Genome-Wide Approaches to Drosophila Heart Development
Abstract
:1. Introduction
2. Genetic Screens for Mutants Affecting Heart Development
2.1. Screens with Deficiency Collections and EMS-Induced Mutations
2.2. Screens with RNA Interference (RNAi)
3. Analyses of the Transcriptome and Proteome of the Drosophila Heart
3.1. Whole Genome Expression Profiling during Remodeling of the Larval into the Adult Cardiac Tube
3.2. Determination of the Proteome of the Adult Fly Heart
3.3. Differential Profiling of Global Gene Expression in Mutant Versus Wild Type Heart
4. Genome-Wide Searches for Binding Sites and Target Genes of Cardiogenic Transcription Factors
5. Computational Analyses Utilizing Genomic Data in Drosophila Cardiogenesis
6. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| CC | Cardial Cell (aka, myocardial cell, cardioblast) |
| ChIP | Chromatin Immunoprecipitation |
| CM | Cardiogenic Mesoderm |
| CRISPR/Cas9 | Clustered Regularly Interspaced Short Palindromic Repeats |
| CRM | Cis-Regulatory Module |
| ECM | Extracellular Matrix |
| PC | Pericardial Cell |
| TF | Transcription Factor |
References
- Bodmer, R.; Frasch, M. Development and Aging of the Drosophila Heart. In Heart Development and Regeneration; Harvey, R.P., Rosenthal, N., Eds.; Heart Development and Regeneration: Academic Press Oxford, UK, 2010; Volume 1, pp. 47–86. [Google Scholar]
- Adams, M.D.; Celniker, S.E.; Holt, R.A.; Evans, C.A.; Gocayne, J.D.; Amanatides, P.G.; Scherer, S.E.; Li, P.W.; Hoskins, R.A.; Galle, R.F.; et al. The genome sequence of Drosophila melanogaster. Science 2000, 287, 2185–2195. [Google Scholar] [CrossRef] [PubMed]
- Nüsslein-Volhard, C.; Wieschaus, E. Mutations affecting segment number and polarity in Drosophila. Nature 1980, 287, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Cook, R.K.; Christensen, S.J.; Deal, J.A.; Coburn, R.A.; Deal, M.E.; Gresens, J.M.; Kaufman, T.C.; Cook, K.R. The generation of chromosomal deletions to provide extensive coverage and subdivision of the Drosophila melanogaster genome. Genome Biol. 2012, 13, R21. [Google Scholar] [CrossRef] [PubMed]
- Vissers, J.H.A.; Manning, S.A.; Kulkarni, A.; Harvey, K.F. A Drosophila RNAi library modulates Hippo pathway-dependent tissue growth. Nat. Commun. 2016, 7, 10368. [Google Scholar] [CrossRef] [PubMed]
- Caussinus, E.; Kanca, O.; Affolter, M. Fluorescent fusion protein knockout mediated by anti-GFP nanobody. Nat. Struct. Mol. Biol. 2012, 19, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Christiansen, A.E.; Schulz, R.A. Second chromosome genes required for heart development in Drosophila melanogaster. Genesis 2007, 45, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Yi, P.; Han, Z.; Li, X.; Olson, E.N. The mevalonate pathway controls heart formation in Drosophila by isoprenylation of Ggamma1. Science 2006, 313, 1301–1303. [Google Scholar] [CrossRef] [PubMed]
