ECG Smart Monitoring versus Implantable Loop Recorders for Atrial Fibrillation Detection after Cryptogenic Stroke—An Overview for Decision Making
Abstract
1. Introduction
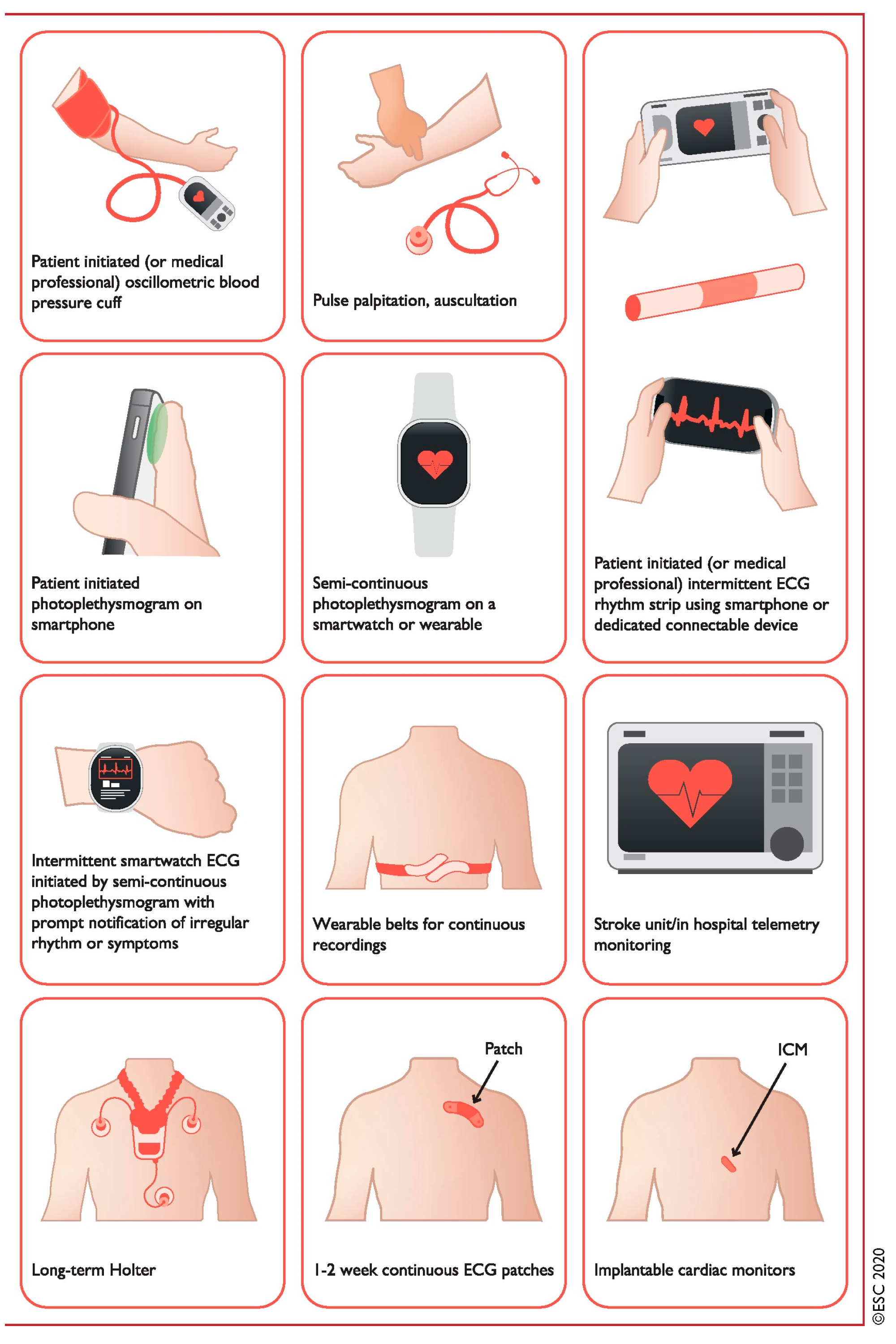
2. Technologies
2.1. Handheld Devices
2.2. Wearable Devices
2.3. Implantable Loop Recorders
2.4. Mobile Platforms and Support Systems
3. Discussion
3.1. Overview of Current Evidence
3.2. AF Duration and Type of Stroke
3.3. Prevention Strategies
3.4. Techniques
3.5. Knowledge Gaps for Atrial-Fibrillation Detection after Cryptogenic Stroke
3.6. Future Directions and Practical Advice
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hart, R.G.; Diener, H.C.; Coutts, S.B.; Easton, J.D.; Granger, C.B.; O’Donnell, M.J.; Sacco, R.L.; Connolly, S.J.; Cryptogenic Stroke, E.I.W.G. Embolic strokes of undetermined source: The case for a new clinical construct. Lancet Neurol. 2014, 13, 429–438. [Google Scholar] [CrossRef]
- Ornello, R.; Degan, D.; Tiseo, C.; Di Carmine, C.; Perciballi, L.; Pistoia, F.; Carolei, A.; Sacco, S. Distribution and Temporal Trends From 1993 to 2015 of Ischemic Stroke Subtypes: A Systematic Review and Meta-Analysis. Stroke 2018, 49, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Wolf, P.A.; Abbott, R.D.; Kannel, W.B. Atrial fibrillation as an independent risk factor for stroke: The Framingham Study. Stroke 1991, 22, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Hohnloser, S.H.; Pajitnev, D.; Pogue, J.; Healey, J.S.; Pfeffer, M.A.; Yusuf, S.; Connolly, S.J.; Investigators, A.W. Incidence of stroke in paroxysmal versus sustained atrial fibrillation in patients taking oral anticoagulation or combined antiplatelet therapy: An ACTIVE W Substudy. J. Am. Coll. Cardiol. 2007, 50, 2156–2161. [Google Scholar] [CrossRef]
- Yiin, G.S.; Howard, D.P.; Paul, N.L.; Li, L.; Luengo-Fernandez, R.; Bull, L.M.; Welch, S.J.; Gutnikov, S.A.; Mehta, Z.; Rothwell, P.M. Age-specific incidence, outcome, cost, and projected future burden of atrial fibrillation-related embolic vascular events: A population-based study. Circulation 2014, 130, 1236–1244. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.G.; Pearce, L.A.; Aguilar, M.I. Meta-analysis: Antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann. Intern. Med. 2007, 146, 857–867. [Google Scholar] [CrossRef]
- Ruff, C.T.; Giugliano, R.P.; Braunwald, E.; Hoffman, E.B.; Deenadayalu, N.; Ezekowitz, M.D.; Camm, A.J.; Weitz, J.I.; Lewis, B.S.; Parkhomenko, A.; et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: A meta-analysis of randomised trials. Lancet 2014, 383, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Sanna, T.; Diener, H.C.; Passman, R.S.; Di Lazzaro, V.; Bernstein, R.A.; Morillo, C.A.; Rymer, M.M.; Thijs, V.; Rogers, T.; Beckers, F.; et al. Cryptogenic stroke and underlying atrial fibrillation. N. Engl. J. Med. 2014, 370, 2478–2486. [Google Scholar] [CrossRef]
- Tsivgoulis, G.; Katsanos, A.H.; Grory, B.M.; Kohrmann, M.; Ricci, B.A.; Tsioufis, K.; Cutting, S.; Krogias, C.; Schellinger, P.D.; Campello, A.R.; et al. Prolonged Cardiac Rhythm Monitoring and Secondary Stroke Prevention in Patients With Cryptogenic Cerebral Ischemia. Stroke 2019, 50, 2175–2180. [Google Scholar] [CrossRef]
- Noubiap, J.J.; Agbaedeng, T.A.; Kamtchum-Tatuene, J.; Fitzgerald, J.L.; Middeldorp, M.E.; Kleinig, T.; Sanders, P. Rhythm monitoring strategies for atrial fibrillation detection in patients with cryptogenic stroke: A systematic review and meta-analysis. Int. J. Cardiol. Heart Vasc. 2021, 34, 100780. [Google Scholar] [CrossRef]
