Abstract
Virtual reality (VR) describes a family of technologies which immerse users in sensorily-stimulating virtual environments. Such technologies have increasingly found applications in the treatment of neurological and mental health disorders. Depression, anxiety, and other mood abnormalities are of concern in the growing older population—especially those who reside in long-term care facilities (LTCFs). The transition from the familiar home environment to the foreign LTCF introduces a number of stressors that can precipitate depression. However, recent studies reveal that VR therapy (VRT) can promote positive emotionality and improve cognitive abilities in older people, both at home and in LTCFs. VR thus holds potential in allowing older individuals to gradually adapt to their new environments—thereby mitigating the detrimental effects of place attachment and social exclusion. Nevertheless, while the current psychological literature is promising, the implementation of VR in LTCFs faces many challenges. LTCF residents must gain trust in VR technologies, care providers require training to maximize the positive effects of VRT, and decision makers must evaluate both the opportunities and obstacles in adopting VR. In this review article, we concisely discuss the implications of depression related to place attachment in LTCFs, and explore the potential therapeutic applications of VR.
1. Overview
Virtual reality (VR) is the use of various technologies to digitally simulate or recreate an environment in which an observer can realistically hear, see, and/or feel as though they are a part of the simulation [1]. This dynamic perceptualization is achieved through technologies such as wearable head-mounted displays (HMDs), noise cancellation headsets, and other multimodal stimuli to immerse the wearer in the virtual atmosphere [1]. While VR has been a critical training tool in the fields of aviation, military combat, and surgery, it has recently shifted towards clinical psychology and therapy [2]. In fact, with the decreasing costs and increasing accessibility of digital media, VR has significant potential in reconstructing conventional approaches to patient care [3].
VR is a popular topic in psychological research studies for its potential in treating various mental disorders such as schizophrenia, post-traumatic stress disorder (PTSD), anxiety, and depression. Psychotherapy experts rank VR as one of the top psychological interventions with an overall positive growth projection in the coming decades [4]. Rothbaum and colleagues first assessed the clinical effects of VR therapy (VRT) on acrophobic college students [5]. Their study established the efficacy of VRT for trauma and anxiety, and provided a basis for further studies on other psychiatric illnesses [6]. With its ability to continuously expose patients to traumatic experiences or threats on a regular basis, VR can train patients to cope with, and eventually eliminate, the fears and negative emotions associated with stressful experiences [6]. One particular demographic that could benefit from the psychological and therapeutic effects of VR is older individuals who reside in long-term care facilities (LTCFs).
An increasing number of older people prefer to age-in-place until they become unable to care for their needs [7]. The choice of moving to a LTCF, however, often becomes inevitable at a certain stage due to deteriorating health conditions. This inevitability, along with demographic changes due to continuous increases in life expectancy, has become a concern [8]. Scholars such as Schwarz and Brent, argue that the proportion of people aged 80 years or older is expected to triple by 2040 [7]. In the United States of America, the age structure of the overall population is projected to change tremendously in the next decade: from 13 percent of the population aged 65 and older in 2010 to around 20 percent in 2030 [9]. Hence, there is a need for social infrastructure to accommodate the physical and psychological needs of this growing population that may transition into LTCFs.
Transitioning into a LTCF may cause severe distress and amplify pre-existing mental health impairments, corresponding to a higher rate of depression of nursing home residents when compared to non-residents [10]. Furthermore, age-related cognitive decline and desires for a familiar environment can further an older individual’s mental deterioration. As mental illnesses can exacerbate other underlying health conditions such as cardiovascular diseases, afflicted individuals may require more demanding and attentive care [11]. VR can serve as a tool to aid the adjustment of older people into new LTCF environments by immersing them in a familiar space to relieve stress, or by familiarizing them with their new surroundings with continuous exposure. VRT can, therefore, mitigate the negative mental health trends seen in individuals in care homes.
This review article explores the potential of VRT in improving mood and alleviating depression in LTCF residents. We first overview the challenges faced by older individuals transitioning to LTCFs. Place attachment bonds are of particular interest, as the disruption of these bonds is a major cause of depression in care homes. Next, we discuss the clinical features and neurological mechanisms of depression related to place attachment, and highlight the difficulties in diagnosing and treating depression in the LTCF setting. We then concisely review the effects of VRT on mood and depression in older individuals—especially those in LTCFs—and conclude by discussing the benefits and limitations of the implementation of VRT in LTCFs.
2. Place Attachment in LTCFs
An individual’s environment shapes their view and perception of life, and therefore plays a critical role in their psychological well-being. Systematic reviews indicate that most older individuals prefer to remain in their current residences rather than transfer into facilities [12]. Alongside the other physical health impairments associated with old age, transitioning from the comfort of their homes into LTCFs causes detrimental cognitive responses in older people. Nevertheless, long-term institutional care is necessary for some older individuals due to the severity of an illness or the lack of an attentive caregiver at home [7]. Moving to a LTCF is extraordinarily stressful and involves nostalgia [13]. In environment–behavior research, nostalgia is considered a result of place attachment, an emotional bond that links a person to a place [8]. A common mechanism that explains this bond is proximity-maintaining behavior [14,15]. As previously mentioned, individuals who exhibit strong attachment to certain places are likely to disregard important opportunities just to physically remain in those places [16].
A newly admitted LTCF resident faces a number of challenges associated with forming new social connections, inadequate privacy, adapting to a new institutional setting, and lack of homelike qualities [17]. Attachment to one’s home complicates the transition into LTCFs. The degree of place attachment and the person’s readiness to adapt to a new environment influence the smoothness of the transition and consequent stress levels. Bernard and Rowles argue that relocating to a LTCF can be traumatizing for some people due to their inexperience in changing homes throughout their life [14]. Jones et al. defined a transition as a passage between two relatively stable periods of time, in which an individual moves from one life phase to another. A transition is a process in which the person must develop new skills, relationships, and coping strategies [14,18].
Manzo and Wright shed light on the relationship between place attachment theory and the ability to transition into a new place. They suggest that in the era of increased mobility, place attachment theory can be used to distinguish mechanisms through which people become attached even to alien places [15]. As Bernard and Rowles wrote, “theories of place attachment and identity emphasize domains of belonging in a more process-oriented and differentiated way. Place attachment is not only related to attitudes, but also to a gamut of processes operating when people form affective, cognitive, behavioral, and social bonds to the environment, thereby, transforming a space into a place” [14]. Place attachment dynamics involving interrelated processes between people and places are called place processes. Examples of these processes include place identity, a process in which people view a place as a significant part of their world, and place interaction, which refers to the typical going-ons in a place [15]. In a well-liked place, all place processes are sophisticatedly interconnected; feeling at-home is being fit in a certain place and being able to continuously engage in place-making processes [15]. In other words, the degree of place attachment pre-determines a person’s willingness and ability to transition into a new place such as a LTCF.