- Hollfelder, D.; Frasch, M.; Reim, I. Distinct functions of the laminin β LN domain and collagen IV during cardiac extracellular matrix formation and stabilization of alary muscle attachments revealed by EMS mutagenesis in Drosophila. BMC Dev. Biol. 2014, 14, 26. [Google Scholar] [CrossRef] [PubMed]
- Hollfelder, D. Implementation of an EMS-Screen to identify genes involved in muscle development in Drosophila melanogaster. Ph.D. Thesis, Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany, January 2014; pp. 1–164. [Google Scholar]
- Drechsler, M.; Schmidt, A.C.; Meyer, H.; Paululat, A. The conserved ADAMTS-like protein Lonely heart mediates matrix formation and cardiac tissue integrity. PLoS Genet. 2013, 9, e1003616. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-O.; Park, S.-J.; Balaban, R.S.; Nirenberg, M.; Kim, Y. A functional genomic screen for cardiogenic genes using RNA interference in developing Drosophila embryos. Proc. Natl. Acad. Sci. USA 2004, 101, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Neely, G.G.; Kuba, K.; Cammarato, A.; Isobe, K.; Amann, S.; Zhang, L.; Murata, M.; Elmén, L.; Gupta, V.; Arora, S.; et al. A global in vivo Drosophila RNAi screen identifies NOT3 as a conserved regulator of heart function. Cell 2010, 141, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Zeitouni, B.; Sénatore, S.; Séverac, D.; Aknin, C.; Sémériva, M.; Perrin, L. Signalling pathways involved in adult heart formation revealed by gene expression profiling in Drosophila. PLoS Genet. 2007, 3, 1907–1921. [Google Scholar] [CrossRef] [PubMed]
- Potier, D.; Seyres, D.; Guichard, C.; Iche-Torres, M.; Aerts, S.; Herrmann, C.; Perrin, L. Identification of cis-regulatory modules encoding temporal dynamics during development. BMC Genomics 2014, 15, 534. [Google Scholar] [CrossRef] [PubMed]
- Cammarato, A.; Ahrens, C.H.; Alayari, N.N.; Qeli, E.; Rucker, J.; Reedy, M.C.; Zmasek, C.M.; Gucek, M.; Cole, R.N.; van Eyk, J.E.; et al. A Mighty Small Heart: The Cardiac Proteome of Adult Drosophila melanogaster. PLoS ONE 2011, 6, e18497. [Google Scholar] [CrossRef] [PubMed]
- Hallier, B.; Hoffmann, J.; Roeder, T.; Tögel, M.; Meyer, H.; Paululat, A. The bHLH Transcription Factor Hand Regulates the Expression of Genes Critical to Heart and Muscle Function in Drosophila melanogaster. PLoS ONE 2015, 10, e0134204. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.M.; Tansey, T.R.; Busser, B.W.; Nolte, M.T.; Jeffries, N.; Gisselbrecht, S.S.; Rusan, N.M.; Michelson, A.M. Two forkhead transcription factors regulate the division of cardiac progenitor cells by a Polo-dependent pathway. Dev. Cell 2012, 23, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Busser, B.W.; Haimovich, J.; Huang, D.; Ovcharenko, I.; Michelson, A.M. Enhancer modeling uncovers transcriptional signatures of individual cardiac cell states in Drosophila. Nucleic Acids Res. 2015, 43, 1726–1739. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-H.; Jakobsen, J.S.; Valentin, G.; Amarantos, I.; Gilmour, D.T.; Furlong, E.E. A Systematic analysis of Tinman function reveals Eya and JAK-STAT signaling as essential regulators of muscle development. Dev. Cell 2009, 16, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Junion, G.; Spivakov, M.; Girardot, C.; Braun, M.; Gustafson, E.H.; Birney, E.; Furlong, E.E. A transcription factor collective defines cardiac cell fate and reflects lineage history. Cell 2012, 148, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Zinzen, R.P.; Girardot, C.; Gagneur, J.; Braun, M.; Furlong, E.E.M. Combinatorial binding predicts spatio-temporal cis-regulatory activity. Nature 2009, 462, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Stojnic, R.; Adryan, B.; Ozdemir, A.; Stathopoulos, A.; Frasch, M. Genome-wide screens for in vivo Tinman binding sites identify cardiac enhancers with diverse functional architectures. PLoS Genet. 2013, 9, e1003195. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.M.; Busser, B.W.; Huang, D.; Cozart, E.J.; Michaud, S.; Zhu, X.; Jeffries, N.; Aboukhalil, A.; Bulyk, M.L.; Ovcharenko, I.; Michelson, A.M. Machine learning classification of cell-specific cardiac enhancers uncovers developmental subnetworks regulating progenitor cell division and cell fate specification. Development 2014, 141, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Haack, T.; Schneider, M.; Schwendele, B.; Renault, A. Drosophila heart cell movement to the midline occurs through both cell autonomous migration and dorsal closure. Dev. Biol. 2014, 396, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Yi, P.; Johnson, A.N.; Han, Z.; Wu, J.; Olson, E.N. Heterotrimeric G proteins regulate a noncanonical function of septate junction proteins to maintain cardiac integrity in Drosophila. Dev. Cell. 2008, 15, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Reim, I.; Hollfelder, D.; Ismat, A.; Frasch, M. The FGF8-related signals Pyramus and Thisbe promote pathfinding, substrate adhesion, and survival of migrating longitudinal gut muscle founder cells. Dev. Biol. 2012, 368, 28–43. [Google Scholar] [CrossRef] [PubMed]
- Koundakjian, E.J.; Cowan, D.M.; Hardy, R.W.; Becker, A.H. The Zuker collection: a resource for the analysis of autosomal gene function in Drosophila melanogaster. Genetics 2004, 167, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Kon, C.; Cadigan, K.M.; da Silva, S.L.; Nusse, R. Developmental roles of the Mi-2/NURD-associated protein p66 in Drosophila. Genetics 2005, 169, 2087–2100. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Venkatesh, T.V.; Bodmer, R. Dual role for the zeste-white3/shaggy-encoded kinase in mesoderm and heart development of Drosophila. Dev. Genet. 1998, 22, 201–211. [Google Scholar] [CrossRef]
