Rationale and Design of the Orencia Atherosclerosis and Rheumatoid Arthritis Study (ORACLE Arthritis Study): Implications of Biologics against Rheumatoid Arthritis and the Vascular Complications, Subclinical Atherosclerosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Target
Patients and Inclusion/Exclusion Criteria
- (1)
- Target patients
- (2)
- Inclusion criteria:
- (i)
- Patients aged 20 years or older at the time of informed consent provision;
- (ii)
- Patients with RA that was poorly controlled with existing anti-rheumatic drugs; at the usual dosage for 3 months or more, in accordance with the criteria stipulated by the Japan College of Rheumatology. Safety criteria: white blood cell count (WBC) > 4000/mm3, lymphocytes > 1000/mm3, and testing negative for blood β-D-glucan;
- (iii)
- Patients who personally provided voluntary written consent prior to participation in the study, upon full understanding of the details of the study.
- (3)
- Exclusion criteria:
- i.
- Patients with active infections or malignancies;
- ii.
- Patients who had been treated with other biological products or molecular-targeted drug therapy;
- iii.
- Pregnant women, lactating women, and patients who desired to become pregnant;
- iv.
- Patients who did not provide consent to participate in the study.
- i.
- Patients with symptomatic heart failure (NYHA class 2 or higher or EF < 40%);
- ii.
- Patients with suspected type 1 diabetes mellitus;
- iii.
- Patients scheduled to undergo coronary revascularization (patients who had undergone percutaneous transluminal coronary angioplasty, including those who underwent drug-eluting stent placement);
- iv.
- Patients who underwent coronary artery bypass surgery;
- v.
- Patients with severe liver or renal disorder;
- vi.
- Patients with a history of allergy to abatacept or drug hypersensitivity;
- vii.
- Patients with arteriosclerosis obliterans (Fontaine class III or higher);
- viii.
- Patients who were considered ineligible by the investigators for reasons other than the above reasons;
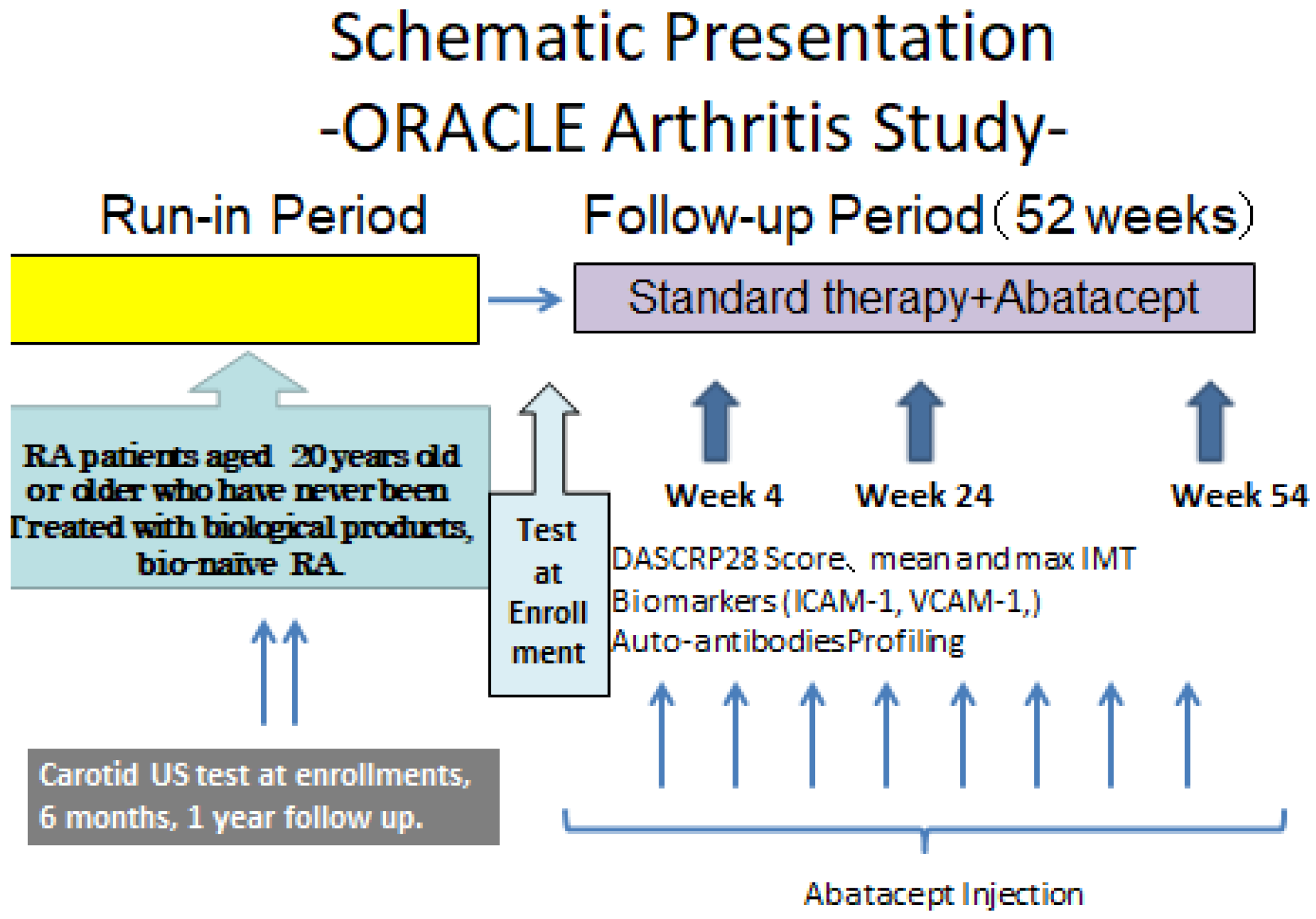
2.2. Study Type and Design
2.3. Outline of the Study
2.3.1. Treatment Regimen Using the Study Drug
2.3.2. Rules for Concomitant Medications (Therapies)
2.3.3. Rules for Dosage Reduction and Treatment Interruption
2.3.4. Patient Enrolment and Randomization Methods (Details Are Presented Below)
2.3.5. Observations and Tests/Data Collection Schedule
2.3.6. Endpoints
- (1)
- Primary endpoints
- (2)
- Secondary endpointsChanges in the following endpoints during treatment and from baseline to post-infusion:
- (i)
- Serum markers of atherosclerosis;
- (ii)
- Atherosclerosis structural markers (FMD and ABI);
- (iii)
- Serum autoantibody profile;
- (iv)
- Blood biochemistry and serological data associated with RA;
- (v)
- Changes in radiography findings based on the Sharp score;
- (vi)
- Joint ultrasonography findings;
- (vii)
- SDAI, CDAI, and Boolean remission rates, and clinical improvement based on the health assessment questionnaire–disability index;
- (viii)
- Arterial stiffness markers (AI or PWV).
2.3.7. Criteria for Study Participation Discontinuation in Individual Patients
