Is a Combination of Melatonin and Amino Acids Useful to Sarcopenic Elderly Patients? A Randomized Trial
Abstract
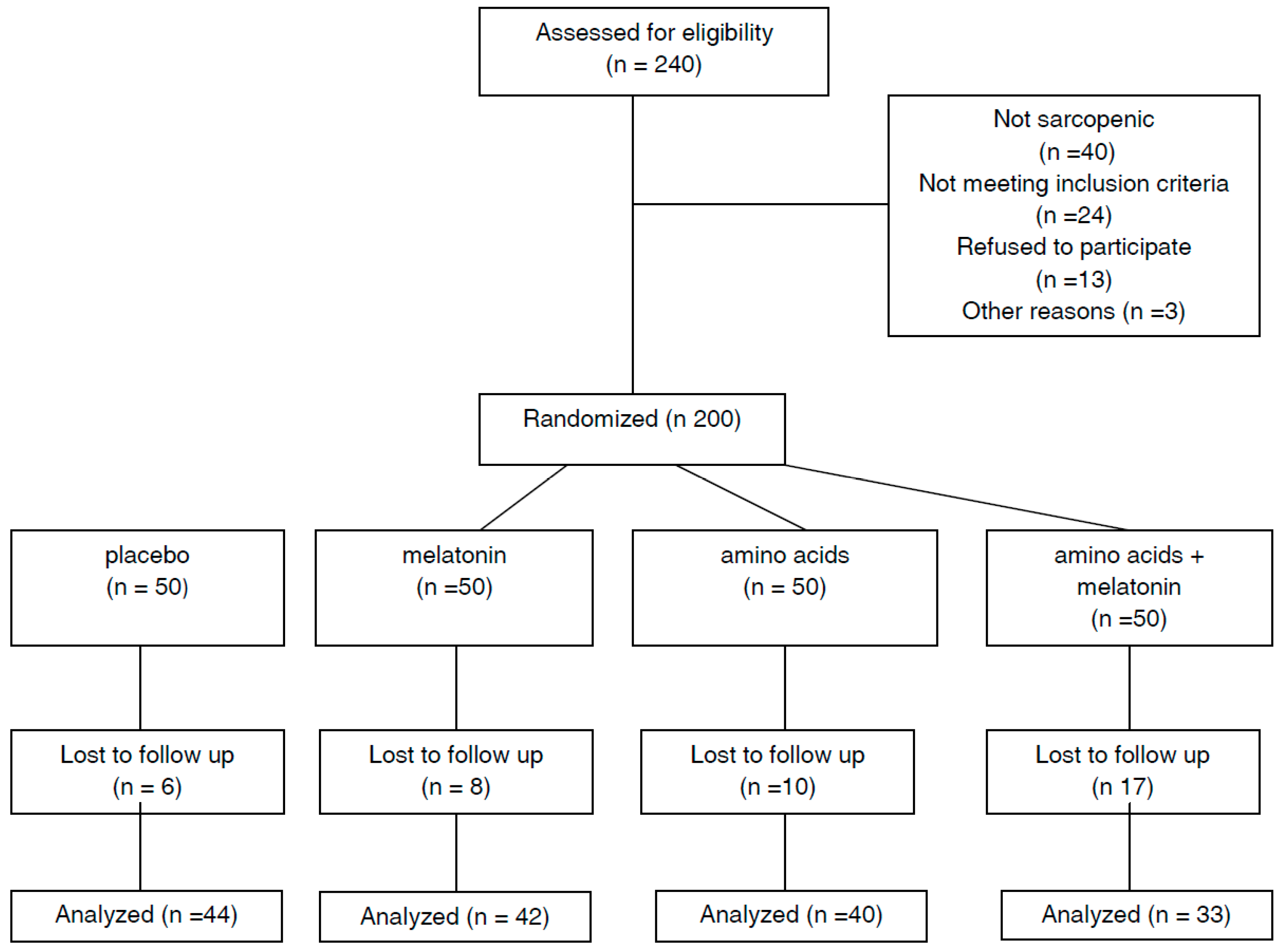
:1. Introduction
2. Materials and Methods
2.1. Setting
2.2. Study Population
2.3. Observed Outcome Variables
2.3.1. Assessment of Functional Performance
2.3.2. Blood Sample Measurements
2.3.3. Assessment of Nutritional Status
Dietary Schedule
2.3.4. Assessment of Cognitive Status
2.4. Randomization
2.5. Intervention and Duration
- (1)
- Placebo: (P)
- (2)
- Melatonin (M) 1 mg/daily 30 min before going to sleep.