3. Depression in LTCFs
3.1. Causation and Neural Mechanisms of Depression in LTCFs
As discussed above, the transition into a LTCF is a significantly stressful event for older people, especially during the first four weeks following admission [19]. A major reason for this transitional stress is the disruption of place attachment bonds that play a major role in an individual’s psychological development and well-being. According to Scannell and Gifford, maintaining proximity to one’s place of attachment can be beneficial in many respects, as this provides emotional healing, cognitive restoration, belongingness, and escape opportunities from daily stressors [20]. Relocation to an entirely foreign place, without the trappings of home and family, can be psychologically devastating. As such, Texas senior home residents reported losses of independence, disruptions of continuity with their past (familiar) lives, and loneliness to be precipitating factors (stressors) in their negative moods and/or depression [10].
The diathesis–stress model of mental disorders holds that certain individuals have genetic, anatomical, and/or psychological predispositions that remain dormant until activated by a source of stress [21]. Predispositions for depression include alterations in brain structure, especially in the bilateral amygdala, hippocampus, and dorsolateral prefrontal cortex (PFC). These neuroanatomical changes have been found in first-degree relatives of patients with depression, suggesting a mechanism of risk [22]. Moreover, stressful events, such as serious housing problems and separation from friends or loved ones, are causally related to depression [23].
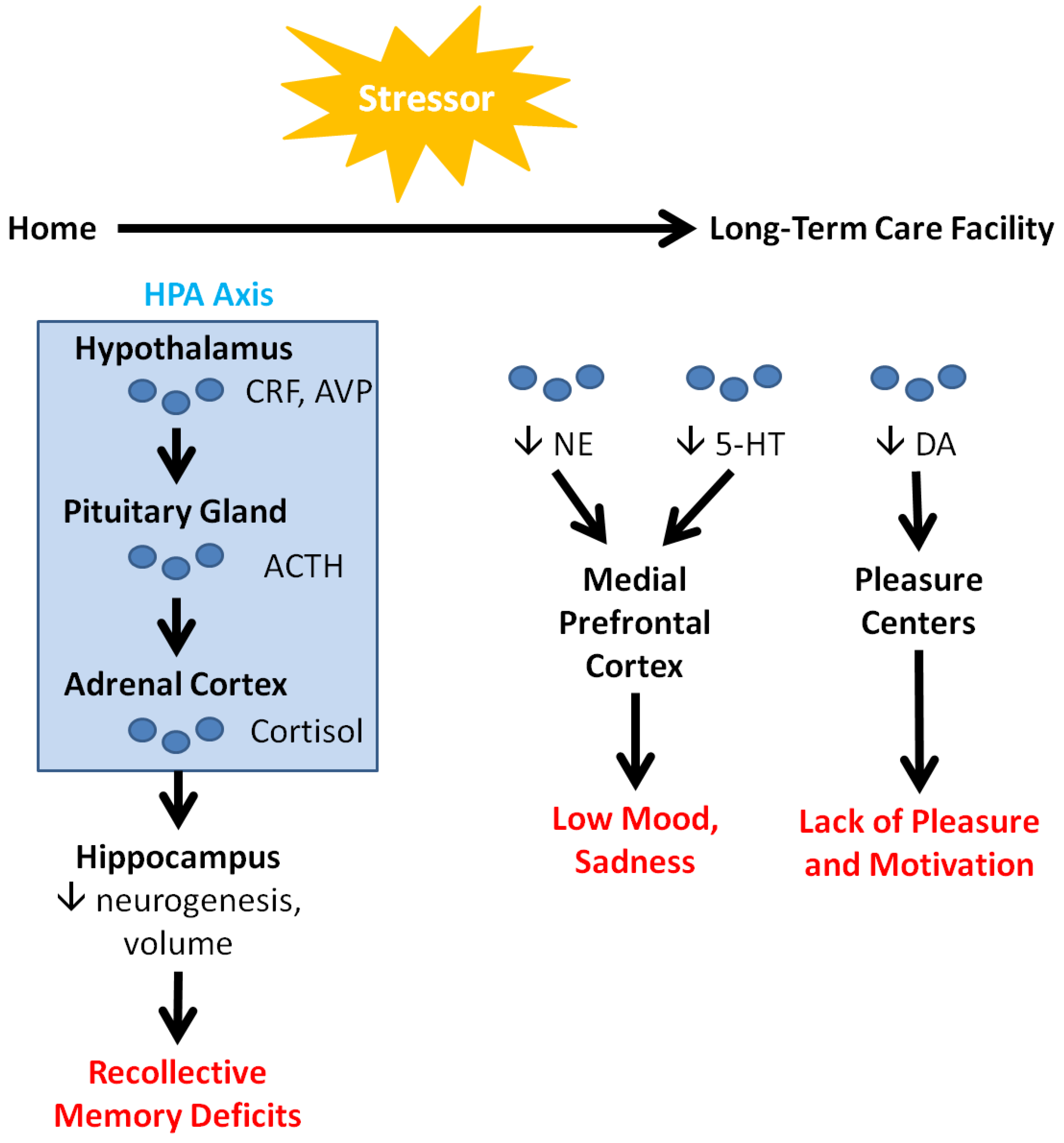
Depression is characterized by symptoms such as persistent disturbances of mood, loss of interest in activities, sleep abnormalities, feelings of guilt, loss of energy, impaired concentration, changes in appetite, psychomotor agitation, and in some cases, suicidal ideation [22]. These psychological and cognitive symptoms may manifest as a result of neurochemical and neuroanatomical changes precipitated by stress. As illustrated in Figure 1, transitional stress immediately activates the hypothalamic–pituitary–adrenal (HPA) axis. The hypothalamus releases corticotropin release factor (CRF) and vasopressin (AVP), which stimulate the release of adrenocorticotropic hormone (ACTH) from the pituitary gland. ACTH causes the adrenal cortex to release cortisol, the stress hormone [24,25]. Among other functions, cortisol downregulates hippocampal neurogenesis, leading to a decrease in hippocampal volume and consequent memory deficits [25].
