SafeHANDS: A Multimodal Hand Hygiene Intervention in a Resource-Limited Neonatal Unit
Abstract
1. Introduction
2. Methods
2.1. Study Design, Population, and Setting
2.2. Hand Hygiene Practices in the Neonatal Unit Prior to the SafeHANDS Intervention
2.3. The SafeHANDS Multimodal Intervention
2.4. Education and Training on Hand Hygiene during the SafeHANDS Intervention
2.5. Data Sources and Outcomes of Interest
- HH compliance rates (by ward and for the neonatal unit overall): for each of the 5 neonatal wards, including NICU, a minimum of 150 direct HH observations were conducted in each study phase by 3 discrete trained observers using the WHO HH observation tool [31] converted to a RedCAP form for mobile devices [32]. HH compliance rates were reported as percentages for each ward and for the neonatal unit overall;
- ABHR usage (by ward and for the neonatal unit overall): the volume of ABHR used by each neonatal ward during each study phase was obtained from the hospital pharmacy dispensing records. To account for ward size and bed occupancy, the volume of ABHR used was divided by the patient days per ward per study phase × 1000. ABHR usage (for each ward and the neonatal unit overall) was reported as the total volume used in litres, as millilitres used per patient day, and as estimated HH actions/patient day (assuming an average of 3 mL ABHR used per opportunity);
- The WHO HH self-assessment framework (HHSAF) score (for the neonatal unit overall) [33,34]: this self-administered validated questionnaire was performed at the study baseline (July 2020) and again following completion of the established study phase (October 2021) to systematically evaluate HH structures, resources, promotion, and practices at the facility. The HHSAF includes 27 indicators in 5 sections, corresponding to the core components of the WHO multimodal HH improvement strategy (system change, training and education, observation and feedback, reminders in the workplace, and hospital safety climate). Question responses were converted to numerical scores per component, producing an overall score sub-categorised into 4 levels of HH practice (inadequate, basic, intermediate, and advanced) [33];
