Under-Recognition of Fractures as Osteoporosis Indicators
Abstract
1. Introduction
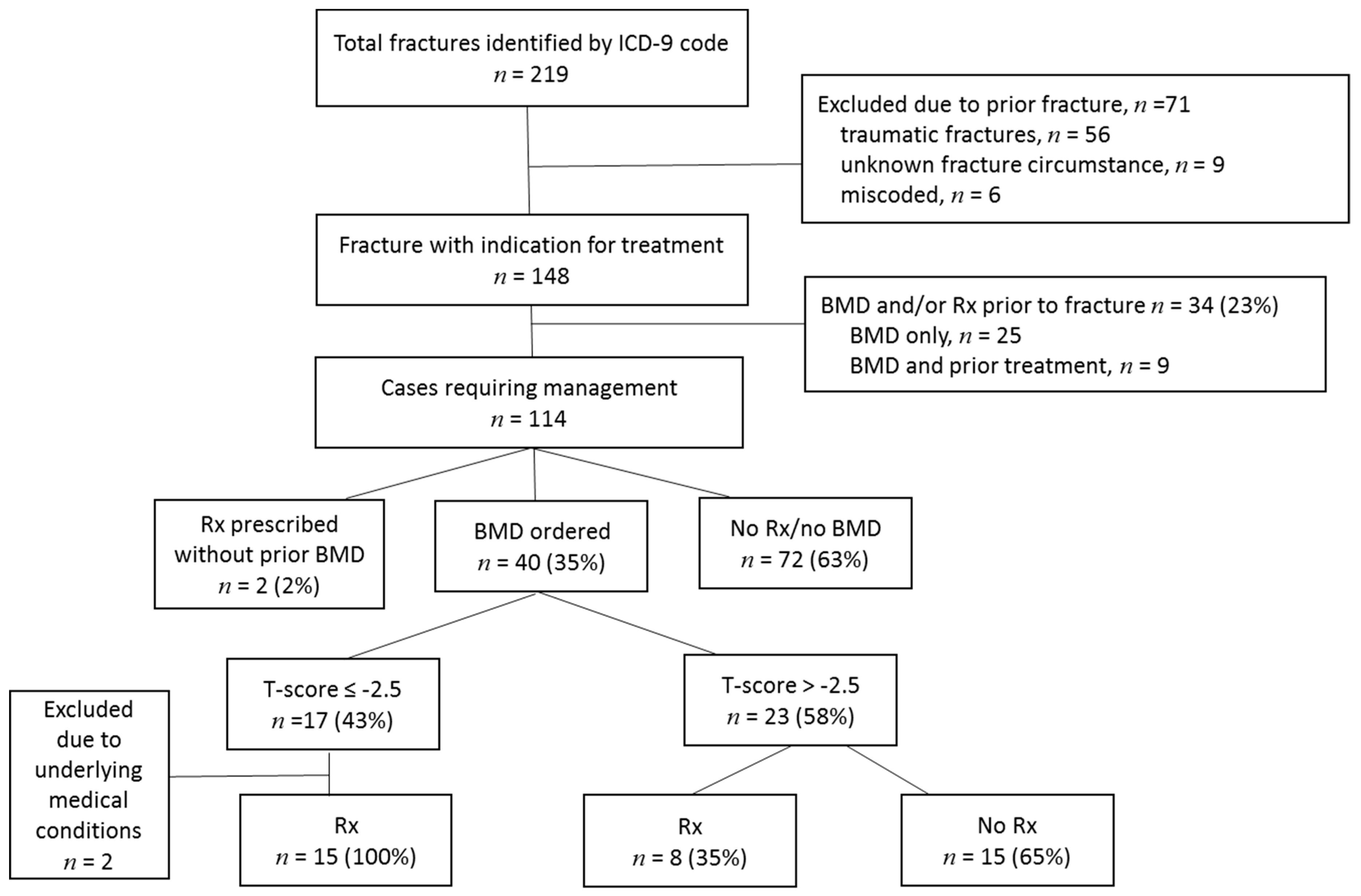
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Freemantle, N.; Cooper, C.; Diez-Perez, A.; Gitlin, M.; Radcliffe, H.; Shepherd, S.; Roux, C. Results of indirect and mixed treatment comparison of fracture efficacy for osteoporosis treatments: A meta-analysis. Osteoporos. Int. 2013, 24, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Cummings, S.R.; Black, D.M.; Thompson, D.E.; Applegate, W.B.; Barrett-Connor, E.; Musliner, T.A.; Palermo, L.; Prineas, R.; Rubin, S.M.; Scott, J.C.; et al. Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: Results from the Fracture Intervention Trial. JAMA 1998, 280, 2077–2082. [Google Scholar] [CrossRef] [PubMed]
- Black, D.M.; Delmas, P.D.; Eastell, R.; Reid, I.R.; Boonen, S.; Cauley, J.A.; Cosman, F.; Lakatos, P.; Leung, P.C.; Man, Z.; et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N. Engl. J. Med. 2007, 356, 1809–1822. [Google Scholar] [CrossRef] [PubMed]
- Cummings, S.R.; San Martin, J.; McClung, M.R.; Siris, E.S.; Eastell, R.; Reid, I.R.; Delmas, P.; Zoog, H.B.; Austin, M.; Wang, A.; et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 2009, 361, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Kiebzak, G.M.; Beinart, G.A.; Perser, K.; Ambrose, C.G.; Siff, S.J.; Heggeness, M.H. Undertreatment of osteoporosis in men with hip fracture. Arch. Intern. Med. 2002, 162, 2217–2222. [Google Scholar] [CrossRef] [PubMed]
- Dhanwal, D.K.; Dennison, E.M.; Harvey, N.C.; Cooper, C. Epidemiology of hip fracture: Worldwide geographic variation. Indian J. Orthop. 2011, 45, 15–22. [Google Scholar] [CrossRef]
- Abrahamsen, B.; van Staa, T.; Ariely, R.; Olson, M.; Cooper, C. Excess mortality following hip fracture: A systematic epidemiological review. Osteoporos. Int. 2009, 20, 1633–1650. [Google Scholar] [CrossRef]
- Huntjens, K.M.; Kosar, S.; van Geel, T.A.; Geusens, P.P.; Willems, P.; Kessels, A.; Winkens, B.; Brink, P.; van Helden, S. Risk of subsequent fracture and mortality within 5 years after a non-vertebral fracture. Osteoporos. Int. 2010, 21, 2075–2082. [Google Scholar] [CrossRef]