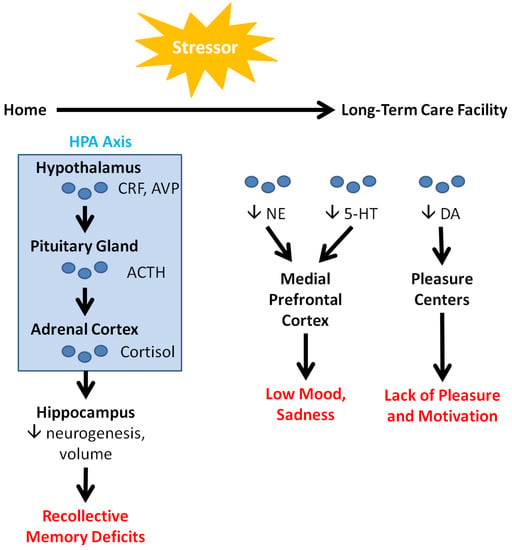
Figure 1.
Neurological factors in depression. Acute and chronic stressors, such as disruptions in place attachment bonds, activate the HPA axis and modulate neurotransmitter levels, leading to cognitive and emotional consequences (highlighted in red). These consequences are alleviated in older people by VRT (see Table 1), encouraging the use of VRT with LTCF residents.
Over a longer term, stressors in LTCFs decrease the levels of circulating neurotransmitters (NTs) such as norepinephrine (NE), serotonin (5-HT), and dopamine (DA). NE and 5-HT ordinarily interact with the medial PFC to regulate mood, sleep, appetite, and other bodily functions. Consequently, downregulation of these NTs leads to the low mood and sadness that are characteristic of depression [26]. These NTs also interact with the amygdala, the portion of the brain responsible for emotions and fear conditioning. Their dysfunction can lead to anxiety, which often co-occurs with depression. In contrast, DA is heavily implicated in learning and the reward system, acting in conjunction with the brain’s pleasure centers. DA’s downregulation correspondingly decreases pleasure and intrinsic motivation, leading to a general loss of interest [26]. Finally, a clinical study on patients with late-life depression by Smith and colleagues detected increased levels of N-acetylaspartate in the posterior cingulate cortex. They also found associations between decreasing levels of glutamate and glutathione, and decreasing symptom severity [27].
While the neural mechanisms of depression are well established, measuring depression in LTCF residents can be a difficult task. As stress is a major causal factor in depression in LTCF residents, measurement of the levels of cortisol, the hormone associated with stress, would ideally be able to confirm diagnosis. Blood cortisol levels can be matched to the degree of depression in some cases of acute and severe depression [28,29]. However, in mild, chronic, or atypical depression, variability exists in the responsiveness of the HPA axis to stressors [30,31,32,33]. As such, other factors may be at play with regards to the HPA axis’ relationship with depression; these include changes in the glucocorticoid receptors, the mineralocorticoid receptors, or the responsiveness of the axis to AVP and CRF [33]. Therefore, the hormone cortisol cannot be used alone as a marker of depression. Studies have suggested that the levels of various NTs and other biomarkers could support the diagnosis and categorization of depression [34,35]. While these theories are promising, further research is needed to make biomarker identification a viable option to diagnose depression [35].
The lack of a simple laboratory test to aid the diagnosis of depression means that other methods are necessary [36]. While many depression-identifying tests have been validated, they have varying sensitivity [37,38,39]. These tests include questions related to physiological variables such as eating habits, weight loss, and sleep, as well as psychological variables such as mood, behavior, and thoughts. Tests can range from short self-reporting tests to detailed interviews involving highly trained medical staff [40,41]. Self-reporting tests are advantageous as they can be administered en masse; as such, they are time-friendly and provide extended coverage, but their accuracy is debatable [42]. There is general agreement in the literature that one-on-one interviews with clients by trained professionals are more exact in diagnosing depression. This accuracy, however, is fully dependent upon the training of those administering the test as well as the amount of time required for each test. These constraints, alongside a lack of administrative support, limit this option in LTCFs [38].
Currently accepted numbers of those with depression in LTCFs are based on a mixture of results from varied tests run by different researchers or organizations on wide-ranging populations. Without a specific biomarker accepted for the diagnosis of depression, multiple depression-identifying tools—including surveys—are heavily relied upon. Self-reporting tests may be used to identify LTCF residents suspected of suffering from depression. However, it is recommended that individuals are then evaluated by trained professionals [36]. Obtaining a true representation of depression in LTCFs requires coordinated mechanisms for standardized testing throughout the whole LTCF community.
3.2. Implications of Depression in LTCFs
Critically, depressive symptoms are present in one third of the older population, with the proportion of depressed older people residing in nursing homes being considerably higher than their community-dwelling counterparts [10,43]. As such, depression and anxiety are the most common mental disorders among older adults in LTCFs, yet have been inadequately studied in social research [44]. Notably, depression in LTCFs is customarily linked to the aging process and therefore overlooked. Important mental effects of depression include reduced well-being, loneliness, functional impairment, and increased mortality [44]. Moreover, depression is a major contributing factor to suicide among older people. In fact, suicide rates among older people are generally higher than those for younger people worldwide [45]. The latter fact is alarming and underscores a need for environment–behavior research concerning the environmental causes of depression in LTCFs.
A major barrier to depression research and treatment in LTCFs are the physical and psychological comorbidities of depression in older people. Age-related depression occurs along with other illnesses, such as dementia, chronic conditions, and functional limitations [46]. The management of depression in people with dementia is particularly challenging as neuronal degeneration complicates treatment responses and prolongs hospitalization [47]. Recovery and rehabilitation are further complicated by depression’s direct relationship with physical illness [48]. Depression may affect cardiovascular endurance: Guinjoan et al. concluded that autonomic function is altered in major depression, indicating decreased parasympathetic activity and increased sympathetic activity. These alterations have implications for the increased risk of cardiac disease found in patients with major depression [49]. In a recent study, depression was found to be associated with decreased heart rate variability (HRV), which is a predictor of cardiovascular morbidity [50].