- Waldron, L.; Steimle, J.D.; Greco, T.M.; Gomez, N.C.; Dorr, K.M.; Kweon, J.; Temple, B.; Yang, X.H.; Wilczewski, C.M.; Davis, I.J.; et al. The cardiac TBX5 interactome reveals a chromatin remodeling network essential for cardiac septation. Dev. Cell 2016, 36, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Dietzl, G.; Chen, D.; Schnorrer, F.; Su, K.-C.; Barinova, Y.; Fellner, M.; Gasser, B.; Kinsey, K.; Oppel, S.; Scheiblauer, S.; et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 2007, 448, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Perkins, L.A.; Holderbaum, L.; Tao, R.; Hu, Y.; Sopko, R.; McCall, K.; Yang-Zhou, D.; Flockhart, I.; Binari, R.; Shim, H.-S.; et al. The transgenic RNAi project at Harvard Medical School: Resources and validation. Genetics 2015, 201, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Zaffran, S.; Reim, I.; Qian, L.; Lo, P.C.; Bodmer, R.; Frasch, M. Cardioblast-intrinsic Tinman activity controls proper diversification and differentiation of myocardial cells in Drosophila. Development 2006, 133, 4073–4083. [Google Scholar] [CrossRef] [PubMed]
- Collart, M.A. The Ccr4-Not complex is a key regulator of eukaryotic gene expression. Wiley Interdiscip. Rev. RNA 2016. [Google Scholar] [CrossRef] [PubMed]
- Schaub, C.; März, J.; Reim, I.; Frasch, M. Org-1-dependent lineage reprogramming generates the ventral longitudinal musculature of the Drosophila heart. Curr. Biol. 2015, 25, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Lehmacher, C.; Abeln, B.; Paululat, A. The ultrastructure of Drosophila heart cells. Arthropod Struct. Dev. 2012, 41, 459–474. [Google Scholar] [CrossRef] [PubMed]
- Bryantsev, A.L.; Cripps, R.M. Purification of cardiac cells from Drosophila embryos. Methods 2011, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Defaye, A.; Perrin, L. Tissue specific RNA isolation in Drosophila embryos: A strategy to analyze context dependent transcriptome landscapes using FACS. Methods Mol. Biol. 2014, 1196, 183–195. [Google Scholar] [PubMed]
- Gay, L.; Miller, M.R.; Ventura, P.B.; Devasthali, V.; Vue, Z.; Thompson, H.L.; Temple, S.; Zong, H.; Cleary, M.D.; Stankunas, K.; et al. Mouse TU tagging: A chemical/genetic intersectional method for purifying cell type-specific nascent RNA. Genes Dev. 2013, 27, 98–115. [Google Scholar] [CrossRef] [PubMed]
- Southall, T.D.; Gold, K.S.; Egger, B.; Davidson, C.M.; Caygill, E.E.; Marshall, O.J.; Brand, A.H. Cell-type-specific profiling of gene expression and chromatin binding without cell isolation: Assaying RNA Pol II occupancy in neural stem cells. Dev. Cell 2013, 26, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Lo, P.C.H.; Skeath, J.B.; Gajewski, K.; Schulz, R.A.; Frasch, M. Homeotic genes autonomously specify the anteroposterior subdivision of the Drosophila dorsal vessel into aorta and heart. Dev. Biol. 2002, 251, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Bousette, N.; Kislinger, T.; Fong, V.; Isserlin, R.; Hewel, J.A.; Emil, A.; Gramolini, A.O. Large-scale characterization and analysis of the murine cardiac proteome. J. Proteome Res. 2009, 8, 1887–1901. [Google Scholar] [CrossRef] [PubMed]
