.; Bandi, V. Drought tolerant plant growth promoting Bacillus spp.: Effect on growth, osmolytes and antioxidante status of maize under droght stress. J. Plant Interact. 2011, 6, 1–14. [Google Scholar] [CrossRef]
- Schlichting, A.F.; Bonfim-Silva, E.M.; Silva, M.C.; Pietro-Souza, W.; Silva, T.J.A.; Farias, L.N. Efficiency of portable chlorophyll meters in assessing the nutritional status of wheat plants. Braz. J. Agric. Environ. Eng. 2015, 19, 1148–1151. [Google Scholar] [CrossRef]
- Croft, H.; Chen, J.M. Leaf pigment content. Compr. Remote Sens. 2018, 3, 117–142. [Google Scholar]
- Freitas, R.M.O.; Dombroski, J.L.D.; Freitas, F.C.L.; Nogueira, N.W.; Pinto, J.R.S. Physiological responses of cowpea under water stress and rewatering in no-tillage and conventional tillage systems. Rev. Caatinga 2017, 30, 559–567. [Google Scholar] [CrossRef]
- Anyia, A.O.; Herzog, H. Water-use efficiency, leaf area and leaf gas exchange of cowpeas under mid-season drought. Eur. J. Agron. 2004, 20, 327–339. [Google Scholar] [CrossRef]
- Guimarães, C.M.; Stone, L.F.; Brunini, O. Adaptation of common bean (Phaseolus vulgaris L.) to drought. Braz. J. Agric. Environ. Eng. 2006, 10, 70–75. [Google Scholar]
- Aalto, M.K.; Helenius, E.; Kariola, T.; Pennanen, V.; Heino, P.; Hõrak, H.; Puzõrjova, I.; Kollist, H.; Palva, E.T. ERD15—An attenuator of plant ABA responses and stomatal aperture. Plant Sci. 2012, 182, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Souza, P.J.O.P.; Ramos, T.F.; Fiel, L.C.S.; Farias, V.D.d.S.; Nunes, H.G.G.C. Yield and water use efficiency of cowpea under water deficit. Rev. Bras. Eng. Agríc. Ambient. 2019, 23, 119–125. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Li, Y.; Zhang, S. Improving water-use efficiency by decreasing stomatal conductance and transpiration rate to maintain higher ear photosyn-thetic rate in drought-resistant wheat. Crop J. 2017, 5, 231–239. [Google Scholar] [CrossRef]
- Souza, L.S.B.; Moura, M.S.B.; Sediyama, G.C.; Silva, T.G.F. Water use efficiency of maize and cowpea crops under exclusive and intercropped planting systems in the Brazilian semiarid region. Bragantia 2011, 70, 715–721. [Google Scholar] [CrossRef]
- Twalla, J.T.; Ding, B.; Cao, G.; Bao, S.; Li, M.; Chen, X.; Xie, X.; Wang, J. Roles of stomata in gramineous crops growth and biomass production. Cereal Res. Commun. 2022, 50, 603–616. [Google Scholar] [CrossRef]
- Silva, C.D.S.; Santos, P.A.A.; Lira, J.M.S.; Santana, M.C.; Júnior, C.D.S. Daily course of gas exchange in cowpea plants subjected to water deficit. Caatinga Mag. 2010, 23, 7–13. [Google Scholar]
- Dutra, A.F.; Melo, A.S.; Filgueiras, L.M.B.; Silva, A.R.F.; Oliveira, I.M.; Brito, M.E.B. Physiological parameters and production components of cowpea cultivated under water deficit. Braz. J. Agrar. Sci. 2015, 10, 189–197. [Google Scholar] [CrossRef][Green Version]
- Bastos, E.A.; Nascimento, S.P.; Silva, E.M.; Filho, F.R.F.; Gomide, R.L. Identification of cowpea genotypes for drought tolerance. Rev. Ciência Agron. 2011, 42, 100–107. [Google Scholar] [CrossRef]