The co-occurrence of depression with dementia in older individuals poses significant challenges with regards to treatment. Antidepressants constitute a canonical therapeutic modality; however, they alter the structure and function of various brain regions, leading to detrimental neural and cognitive side effects that are particularly problematic in those with dementia [47]. Psychosocial treatments such as reminiscence and behavior therapy are constrained by their limited efficacy [47]. As such, it is necessary to explore technologically-driven treatment modalities, such as VRT, in alleviating depression and place attachment in LTCF residents.
4. VR’s Effect on Depression and Mood in Older People
The medical applications of VR have been extensively reviewed [51]. Applications span numerous disciplines—including medical education and training, robotic surgery, molecular biology, forensic pathology, and others [52,53]. We will focus primarily on the applications of VR in psychology, specifically as a therapeutic intervention for depression and mood in LTCFs. Given the limitations of behavioral therapy due to the complex psychological states of older individuals, the application of VR for mental health treatment is critical.
It is evident that depression is associated with increased stress levels related to place attachment and relocation. A number of studies investigated reminiscence therapy using digital media and new technologies, such as VR, as a treatment for memory loss in people with dementia [54]. In the cognitive and behavioral sciences, VR is defined as “an advanced form of human–computer interface that allows the user to interact with and become immersed in a computer-generated environment in a naturalistic fashion,” meaning that patients can “live” the experience of being in completely different spaces during the VR session (telepresence) [55]. However, the use and implementation of the technology is debated amongst researchers and healthcare practitioners.
Describing the spectrum and limitations of VR technologies, Klein et al. write that “virtual reality worlds with a high level of interaction demands are often too hard to grasp for people with dementia in terms of cognitive elaboration and comprehension of the metaphors used. Nevertheless, there is indication that simulated locations and objects can have a positive impact for reminiscence therapy” [54]. Such positive effects of VR are demonstrated by a number of recent cognitive and psychological studies. For instance, Yang and colleagues examined the impact of VR technology on falls and depression among older individuals with mild depression, with their results showing that depression and internal stress scores were reduced after the intervention. They concluded that the VR exercise program exerted a positive effect on the psychological function of the aged and could be potentially utilized as a therapeutic intervention for reducing depression and internal stress among older people [56].
While literature of VRT as an intervention for depression are currently limited, numerous recent trials demonstrate the value of VRT in enhancing moods and cognition in older people (Table 1). These include several ambulatory studies. For instance, Chan et al. studied 236 healthy members (aged 60 and above) of community centers in Hong Kong. Members of the trial group were exposed to a VR-based tour of Hong Kong’s landmark sites, which included both present-day and 20-year-old images. After a single exposure, they exhibited increases in overall positive emotions—including increased interest, enthusiasm, and excitement. They also exhibited decreases in overall negative affect and its components, such as distress, hostility, and guilt [57]. In another study, Graf et al. exposed 14 home-residing pensioners to a VR “forest walk” experience via the Oculus Go headset system. The experience included a virtual dog as a companion, and several cognitively stimulating mini-games. After the VR exercise, participants demonstrated an increased overall positive affect [58]. Moreover, Banos and colleagues exposed 18 Spanish Senior University participants (aged 58 to 79 years) to VR nature walks meant to elicit joy and/or relaxation. After one, two, or three exposures, both the joy-inducing and relaxation-inducing virtual environments (VE) increased perceived joy and relaxation, and reduced sadness and anxiety [59]. These trials underscore the ability of a single VRT session to influence mood among older individuals.

Table 1.
Effects of VR on mood and cognitive function in older people.
Other longer-term ambulatory studies also corroborate the potential of VRT to induce positive moods among older people. Barsasella et al. exposed 29 Taipei Medical University Aging Center patients (ages 60–94) to VR biweekly for six weeks (twelve total exposures). VR experiences were delivered through several applications on the High-Tech Computer Corporation’s Vive platform. Notably, participants exhibited statistically significant increases in happiness, as measured through the Chinese Happiness Inventory, post-intervention [60]. Additionally, Gamito and colleagues exposed Portuguese senior daycare center users to biweekly VR interventions over the course of six weeks. The intervention was a city-based VR in which participants carried out everyday activities, such as household organization, shopping, and watching television. Intervention group members exhibited increases in general and visual memory as well as attention [61].
Similar results to those of the aforementioned trials were obtained from studies in LTCFs. Brimelow et al. exposed 13 Australian residential aged care facility (RACF) residents, aged 66–93 years, to relaxing scenes through the Samsung Galaxy S7 smartphone-Samsung Gear VR headset system. After a single exposure, participants exhibited decreased total apathy and increased levels of facial expression, eye contact, physical engagement, verbal tone, and verbal expression [62]. In another study by Saredakis and colleagues, 17 RACF residents were exposed twice to a wandering experience based on Google Street View. This VR intervention utilized the Wander application through YouTube VR and the Oculus Go headset. The investigators found correlations between decreased apathy after the VR experiences and increased semantic fluency [63]. Finally, D’Cunha et al. exposed 11 RACF residents to an immersive bicycling experience through projected footage and stationary pedal exercisers. While no significant changes in mood or apathy were observed, the majority of participants enjoyed the experience, which allowed them to reminisce on cycling memories [64].
5. Concluding Remarks
Numerous studies substantiate the claim that VRT has clear potential in alleviating negative feelings while promoting improved cognitive ability and positive emotions in older patients. They indicate that VRT reduces overall negative feelings—such as apathy, distress, and anxiety—while increasing overall positive outlook compared to pre-exposed VRT patients [57,58,60]. It is worth noting here that, while anatomical and molecular factors such as NTs were not assessed in the reviewed studies, the described psychological findings suggest that VRT modulates the neurological mechanisms underlying mood and depression (Figure 1). Moreover, not only does VRT correlate with psychological relief, but evidence also suggests that VRT can improve both physical and cognitive functionality in older patients. As shown by Brimelow et al. and Saredakis et al., patients who underwent VRT exhibited improvements in visual memory, linguistic fluency and expression, and physical engagement [62,63]. These heightened abilities can alleviate the feelings of social exclusion felt by many LTCF residents, furthering the positive effects of VR on a patient’s overall psychological well-being. In this light, VRT has the potential to improve mood and cognition, and mitigate the effects of depression—including those related to place attachment—in residents of care homes.