- Healthcare-associated bloodstream infection (HA-BSI) rate (for the neonatal unit overall): a laboratory-confirmed HA-BSI episode was defined as a blood culture collected > 72 h after unit admission with the isolation of a known pathogen. Organisms were classified using the United States Centers for Disease Control (CDC) list of pathogens and contaminants [35]. Repeat blood cultures isolating the same pathogen within 14 days of the original specimen were considered to represent a single episode of infection. Patients who isolated coagulase-negative staphylococci (CoNS) from two separate blood cultures taken 24–48 h apart were included as pathogens. All other contaminants were excluded. The HA-BSI rate was calculated for each study phase by dividing the total HA-BSI episodes by the total patient days for the neonatal unit in that 5-month period × 1000.
2.6. Statistical Analysis
3. Results
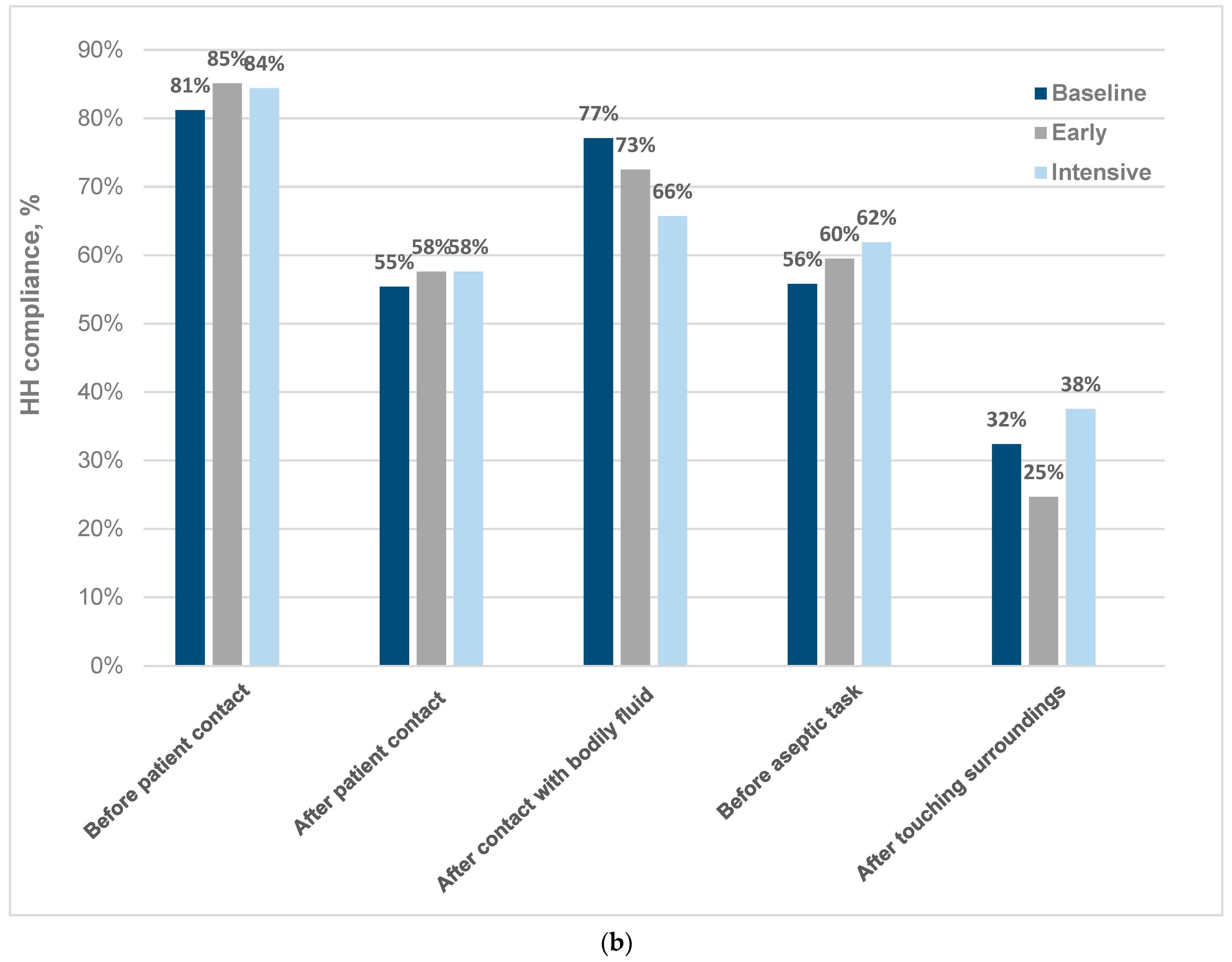
3.1. Baseline Observations
3.2. Changes after the SafeHANDS Intervention
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABHR | ABHR |
| HH | Hand hygiene |
| HHSAF | HH self-assessment framework |
| IPC | Infection Prevention and Control |
| LMIC | Low- and Middle-Income Countries |
| WHO | World Health Organization |
References
- Badia, J.M.; Casey, A.L.; Petrosillo, N.; Hudson, P.M.; Mitchell, S.A.; Crosby, C. Impact of surgical site infection on healthcare costs and patient outcomes: A systematic review in six European countries. J. Hosp. Infect. 2017, 96, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Public Health Agency of Canada. Canadian Antimicrobial Resistance Surveillance System Report 2016; Public Health Agency of Canada: Ottawa, ON, Canada, 2009; Available online: https://www.canada.ca/en/public-health/services/publications/drugs-health-products/canadian-antimicrobial-resistance-surveillance-system-report-2016.html (accessed on 21 August 2022).
