The Rise of Imported Dengue Infections in Victoria, Australia, 2010–2016
Abstract
1. Introduction
2. Methods
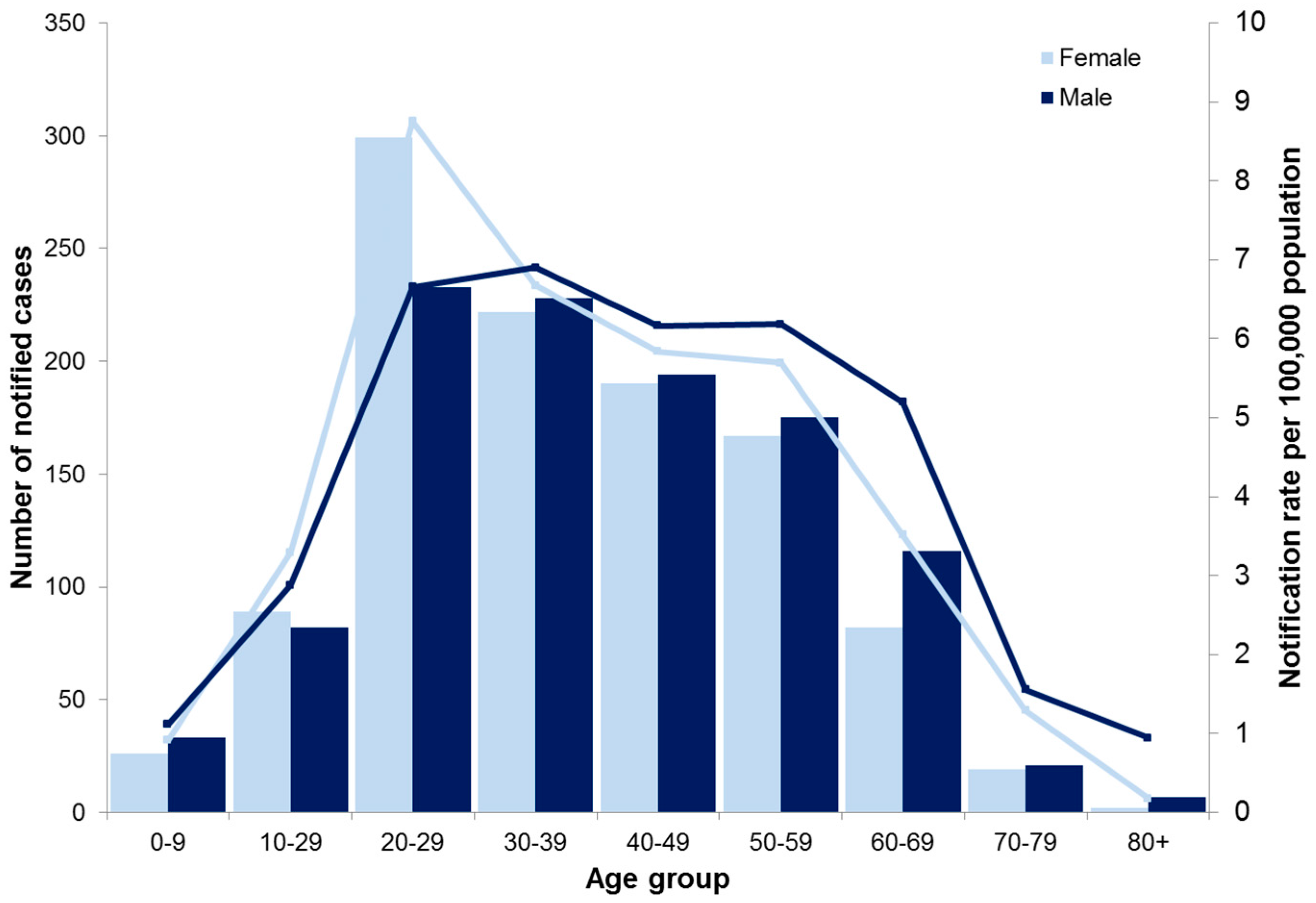
3. Results
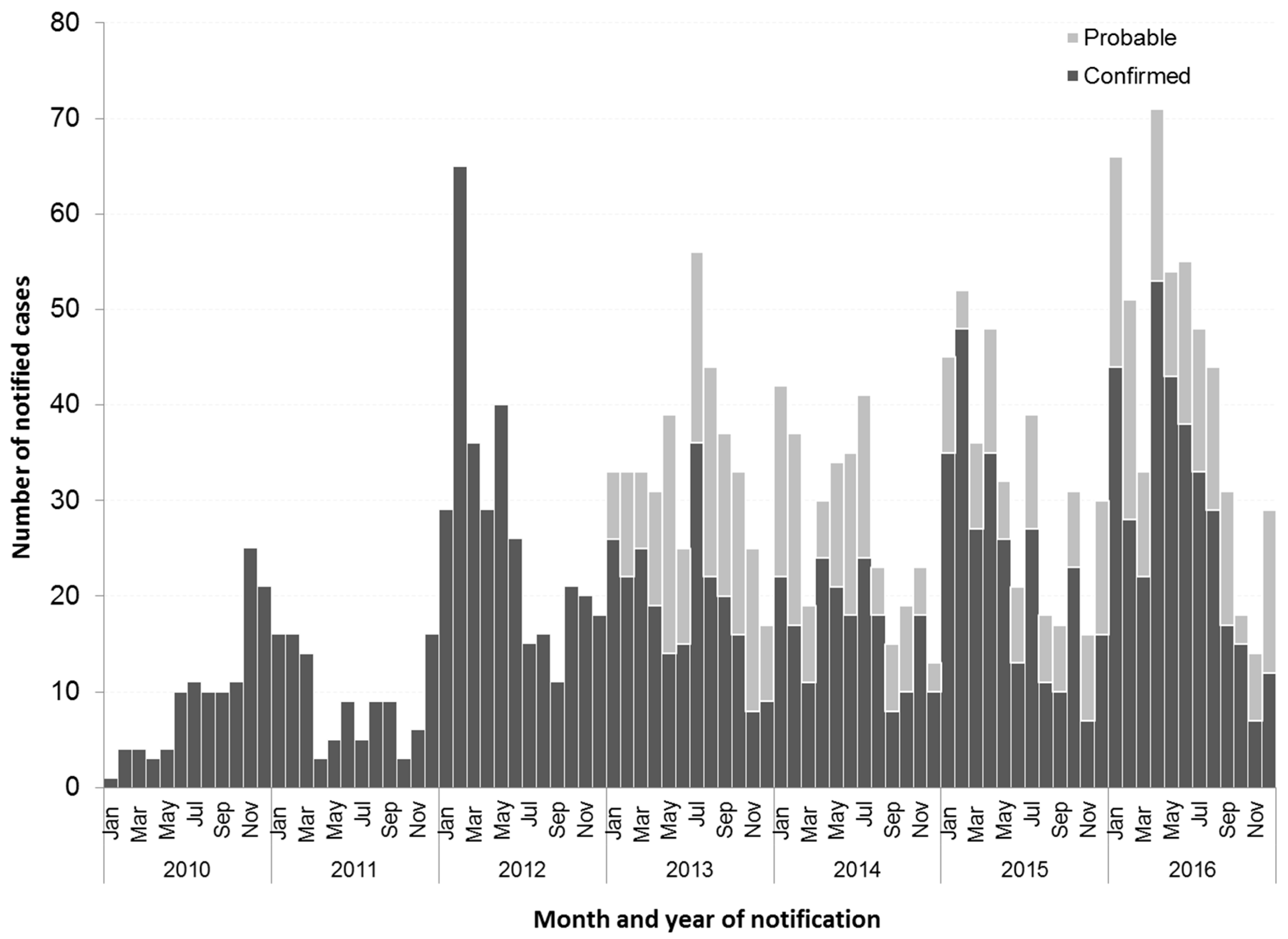
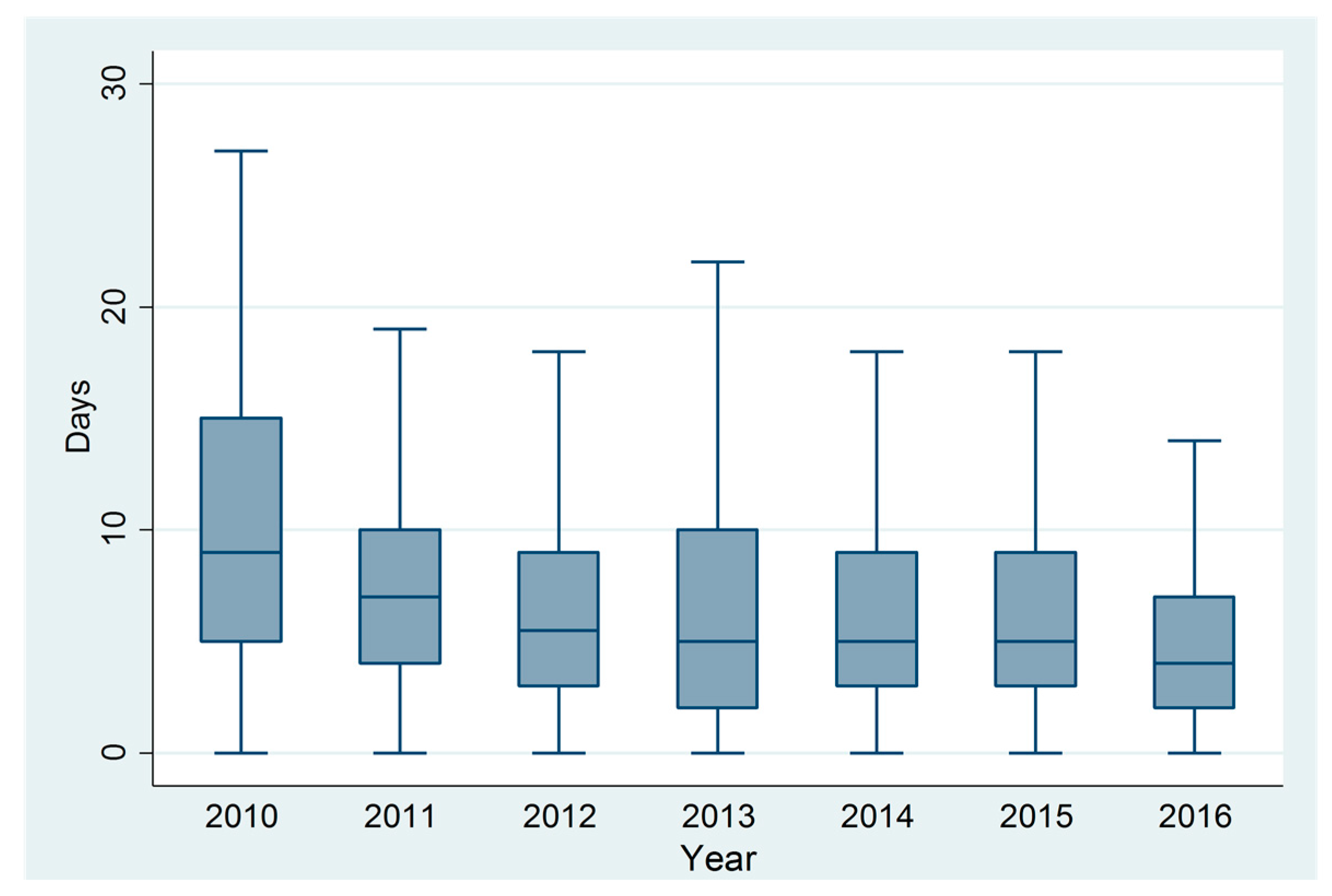
3.1. Increase in Dengue Notifications
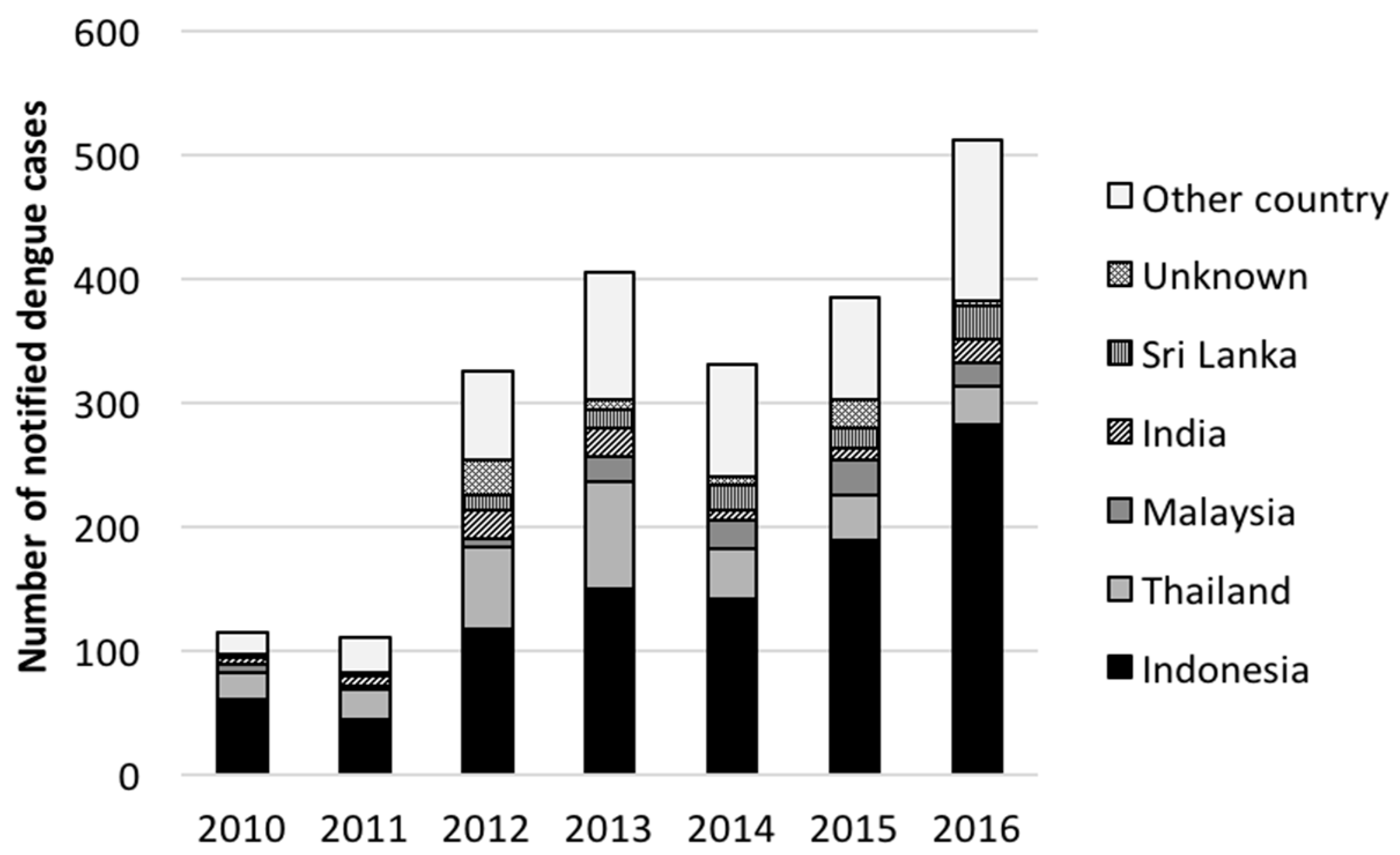
3.2. Country of Acquisition
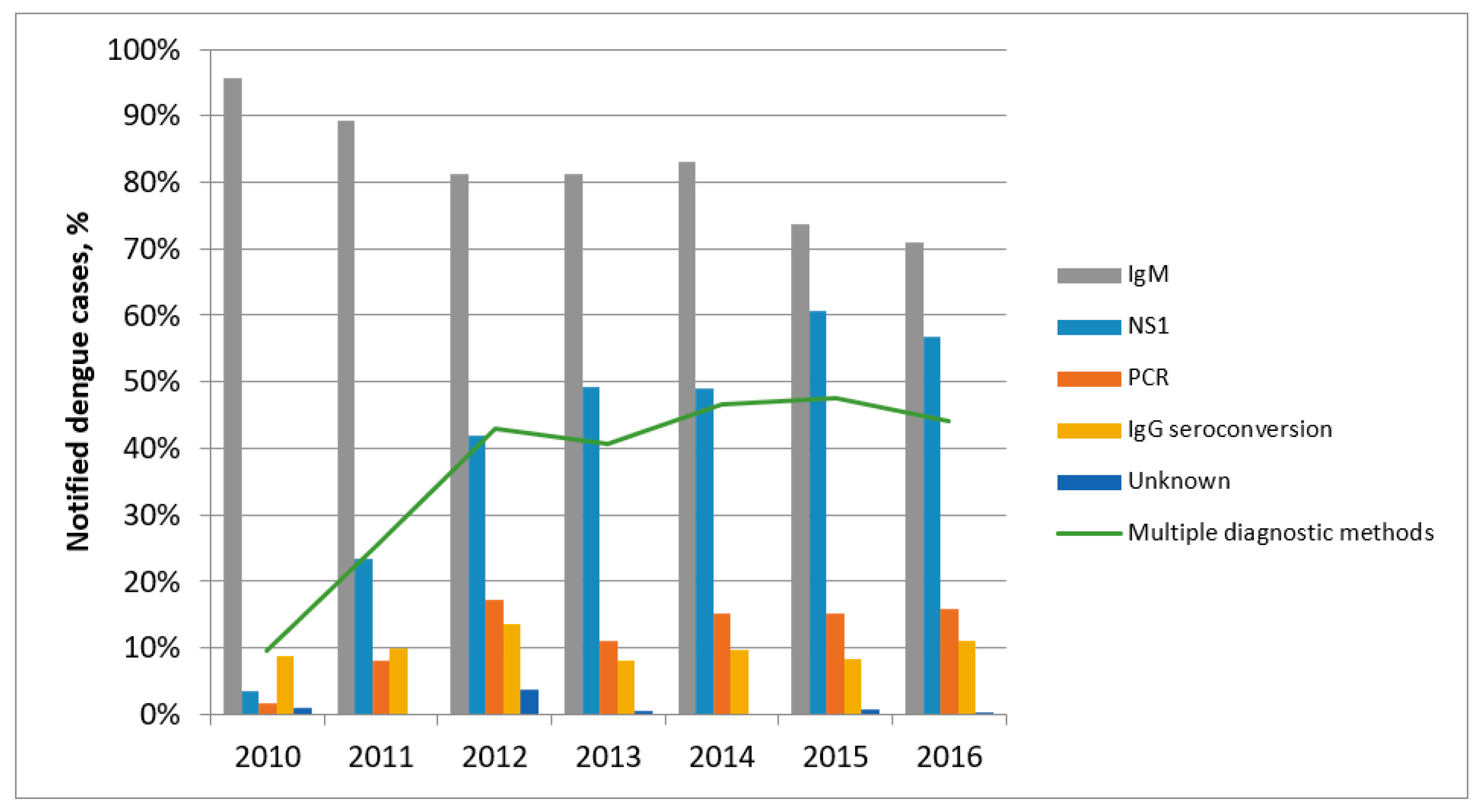
3.3. Diagnostic Tests
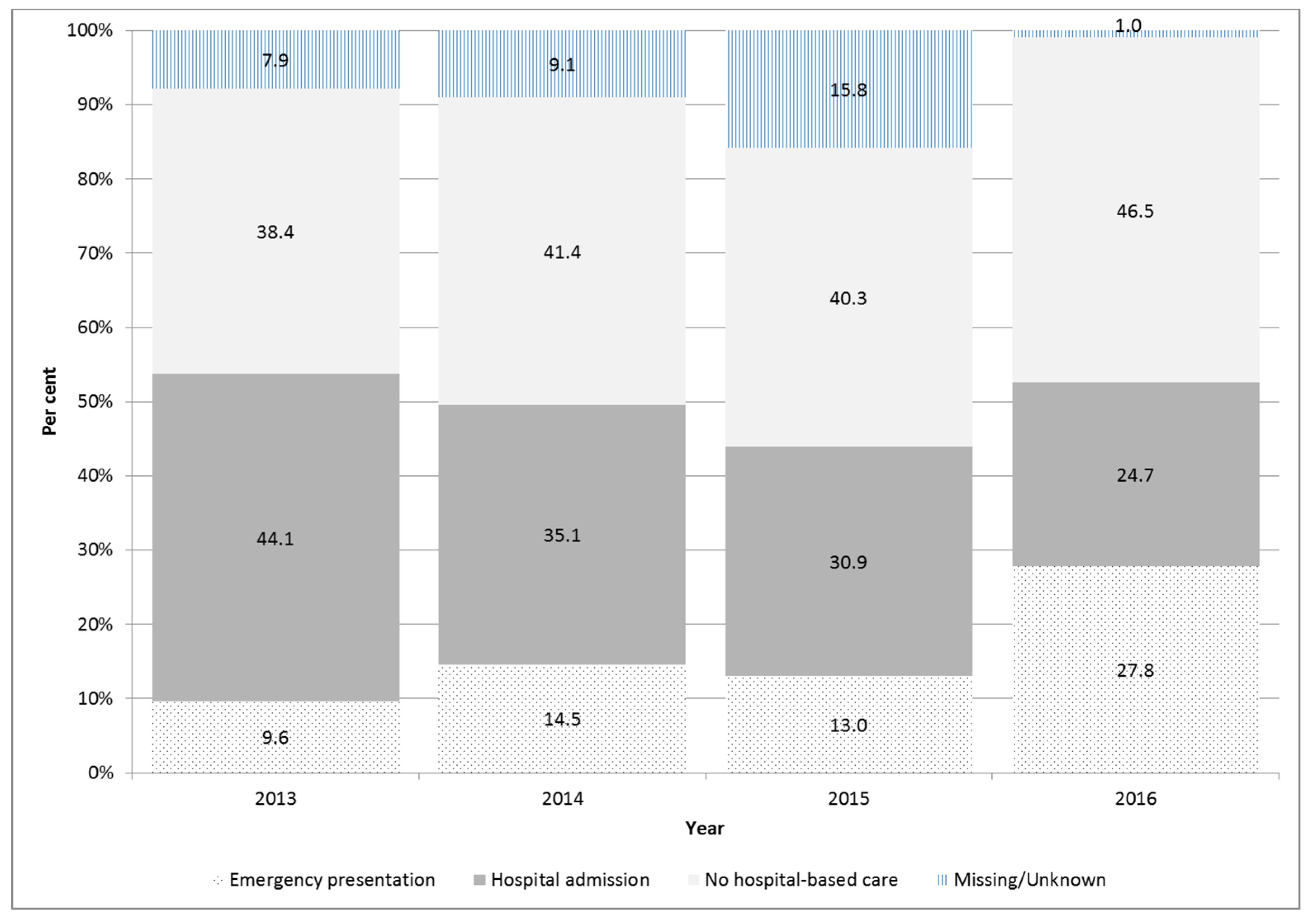
3.4. Clinical Presentation