- Kölsch, V.; Paululat, A. The highly conserved cardiogenic bHLH factor Hand is specifically expressed in circular visceral muscle progenitor cells and in all cell types of the dorsal vessel during Drosophila embryogenesis. Dev. Genes Evol. 2002, 212, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Lo, P.C.H.; Zaffran, S.; Sénatore, S.; Frasch, M. The Drosophila Hand gene is required for remodeling of the developing adult heart and midgut during metamorphosis. Dev. Biol. 2007, 311, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Elhanany-Tamir, H.; Yu, Y.V.; Shnayder, M.; Jain, A.; Welte, M.; Volk, T. Organelle positioning in muscles requires cooperation between two KASH proteins and microtubules. J. Cell Biol. 2012, 198, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Nowak, S.J.; Aihara, H.; Gonzalez, K.; Nibu, Y.; Baylies, M.K. Akirin links twist-regulated transcription with the Brahma chromatin remodeling complex during embryogenesis. PLoS Genet. 2012, 8, e1002547. [Google Scholar] [CrossRef] [PubMed]
- Jani, K.; Schöck, F. Zasp is required for the assembly of functional integrin adhesion sites. J. Cell Biol. 2007, 179, 1583–1597. [Google Scholar] [CrossRef] [PubMed]
- Harpaz, N.; Ordan, E.; Ocorr, K.; Bodmer, R.; Volk, T. Multiplexin promotes heart but not aorta morphogenesis by polarized enhancement of Slit/Robo activity at the heart lumen. PLoS Genet. 2013, 9, e1003597. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.; Weiszmann, R.; Frise, E.; Hammonds, A.; Tomancak, P.; Beaton, A.; Berman, B.; Quan, E.; Shu, S.; Lewis, S.; et al. BDGP in situ Homepage. Available online: http://insitu.fruitfly.org/cgi-bin/ex/insitu.pl (accessed on 7 April, 2016).
- Gajewski, K.; Kim, Y.; Lee, Y.M.; Olson, E.N.; Schulz, R.A. D-mef2 is a target for Tinman activation during Drosophila heart development. EMBO J. 1997, 16, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Gajewski, K.; Zhang, Q.; Choi, C.Y.; Fossett, N.; Dang, A.; Kim, Y.H.; Kim, Y.; Schulz, R.A. pannier is a transcriptional target and partner of Tinman during Drosophila cardiogenesis. Dev. Biol. 2001, 233, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Olson, E.N. Hand is a direct target of Tinman and GATA factors during Drosophila cardiogenesis and hematopoiesis. Development 2005, 132, 3525–3536. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.M.; Hendren, J.D.; Helander, L.A.; Cripps, R.M. The NK homeodomain transcription factor Tinman is a direct activator of seven-up in the Drosophila dorsal vessel. Dev. Biol. 2007, 302, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Wang, J.; Tokusumi, T.; Gajewski, K.; Schulz, R.A. Requirement of the LIM homeodomain transcription factor Tailup for normal heart and hematopoietic organ formation in Drosophila melanogaster. Mol. Cell. Biol. 2007, 27, 3962–3969. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.-R.; Najand, N.; Brook, W.J. Tinman is a direct activator of midline in the Drosophila dorsal vessel. Dev. Dyn. 2011, 240, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Lovato, T.L.; Sensibaugh, C.A.; Swingle, K.L.; Martinez, M.M.; Cripps, R.M. The Drosophila transcription factors Tinman and Pannier activate and collaborate with Myocyte enhancer factor-2 to promote heart cell fate. PLoS ONE 2015, 10, e0132965. [Google Scholar] [CrossRef] [PubMed]