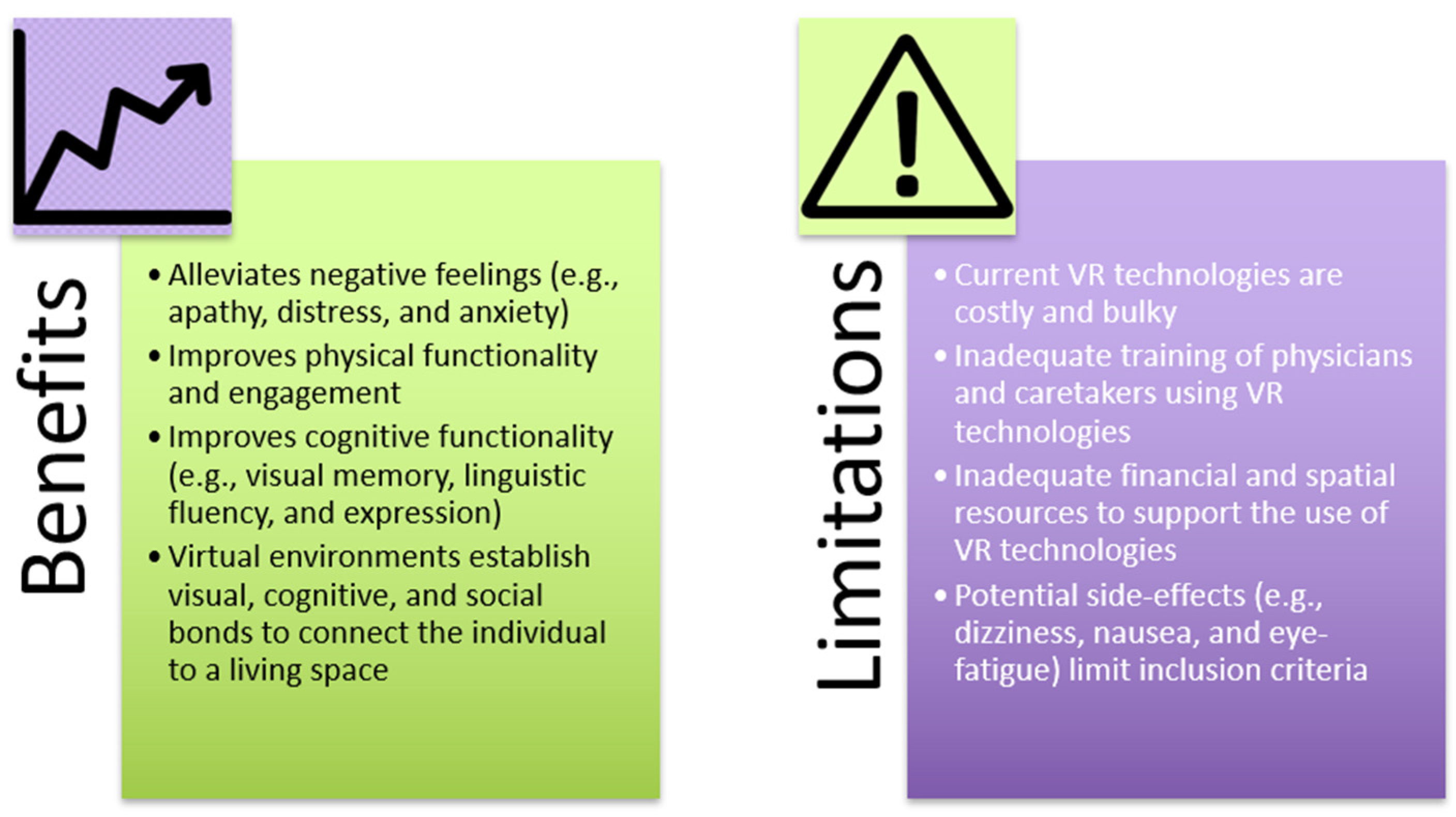
While VRT offers many benefits, there are various challenges involved in its large-scale implementation in mental rehabilitation and therapy (Figure 2). Current technologies are costly and bulky, limiting their applicability in care homes due to inadequate financial resources or physical space. Furthermore, adequate training for using the technologies is required for physicians and caretakers to maximize benefit from the treatments. VRT has been attributed to various side-effects, including dizziness, nausea, and eye-fatigue, effectively limiting the population who can participate in the treatments [65,66]. Given that older people already have decreased cognitive ability due to mental deterioration, VRT may cause further aggravation and discomfort. Moreover, lack of technological understanding and insufficient physical ability may also make older individuals apprehensive of VRT, making it harder to implement as a therapeutic intervention [67]. In addition to user concern, further research is necessary to consolidate the benefits of VRT so to make it a more accepted long-term intervention within the medical field [68]. In the context of broader future applications of VRT, as well as being a potential treatment for psychological ailments in older people, VRT can also be suggested for other age groups—including children, adolescents, and middle-aged individuals. A recent study by Mesa-Gresa and colleagues yielded positive results for the use of VR for autism and phobias in children and adolescents, exemplifying the extent to which VRT can aid in psychological rehabilitation in various populations [69]. This study found that VRT increased the emotional understanding and enhanced the communication skills of individuals on the autism spectrum. With this, prospects for the future application of VRT for psychological disorders are endless.
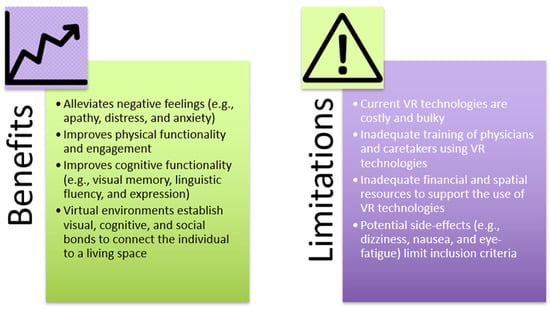
Figure 2.
Potential benefits and limitations of the use of VRT in LTCFs.
Numerous studies support that VR provides an opportunity for LTCF residents to adapt to their new environments in a controlled atmosphere, ultimately alleviating distress and subsequent psychological turmoil, and instead promoting positive feelings and emotions. Virtual environments can elicit positive psychological responses due to their ability to establish the necessary visual, cognitive, and social bonds to connect the individual to the living space. The neuroscientific influences of these virtual environments that simulate real structural environments are profound. The National Human Activity Pattern Survey finds that approximately 90% of an individual’s life is spent within a built system [70]. Built systems are necessary for one’s social, mental, and physical development—ultimately influencing one’s perception and psychological state. As the process of aging starts to deteriorate the cognitive mind, the ability to emotionally connect to a new environment is also hindered, contributing to social exclusion and depression in older individuals. Hence, social planners, architects, neuroscientists, and psychologists must collaborate when designing infrastructures catered to older people due to the influence of these structured environments on psychological well-being [71]. To maximize the positive effects of VRT, proper infrastructure for VR equipment and adequate training of healthcare providers are necessary. Educational programs to destigmatize VR may also prove beneficial for establishing end user trust [67]. Finally, further quantitative and qualitative research is needed in the fields of environmental psychology and environmental neuroscience to explore and substantiate the potential of VR technology in emotional and mental health.