- Empana, J.P.; Dargent-Molina, P.; Breart, G.; Group, E. Effect of hip fracture on mortality in elderly women: The EPIDOS prospective study. J. Am. Geriatr. Soc. 2004, 52, 685–690. [Google Scholar] [CrossRef]
- Solomon, D.H.; Johnston, S.S.; Boytsov, N.N.; McMorrow, D.; Lane, J.M.; Krohn, K.D. Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J. Bone Miner. Res. 2014, 29, 1929–1937. [Google Scholar] [CrossRef]
- Kolata, G. Fearing Rrare Side Eeffects, Millions Take Their Chances With Osteoporosis. New York Times. 1 June 2016. Available online: https://www.nytimes.com/2016/06/02/health/osteoporosis (accessed on 1 July 2018).
- Center, J.R.; Bliuc, D.; Nguyen, T.V.; Eisman, J.A. Risk of subsequent fracture after low-trauma fracture in men and women. JAMA 2007, 297, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Eisman, J.A.; Bogoch, E.R.; Dell, R.; Harrington, J.T.; McKinney, R.E., Jr.; McLellan, A.; Mitchell, P.J.; Silverman, S.; Singleton, R.; Siris, E.; et al. Making the first fracture the last fracture: ASBMR task force report on secondary fracture prevention. J. Bone Miner. Res. 2012, 27, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Capture the Fracture. Available online: http://www.capturethe-fracture.org/programme-overview (accessed on 1 July 2018).
- Ekman, E.F. The role of the orthopaedic surgeon in minimizing mortality and morbidity associated with fragility fractures. J. Am. Acad. Orthop. Surg. 2010, 18, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Profile of Veterans: 2014, Data from the American Community Survey; United States Department of Veterans Affairs. Available online: https://www.va.gov/vetdata/docs/SpecialReports/Profile_of_Veterans_2014.pdf (accessed on 1 July 2018).
- Office of Inspector General (OIG). Management of osteoporosis in veterans with fractures. Fed. Regist. 2010. Available online: http://www.va.gov/oig/publications/reports-list.asp (accessed on 1 July 2018).