- Hermans, A.N.L.; Gawalko, M.; Dohmen, L.; van der Velden, R.M.J.; Betz, K.; Duncker, D.; Verhaert, D.V.M.; Heidbuchel, H.; Svennberg, E.; Neubeck, L.; et al. Mobile health solutions for atrial fibrillation detection and management: A systematic review. Clin. Res. Cardiol. Off. J. Ger. Card. Soc. 2022, 111, 479–491. [Google Scholar] [CrossRef]
- Maines, M.; Zorzi, A.; Tomasi, G.; Angheben, C.; Catanzariti, D.; Piffer, L.; Del Greco, M. Clinical impact, safety, and accuracy of the remotely monitored implantable loop recorder Medtronic Reveal LINQTM. EP Eur. 2018, 20, 1050–1057. [Google Scholar] [CrossRef]
- Gladstone, D.J.; Spring, M.; Dorian, P.; Panzov, V.; Thorpe, K.E.; Hall, J.; Vaid, H.; O’Donnell, M.; Laupacis, A.; Cote, R.; et al. Atrial fibrillation in patients with cryptogenic stroke. N. Engl. J. Med. 2014, 370, 2467–2477. [Google Scholar] [CrossRef]
- Purerfellner, H.; Pokushalov, E.; Sarkar, S.; Koehler, J.; Zhou, R.; Urban, L.; Hindricks, G. P-wave evidence as a method for improving algorithm to detect atrial fibrillation in insertable cardiac monitors. Heart Rhythm Off. J. Heart Rhythm Soc. 2014, 11, 1575–1583. [Google Scholar] [CrossRef]
- Kleindorfer, D.O.; Towfighi, A.; Chaturvedi, S.; Cockroft, K.M.; Gutierrez, J.; Lombardi-Hill, D.; Kamel, H.; Kernan, W.N.; Kittner, S.J.; Leira, E.C.; et al. 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline From the American Heart Association/American Stroke Association. Stroke 2021, 52, e364–e467. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.J.; Wakefield, V.; Jhita, T.; Kew, K.; Cain, P.; Marceniuk, G. Implantable cardiac monitors to detect atrial fibrillation after cryptogenic stroke: A systematic review and economic evaluation. Health Technol. Assess 2020, 24, 1–184. [Google Scholar] [CrossRef] [PubMed]
- Seet, R.C.; Friedman, P.A.; Rabinstein, A.A. Prolonged rhythm monitoring for the detection of occult paroxysmal atrial fibrillation in ischemic stroke of unknown cause. Circulation 2011, 124, 477–486. [Google Scholar] [CrossRef]
- Chew, D.S.; Rennert-May, E.; Quinn, F.R.; Buck, B.; Hill, M.D.; Spackman, E.; Manns, B.J.; Exner, D.V. Economic evaluation of extended electrocardiogram monitoring for atrial fibrillation in patients with cryptogenic stroke. Int. J. Stroke 2021, 16, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498. [Google Scholar]
- Rozen, G.; Vaid, J.; Hosseini, S.M.; Kaadan, M.I.; Rafael, A.; Roka, A.; Poh, Y.C.; Poh, M.Z.; Heist, E.K.; Ruskin, J.N. Diagnostic Accuracy of a Novel Mobile Phone Application for the Detection and Monitoring of Atrial Fibrillation. Am. J. Cardiol. 2018, 121, 1187–1191. [Google Scholar] [CrossRef]
- Vandenberk, T.; Stans, J.; Mortelmans, C.; Van Haelst, R.; Van Schelvergem, G.; Pelckmans, C.; Smeets, C.J.; Lanssens, D.; De Canniere, H.; Storms, V.; et al. Clinical Validation of Heart Rate Apps: Mixed-Methods Evaluation Study. JMIR Mhealth Uhealth 2017, 5, e129. [Google Scholar] [CrossRef]