Author Contributions
Conceptualization, M.A.; literature review and resources, K.Z.; writing—original draft preparation, K.Z., A.D., M.S.Y., S.H., H.E.-H. and M.A.; writing—review and editing, K.Z., A.D., M.S.Y., S.H., H.E.-H. and M.A.; figure preparation and editing, K.Z.; supervision, K.Z., S.H., H.E.-H. and M.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Virginia Commonwealth University, grant number FRG 2020/1729.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Li, A.; Montano, Z.; Chen, V.J.; Gold, J.I. Virtual reality and pain management: Current trends and future directions. Pain Manag. 2011, 1, 147–157. [Google Scholar] [CrossRef]
- Bouchard, S.; Rizzo, A. Applications of Virtual Reality in Clinical Psychology and Clinical Cognitive Neuroscience–An Introduction. In Virtual Reality for Psychological and Neurocognitive Interventions; Rizzo, A., Bouchard, S., Eds.; Springer: New York, NY, USA, 2019; pp. 1–13. [Google Scholar] [CrossRef]
- Valmaggia, L.R.; Latif, L.; Kempton, M.J.; Rus-Calafell, M. Virtual reality in the psychological treatment for mental health problems: An systematic review of recent evidence. Psychiatry Res. 2016, 236, 189–195. [Google Scholar] [CrossRef]
- Norcross, J.C.; Pfund, R.A.; Prochaska, J.O. Psychotherapy in 2022: A Delphi poll on its future. Prof. Psychol. Res. Pract. 2013, 44, 363–370. [Google Scholar] [CrossRef]
- Rothbaum, B.O.; Hodges, L.F.; Kooper, R.; Opdyke, D.; Williford, J.S.; North, M. Effectiveness of computer-generated (virtual reality) graded exposure in the treatment of acrophobia. Am. J. Psychiatry 1995, 152, 626–628. [Google Scholar] [CrossRef]
- Park, M.J.; Kim, D.J.; Lee, U.; Na, E.J.; Jeon, H.J. A Literature Overview of Virtual Reality (VR) in Treatment of Psychiatric Disorders: Recent Advances and Limitations. Front. Psychiatry 2019, 10, 505. [Google Scholar] [CrossRef] [PubMed]
- Aging, Autonomy, and Architecture: Advances in Assisted Living; Schwarz, B., Brent, R., Eds.; The Johns Hopkins University Press: Baltimore, MD, USA, 1999. [Google Scholar]
- Kopec, D. Environmental Psychology for Design, 1st ed.; Fairchild Books: New York, NY, USA, 2012. [Google Scholar]
- Ortman, J.M.; Velkoff, V.A.; Hogan, H. An Aging Nation: The Older Population in the United States; US Census Bureau: Suitland, MD, USA, 2014. [Google Scholar]
- Choi, N.G.; Ransom, S.; Wyllie, R.J. Depression in older nursing home residents: The influence of nursing home environmental stressors, coping, and acceptance of group and individual therapy. Aging Ment. Health 2008, 12, 536–547. [Google Scholar] [CrossRef] [PubMed]
- De Mendonça Lima, C.A.; Ivbijaro, G. Mental health and wellbeing of older people: Opportunities and challenges. Ment. Health Fam. Med. 2013, 10, 125–127. [Google Scholar]
- Roy, N.; Dube, R.; Despres, C.; Freitas, A.; Legare, F. Choosing between staying at home or moving: A systematic review of factors influencing housing decisions among frail older adults. PLoS ONE 2018, 13, e0189266. [Google Scholar] [CrossRef]
- Dictionary.com. Nostalgia. Available online: https://www.dictionary.com/browse/nostalgia#:~:text=a%20wistful%20desire%20to%20return,that%20elicits%20or%20displays%20nostalgia (accessed on 28 February 2021).
- Environmental Gerontology: Making Meaningful Places in Old Age; Rowles, G.D., Bernard, M., Eds.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Place Attachment: Advances in Theory, Methods and Applications; Manzo, L.C., Devine-Wright, P., Eds.; Routledge Inc.: London, UK, 2014. [Google Scholar]
- Scannell, L.; Gifford, R. Defining place attachment: A tripartite organizing framework. J. Environ. Psychol. 2010, 30, 1–10. [Google Scholar] [CrossRef]
- Wright, C.J. Homelike Variables and Rates of Depression among Assisted Living Facility Residents; St. Catherine University: St. Paul, MN, USA, 2014. [Google Scholar]
- Schumacher, K.L.; Jones, P.S.; Meleis, A.I. Helping Elderly Persons in Transition: A Framework for Research and Practice. In Life Transitions in the Older Adult: Issues for Nurses and Other Health Professionals; Swanson, E.A., Tripp-Reimer, T., Eds.; Springer: New York, NY, USA, 1999; pp. 1–26. [Google Scholar]
- Stevens, A.K.; Raphael, H.; Green, S.M. A qualitative study of older people with minimal care needs experiences of their admission to a nursing home with Registered Nurse care. Qual. Ageing Older Adults 2015, 16, 94–105. [Google Scholar] [CrossRef]
- Scannell, L.; Gifford, R. The experienced psychological benefits of place attachment. J. Environ. Psychol. 2017, 51, 256–269. [Google Scholar] [CrossRef]
- Abela, J.R.; D’Alessandro, D.U. Beck’s cognitive theory of depression: A test of the diathesis-stress and causal mediation components. Br. J. Clin. Psychol. 2002, 41, 111–128. [Google Scholar] [CrossRef]
- Singh, M.K.; Gotlib, I.H. The neuroscience of depression: Implications for assessment and intervention. Behav. Res. Ther. 2014, 62, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Kendler, K.S.; Karkowski, L.M.; Prescott, C.A. Causal relationship between stressful life events and the onset of major depression. Am. J. Psychiatry 1999, 156, 837–841. [Google Scholar] [CrossRef]