- Cosman, F.; de Beur, S.J.; LeBoff, M.S.; Lewiecki, E.M.; Tanner, B.; Randall, S.; Lindsay, R. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Osteoporos. Int. 2014, 25, 2359–2381. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A. Osteoporosis and osteopenia. J. Bone Miner. Res. 1990, 5, 209–211. [Google Scholar] [CrossRef] [PubMed]
- De Laet, C.E.; van Hout, B.A.; Burger, H.; Hofman, A.; Pols, H.A. Bone density and risk of hip fracture in men and women: Cross sectional analysis. BMJ 1997, 315, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Schroder, H.M.; Petersen, K.K.; Erlandsen, M. Occurrence and incidence of the second hip fracture. Clin. Orthop. Relat. Res. 1993, 166–169. [Google Scholar]
- Recker, R.R.; Hinders, S.; Davies, K.M.; Heaney, R.P.; Stegman, M.R.; Lappe, J.M.; Kimmel, D.B. Correcting calcium nutritional deficiency prevents spine fractures in elderly women. J. Bone Miner. Res. 1996, 11, 1961–1966. [Google Scholar] [CrossRef]
- Stone, N.J.; Robinson, J.G.; Lichtenstein, A.H.; Bairey Merz, C.N.; Blum, C.B.; Eckel, R.H.; Goldberg, A.C.; Gordon, D.; Levy, D.; Lloyd-Jones, D.M.; et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129 (Suppl. 2), S1–S45. [Google Scholar] [CrossRef]
- Chapuy, M.C.; Arlot, M.E.; Duboeuf, F.; Brun, J.; Crouzet, B.; Arnaud, S.; Delmas, P.D.; Meunier, P.J. Vitamin D3 and calcium to prevent hip fractures in elderly women. N. Engl. J. Med. 1992, 327, 1637–1642. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Harris, S.S.; Krall, E.A.; Dallal, G.E. Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. N. Engl. J. Med. 1997, 337, 670–676. [Google Scholar] [CrossRef]
- Black, D.M.; Cummings, S.R.; Karpf, D.B.; Cauley, J.A.; Thompson, D.E.; Nevitt, M.C.; Bauer, D.C.; Genant, H.K.; Haskell, W.L.; Marcus, R.; et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 1996, 348, 1535–1541. [Google Scholar] [CrossRef]
- Lyles, K.W.; Colon-Emeric, C.S.; Magaziner, J.S.; Adachi, J.D.; Pieper, C.F.; Mautalen, C.; Hyldstrup, L.; Recknor, C.; Nordsletten, L.; Moore, K.A.; et al. Zoledronic acid and clinical fractures and mortality after hip fracture. N. Engl. J. Med. 2007, 357, 1799–1809. [Google Scholar] [CrossRef] [PubMed]
- Reginster, J.; Minne, H.W.; Sorensen, O.H.; Hooper, M.; Roux, C.; Brandi, M.L.; Lund, B.; Ethgen, D.; Pack, S.; Roumagnac, I.; et al. Randomized trial of the effects of risedronate on vertebral fractures in women with established postmenopausal osteoporosis. Vertebral Efficacy with Risedronate Therapy (VERT) Study Group. Osteoporos. Int. 2000, 11, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Black, D.M.; Reid, I.R.; Cauley, J.A.; Cosman, F.; Leung, P.C.; Lakatos, P.; Lippuner, K.; Cummings, S.R.; Hue, T.F.; Mukhopadhyay, A.; et al. The effect of 6 versus 9 years of zoledronic acid treatment in osteoporosis: A randomized second extension to the HORIZON-Pivotal Fracture Trial (PFT). J. Bone Miner. Res. 2015, 30, 934–944. [Google Scholar] [CrossRef] [PubMed]
- Camacho, P.M.; Petak, S.M.; Binkley, N.; Clarke, B.L.; Harris, S.T.; Hurley, D.L.; Kleerekoper, M.; Lewiecki, E.M.; Miller, P.D.; Narula, H.S.; et al. American Association of Clinical Endocrinologists and American College of Endocrinology Clinical Practice Guidelines for the Diagnosis and Treatment of Postmenopausal Osteoporosis—2016. Endocr. Pract. 2016, 22 (Suppl. 4), 1–42. [Google Scholar] [CrossRef]
- Watts, N.B.; Adler, R.A.; Bilezikian, J.P.; Drake, M.T.; Eastell, R.; Orwoll, E.S.; Finkelstein, J.S.; Endocrine, S. Osteoporosis in men: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2012, 97, 1802–1822. [Google Scholar] [CrossRef]