- Kremser, T.; Gajewski, K.; Schulz, R.A.; Renkawitz-Pohl, R. Tinman regulates the transcription of the beta3 tubulin gene (betaTub60D) in the dorsal vessel of Drosophila. Dev. Biol. 1999, 216, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tao, Y.; Reim, I.; Gajewski, K.; Frasch, M.; Schulz, R.A. Expression, regulation, and requirement of the Toll transmembrane protein during dorsal vessel formation in Drosophila melanogaster. Mol. Cell. Biol. 2005, 25, 4200–4210. [Google Scholar] [CrossRef] [PubMed]
- Akasaka, T.; Klinedinst, S.; Ocorr, K.; Bustamante, E.L.; Kim, S.K.; Bodmer, R. The ATP-sensitive potassium (KATP) channel-encoded dSUR gene is required for Drosophila heart function and is regulated by tinman. Proc. Natl. Acad. Sci. USA 2006, 103, 11999–12004. [Google Scholar] [CrossRef] [PubMed]
- Hendren, J.D.; Shah, A.P.; Arguelles, A.M.; Cripps, R.M. Cardiac expression of the Drosophila Sulphonylurea receptor gene is regulated by an intron enhancer dependent upon the NK homeodomain factor Tinman. Mech. Dev. 2007, 124, 416–426. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bonn, S.; Zinzen, R.P.; Girardot, C.; Gustafson, E.H.; Perez-Gonzalez, A.; Delhomme, N.; Ghavi-Helm, Y.; Wilczyński, B.; Riddell, A.; Furlong, E.E.M. Tissue-specific analysis of chromatin state identifies temporal signatures of enhancer activity during embryonic development. Nat. Genet. 2012, 44, 148–156. [Google Scholar] [CrossRef] [PubMed]
- McClure, C.D.; Southall, T.D. Getting down to specifics: Profiling gene expression and protein-DNA interactions in a cell type-specific manner. Adv. Genet. 2015, 91, 103–151. [Google Scholar] [PubMed]
- Schauer, T.; Schwalie, P.C.; Handley, A.; Margulies, C.E.; Flicek, P.; Ladurner, A.G. CAST-ChIP maps cell-type-specific chromatin states in the Drosophila central nervous system. Cell Rep. 2013, 5, 271–282. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nagarkar-Jaiswal, S.; Lee, P.-T.; Campbell, M.E.; Chen, K.; Anguiano-Zarate, S.; Gutierrez, M.C.; Busby, T.; Lin, W.-W.; He, Y.; Schulze, K.L.; et al. A library of MiMICs allows tagging of genes and reversible, spatial and temporal knockdown of proteins in Drosophila. Elife 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Sarov, M.; Barz, C.; Jambor, H.; Hein, M.Y.; Schmied, C.; Suchold, D.; Stender, B.; Janosch, S.; Vikas, V.K.J.; Krisnan, R.T.; et al. A genome-wide resource for the analysis of protein localisation in Drosophila. Elife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yin, Z.; Hudson, J.B.; Ferguson, E.L.; Frasch, M. Smad proteins act in combination with synergistic and antagonistic regulators to target Dpp responses to the Drosophila mesoderm. Genes Dev. 1998, 12, 2354–2370. [Google Scholar] [CrossRef] [PubMed]