- Pariante, C.M.; Lightman, S.L. The HPA axis in major depression: Classical theories and new developments. Trends Neurosci. 2008, 31, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Cowen, P.J. Not fade away: The HPA axis and depression. Psychol. Med. 2010, 40, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Nutt, D.; Demyttenaere, K.; Janka, Z.; Aarre, T.; Bourin, M.; Canonico, P.L.; Carrasco, J.L.; Stahl, S. The other face of depression, reduced positive affect: The role of catecholamines in causation and cure. J. Psychopharmacol. 2007, 21, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.S.; Oeltzschner, G.; Gould, N.F.; Leoutsakos, J.S.; Nassery, N.; Joo, J.H.; Kraut, M.A.; Edden, R.A.E.; Barker, P.B.; Wijtenburg, S.A.; et al. Neurotransmitters and Neurometabolites in Late-Life Depression: A Preliminary Magnetic Resonance Spectroscopy Study at 7T. J. Affect. Disord. 2021, 279, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Vreeburg, S.A.; Hoogendijk, W.J.; van Pelt, J.; Derijk, R.H.; Verhagen, J.C.; van Dyck, R.; Smit, J.H.; Zitman, F.G.; Penninx, B.W. Major depressive disorder and hypothalamic-pituitary-adrenal axis activity: Results from a large cohort study. Arch. Gen. Psychiatry 2009, 66, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Holsen, L.M.; Lancaster, K.; Klibanski, A.; Whitfield-Gabrieli, S.; Cherkerzian, S.; Buka, S.; Goldstein, J.M. HPA-axis hormone modulation of stress response circuitry activity in women with remitted major depression. Neuroscience 2013, 250, 733–742. [Google Scholar] [CrossRef]
- Krogh, J.; Videbech, P.; Renvillard, S.G.; Garde, A.H.; Jorgensen, M.B.; Nordentoft, M. Cognition and HPA axis reactivity in mildly to moderately depressed outpatients: A case-control study. Nord. J. Psychiatry 2012, 66, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.; Gallagher, P.; Del-Estal, D.; Hearn, A.; Ferrier, I.N.; Young, A.H. Hypothalamic-pituitary-adrenal axis function in patients with chronic depression. Psychol. Med. 2002, 32, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Stetler, C.; Miller, G.E. Depression and hypothalamic-pituitary-adrenal activation: A quantitative summary of four decades of research. Psychosom. Med. 2011, 73, 114–126. [Google Scholar] [CrossRef]
- Nandam, L.S.; Brazel, M.; Zhou, M.; Jhaveri, D.J. Cortisol and Major Depressive Disorder-Translating Findings from Humans to Animal Models and Back. Front. Psychiatry 2019, 10, 974. [Google Scholar] [CrossRef] [PubMed]
- Peacock, B.N.; Scheiderer, D.J.; Kellermann, G.H. Biomolecular aspects of depression: A retrospective analysis. Compr. Psychiatry 2017, 73, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Strawbridge, R.; Young, A.H.; Cleare, A.J. Biomarkers for depression: Recent insights, current challenges and future prospects. Neuropsychiatr. Dis. Treat. 2017, 13, 1245–1262. [Google Scholar] [CrossRef] [PubMed]
- Maurer, D.M.; Raymond, T.J.; Davis, B.N. Depression: Screening and diagnosis. Am. Fam. Physician 2018, 98, 508–515. [Google Scholar] [PubMed]
- Eaton, W.W.; Neufeld, K.; Chen, L.S.; Cai, G. A comparison of self-report and clinical diagnostic interviews for depression: Diagnostic interview schedule and schedules for clinical assessment in neuropsychiatry in the Baltimore epidemiologic catchment area follow-up. Arch. Gen. Psychiatry 2000, 57, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Chun, A.; Reinhardt, J.P.; Ramirez, M.; Ellis, J.M.; Silver, S.; Burack, O.; Eimicke, J.P.; Cimarolli, V.; Teresi, J.A. Depression recognition and capacity for self-report among ethnically diverse nursing homes residents: Evidence of disparities in screening. J. Clin. Nurs. 2017, 26, 4915–4926. [Google Scholar] [CrossRef]
- Goodarzi, Z.S.; Mele, B.S.; Roberts, D.J.; Holroyd-Leduc, J. Depression Case Finding in Individuals with Dementia: A Systematic Review and Meta-Analysis. J. Am. Geriatr. Soc. 2017, 65, 937–948. [Google Scholar] [CrossRef]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef]
- Alexopoulos, G.S.; Abrams, R.C.; Young, R.C.; Shamoian, C.A. Cornell scale for depression in dementia. Biol. Psychiatry 1988, 23, 271–284. [Google Scholar] [CrossRef]
- Balsamo, M.; Cataldi, F.; Carlucci, L.; Padulo, C.; Fairfield, B. Assessment of late-life depression via self-report measures: A review. Clin. Interv. Aging 2018, 13, 2021–2044. [Google Scholar] [CrossRef] [PubMed]
- Most, E.I.; Scheltens, P.; Van Someren, E.J. Prevention of depression and sleep disturbances in elderly with memory-problems by activation of the biological clock with light—A randomized clinical trial. Trials 2010, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Drageset, J.; Eide, G.E.; Ranhoff, A.H. Anxiety and depression among nursing home residents without cognitive impairment. Scand. J. Caring Sci. 2013, 27, 872–881. [Google Scholar] [CrossRef]
- Conejero, I.; Olie, E.; Courtet, P.; Calati, R. Suicide in older adults: Current perspectives. Clin. Interv. Aging 2018, 13, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Schimmele, C.M.; Chappell, N.L. Aging and late-life depression. J. Aging Health 2012, 24, 3–28. [Google Scholar] [CrossRef]
- Hickman, S.E.; Barrick, A.L.; Williams, C.S.; Zimmerman, S.; Connell, B.R.; Preisser, J.S.; Mitchell, C.M.; Sloane, P.D. The effect of ambient bright light therapy on depressive symptoms in persons with dementia. J. Am. Geriatr. Soc. 2007, 55, 1817–1824. [Google Scholar] [CrossRef]
- Huang, Y.; Carpenter, I. Identifying elderly depression using the Depression Rating Scale as part of comprehensive standardised care assessment in nursing homes. Aging Ment. Health 2011, 15, 1045–1051. [Google Scholar] [CrossRef]