- Fan, Y.Y.; Li, Y.G.; Li, J.; Cheng, W.K.; Shan, Z.L.; Wang, Y.T.; Guo, Y.T. Diagnostic Performance of a Smart Device With Photoplethysmography Technology for Atrial Fibrillation Detection: Pilot Study (Pre-mAFA II Registry). JMIR Mhealth Uhealth 2019, 7, e11437. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, J.W.; Grigg, S.; Crawford, W.; Turakhia, M.P.; Perez, M.; Ingelsson, E.; Wheeler, M.T.; Ioannidis, J.P.A.; Ashley, E.A. Accuracy of Smartphone Camera Applications for Detecting Atrial Fibrillation: A Systematic Review and Meta-analysis. JAMA Netw. Open 2020, 3, e202064. [Google Scholar] [CrossRef]
- Hall, A.; Mitchell, A.R.J.; Ashmore, L.; Holland, C. Atrial fibrillation prevalence and predictors in patients with diabetes: A cross-sectional screening study. Br. J. Cardiol. 2022, 29, 8. [Google Scholar] [CrossRef] [PubMed]
- Pezawas, T.; Stix, G.; Kastner, J.; Schneider, B.; Wolzt, M.; Schmidinger, H. Implantable loop recorder in unexplained syncope: Classification, mechanism, transient loss of consciousness and role of major depressive disorder in patients with and without structural heart disease. Heart 2008, 94, e17. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Chen, Y.; Lane, D.A.; Liu, L.; Wang, Y.; Lip, G.Y.H. Mobile Health Technology for Atrial Fibrillation Management Integrating Decision Support, Education, and Patient Involvement: mAF App Trial. Am. J. Med. 2017, 130, 1388–1396.e6. [Google Scholar] [CrossRef] [PubMed]
- Dussault, C.; Toeg, H.; Nathan, M.; Wang, Z.J.; Roux, J.F.; Secemsky, E. Electrocardiographic monitoring for detecting atrial fibrillation after ischemic stroke or transient ischemic attack: Systematic review and meta-analysis. Circ. Arrhythm. Electrophysiol. 2015, 8, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Sposato, L.A.; Cipriano, L.E.; Saposnik, G.; Ruiz Vargas, E.; Riccio, P.M.; Hachinski, V. Diagnosis of atrial fibrillation after stroke and transient ischaemic attack: A systematic review and meta-analysis. Lancet Neurol. 2015, 14, 377–387. [Google Scholar] [CrossRef]
- Kishore, A.; Vail, A.; Majid, A.; Dawson, J.; Lees, K.R.; Tyrrell, P.J.; Smith, C.J. Detection of atrial fibrillation after ischemic stroke or transient ischemic attack: A systematic review and meta-analysis. Stroke 2014, 45, 520–526. [Google Scholar] [CrossRef]
- Kamel, H.; Navi, B.B.; Elijovich, L.; Josephson, S.A.; Yee, A.H.; Fung, G.; Johnston, S.C.; Smith, W.S. Pilot randomized trial of outpatient cardiac monitoring after cryptogenic stroke. Stroke 2013, 44, 528–530. [Google Scholar] [CrossRef]
- Higgins, P.; MacFarlane, P.W.; Dawson, J.; McInnes, G.T.; Langhorne, P.; Lees, K.R. Noninvasive cardiac event monitoring to detect atrial fibrillation after ischemic stroke: A randomized, controlled trial. Stroke 2013, 44, 2525–2531. [Google Scholar] [CrossRef]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castella, M.; Diener, H.C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. EP Eur. 2016, 18, 1609–1678. [Google Scholar] [CrossRef]
- Rizos, T.; Quilitzsch, A.; Busse, O.; Haeusler, K.G.; Endres, M.; Heuschmann, P.; Veltkamp, R. Diagnostic work-up for detection of paroxysmal atrial fibrillation after acute ischemic stroke: Cross-sectional survey on German stroke units. Stroke 2015, 46, 1693–1695. [Google Scholar] [CrossRef] [PubMed]
- Ritter, M.A.; Kochhauser, S.; Duning, T.; Reinke, F.; Pott, C.; Dechering, D.G.; Eckardt, L.; Ringelstein, E.B. Occult atrial fibrillation in cryptogenic stroke: Detection by 7-day electrocardiogram versus implantable cardiac monitors. Stroke 2013, 44, 1449–1452. [Google Scholar] [CrossRef] [PubMed]