- Halfon, M.S.; Carmena, A.; Gisselbrecht, S.; Sackerson, C.M.; Jiménez, F.; Baylies, M.K.; Michelson, A.M. Ras pathway specificity is determined by the integration of multiple signal-activated and tissue-restricted transcription factors. Cell 2000, 103, 63–74. [Google Scholar] [CrossRef]
- Reim, I.; Frasch, M. The Dorsocross T-box genes are key components of the regulatory network controlling early cardiogenesis in Drosophila. Development 2005, 132, 4911–4925. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-H.; Frasch, M. Nuclear integration of positive Dpp signals, antagonistic Wg inputs and mesodermal competence factors during Drosophila visceral mesoderm induction. Development 2005, 132, 1429–1442. [Google Scholar] [CrossRef] [PubMed]
- Zaffran, S.; Küchler, A.; Lee, H.H.; Frasch, M. biniou (FoxF), a central component in a regulatory network controlling visceral mesoderm development and midgut morphogenesis in Drosophila. Genes Dev. 2001, 15, 2900–2915. [Google Scholar] [PubMed]
- Jakobsen, J.S.; Braun, M.; Astorga, J.; Gustafson, E.H.; Sandmann, T.; Karzynski, M.; Carlsson, P.; Furlong, E.E.M. Temporal ChIP-on-chip reveals Biniou as a universal regulator of the visceral muscle transcriptional network. Genes Dev. 2007, 21, 2448–2460. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Pomares, J.M.; González-Rosa, J.M.; Muñoz-Chápuli, R. Building the vertebrate heart - an evolutionary approach to cardiac development. Int. J. Dev. Biol. 2009, 53, 1427–1443. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Y.; MacArthur, S.; Bourgon, R.; Nix, D.; Pollard, D.A.; Iyer, V.N.; Hechmer, A.; Simirenko, L.; Stapleton, M.; Luengo Hendriks, C.L.; et al. Transcription factors bind thousands of active and inactive regions in the Drosophila blastoderm. PLoS Biol. 2008, 6, e27. [Google Scholar]
- Fisher, W.W.; Li, J.J.; Hammonds, A.S.; Brown, J.B.; Pfeiffer, B.D.; Weiszmann, R.; MacArthur, S.; Thomas, S.; Stamatoyannopoulos, J.A.; Eisen, M.B.; et al. DNA regions bound at low occupancy by transcription factors do not drive patterned reporter gene expression in Drosophila. Proc. Natl. Acad. Sci. USA 2012, 109, 21330–21335. [Google Scholar] [CrossRef] [PubMed]
- Asadzadeh, J.; Neligan, N.; Canabal-Alvear, J.J.; Daly, A.C.; Kramer, S.G.; Labrador, J.-P. The Unc-5 receptor is directly regulated by Tinman in the developing Drosophila dorsal vessel. PLoS ONE 2015, 10, e0137688. [Google Scholar] [CrossRef] [PubMed]
- Remeseiro, S.; Hörnblad, A.; Spitz, F. Gene regulation during development in the light of topologically associating domains. Wiley Interdiscip. Rev. Dev. Biol. 2016, 5, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Narlikar, L.; Sakabe, N.J.; Blanski, A.A.; Arimura, F.E.; Westlund, J.M.; Nobrega, M.A.; Ovcharenko, I. Genome-wide discovery of human heart enhancers. Genome Res. 2010. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, B.; Reim, I.; Friedrich-Alexander-University of Erlangen-Nürnberg, Erlangen, Germany. Personal communication, 2016.