- Zaidi, A.K.M.; Huskins, W.C.; Thaver, D.; Bhutta, Z.A.; Abbas, Z.; Goldmann, D.A. Hospital-acquired neonatal infections in developing countries. Lancet 2005, 365, 1175–1188. [Google Scholar] [CrossRef] [PubMed]
- Dramowski, A.; Velaphi, S.; Reubenson, G.; Bekker, A.; Perovic, O.; Finlayson, H.; Duse, A.; Rhoda, N.R.; Govender, N.P. National Neonatal Sepsis Task Force launch: Supporting infection prevention and surveillance, outbreak investigation and antimicrobial stewardship in neonatal units in South Africa. S. Afr. Med. J. 2020, 110, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Gould, D.J.; Moralejo, D.; Drey, N.; Chudleigh, J.H.; Taljaard, M. Interventions to improve hand hygiene compliance in patient care. Cochrane Database Syst. Rev. 2017, 9, 1–94. [Google Scholar] [CrossRef]
- Peters, A.; Masson-Roy, S.; Pittet, D. Healthcare-associated sepsis and the role of clean hands: When we do not see the trees for the forest. Int. J. Infect. Dis. 2018, 70, 101–103. [Google Scholar] [CrossRef]
- Pittet, D. The Lowbury lecture: Behaviour in infection control. J. Hosp. Infect. 2004, 58, 1–13. [Google Scholar] [CrossRef]
- Santosaningsih, D.; Erikawati, D.; Santoso, S.; Noorhamdani, N.; Ratridewi, I.; Candradikusuma, D.; Chozin, I.N.; Huwae, T.E.C.J.; van der Donk, G.; van Boven, E.; et al. Intervening with healthcare workers’ hand hygiene compliance, knowledge, and perception in a limited-resource hospital in Indonesia: A randomized controlled trial study. Antimicrob. Resist. Infect. Control 2017, 6, 23. [Google Scholar] [CrossRef]
- Pittet, D.; Allegranzi, B.; Boyce, J.; World Health Organization World Alliance for Patient Safety First Global Patient Safety Challenge Core Group of Experts. The World Health Organization Guidelines on Hand Hygiene in Health Care and their consensus recommendations. Infect. Control Hosp. Epidemiol. 2009, 30, 611–622. [Google Scholar] [CrossRef]
- Pittet, D.; Hugonnet, S.; Harbarth, S.; Mourouga, P.; Sauvan, V.; Touveneau, S.; Perneger, T.V. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet 2000, 356, 1307–1312. [Google Scholar] [CrossRef]
- Erasmus, V.; Daha, T.J.; Brug, H.; Richardus, J.H.; Behrendt, M.D.; Vos, M.C.; Van Beeck, E.F. Systematic review of studies on compliance with hand hygiene guidelines in hospital care. Infect. Control Hosp. Epidemiol. 2010, 31, 283–294. [Google Scholar] [CrossRef]
- Amazian, K.; Abdelmoumène, T.; Sekkat, S.; Terzaki, S.; Njah, M.; Dhidah, L.; Caillat-Vallet, E.; Saadatian-Elahi, M.; Fabry, J.; Members of the NosoMed Network. Multicentre study on hand hygiene facilities and practice in the Mediterranean area: Results from the NosoMed Network. J. Hosp. Infect. 2006, 62, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Polin, R.A.; Denson, S.; Brady, M.T. Committee on Fetus and Newborn, Committee on Infectious Diseases. Strategies for prevention of health care-associated infections in the NICU. Pediatrics 2012, 129, e1085–e1093. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines on Hand Hygiene in Health Care: First Global Patient Safety Challenge: Clean Care Is Safer Care; World Health Organization: Geneva, Switzerland, 2009; Available online: https://www.who.int/gpsc/5may/tools/who_guidelines-handhygiene_summary.pdf (accessed on 2 January 2022).
- World Health Organization. Guide to Implementation: A Guide to the Implementation of the WHO Multimodal Hand Hygiene Improvement Strategy; World Health Organization: Geneva, Switzerland, 2009; Available online: https://www.who.int/gpsc/5may/Guide_to_Implementation.pdf (accessed on 2 January 2022).