- Gupta, M.J.; Shah, S.; Peterson, S.; Baim, S. Rush Fracture Liaison Service for capturing “missed opportunities” to treat osteoporosis in patients with fragility fractures. Osteoporos. Int. 2018, 29, 1861–1874. [Google Scholar] [CrossRef]
- Lee, R.H.; Lyles, K.W.; Pearson, M.; Barnard, K.; Colon-Emeric, C. Osteoporosis screening and treatment among veterans with recent fracture after implementation of an electronic consult service. Calcif. Tissue Int. 2014, 94, 659–664. [Google Scholar] [CrossRef]
- Singh, I.; Fletcher, R.; Scanlon, L.; Tyler, M.; Aithal, S. A quality improvement initiative on the management of osteoporosis in older people with Parkinsonism. BMJ Qual. Improv. Rep. 2016, 5. [Google Scholar] [CrossRef]
- Bliuc, D.; Eisman, J.A.; Center, J.R. A randomized study of two different information-based interventions on the management of osteoporosis in minimal and moderate trauma fractures. Osteoporos. Int. 2006, 17, 1309–1317. [Google Scholar] [CrossRef] [PubMed]
- Cranney, A.; Lam, M.; Ruhland, L.; Brison, R.; Godwin, M.; Harrison, M.M.; Harrison, M.B.; Anastassiades, T.; Grimshaw, J.M.; Graham, I.D. A multifaceted intervention to improve treatment of osteoporosis in postmenopausal women with wrist fractures: A cluster randomized trial. Osteoporos. Int. 2008, 19, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Cuddihy, M.T.; Amadio, P.C.; Gabriel, S.E.; Pankratz, V.S.; Kurland, R.L.; Melton, L.J., 3rd. A prospective clinical practice intervention to improve osteoporosis management following distal forearm fracture. Osteoporos. Int. 2004, 15, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Johnell, O.; Kanis, J. Epidemiology of osteoporotic fractures. Osteoporos. Int. 2005, 16 (Suppl. 2), S3–S7. [Google Scholar] [CrossRef]
- Elliot-Gibson, V.; Bogoch, E.R.; Jamal, S.A.; Beaton, D.E. Practice patterns in the diagnosis and treatment of osteoporosis after a fragility fracture: A systematic review. Osteoporos. Int. 2004, 15, 767–778. [Google Scholar] [CrossRef] [PubMed]
| Variable | n (%) |
|---|---|
| Age, mean (SD) | 68 (9.5) |
| Gender, male (n, %) | 105 (92) |
| Race/ethnicity (n, %) | |
| Non-Hispanic black | 24 (21) |
| Non-Hispanic white | 64 (56) |
| Hispanic white | 20 (18) |
| Unknown | 6 (5) |
| Comorbidities (n, %) | |
| Diabetes | 21 (18) |
| Tobacco | 17 (15) |
| COPD | 11 (10) |
| Alcohol abuse | 17 (11) |
| Seizure disorder | 5 (4) |
| GFR < 30 mL/min per 1.73 m2 | 4 (4) |
| Fracture Type | n (%) |
|---|---|
| Rib | 21 (18) |
| Radius | 19 (17) |
| Vertebral | 17 (15) |
| Femoral neck | 17 (15) |
| Humerus | 8 (7) |
| Fibula | 8 (7) |
| Patella | 7 (6) |
| Ulna | 4 (4) |
| Tibia | 4 (4) |
| Sacrum | 3 (3) |
| Clavicle | 3 (3) |
| Femoral shaft | 3 (3) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lagari, V.S.; Al-Yatama, F.; Rodriguez, G.; Berger, H.R.; Levis, S. Under-Recognition of Fractures as Osteoporosis Indicators. Geriatrics 2019, 4, 9. https://doi.org/10.3390/geriatrics4010009
Lagari VS, Al-Yatama F, Rodriguez G, Berger HR, Levis S. Under-Recognition of Fractures as Osteoporosis Indicators. Geriatrics. 2019; 4(1):9. https://doi.org/10.3390/geriatrics4010009
Chicago/Turabian StyleLagari, Violet S., Fatima Al-Yatama, Gracielena Rodriguez, Hara R. Berger, and Silvina Levis. 2019. "Under-Recognition of Fractures as Osteoporosis Indicators" Geriatrics 4, no. 1: 9. https://doi.org/10.3390/geriatrics4010009
APA StyleLagari, V. S., Al-Yatama, F., Rodriguez, G., Berger, H. R., & Levis, S. (2019). Under-Recognition of Fractures as Osteoporosis Indicators. Geriatrics, 4(1), 9. https://doi.org/10.3390/geriatrics4010009