- Grigorian, M.; Mandal, L.; Hakimi, M.; Ortiz, I.; Hartenstein, V. The convergence of Notch and MAPK signaling specifies the blood progenitor fate in the Drosophila mesoderm. Dev. Biol. 2011, 353, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Chartier, A.; Zaffran, S.; Astier, M.; Sémériva, M.; Gratecos, D. Pericardin, a Drosophila type IV collagen-like protein is involved in the morphogenesis and maintenance of the heart epithelium during dorsal ectoderm closure. Development 2002, 129, 3241–3253. [Google Scholar] [PubMed]
- Gratz, S.J.; Harrison, M.M.; Wildonger, J.; O'Connor-Giles, K.M. Precise Genome Editing of Drosophila with CRISPR RNA-Guided Cas9. Methods Mol. Biol. 2015, 1311, 335–348. [Google Scholar] [PubMed]
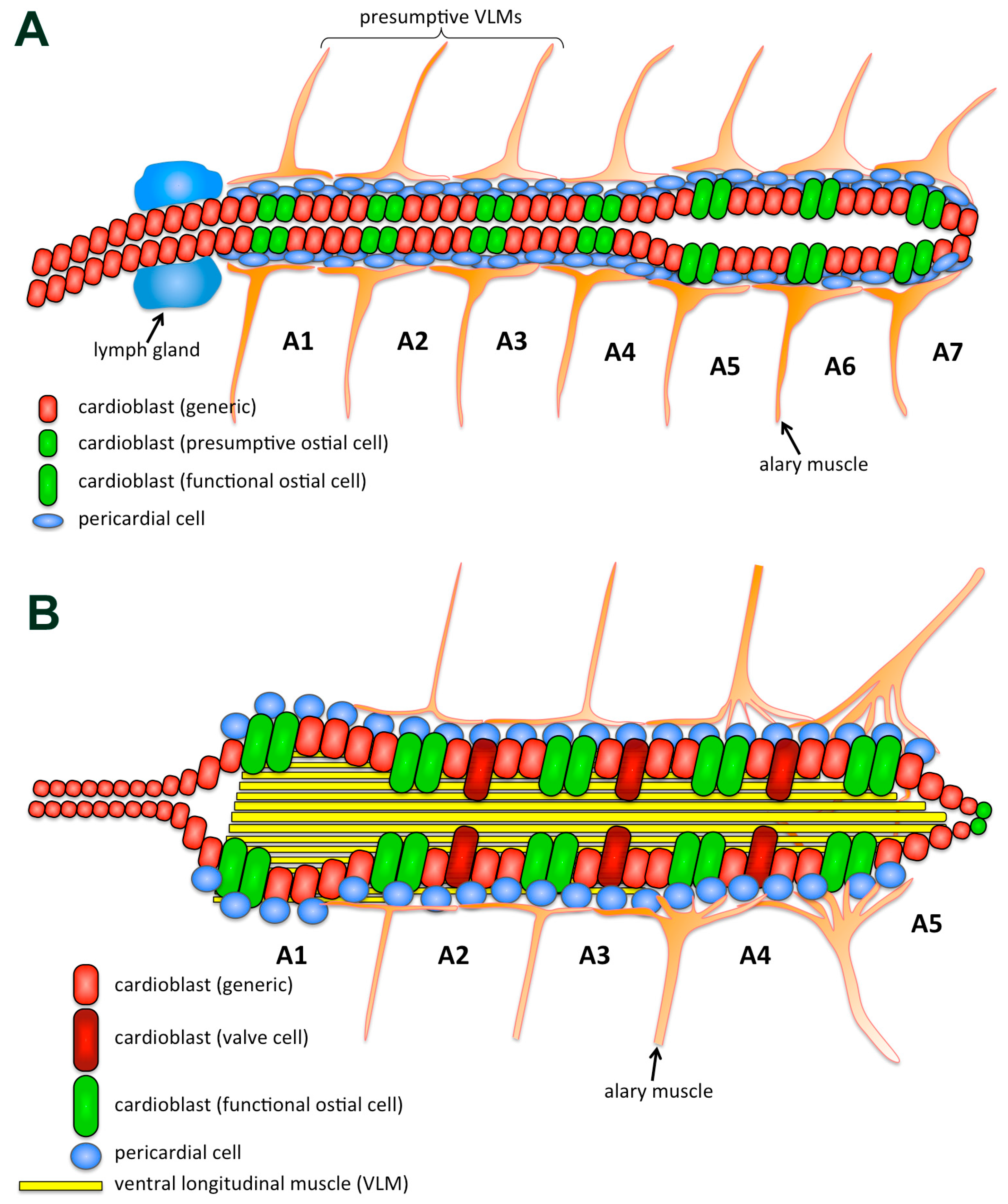
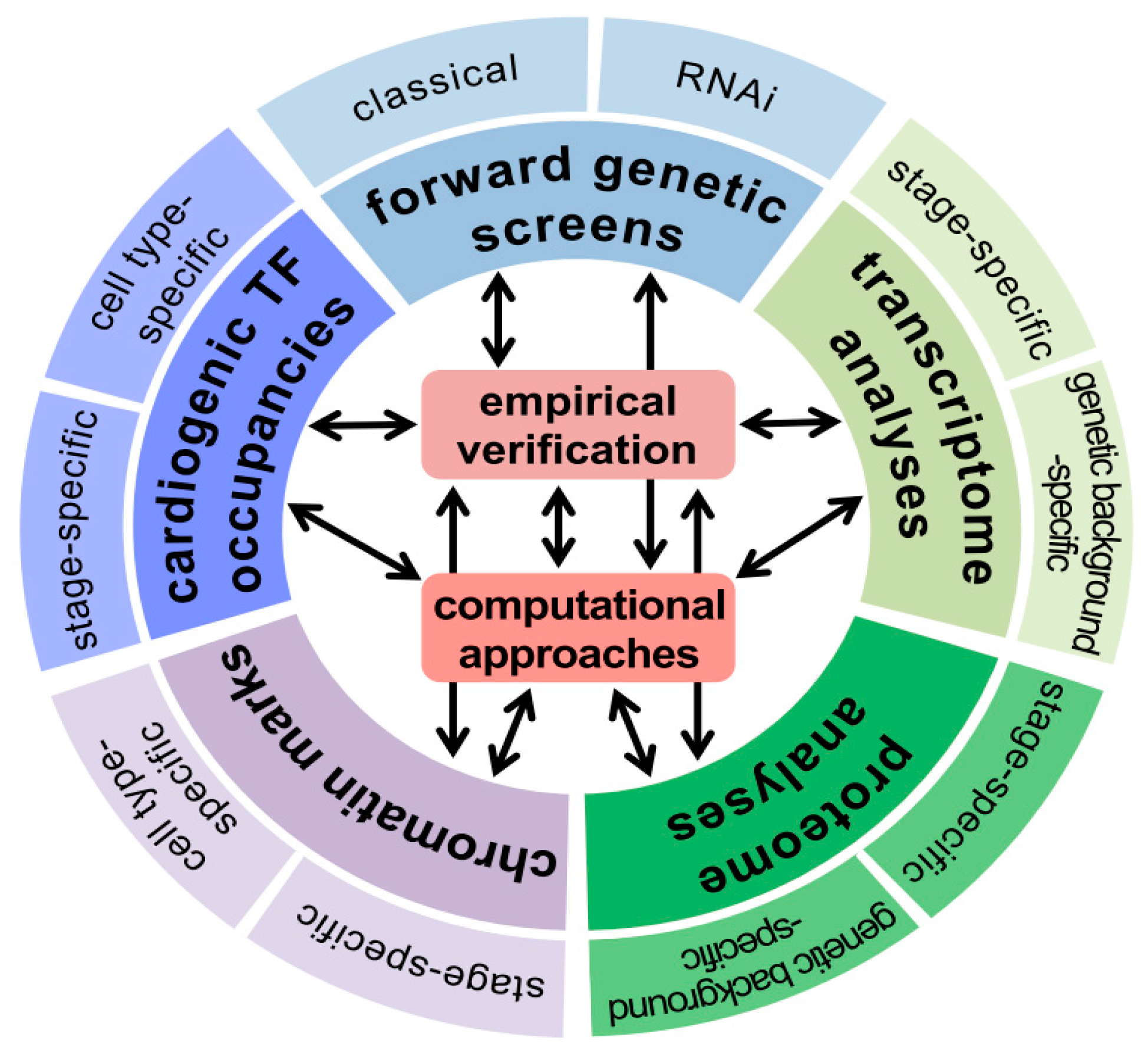
| Mutational Screens | Type | Heart Markers Used | Chromosome |
| Tao et al., 2007 [7] | deficiencies | tup-GFP or Hand-GFP | 2nd |
| Yi et al., 2006 [8] | avail. lethals | Hand-GFP | various |
| Hollfelder et al., 2014 [9,10] | EMS | tin-GFP + org-1-RFP | 2nd |
| Drechsler et al., 2013 [11] | EMS (Zuker) | Hand-GFP | 2nd, 3rd |
| RNAi Screens | Type | Tissue | |
| Kim et al., 2004 [12] | injections | embryonic heart (Mef2-lacZ-marked) | |
| Neely et al., 2010 [13] | GAL4-induced | developing adult heart | |
| Transcriptome, Proteome Screens | Type | Tissue | Genotypes |
| Zeitouni et al., 2007 [14]; Potier et al., 2014 [15] | transcriptome | developing adult heart | WT |
| Cammarato et al., 2011 [16] | proteome | adult heart | WT |
| Hallier et al., 2015 [17] | transcriptome | larval heart 3rd instar | Hand/WT |
| Ahmad et al., 2012 [18]; Busser et al., 2015 [19] | transcriptome | embryo, dorsal mesoderm | various heart mutants/WT |
| Chromatin Immunoprecipitation Screens | Type | Transcription factors | |
| Liu et al., 2009 [20] | ChIP-chip | Tin | |
| Junion et al., 2012 [21]; Zinzen et al., 2009 [22] | ChIP-chip | Tin, Doc, Pnr, Mad, dTCF | |
| Jin et al., 2013 [23] | ChIP-chip | Tin | |
| Computational Screens | Type | Aims | |
| Ahmad et al., 2014 [24]; Busser et al., 2015 [19] | machine learning | cardiac enhancer identification | |
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frasch, M. Genome-Wide Approaches to Drosophila Heart Development. J. Cardiovasc. Dev. Dis. 2016, 3, 20. https://doi.org/10.3390/jcdd3020020
Frasch M. Genome-Wide Approaches to Drosophila Heart Development. Journal of Cardiovascular Development and Disease. 2016; 3(2):20. https://doi.org/10.3390/jcdd3020020
Chicago/Turabian StyleFrasch, Manfred. 2016. "Genome-Wide Approaches to Drosophila Heart Development" Journal of Cardiovascular Development and Disease 3, no. 2: 20. https://doi.org/10.3390/jcdd3020020
APA StyleFrasch, M. (2016). Genome-Wide Approaches to Drosophila Heart Development. Journal of Cardiovascular Development and Disease, 3(2), 20. https://doi.org/10.3390/jcdd3020020