- Pittet, D.; Allegranzi, B.; Storr, J. The WHO Clean Care is Safer Care programme: Field-testing to enhance sustainability and spread of hand hygiene improvements. J. Infect. Public Health 2008, 1, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Pfäfflin, F.; Tufa, T.B.; Getachew, M.; Nigussie, T.; Schönfeld, A.; Häussinger, D.; Feldt, T.; Schmidt, N. Implementation of the WHO multimodal Hand Hygiene Improvement Strategy in a University Hospital in Central Ethiopia. Antimicrob. Resist. Infect. Control 2017, 6, 3. [Google Scholar] [CrossRef]
- Farhoudi, F.; Sanaei Dashti, A.; Hoshangi Davani, M.; Ghalebi, N.; Sajadi, G.; Taghizadeh, R. Impact of WHO Hand Hygiene Improvement Program Implementation: A Quasi-Experimental Trial. Biomed. Res. Int. 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Lee, S.S.; Park, S.J.; Chung, M.J.; Lee, J.H.; Kang, H.J.; Lee, J.A.; Kim, Y.K. Improved Hand Hygiene Compliance is Associated with the Change of Perception toward Hand Hygiene among Medical Personnel. Infect. Chemother. 2014, 46, 165–171. [Google Scholar] [CrossRef]
- Mahfouz, A.A.; Al-Zaydani, I.A.; Abdelaziz, A.O.; El-Gamal, M.N.; Assiri, A.M. Changes in hand hygiene compliance after a multimodal intervention among health-care workers from intensive care units in Southwestern Saudi Arabia. J. Epidemiol. Glob. Health 2014, 4, 315–321. [Google Scholar] [CrossRef]
- Mestre, G.; Berbel, C.; Tortajada, P.; Alarcia, M.; Coca, R.; Gallemi, G.; Garcia, I.; Fernández, M.M.; Aguilar, M.C.; Martínez, J.A. “The 3/3 strategy”: A successful multifaceted hospital wide hand hygiene intervention based on WHO and continuous quality improvement methodology. PLoS ONE 2012, 7, 1–12. [Google Scholar] [CrossRef]
- Monistrol, O.; Calbo, E.; Riera, M.; Nicolás, C.; Font, R.; Freixas, N.; Garau, J. Impact. of a hand hygiene educational programme on hospital-acquired infections in medical wards. Clin. Microbiol. Infect. 2012, 18, 1212–1218. [Google Scholar] [CrossRef]
- Allegranzi, B.; Sax, H.; Bengaly, L.; Richet, H.; Minta, D.K.; Chraiti, M.N.; Sokona, F.M.; Gayet-Ageron, A.; Bonnabry, P.; Pittet, D.; et al. Successful implementation of the World Health Organization hand hygiene improvement strategy in a referral hospital in Mali, Africa. Infect. Control Hosp. Epidemiol. 2010, 31, 133–141. [Google Scholar] [CrossRef]
- Schweizer, M.L.; Reisinger, H.S.; Ohl, M.; Formanek, M.B.; Blevins, A.; Ward, M.A.; Perencevich, E.N. Searching for an optimal hand hygiene bundle: A meta-analysis. Clin. Infect. Dis. 2014, 58, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Luangasanatip, N.; Hongsuwan, M.; Limmathurotsakul, D.; Lubell, Y.; Lee, A.S.; Harbarth, S.; Day, N.P.J.; Graves, N.; Cooper, B. Comparative efficacy of interventions to promote hand hygiene in hospital: Systematic review and network meta-analysis. BMJ 2015, 351, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Aiello, A.E.; Cimiotti, J.; Della-Latta, P.; Larson, E.L. A comparison of the bacteria found on the hands of “homemakers” and neonatal intensive care unit nurses. J. Hosp. Infect. 2003, 54, 310–315. [Google Scholar] [CrossRef]
- Biswal, M.; Angrup, A.; Rajpoot, S.; Kaur, R.; Kaur, K.; Kaur, H.; Dhaliwal, N.; Arora, P.; Gupta, A. Hand hygiene compliance of patients’ family members in India: Importance of educating the unofficial “fourth category” of healthcare personnel. J. Hosp. Infect. 2020, 104, 425–429. [Google Scholar] [CrossRef]
- WHO. Hand Hygiene Self-Assessment Framework; World Health Organization: Geneva, Switzerland, 2010; Available online: https://www.who.int/gpsc/5may/tools (accessed on 12 June 2022).
- Dramowski, A.; Aucamp, M.; Bekker, A.; Mehtar, S. Infectious disease exposures and outbreaks at a South African neonatal unit with review of neonatal outbreak epidemiology in Africa. Int. J. Infect. Dis. 2017, 57, 79–85. [Google Scholar] [CrossRef]
- Jassat, W.; Abdool Karim, S.; Mudara, C.; Welch, R.; Ozougwu, L.; Groome, M.; Govender, N.; von Gottberg, A.; Wolter, N.; Wolmarans, M.; et al. Clinical severity of COVID-19 in patients admitted to hospital during the omicron wave in South Africa: A retrospective observational study. Lancet Glob. Health 2022, 10, e961–e969. [Google Scholar] [CrossRef] [PubMed]
- WHO. Hand Hygiene Technical Reference Manual. World Health Organization: Geneva, Switzerland, 2009; Available online: https://cdn.who.int/media/docs/default-source/integrated-health-services-(ihs)/hand-hygiene/monitoring/surveyform/observationform.doc?sfvrsn=39b780c9_6 (accessed on 15 August 2022).