- Rufini, M.; Ferreira, P.A.A.; Soares, B.L.; Oliveira, D.P.; Andrade, M.J.B.; Moreira, F.M.S. Symbiosis of nitrogen-fixing bacteria with common bean at different pH values. Braz. Agric. Res. 2011, 46, 81–88. [Google Scholar] [CrossRef]
- Silva, H.A.P.; Caetano, V.S.; Bezerra, A.C.M.; Vidal, M.S.; Araújo, J.L.S. Effect of Water Deficit on Biological N2 Fixation in Common Bean Cultivars; Research and Development Bulletin, 94; EM-BRAPA: Rio de Janeiro, Brazil, 2012; 24p. [Google Scholar]
- Silva, C.F.; Moura, M.F.; Vilela, A.R.; Araújo, M.B.; Marques, J.D.S. Macassar bean production in the function of inoculant employment and organic and mineral fertilizers. Divers. J. 2019, 4, 1130–1145. [Google Scholar] [CrossRef]
| Treatment | Plant Height at 24 DAEs (cm) |
|---|---|
| Not Inoculated | 8.68 ab |
| Inoculated | 7.84 b |
| Coinoculated | 8.65 ab |
| Nitrogen Fertilizer | 9.56 a |
| Average | 8.68 |
| Treatment | Stem Diameter at 39 DAEs (mm) | Stem Diameter at 54 DAEs (mm) |
|---|---|---|
| Not Inoculated | 3.50 b | 3.03 b |
| Inoculated | 3.47 b | 2.96 b |
| Coinoculated | 3.69 ab | 3.26 a |
| Nitrogen Fertilizer | 3.85 a | 3.31 a |
| Average | 3.63 | 3.14 |
| Treatment | SPAD Index at 24 DAEs | SPAD Index at 39 DAEs |
|---|---|---|
| Not Inoculated | 52.10 b | 47.36 c |
| Inoculated | 51.61 b | 49.44 bc |
| Coinoculated | 55.44 a | 54.91 a |
| Nitrogen Fertilizer | 56.82 a | 53.15 ab |
| Average | 53.99 | 51.22 |
| Treatment | Length of Pods (cm) |
|---|---|
| Not Inoculated | 7.48 a |
| Inoculated | 6.86 a |
| Coinoculated | 7.47 a |
| Nitrogen Fertilizer | 5.78 b |
| Average | 6.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nonato, J.J.; da Silva, T.J.A.; Schlichting, A.F.; Meneghetti, L.A.M.; de Oliveira, N.P.R.; Duarte, T.F.; Guimarães, S.L.; Koetz, M.; Campos e Silva, I.A.; da Silva, P.F.; et al. Water Availability Associated with Coinoculation with Growth-Promoting Rhizobacteria in Cowpea. Nitrogen 2025, 6, 74. https://doi.org/10.3390/nitrogen6030074
Nonato JJ, da Silva TJA, Schlichting AF, Meneghetti LAM, de Oliveira NPR, Duarte TF, Guimarães SL, Koetz M, Campos e Silva IA, da Silva PF, et al. Water Availability Associated with Coinoculation with Growth-Promoting Rhizobacteria in Cowpea. Nitrogen. 2025; 6(3):74. https://doi.org/10.3390/nitrogen6030074
Chicago/Turabian StyleNonato, Júlio José, Tonny José Araújo da Silva, Alessana Franciele Schlichting, Luana Aparecida Menegaz Meneghetti, Niclene Ponce Rodrigues de Oliveira, Thiago Franco Duarte, Salomão Lima Guimarães, Marcio Koetz, Ivis Andrei Campos e Silva, Patrícia Ferreira da Silva, and et al. 2025. "Water Availability Associated with Coinoculation with Growth-Promoting Rhizobacteria in Cowpea" Nitrogen 6, no. 3: 74. https://doi.org/10.3390/nitrogen6030074
APA StyleNonato, J. J., da Silva, T. J. A., Schlichting, A. F., Meneghetti, L. A. M., de Oliveira, N. P. R., Duarte, T. F., Guimarães, S. L., Koetz, M., Campos e Silva, I. A., da Silva, P. F., Pacheco, A. B., & Bonfim-Silva, E. M. (2025). Water Availability Associated with Coinoculation with Growth-Promoting Rhizobacteria in Cowpea. Nitrogen, 6(3), 74. https://doi.org/10.3390/nitrogen6030074