- (1)
- Actions to be taken at study discontinuation
- (2)
- Criteria for discontinuation:
- (i)
- When a patient request to opt out of the study or withdraws consent;
- (ii)
- When serious adverse events occur, and treatment continuation is determined impossible;
- (iii)
- When the entire study is prematurely terminated;
- (iv)
- When the investigators consider it appropriate to discontinue the study for other reasons.
2.3.8. Target Sample Size, Rationale for Its Determination, and Statistical Analysis Methods
- (1)
- Target sample size (30 patients) and rationale for its determination
- (2)
- Statistical analysis methods
2.3.9. Consideration of Patients’ Rights and Method for the Protection of Personal Information
2.3.10. Obtaining Informed Consent
- (i)
- Participation in the study is voluntary; patients’ refusal to participate in the study is not associated with disadvantages, and the patient is allowed to withdraw from the study at any time;
- (ii)
- Significance (background), objectives, subjects, method, period, and planned sample size of the study;
- (iii)
- Anticipated benefits and foreseeable disadvantages of participation in the study;
- (iv)
- Handling, storage period, and method of sample disposal, including patients’ personal information, and study methods are available for inspection;
- (v)
- Handling in the cases of publication of study results and patent acquisition;
- (vi)
- Burden of study-related costs by the patients, sources of study funds, and conflict of interest;
- (vii)
- Study organization structure and consultation office (contact information) for inquiries on the study and for lodging complaints, and
- (viii)
- Actions to be taken and the availability of compensation in the event of health injuries to patients.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kaplan, M.J. Cardiovascular complications of rheumatoid arthritis: Assessment, prevention, and treatment. Rheum. Dis. Clin. 2010, 36, 405–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szekanecz, Z.; Kerekes, G.; Dér, H.; Sándor, Z.; Szabó, Z.; Végvári, A.; Simkovics, E.; Soós, L.; Szentpétery, A.; Besenyei, T.; et al. Accelerated atherosclerosis in rheumatoid arthritis. Ann. N. Y. Acad. Sci. 2007, 1108, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.J.; Symmons, D.P.; McCarey, D.; Dijkmans, B.A.; Nicola, P.; Kvien, T.K.; McInnes, I.B.; Haentzschel, H.; Gonzalez-Gay, M.A.; Provan, S.; et al. EULAR evidence-based recommendations for cardiovascular risk management in patients with rheumatoid arthritis and other forms of inflammatory arthritis. Ann. Rheum. Dis. 2010, 69, 325–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsuoka, K.; Komori, H.; Nose, M.; Endo, Y.; Sawasaki, T. Simple screening method for autoantigen proteins using the N-terminal biotinylated protein library produced by wheat cell-free synthesis. J. Proteome Res. 2010, 9, 4264–4273. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, T.; Abe, K.; Aoki, I.; Minegishi, S.; Ryo, A.; Matsunaga, S.; Matsuoka, K.; Takeda, H.; Sawasaki, T.; Umemura, S.; et al. Anti-interleukin-5 and multiple autoantibodies are associated with human atherosclerotic diseases and serum interleukin-5 levels. FASEB J. 2013, 27, 3437–3445. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Eng. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Nicola, P.J.; Maradit-Kremers, H.; Roger, V.L.; Jacobsen, S.J.; Crowson, C.S.; Ballman, K.V.; Gabriel, S.E. The risk of congestive heart failure in rheumatoid arthritis: A population-based study over 46 years. Arthritis Rheumatol. 2005, 52, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Widdifield, J.; Paterson, J.M.; Huang, A.; Bernatsky, S. Causes of death in rheumatoid arthritis: How do they compare to the general population? Arthritis Care Res. (Hoboken) 2018, 70, 1748–1755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crowson, C.S.; Therneau, T.M.; Davis, J.M., 3rd; Roger, V.L.; Matteson, E.L.; Gabriel, S.E. Brief report: Accelerated aging influences cardiovascular disease risk in rheumatoid arthritis. Arthritis Rheumatol. 2013, 65, 2562–2566. [Google Scholar] [CrossRef] [Green Version]
- Aubry, M.C.; Maradit-Kremers, H.; Reinalda, M.S.; Crowson, C.S.; Edwards, W.D.; Gabriel, S.E. Differences in atherosclerotic coronary heart disease between subjects with and without rheumatoid arthritis. J. Rheumatol. 2007, 34, 937–942. [Google Scholar] [PubMed]
- Albrecht, K.; Luque Ramos, A.; Hoffmann, F.; Redeker, I.; Zink, A. High prevalence of diabetes in patients with rheumatoid arthritis: Results from a questionnaire survey linked to claims data. Rheumatology (Oxford) 2018, 57, 329–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escalante, A.; Haas, R.W.; del Rincon, I. Paradoxical effect of body mass index on survival in rheumatoid arthritis: Role of comorbidity and systemic inflammation. Arch. Intern. Med. 2005, 165, 1624–1629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myasoedova, E.; Crowson, C.S.; Kremers, H.M.; Roger, V.L.; Fitz-Gibbon, P.D.; Therneau, T.M.; Gabriel, S.E. Lipid paradox in rheumatoid arthritis: The impact of serum lipid measures and systemic inflammation on the risk of cardiovascular disease. Ann. Rheum. Dis. 2011, 70, 482–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Solomon, D.H.; Reed, G.W.; Kremer, J.M.; Curtis, J.R.; Farkouh, M.E.; Harrold, L.R.; Hochberg, M.C.; Tsao, P.; Greenberg, J.D. Disease activity in rheumatoid arthritis and the risk of cardiovascular events. Arthritis Rheumatol. 2015, 67, 1449–1455. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Longo, F.J.; Oliver-Minarro, D.; de la Torre, I.; Gonzalez-Diaz de Rabago, E.; Sanchez-Ramon, S.; Rodriguez-Mahou, M.; Paravisini, A.; Monteagudo, I.; Gonzalez, C.M.; Garcia-Castro, M.; et al. Association between anti-cyclic citrullinated peptide antibodies and ischemic heart disease in patients with rheumatoid arthritis. Arthritis Rheumatol. 2009, 61, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Lv, S.; Liu, B.; Liu, Z.; Luo, Y.; Kong, W.; Xu, Q.; Feng, J.; Wang, X. CTLA4-IgG ameliorates homocysteine-accelerated atherosclerosis by inhibiting T-cell overactivation in apoE(-/-) mice. Cardiovasc. Res. 2013, 97, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Ewing, M.M.; Karper, J.C.; Abdul, S.; de Jong, R.C.; Peters, H.A.; de Vries, M.R.; Redeker, A.; Kuiper, J.; Toes, R.E.; Arens, R.; et al. T-cell co-stimulation by CD28-CD80/86 and its negative regulator CTLA-4 strongly influence accelerated atherosclerosis development. Int. J. Cardiol. 