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Ratnam, I.; Black, J.; Leder, K.; Biggs, B.A.; Matchett, E.; Padiglione, A.; Woolley, I.; Panagiotidis, T.; Gherardin, T.; Pollissard, L.; et al. Incidence and seroprevalence of dengue virus infections in Australian travellers to Asia. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Jelinek, T.; Muhlberger, N.; Harms, G.; Corachan, M.; Grobusch, M.P.; Knobloch, J.; Bronner, U.; Laferl, H.; Kapaun, A.; Bisoffi, Z.; et al. Epidemiology and clinical features of imported dengue fever in Europe: Sentinel surveillance data from TropNetEurop. Clin. Infect. Dis. 2002, 35, 1047–1052. [Google Scholar] [CrossRef] [PubMed]
- Gubler, D.J. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 1998, 11, 480–496. [Google Scholar] [PubMed]
- Ratnam, I.; Leder, K.; Black, J.; Torresi, J. Dengue fever and international travel. J. Travel Med. 2013, 20, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Guzman, M.G.; Kouri, G. Dengue: An update. Lancet Infect. Dis. 2002, 2, 33–42. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Tambyah, P.A. Severe dengue virus infection in travelers. J. Infect. Dis. 2007, 195, 1081–1083. [Google Scholar] [CrossRef] [PubMed]
- Jelinek, T. Dengue fever in international travelers. Clin. Infect. Dis. 2000, 31, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Schaffner, F.; Fontenille, D.; Mathis, A. Autochthonous dengue emphasises the threat of arbovirosis in Europe. Lancet Infect. Dis. 2014, 14, 1044. [Google Scholar] [CrossRef]
- Freedman, D.O.; Weld, L.H.; Kozarsky, P.E.; Fisk, T.; Robins, R.; von Sonnenburg, F.; Keystone, J.S.; Pandey, P.; Cetron, M.S.; GeoSentinel Surveillance Network. Spectrum of disease and relation to place of exposure among ill returned travelers. N. Engl. J. Med. 2006, 354, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Knope, K.; National, A.; Malaria Advisory, C.; Giele, C. Increasing notifications of dengue in Australia related to overseas travel, 1991 to 2012. Commun. Dis. Intell. Q. Rep. 2013, 37, E55–E59. [Google Scholar] [PubMed]
- Australian Bureau of Statistics. Australian Demographic Statistics Cat No. 3235.0. Population by Age and Sex, Regions of Australia. 2014. Available online: http://www.abs.gov.au/AUSSTATS/abs@nsf/DetailsPage/3235.02016?OpenDocument (accessed on 1 January 2018).