- (3)
- Essential amino acids (eAA) 4 g/daily every morning during breakfast
- (4)
- Essential amino acids (eAA) 4 g/daily every morning during breakfast + melatonin 1 mg/daily 30 min before going to sleep.
2.5.1. Amino Acid Supplementation
2.5.2. Melatonin Supplementation
2.5.3. Placebo Supplementation
2.6. Physical Activity
2.7. Sample Size
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, S.; Moon, C.S.; Khogali, A. Biomarkers in Parkinson’s disease (recent update). Neurochem. Int. 2013, 63, 201–229. [Google Scholar] [CrossRef] [PubMed]
- Solerte, S.B.; Gazzaruso, C.; Bonacasa, R. Nutritional supplements with oral aminoacid mixtures increases whole-body lean mass and insulin sensitivity in elderly subjects with sarcopenia. Am. J. Cardiol. 2008, 101, 69E–77E. [Google Scholar] [CrossRef] [PubMed]
- Beasley, J.M.; Shikanym, J.M.; Thomson, C.A. The role of dietary protein intake in the prevention of sarcopenia of aging. Nutr. Clin. Pract. 2013, 28, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Kim, J.H.; Lee, D.C. Urine melatonin levels are inversely associated with sarcopenia in postmenopausal women. Menopause 2014, 21, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Oner, J.; Oner, H.; Sahin, Z. Melatonin is as effective as testosterone in the prevention of soleus muscle atrophy induced by castration in rats. Anat. Rec. (Hoboken) 2008, 29, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, D.X.; Lv, J.W. Melatonin attenuates lipopolysaccharide (LPS)-induced apoptotic liver damage in D-galactosamine-sensitized mice. Toxicology 2007, 237, 49–57. [Google Scholar] [CrossRef]
- Park, J.H.; Chung, E.J.; Kwon, H.J. Protective effect of melatonin on TNF-α-induced muscle atrophy in L6 myotubes. J. Pineal. Res. 2013, 54, 417–425. [Google Scholar] [CrossRef]
- Hibaoui, Y.; Reutenauer-Patte, J.; Patthey-Vuadens, O. Melatonin improves muscle function of the dystrophic mdx5Cv mouse, a model for Duchenne muscular dystrophy. J. Pineal. Res. 2011, 51, 163–171. [Google Scholar] [CrossRef]
- Pozo, M.J.; Gomez-Pinilla, P.J.; Camello-Almaraz, C. Melatonin, a potential therapeutic agent for smooth muscle-related pathological conditions and aging. Curr. Med. Chem. 2010, 17, 4150–4165. [Google Scholar] [CrossRef]
- Chahbouni, M.; Escames, G.; Venegas, C. Melatonin treatment normalizes plasma pro-inflammatory cytokines and nitrosative/oxidative stress in patients suffering from Duchenne muscular dystrophy. J. Pineal. Res. 2010, 48, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Coto-Montes, A.; Boga, J.A.; Tan, D.X. Melatonin as a Potential Agent in the Treatment of Sarcopenia. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Tuomi, T.; Nagorny, C.L.; Singh, P. Increased Melatonin Signaling Is a Risk Factor for Type 2 Diabetes. Cell Metab. 2016, 23, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Bouatia-Naji, N.; Bonnefond, A.; Cavalcanti-Proença, C. A variant near MTNR1B is associated with increased fasting plasma glucose levels and type 2 diabetes risk. Nat. Genet. 2009, 41, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, V.; Nagorny, C.L.; Erdos, M.R. Common variant in MTNR1B associated with increased risk of type 2 diabetes and impaired early insulin secretion. Nat. Genet. 2009, 41, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Prokopenko, I.; Langenberg, C.; Florez, J.C. Variants in MTNR1B influence fasting glucose levels. Nat. Genet. 2009, 41, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, A.; Ladenvall, C.; Ahluwalia, T.S. Effects of common genetic variants associated with type 2 diabetes and glycemic traits on α- and β-cell function and insulin action in humans. Diabetes 2013, 62, 2978–2983. [Google Scholar] [CrossRef] [PubMed]