- Stahrenberg, R.; Weber-Kruger, M.; Seegers, J.; Edelmann, F.; Lahno, R.; Haase, B.; Mende, M.; Wohlfahrt, J.; Kermer, P.; Vollmann, D.; et al. Enhanced detection of paroxysmal atrial fibrillation by early and prolonged continuous holter monitoring in patients with cerebral ischemia presenting in sinus rhythm. Stroke 2010, 41, 2884–2888. [Google Scholar] [CrossRef] [PubMed]
- Barthelemy, J.C.; Feasson-Gerard, S.; Garnier, P.; Gaspoz, J.M.; Da Costa, A.; Michel, D.; Roche, F. Automatic cardiac event recorders reveal paroxysmal atrial fibrillation after unexplained strokes or transient ischemic attacks. Ann. Noninvasive Electrocardiol. 2003, 8, 194–199. [Google Scholar] [CrossRef]
- Suissa, L.; Lachaud, S.; Mahagne, M.H. Optimal timing and duration of continuous electrocardiographic monitoring for detecting atrial fibrillation in stroke patients. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2013, 22, 991–995. [Google Scholar] [CrossRef]
- Haeusler, K.G.; Kirchhof, P.; Kunze, C.; Tütüncü, S.; Fiessler, C.; Malsch, C.; Olma, M.C.; Jawad-Ul-Qamar, M.; Krämer, M.; Wachter, R.; et al. Systematic monitoring for detection of atrial fibrillation in patients with acute ischaemic stroke (MonDAFIS): A randomised, open-label, multicentre study. Lancet Neurol. 2021, 20, 426–436. [Google Scholar] [CrossRef]
- Wachter, R.; Groschel, K.; Gelbrich, G.; Hamann, G.F.; Kermer, P.; Liman, J.; Seegers, J.; Wasser, K.; Schulte, A.; Jurries, F.; et al. Holter-electrocardiogram-monitoring in patients with acute ischaemic stroke (Find-AF(RANDOMISED)): An open-label randomised controlled trial. Lancet Neurol. 2017, 16, 282–290. [Google Scholar] [CrossRef]
- Etgen, T.; Hochreiter, M.; Mundel, M.; Freudenberger, T. Insertable cardiac event recorder in detection of atrial fibrillation after cryptogenic stroke: An audit report. Stroke 2013, 44, 2007–2009. [Google Scholar] [CrossRef]
- Bernstein, R.A.; Kamel, H.; Granger, C.B.; Piccini, J.P.; Sethi, P.P.; Katz, J.M.; Vives, C.A.; Ziegler, P.D.; Franco, N.C.; Schwamm, L.H. Effect of Long-term Continuous Cardiac Monitoring vs. Usual Care on Detection of Atrial Fibrillation in Patients With Stroke Attributed to Large- or Small-Vessel Disease: The STROKE-AF Randomized Clinical Trial. JAMA J. Am. Med. Assoc. 2021, 325, 2169–2177. [Google Scholar] [CrossRef]
- Buck, B.H.; Hill, M.D.; Quinn, F.R.; Butcher, K.S.; Menon, B.K.; Gulamhusein, S.; Siddiqui, M.; Coutts, S.B.; Jeerakathil, T.; Smith, E.E.; et al. Effect of Implantable vs. Prolonged External Electrocardiographic Monitoring on Atrial Fibrillation Detection in Patients With Ischemic Stroke: The PER DIEM Randomized Clinical Trial. JAMA J. Am. Med. Assoc. 2021, 325, 2160–2168. [Google Scholar] [CrossRef] [PubMed]
- Burger, A.L.; Roesler, C.; Ebner, J.; Sommer, P.; Mutzenbach, S.; Winkler, W.B.; Weidinger, F.; Ristl, R.; Pezawas, T.; Greisenegger, S. Gapless Electrocardiogram-Monitoring in stroke at high risk of atrial fibrillation. Eur. J. Neurol. Off. J. Eur. Fed. Neurol. Soc. 2023, 30, 2092–2098. [Google Scholar] [CrossRef]
- Diener, H.C. To monitor or to not monitor for paroxysmal atrial fibrillation after transient ischemic attack or stroke: This is the question. Stroke 2014, 45, 355–356. [Google Scholar] [CrossRef]
- Yiin, G.S.C.; Li, L.; Bejot, Y.; Rothwell, P.M. Time Trends in Atrial Fibrillation-Associated Stroke and Premorbid Anticoagulation. Stroke 2018, 50, 21–27. [Google Scholar] [CrossRef]