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Tartari, E.; Muthukumaran, P.; Peters, A.; Allegranzi, B.; Pittet, D. Monitoring Your institution: The WHO Hand Hygiene self-assessment Framework—Is it worth it? Clin. Microbiol. Infect. 2019, 25, 925–928. [Google Scholar] [CrossRef] [PubMed]
- Stewardson, A.J.; Allegranzi, B.; Perneger, T.V.; Attar, H.; Pittet, D. Testing the WHO Hand Hygiene Self-Assessment Framework for usability and reliability. J. Hosp. Infect. 2013, 83, 30–35. [Google Scholar] [CrossRef]
- Centres for Disease Control and Prevention. CDC List of Common Skin Contaminants/Common Commensals. 2011. Available online: http://www.cdc.gov/nhsn/XLS/Common-SkinContaminant-List-June-2011.xlsx (accessed on 15 August 2022).
- Lambe, K.A.; Lydon, S.; Madden, C.; Vellinga, A.; Hehir, A.; Walsh, M.; O’Connor, P. Hand Hygiene Compliance in the ICU: A Systematic Review. Crit. Care Med. 2019, 47, 1251–1257. [Google Scholar] [CrossRef]
- Lam, B.C.C.; Lee, J.; Lau, Y.L. Hand hygiene practices in a neonatal intensive care unit: A multimodal intervention and impact on nosocomial infection. Pediatrics 2004, 114, e565–e571. [Google Scholar] [CrossRef] [PubMed]
- Won, S.P.; Chou, H.C.; Hsieh, W.S.; Chen, C.Y.; Huang, S.M.; Tsou, K.I.; Tsao, P.-N. Handwashing program for the prevention of nosocomial infections in a neonatal intensive care unit. Infect. Control Hosp. Epidemiol. 2004, 25, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.; Sethi, A.; Kaur, A.; Kaur, G.; Vardhan Gupta, H.; Kaur, S.; Parihar, M.S.; Singh, S.M.P. Quality Improvement Initiative to Improve Hand Hygiene Compliance in Indian Special Newborn Care Unit. Pediatr. Qual. Saf. 2021, 6, e492. [Google Scholar] [CrossRef] [PubMed]
- Kallam, B.; Pettitt-Schieber, C.; Owen, M.; Agyare Asante, R.; Darko, E.; Ramaswamy, R. Implementation science in low-resource settings: Using the interactive systems framework to improve hand hygiene in a tertiary hospital in Ghana. Int. J. Qual. Health Care 2018, 30, 724–730. [Google Scholar] [CrossRef]
- Sadeghi-Moghaddam, P.; Arjmandnia, M.; Shokrollahi, M.; Aghaali, M. Does training improve compliance with hand hygiene and decrease infections in the neonatal intensive care unit? A prospective study. J. Neonatal Perinatal Med. 2015, 8, 221–225. [Google Scholar] [CrossRef]
- Chhapola, V.; Brar, R. Impact of an educational intervention on hand hygiene compliance and infection rate in a developing country neonatal intensive care unit. Int. J. Nurs. Pract. 2015, 21, 486–492. [Google Scholar] [CrossRef]
- White, K.M.; Jimmieson, N.L.; Obst, P.L.; Graves, N.; Barnett, A.; Cockshaw, W.; Gee, P.; Haneman, L.; Page, K.; Campbell, M.; et al. Using a theory of planned behaviour framework to explore hand hygiene beliefs at the “5 critical moments” among Australian hospital-based nurses. BMC Health Serv. Res. 2015, 15, 59. [Google Scholar] [CrossRef]
- Allegranzi, B.; Stewardson, A.; Pittet, D. Compliance with hand hygiene best practices. In Hand Hygiene: A Handbook for Medical Professionals; Pittet, D., Boyce, J.M., Allegranzi, B., Eds.; Wiley: Hoboken, NJ, USA, 2017; pp. 76–84. [Google Scholar]
- Pittet, D.; Mourouga, P.; Perneger, T.V. Compliance with handwashing in a teaching hospital. Infection Control Program. Ann. Intern. Med. 1999, 130, 126–130. [Google Scholar] [CrossRef]