- Chief Health Officer Advisory. Mosquito-Borne Disease in Returned Travellers-Alert for Health Professionals-26 February 2013. Available online: https://www2.health.vic.gov.au/about/publications/researchandreports/Mosquito-borne-disease-in-returned-travellers---alert-for-health-professionals---26-February-2013 (accessed on 1 January 2018).
- Tai, A.Y.; McGuinness, S.L.; Robosa, R.; Turner, D.; Huang, G.K.; Leder, K.; Korman, T.M.; Thevarajan, I.; Stewardson, A.J.; Padiglione, A.A.; et al. Management of dengue in Australian travellers: A retrospective multicentre analysis. Med. J. Aust. 2017, 206, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Kutsuna, S.; Kato, Y.; Moi, M.L.; Kotaki, A.; Ota, M.; Shinohara, K.; Kobayashi, T.; Yamamoto, K.; Fujiya, Y.; Mawatari, M.; et al. Autochthonous dengue fever, Tokyo, Japan, 2014. Emerg. Infect. Dis. 2015, 21, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Succo, T.; Leparc-Goffart, I.; Ferre, J.B.; Roiz, D.; Broche, B.; Maquart, M.; Noel, H.; Catelinois, O.; Entezam, F.; Caire, D.; et al. Autochthonous dengue outbreak in Nimes, South of France, July to September 2015. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Nicholson, J.; Beebe, N.W.; Davis, J.; Muzari, O.M.; Russell, R.C.; Devine, G.J.; Ritchie, S.A. Ten years of the tiger: Aedes albopictus presence in Australia since its discovery in the Torres Strait in 2005. One Health 2016, 2, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Saez-Llorens, X.; Tricou, V.; Yu, D.; Rivera, L.; Tuboi, S.; Garbes, P.; Borkowski, A.; Wallace, D. Safety and immunogenicity of one versus two doses of Takeda's tetravalent dengue vaccine in children in Asia and Latin America: Interim results from a phase 2, randomised, placebo-controlled study. Lancet Infect. Dis. 2017, 17, 615–625. [Google Scholar] [CrossRef]
- Capeding, M.R.; Tran, N.H.; Hadinegoro, S.R.; Ismail, H.I.; Chotpitayasunondh, T.; Chua, M.N.; Luong, C.Q.; Rusmil, K.; Wirawan, D.N.; Nallusamy, R.; et al. Clinical efficacy and safety of a novel tetravalent dengue vaccine in healthy children in Asia: A phase 3, randomised, observer-masked, placebo-controlled trial. Lancet 2014, 384, 1358–1365. [Google Scholar] [CrossRef]
Sample Availability: The data analysed were collected under the auspices of the Public Health and Wellbeing Regulations (2008). Requests for data may be made to the Department of Health and Human Services: https://www2.health.vic.gov.au/public-health/infectious-diseases/infectious-diseases-surveillance/infectious-diseases-surveillance-in-victoria. |
| Country of Acquisition | IRR * | [95% Confidence Interval] |
|---|---|---|
| All | 1.22 | [1.19–1.25] |
| Indonesia | 1.30 | [1.26–1.34] |
| Thailand | 1.02 | [0.97–1.08] |
| Other (excl. Indonesia or Thailand) | 1.25 | [1.21–1.30] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rowe, S.L.; Thevarajan, I.; Richards, J.; Gibney, K.; Simmons, C.P. The Rise of Imported Dengue Infections in Victoria, Australia, 2010–2016. Trop. Med. Infect. Dis. 2018, 3, 9. https://doi.org/10.3390/tropicalmed3010009
Rowe SL, Thevarajan I, Richards J, Gibney K, Simmons CP. The Rise of Imported Dengue Infections in Victoria, Australia, 2010–2016. Tropical Medicine and Infectious Disease. 2018; 3(1):9. https://doi.org/10.3390/tropicalmed3010009
Chicago/Turabian StyleRowe, Stacey L., Irani Thevarajan, Jack Richards, Katherine Gibney, and Cameron P. Simmons. 2018. "The Rise of Imported Dengue Infections in Victoria, Australia, 2010–2016" Tropical Medicine and Infectious Disease 3, no. 1: 9. https://doi.org/10.3390/tropicalmed3010009
APA StyleRowe, S. L., Thevarajan, I., Richards, J., Gibney, K., & Simmons, C. P. (2018). The Rise of Imported Dengue Infections in Victoria, Australia, 2010–2016. Tropical Medicine and Infectious Disease, 3(1), 9. https://doi.org/10.3390/tropicalmed3010009