- Li, L.; Yiin, G.S.; Geraghty, O.C.; Schulz, U.G.; Kuker, W.; Mehta, Z.; Rothwell, P.M.; Oxford Vascular, S. Incidence, outcome, risk factors, and long-term prognosis of cryptogenic transient ischaemic attack and ischaemic stroke: A population-based study. Lancet Neurol. 2015, 14, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Perera, K.S.; Vanassche, T.; Bosch, J.; Giruparajah, M.; Swaminathan, B.; Mattina, K.R.; Berkowitz, S.D.; Arauz, A.; O’Donnell, M.J.; Ameriso, S.F.; et al. Embolic strokes of undetermined source: Prevalence and patient features in the ESUS Global Registry. Int. J. Stroke 2016, 11, 526–533. [Google Scholar] [CrossRef]
- Henriksson, K.M.; Farahmand, B.; Asberg, S.; Terent, A.; Edvardsson, N. First-ever atrial fibrillation documented after hemorrhagic or ischemic stroke: The role of the CHADS(2) score at the time of stroke. Clin. Cardiol. 2011, 34, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Baturova, M.A.; Lindgren, A.; Carlson, J.; Shubik, Y.V.; Olsson, S.B.; Platonov, P.G. Predictors of new onset atrial fibrillation during 10-year follow-up after first-ever ischemic stroke. Int. J. Cardiol. 2015, 199, 248–252. [Google Scholar] [CrossRef]
- Sharma, M.; Smith, E.E.; Pearce, L.A.; Shoamanesh, A.; Perera, K.S.; Coutts, S.B.; Damgaard, D.; Ameriso, S.F.; Rha, J.H.; Modrau, B.; et al. Frequency and Patterns of Brain Infarction in Patients With Embolic Stroke of Undetermined Source: NAVIGATE ESUS Trial. Stroke 2022, 53, 45–52. [Google Scholar] [CrossRef]
- Tirschwell, D.; Akoum, N. Detection of Subclinical Atrial Fibrillation After Stroke: Is There Enough Evidence to Treat? JAMA J. Am. Med. Assoc. 2021, 325, 2157–2159. [Google Scholar] [CrossRef]
- Tsivgoulis, G.; Triantafyllou, S.; Palaiodimou, L.; Grory, B.M.; Deftereos, S.; Kohrmann, M.; Dilaveris, P.; Ricci, B.; Tsioufis, K.; Cutting, S.; et al. Prolonged Cardiac Monitoring and Stroke Recurrence: A Meta-analysis. Neurology 2022, 98, e1942–e1952. [Google Scholar] [CrossRef]
- Sharma, M.; Hart, R.G.; Smith, E.E.; Bosch, J.; Eikelboom, J.W.; Connolly, S.J.; Dyal, L.; Reeh, K.W.; Casanova, A.; Diaz, R.; et al. Rivaroxaban for Prevention of Covert Brain Infarcts and Cognitive Decline: The COMPASS MRI Substudy. Stroke 2020, 51, 2901–2909. [Google Scholar] [CrossRef]
- Shoamanesh, A.; Mundl, H.; Smith, E.E.; Masjuan, J.; Milanov, I.; Hirano, T.; Agafina, A.; Campbell, B.; Caso, V.; Mas, J.L.; et al. Factor XIa inhibition with asundexian after acute non-cardioembolic ischaemic stroke (PACIFIC-Stroke): An international, randomised, double-blind, placebo-controlled, phase 2b trial. Lancet 2022, 400, 997–1007. [Google Scholar] [CrossRef]
- Meinel, T.R.; Kaesmacher, J.; Roten, L.; Fischer, U. Covert Brain Infarction: Towards Precision Medicine in Research, Diagnosis, and Therapy for a Silent Pandemic. Stroke 2020, 51, 2597–2606. [Google Scholar] [CrossRef] [PubMed]
- Svennberg, E.; Friberg, L.; Frykman, V.; Al-Khalili, F.; Engdahl, J.; Rosenqvist, M. Clinical outcomes in systematic screening for atrial fibrillation (STROKESTOP): A multicentre, parallel group, unmasked, randomised controlled trial. Lancet 2021, 398, 1498–1506. [Google Scholar] [CrossRef] [PubMed]