- Cohen, B.; Saiman, L.; Cimiotti, J.; Larson, E. Factors associated with hand hygiene practices in two neonatal intensive care units. Pediatr. Infect. Dis. J. 2003, 22, 494–499. [Google Scholar] [CrossRef]
- Kouni, S.; Kourlaba, G.; Mougkou, K.; Maroudi, S.; Chavela, B.; Nteli, C.; Lourida, A.; Spyridis, N.; Zaoutis, T.; Coffin, S. Assessment of hand hygiene resources and practices at the 2 children’s hospitals in Greece. Pediatr. Infect. Dis. J. 2014, 33, e247–e251. [Google Scholar] [CrossRef]
- Pittet, D. Improving adherence to hand hygiene practice: A multidisciplinary approach. Emerg. Infect. Dis. 2001, 7, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Olivier, C.; Kunneke, H.; O’Connell, N.; von Delft, E.; Wates, M.; Dramowski, A. Healthcare-associated infections in paediatric and neonatal wards: A point prevalence survey at four South African hospitals. SAMJ 2018, 108, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, S.; Chaurasia, S.; Sankar, M.J.; Paul, V.K.; Deorari, A.K.; Joshi, M.; Agarwal, R. Stepwise interventions for improving hand hygiene compliance in a level 3 academic neonatal intensive care unit in north India. J Perinatol. 2021, 41, 2834–2839. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Bhattacharya, S.D.; Singh, A.K.; Saha, M. Addressing Hand Hygiene Compliance in a Low-Resource Neonatal Intensive Care Unit: A Quality Improvement Project. J. Pediatr. Infect. Dis. Soc. 2019, 8, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Wetzker, W.; Bunte-Schönberger, K.; Walter, J.; Pilarski, G.; Gastmeier, P.; Reichardt, C. Compliance with hand hygiene: Reference data from the national hand hygiene campaign in Germany. J. Hosp. Infect. 2016, 92, 328–331. [Google Scholar] [CrossRef] [PubMed]
- de Kraker, M.E.A.; Tartari, E.; Tomczyk, S.; Twyman, A.; Francioli, L.C.; Cassini, A.; Allegranzi, B.; Pittet, D. Implementation of hand hygiene in health-care facilities: Results from the WHO Hand Hygiene Self-Assessment Framework global survey 2019. Lancet Infect. Dis. 2022, 22, 835–844. [Google Scholar] [CrossRef]
- Allegranzi, B.; Stewardson, A.; Pittet, D. Monitoring your institution (hand hygiene self-assessment framework). In Hand Hygiene: A Handbook for Medical Professionals; Pittet, D., Boyce, J.M., Allegranzi, B., Eds.; Wiley: Hoboken, NJ, USA, 2017; pp. 244–248. [Google Scholar]
- Loftus, M.J.; Guitart, C.; Tartari, E.; Stewardson, A.J.; Amer, F.; Bellissimo-Rodrigues, F.; Lee, Y.F.; Mehtar, S.; Sithole, B.L.; Pittet, D. Hand hygiene in low- and middle-income countries. Int. J. Infect. Dis. 2019, 86, 25–30. [Google Scholar] [CrossRef]
- Dramowski, A.; Aucamp, M.; Beales, E.; Bekker, A.; Cotton, M.F.; Fitzgerald, F.C.; Labi, A.-K.; Russell, N.; Strysko, J.; Whitelaw, A.; et al. Healthcare-Associated Infection Prevention Interventions for Neonates in Resource-Limited Settings. Front. Pediatr. 2022, 10, 919403. [Google Scholar] [CrossRef]

| Phase Ward | Baseline Phase HH Compliance N (%) | Early Phase HH Compliance N (%) | Intensive Phase HH Compliance N (%) | % Change in HH Compliance * | p-Value | HH Target Level Achieved # |
|---|---|---|---|---|---|---|
| Overall unit | 617/1002 (61.6%) | 369/630 (58.6%) | 497/798 (62.3%) | +0.7% | 0.797 | silver |