2013, 168, 1965–1974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roubille, C.; Richer, V.; Starnino, T.; McCourt, C.; McFarlane, A.; Fleming, P.; Siu, S.; Kraft, J.; Lynde, C.; Pope, J.; et al. The effects of tumour necrosis factor inhibitors, methotrexate, non-steroidal anti-inflammatory drugs and corticosteroids on cardiovascular events in rheumatoid arthritis, psoriasis and psoriatic arthritis: A systematic review and meta-analysis. Ann. Rheum. Dis. 2015, 74, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Kang, E.H.; Brill, G.; Desai, R.J.; Kim, S.C. Cardiovascular (CV) risk after initiation of abatacept versus TNF inhibi tors in rheumatoid arthritis patients with and without baseline CV disease. J. Rheumatol. 2018, 45, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
| Body Weight of Patient | Dose | Number of Vials |
|---|---|---|
| Less than 60 kg | 500 mg | 2 |
| 60 to 100 kg | 750 mg | 3 |
| More than 100 kg | 1 g | 4 |
| Follow-Up Period (52 ± 4 Weeks) | ||||||
|---|---|---|---|---|---|---|
| Time point | Baseline | 0 W | 4 W | 12 ± 2 W | 26 ± 4 W | 52 ± 4 W |
| Informed consent | ◯ | |||||
| Patient background characteristics/RA activity | ◯ | ◯ | ◯ | ◯ | ◯ | |
| Blood pressure and pulse rate | ◯ | ◯ | ◯ | ◯ | ◯ | ◯ |
| Blood biochemistry | ◯ | ◯ | ◯ | ◯ | ◯ | |
| Mean IMT | ◯ | ◯ | ◯ | |||
| Radiography examination (hands and feet)/joint ultrasonography findings | ◯ | ◯ | ◯ | ◯ | ||
| Biomarker autoantibody | ◯ | ◯ | ◯ | ◯ | ||
| Adverse events | ← ◯ → | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishigami, T.; Nanki, T.; Sugawara, T.; Uchida, K.; Takeda, H.; Sawasaki, T.; Chen, L.; Doi, H.; Arakawa, K.; Saigo, S.; et al. Rationale and Design of the Orencia Atherosclerosis and Rheumatoid Arthritis Study (ORACLE Arthritis Study): Implications of Biologics against Rheumatoid Arthritis and the Vascular Complications, Subclinical Atherosclerosis. Methods Protoc. 2021, 4, 83. https://doi.org/10.3390/mps4040083
Ishigami T, Nanki T, Sugawara T, Uchida K, Takeda H, Sawasaki T, Chen L, Doi H, Arakawa K, Saigo S, et al. Rationale and Design of the Orencia Atherosclerosis and Rheumatoid Arthritis Study (ORACLE Arthritis Study): Implications of Biologics against Rheumatoid Arthritis and the Vascular Complications, Subclinical Atherosclerosis. Methods and Protocols. 2021; 4(4):83. https://doi.org/10.3390/mps4040083
Chicago/Turabian StyleIshigami, Tomoaki, Toshihiro Nanki, Takuya Sugawara, Kotaro Uchida, Hiroyuki Takeda, Tatsuya Sawasaki, Lin Chen, Hiroshi Doi, Kentaro Arakawa, Sae Saigo, and et al. 2021. "Rationale and Design of the Orencia Atherosclerosis and Rheumatoid Arthritis Study (ORACLE Arthritis Study): Implications of Biologics against Rheumatoid Arthritis and the Vascular Complications, Subclinical Atherosclerosis" Methods and Protocols 4, no. 4: 83. https://doi.org/10.3390/mps4040083