- Diener, H.C.; Eikelboom, J.; Connolly, S.J.; Joyner, C.D.; Hart, R.G.; Lip, G.Y.; O’Donnell, M.; Hohnloser, S.H.; Hankey, G.J.; Shestakovska, O.; et al. Apixaban versus aspirin in patients with atrial fibrillation and previous stroke or transient ischaemic attack: A predefined subgroup analysis from AVERROES, a randomised trial. Lancet Neurol. 2012, 11, 225–231. [Google Scholar] [CrossRef]
- Healey, J.S.; Connolly, S.J.; Gold, M.R.; Israel, C.W.; Van Gelder, I.C.; Capucci, A.; Lau, C.P.; Fain, E.; Yang, S.; Bailleul, C.; et al. Subclinical atrial fibrillation and the risk of stroke. N. Engl. J. Med. 2012, 366, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Kamel, H.; Okin, P.M.; Elkind, M.S.; Iadecola, C. Atrial Fibrillation and Mechanisms of Stroke: Time for a New Model. Stroke 2016, 47, 895–900. [Google Scholar] [CrossRef]
- Singer, D.E.; Ziegler, P.D.; Koehler, J.L.; Sarkar, S.; Passman, R.S. Temporal Association Between Episodes of Atrial Fibrillation and Risk of Ischemic Stroke. JAMA Cardiol. 2021, 6, 1364–1369. [Google Scholar] [CrossRef]
- Lip, G.Y.; Nieuwlaat, R.; Pisters, R.; Lane, D.A.; Crijns, H.J. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: The euro heart survey on atrial fibrillation. Chest 2010, 137, 263–272. [Google Scholar] [CrossRef]
- Kamel, H.; Johnson, D.R.; Hegde, M.; Go, A.S.; Sidney, S.; Sorel, M.; Hills, N.K.; Johnston, S.C. Detection of atrial fibrillation after stroke and the risk of recurrent stroke. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2012, 21, 726–731. [Google Scholar] [CrossRef]
- Kim, A.S.; Kamel, H.; Bernstein, R.A.; Manchanda, M.; Caprio, F.Z. Controversies in Stroke: Should Patients With Embolic Stroke of Undetermined Source Undergo Intensive Heart Rhythm Monitoring With an Implantable Loop Recorder? Stroke 2022, 53, 3243–3247. [Google Scholar] [CrossRef] [PubMed]
- Veltkamp, R.; Pearce, L.A.; Korompoki, E.; Sharma, M.; Kasner, S.E.; Toni, D.; Ameriso, S.F.; Mundl, H.; Tatlisumak, T.; Hankey, G.J.; et al. Characteristics of Recurrent Ischemic Stroke After Embolic Stroke of Undetermined Source: Secondary Analysis of a Randomized Clinical Trial. JAMA Neurol. 2020, 77, 1233–1240. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Al-Khatib, S.M.; Desvigne-Nickens, P.; Alonso, A.; Djousse, L.; Forman, D.E.; Gillis, A.M.; Hendriks, J.M.L.; Hills, M.T.; Kirchhof, P.; et al. Research Priorities in the Secondary Prevention of Atrial Fibrillation: A National Heart, Lung, and Blood Institute Virtual Workshop Report. J. Am. Heart Assoc. 2021, 10, e021566. [Google Scholar] [CrossRef]
- Gage, B.F.; van Walraven, C.; Pearce, L.; Hart, R.G.; Koudstaal, P.J.; Boode, B.S.; Petersen, P. Selecting patients with atrial fibrillation for anticoagulation: Stroke risk stratification in patients taking aspirin. Circulation 2004, 110, 2287–2292. [Google Scholar] [CrossRef]
- Brambatti, M.; Connolly, S.J.; Gold, M.R.; Morillo, C.A.; Capucci, A.; Muto, C.; Lau, C.P.; Van Gelder, I.C.; Hohnloser, S.H.; Carlson, M.; et al. Temporal relationship between subclinical atrial fibrillation and embolic events. Circulation 2014, 129, 2094–2099. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.M.; Koehler, J.; Ziegler, P.D.; Sarkar, S.; Zweibel, S.; Passman, R.S. Stroke Risk as a Function of Atrial Fibrillation Duration and CHA(2)DS(2)-VASc Score. Circulation 2019, 140, 1639–1646. [Google Scholar] [CrossRef]
- Turakhia, M.P.; Ziegler, P.D.; Schmitt, S.K.; Chang, Y.; Fan, J.; Than, C.T.; Keung, E.K.; Singer, D.E. Atrial Fibrillation Burden and Short-Term Risk of Stroke: Case-Crossover Analysis of Continuously Recorded Heart Rhythm from Cardiac Electronic Implanted Devices. Circ. Arrhythm. Electrophysiol. 2015, 8, 1040–1047. [Google Scholar] [CrossRef]