| Neonatal intensive care unit | 128/182 (70.3%) | 67/106 (63.2%) | 157/227 (69.2%) | −1.1% | 0.883 | silver |
| Ward 1 | 118/212 (55.7%) | 66/127 (52%) | 105/155 (67.7%) | +12.0% | 0.025 | silver |
| Ward 2 | 142/206 (68.9%) | 87/143 (60.8%) | 81/134 (60.4%) | −8.5% | 0.135 | silver |
| Ward 3 | 116/201 (57.7%) | 108/178 (60.7%) | 94/144 (65.3%) | +7.6% | 0.190 | silver |
| Kangaroo Mother care ward | 113/201 (56.2%) | 41/76 (53.9%) | 60/136 (44.1%) | −12.1% | 0.038 | bronze |
| Intervention Phase | Baseline | Early | Intensive | Change in Volume of ABHR Used # (Percentage) |
|---|---|---|---|---|
| Overall neonatal unit ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 1203.5 70 23 | 1111 61 20.3 | 1337.5 73 24 | +11% |
| Ward 1 ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 301 67 22 | 276 61 20 | 301 67 22 | 0% |
| Ward 2 ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 283.5 63 21 | 261 68 19 | 376 84 28 | +33% |
| Ward 3 ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 281 62 21 | 341 76 25 | 396 88 29 | +42% |
| Kangaroo Mother care ward ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 188 42 14 | 128 28 9 | 118 26 9 | −38% |
| Neonatal ICU ABHR volume used (litres) ABHR used (mL/patient day) Estimated HH actions/patient day * | 150 83 27 | 105 58 19 | 146.5 81 27 | −2.4% |
| Component | Pre-Implementation (November 2020) | Post-Implementation (October 2021) |
|---|---|---|
| 1. System change | 75/100 | 75/100 |
| 2. Educational and training | 15/100 | 40/100 |
| 3. Evaluation and feedback | 40/100 | 45/100 |
| 4. Reminders in the workplace | 20/100 | 52.5/100 |
| 5. Institutional Safety Climate | 15/100 | 50/100 |
| Total score | 165 (Basic level) | 262.5 (Intermediate level) |
| Metric | Baseline | Early | Intensive |
|---|---|---|---|
| Neonatal unit bed occupancy per study phase | 85.6% | 90.5% | 91.0% |
| Neonatal unit HA-BSI episodes | 49 | 66 | 56 |
| Neonatal unit patient days per study phase | 17,239 | 18,218 | 18,314 |
| Neonatal HA-BSI rate per 1000 patient days | 2.9 | 3.6 | 3.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dramowski, A.; Erasmus, L.M.; Aucamp, M.; Fataar, A.; Cotton, M.F.; Coffin, S.E.; Bekker, A.; Whitelaw, A.C. SafeHANDS: A Multimodal Hand Hygiene Intervention in a Resource-Limited Neonatal Unit. Trop. Med. Infect. Dis. 2023, 8, 27. https://doi.org/10.3390/tropicalmed8010027
Dramowski A, Erasmus LM, Aucamp M, Fataar A, Cotton MF, Coffin SE, Bekker A, Whitelaw AC. SafeHANDS: A Multimodal Hand Hygiene Intervention in a Resource-Limited Neonatal Unit. Tropical Medicine and Infectious Disease. 2023; 8(1):27. https://doi.org/10.3390/tropicalmed8010027
Chicago/Turabian StyleDramowski, Angela, Louisa M. Erasmus, Marina Aucamp, Aaqilah Fataar, Mark F. Cotton, Susan E. Coffin, Adrie Bekker, and Andrew C. Whitelaw. 2023. "SafeHANDS: A Multimodal Hand Hygiene Intervention in a Resource-Limited Neonatal Unit" Tropical Medicine and Infectious Disease 8, no. 1: 27. https://doi.org/10.3390/tropicalmed8010027
APA StyleDramowski, A., Erasmus, L. M., Aucamp, M., Fataar, A., Cotton, M. F., Coffin, S. E., Bekker, A., & Whitelaw, A. C. (2023). SafeHANDS: A Multimodal Hand Hygiene Intervention in a Resource-Limited Neonatal Unit. Tropical Medicine and Infectious Disease, 8(1), 27. https://doi.org/10.3390/tropicalmed8010027