- Guinjoan, S.M.; Bernabo, J.L.; Cardinali, D.P. Cardiovascular tests of autonomic function and sympathetic skin responses in patients with major depression. J. Neurol. Neurosurg. Psychiatry 1995, 59, 299–302. [Google Scholar] [CrossRef]
- Borrione, L.; Brunoni, A.R.; Sampaio-Junior, B.; Aparicio, L.M.; Kemp, A.H.; Bensenor, I.; Lotufo, P.A.; Fraguas, R. Associations between symptoms of depression and heart rate variability: An exploratory study. Psychiatry Res. 2018, 262, 482–487. [Google Scholar] [CrossRef]
- Dascal, J.; Reid, M.; Ishak, W.W.; Spiegel, B.; Recacho, J.; Rosen, B.; Danovitch, I. Virtual reality and medical inpatients: A systematic review of randomized, controlled trials. Innov. Clin. Neurosci. 2017, 14, 14–21. [Google Scholar]
- Madrigal, E.; Prajapati, S.; Hernandez-Prera, J.C. Introducing a Virtual Reality Experience in Anatomic Pathology Education. Am. J. Clin. Pathol. 2016, 146, 462–468. [Google Scholar] [CrossRef]
- Pottle, J. Virtual reality and the transformation of medical education. Future Healthc. J. 2019, 6, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Klein, P.; Uhlig, M.; Will, H. The Touch and Feel of the Past—Using Haptic and VR Artefacts to Enrich Reminiscence Therapy for People with Dementia. Technologies 2018, 6, 104. [Google Scholar] [CrossRef]
- Schultheis, M.T.; Rizzo, A.A. The application of virtual reality technology in rehabilitation. Rehabil. Psychol. 2001, 46, 296–311. [Google Scholar] [CrossRef]
- Yang, J.E.; Lee, T.Y.; Kim, J.K. The effect of a VR exercise program on falls and depression in the elderly with mild depression in the local community. J. Phys. Ther. Sci. 2017, 29, 2157–2159. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chan, J.Y.C.; Chan, T.K.; Wong, M.P.F.; Cheung, R.S.M.; Yiu, K.K.L.; Tsoi, K.K.F. Effects of virtual reality on moods in community older adults. A multicenter randomized controlled trial. Int. J. Geriatr. Psychiatry 2020, 35, 926–933. [Google Scholar] [CrossRef]
- Graf, L.; Liszio, S.; Masuch, M. Playing in virtual nature: Improving mood of elderly people using VR technology. In Proceedings of the Conference on Mensch und Computer (MuC ’20), Munich, Germany, 6 September 2020; pp. 155–164. [Google Scholar]
- Baños, R.M.; Etchemendy, E.; Castilla, D.; García-Palacios, A.; Quero, S.; Botella, C. Positive mood induction procedures for virtual environments designed for elderly people. Interact. Comput. 2012, 24, 131–138. [Google Scholar] [CrossRef]
- Barsasella, D.; Liu, M.F.; Malwade, S.; Galvin, C.J.; Dhar, E.; Chang, C.C.; Li, Y.J.; Syed-Abdul, S. Effects of Virtual Reality Sessions on the Quality of Life, Happiness, and Functional Fitness among the Older People: A Randomized Controlled Trial from Taiwan. Comput. Methods Programs Biomed. 2020. [Google Scholar] [CrossRef]
- Gamito, P.; Oliveira, J.; Alves, C.; Santos, N.; Coelho, C.; Brito, R. Virtual Reality-Based Cognitive Stimulation to Improve Cognitive Functioning in Community Elderly: A Controlled Study. Cyberpsychol. Behav. Soc. Netw. 2020, 23, 150–156. [Google Scholar] [CrossRef]
- Brimelow, R.E.; Dawe, B.; Dissanayaka, N. Preliminary Research: Virtual Reality in Residential Aged Care to Reduce Apathy and Improve Mood. Cyberpsychol. Behav. Soc. Netw. 2020, 23, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Saredakis, D.; Keage, H.A.; Corlis, M.; Loetscher, T. Using Virtual Reality to Improve Apathy in Residential Aged Care: Mixed Methods Study. J. Med. Internet Res. 2020, 22, e17632. [Google Scholar] [CrossRef] [PubMed]
- D’Cunha, N.M.; Isbel, S.T.; Frost, J.; Fearon, A.; McKune, A.J.; Naumovski, N.; Kellett, J. Effects of a virtual group cycling experience on people living with dementia: A mixed method pilot study. Dement. Lond. 2020. [Google Scholar] [CrossRef]
- Lavoie, R.; Main, K.; King, C.; King, D. Virtual experience, real consequences: The potential negative emotional consequences of virtual reality gameplay. Virtual Real. 2020, 25, 69–81. [Google Scholar] [CrossRef]
- Garrett, B.; Taverner, T.; Gromala, D.; Tao, G.; Cordingley, E.; Sun, C. Virtual Reality Clinical Research: Promises and Challenges. JMIR Serious Games 2018, 6, e10839. [Google Scholar] [CrossRef]
- Baniasadi, T.; Ayyoubzadeh, S.M.; Mohammadzadeh, N. Challenges and Practical Considerations in Applying Virtual Reality in Medical Education and Treatment. Oman Med. J. 2020, 35, e125. [Google Scholar] [CrossRef]
- Wiederhold, B.K.; Riva, G. Virtual Reality Therapy: Emerging Topics and Future Challenges. Cyberpsychol. Behav. Soc. Netw. 2019, 22, 3–6. [Google Scholar] [CrossRef]
- Mesa-Gresa, P.; Gil-Gomez, H.; Lozano-Quilis, J.A.; Gil-Gomez, J.A. Effectiveness of Virtual Reality for Children and Adolescents with Autism Spectrum Disorder: An Evidence-Based Systematic Review. Sensors 2018, 18, 2486. [Google Scholar] [CrossRef]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V.; Hern, S.C.; Engelmann, W.H. The National Human Activity Pattern Survey (NHAPS): A resource for assessing exposure to environmental pollutants. J. Expo. Anal. Environ. Epidemiol. 2001, 11, 231–252. [Google Scholar] [CrossRef]
- Chiamulera, C.; Ferrandi, E.; Benvegnu, G.; Ferraro, S.; Tommasi, F.; Maris, B.; Zandonai, T.; Bosi, S. Virtual Reality for Neuroarchitecture: Cue Reactivity in Built Spaces. Front. Psychol. 2017, 8, 185. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).