- Lin, H.J.; Wolf, P.A.; Kelly-Hayes, M.; Beiser, A.S.; Kase, C.S.; Benjamin, E.J.; D’Agostino, R.B. Stroke severity in atrial fibrillation. The Framingham Study. Stroke 1996, 27, 1760–1764. [Google Scholar] [CrossRef]
- Diener, H.C.; Sacco, R.L.; Easton, J.D.; Granger, C.B.; Bernstein, R.A.; Uchiyama, S.; Kreuzer, J.; Cronin, L.; Cotton, D.; Grauer, C.; et al. Dabigatran for Prevention of Stroke after Embolic Stroke of Undetermined Source. N. Engl. J. Med. 2019, 380, 1906–1917. [Google Scholar] [CrossRef]
- Hart, R.G.; Sharma, M.; Mundl, H.; Kasner, S.E.; Bangdiwala, S.I.; Berkowitz, S.D.; Swaminathan, B.; Lavados, P.; Wang, Y.; Wang, Y.; et al. Rivaroxaban for Stroke Prevention after Embolic Stroke of Undetermined Source. N. Engl. J. Med. 2018, 378, 2191–2201. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, I.C.; Healey, J.S.; Crijns, H.; Wang, J.; Hohnloser, S.H.; Gold, M.R.; Capucci, A.; Lau, C.P.; Morillo, C.A.; Hobbelt, A.H.; et al. Duration of device-detected subclinical atrial fibrillation and occurrence of stroke in ASSERT. Eur. Heart J. 2017, 38, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, J.H.; Diederichsen, S.Z.; Hojberg, S.; Krieger, D.W.; Graff, C.; Kronborg, C.; Olesen, M.S.; Nielsen, J.B.; Holst, A.G.; Brandes, A.; et al. Implantable loop recorder detection of atrial fibrillation to prevent stroke (The LOOP Study): A randomised controlled trial. Lancet 2021, 398, 1507–1516. [Google Scholar] [CrossRef]
- Chaisinanunkul, N.; Khurshid, S.; Buck, B.H.; Rabinstein, A.A.; Anderson, C.D.; Hill, M.D.; Fugate, J.E.; Saver, J.L. How often is occult atrial fibrillation in cryptogenic stroke causal vs. incidental? A meta-analysis. Front. Neurol. 2023, 14, 1103664. [Google Scholar] [CrossRef] [PubMed]
- Healey, J.S.; Gladstone, D.J.; Swaminathan, B.; Eckstein, J.; Mundl, H.; Epstein, A.E.; Haeusler, K.G.; Mikulik, R.; Kasner, S.E.; Toni, D.; et al. Recurrent Stroke With Rivaroxaban Compared With Aspirin According to Predictors of Atrial Fibrillation: Secondary Analysis of the NAVIGATE ESUS Randomized Clinical Trial. JAMA Neurol. 2019, 76, 764–773. [Google Scholar] [CrossRef]
- Hart, R.G.; Veltkamp, R.C.; Sheridan, P.; Sharma, M.; Kasner, S.E.; Bangdiwala, S.I.; Ntaios, G.; Shoamanesh, A.; Ameriso, S.F.; Toni, D.; et al. Predictors of Recurrent Ischemic Stroke in Patients with Embolic Strokes of Undetermined Source and Effects of Rivaroxaban Versus Aspirin According to Risk Status: The NAVIGATE ESUS Trial. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2019, 28, 2273–2279. [Google Scholar] [CrossRef]
- Thijs, V.N.; Brachmann, J.; Morillo, C.A.; Passman, R.S.; Sanna, T.; Bernstein, R.A.; Diener, H.C.; Di Lazzaro, V.; Rymer, M.M.; Hogge, L.; et al. Predictors for atrial fibrillation detection after cryptogenic stroke: Results from CRYSTAL AF. Neurology 2016, 86, 261–269. [Google Scholar] [CrossRef]
- Kamel, H.; Longstreth, W.T., Jr.; Tirschwell, D.L.; Kronmal, R.A.; Broderick, J.P.; Palesch, Y.Y.; Meinzer, C.; Dillon, C.; Ewing, I.; Spilker, J.A.; et al. The AtRial Cardiopathy and Antithrombotic Drugs In prevention after cryptogenic stroke randomized trial: Rationale and methods. Int. J. Stroke 2019, 14, 207–214. [Google Scholar] [CrossRef]
- Koh, K.T.; Law, W.C.; Zaw, W.M.; Foo, D.H.P.; Tan, C.T.; Steven, A.; Samuel, D.; Fam, T.L.; Chai, C.H.; Wong, Z.S.; et al. Smartphone electrocardiogram for detecting atrial fibrillation after a cerebral ischaemic event: A multicentre randomized controlled trial. EP Eur. 2021, 23, 1016–1023. [Google Scholar] [CrossRef]
- Magnusson, P.; Lyren, A.; Mattsson, G. Diagnostic yield of chest and thumb ECG after cryptogenic stroke, Transient ECG Assessment in Stroke Evaluation (TEASE): An observational trial. BMJ Open 2020, 10, e037573. [Google Scholar] [CrossRef]
- Magnusson, P.; Lyren, A.; Mattsson, G. Patient-reported feasibility of chest and thumb ECG after cryptogenic stroke in Sweden: An observational study. BMJ Open 2020, 10, e037360. [Google Scholar] [CrossRef] [PubMed]
- Rizas, K.D.; Freyer, L.; Sappler, N.; von Stulpnagel, L.; Spielbichler, P.; Krasniqi, A.; Schreinlechner, M.; Wenner, F.N.; Theurl, F.; Behroz, A.; et al. Smartphone-based screening for atrial fibrillation: A pragmatic randomized clinical trial. Nat. Med. 2022, 28, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
| Method | Pros | Cons |
|---|---|---|
| Noninvasive | ||
| (1) Continuous in-hospital telemetry | - Accurate diagnosis - Detects asymptomatic events | - Requires inpatient monitoring - Restricts patient movement - Expensive |
| (2) Holter-ECG (24–72 h) (3) Handheld devices | - Accurate diagnosis - Detects asymptomatic events | - Short monitoring period - Symptom diary required |
| Patient triggered/event recorder | - Correlation with symptoms - Longer monitoring periods | - No detection of asymptomatic events - Patient participation required |
| (4) Wearables Mobile automatic/wearable cardiovascular telemetry | - Continuous recording - Detects asymptomatic events | - Patient compliance, skin irritation - Expensive |
| Invasive | ||
| (5) Implantable loop recorder | - Follow-up up to 5a - Internet-based data transmission - Detects asymptomatic events - Correlation with symptoms | - False-positive/-negative detection - Initially expensive and invasive |
| (6) Already implanted PM or ICD | - Endless follow-up - Internet-based data transmission - Detects asymptomatic events - Correlation with symptoms | - Restricted to small population group |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pezawas, T. ECG Smart Monitoring versus Implantable Loop Recorders for Atrial Fibrillation Detection after Cryptogenic Stroke—An Overview for Decision Making. J. Cardiovasc. Dev. Dis. 2023, 10, 306. https://doi.org/10.3390/jcdd10070306
Pezawas T. ECG Smart Monitoring versus Implantable Loop Recorders for Atrial Fibrillation Detection after Cryptogenic Stroke—An Overview for Decision Making. Journal of Cardiovascular Development and Disease. 2023; 10(7):306. https://doi.org/10.3390/jcdd10070306
Chicago/Turabian StylePezawas, Thomas. 2023. "ECG Smart Monitoring versus Implantable Loop Recorders for Atrial Fibrillation Detection after Cryptogenic Stroke—An Overview for Decision Making" Journal of Cardiovascular Development and Disease 10, no. 7: 306. https://doi.org/10.3390/jcdd10070306
APA StylePezawas, T. (2023). ECG Smart Monitoring versus Implantable Loop Recorders for Atrial Fibrillation Detection after Cryptogenic Stroke—An Overview for Decision Making. Journal of Cardiovascular Development and Disease, 10(7), 306. https://doi.org/10.3390/jcdd10070306