- Renström, F.; Koivula, R.W.; Varga, T.V. Season-dependent associations of circadian rhythm-regulating loci (CRY1, CRY2 and MTNR1B) and glucose homeostasis: the GLACIER Study. Diabetologia 2015, 58, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Rönn, T.; Wen, J.; Yang, Z. A common variant in MTNR1B, encoding melatonin receptor 1B, is associated with type 2 diabetes and fasting plasma glucose in Han Chinese individuals. Diabetologia 2009, 52, 830–833. [Google Scholar] [CrossRef] [Green Version]
- Peschke, E. Melatonin, endocrine pancreas and diabetes. J. Pineal. Res. 2008, 44, 26–40. [Google Scholar] [CrossRef]
- Bailey, C.J.; Atkins, T.W.; Matty, A.J. Melatonin inhibition of insulin secretion in the rat and mouse. Horm. Res. 1974, 5, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Prod’homme, M.; Rieu, I.; Balage, M. Insulin and amino acids both strongly participate to the regulation of protein metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Volpi, E.; Lucidi, P.; Cruciani, G. Contribution of amino acids and insulin to protein anabolism during meal absorption. Diabetes 1996, 45, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Dardevet, D.; Rémond, D.; Peyron, M.A. Muscle wasting and resistance of muscle anabolism: the “anabolic threshold concept” for adapted nutritional strategies during sarcopenia. Sci. World J. 2012, 2012, 269531. [Google Scholar] [CrossRef]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Liperoti, R.; Russo, A. Sarcopenia as a risk factor for falls in elderly individuals: Results from the il SIRENTE study. Clin. Nutr. 2012, 31, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Spiekeman, A.M. Nutritional assessment (protein nutriture). Anal. Chem. 1995, 67, 429R–436R. [Google Scholar] [CrossRef]
- Guigoz, Y.; Vellas, B.J. Malnutrition in the elderly: The Mini Nutritional Assessment (MNA). Ther. Umsch. 1997, 54, 345–350. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 1, 189–198. [Google Scholar] [CrossRef]
- Rondanelli, M.; Klersy, C.; Terracol, G.; Talluri, J.; Maugeri, R.; Guido, D.; Faliva, M.A.; Solerte, B.S.; Fioravanti, M.; Lukaski, H.; et al. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am. J. Clin. Nutr. 2016, 103, 830–840. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.K.; Suzuki, T.; Saito, K. Effects of exercise and aminoacid supplementation on body composition and physical function in community-dwelling elderly Japanese sarcopenic women: A randomized controlled trial. J. Am. Geriatr. Soc. 2012, 60, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Trabal, J.; Forga, M.; Leyes, P. Effects of free leucine supplementation and resistance training on muscle strength and functional status in older adults: a randomized controlled trial. Clin. Interv. Aging 2015, 10, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Ispoglou, T.; White, H.; Preston, T. Double-blind, placebo-controlled pilot trial of L-Leucine-enriched amino-acid mixtures on body composition and physical performance in men and women aged 65–75 years. Eur. J. Clin. Nutr. 2016, 70, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Frankel, J.E.; Bean, J.F.; Frontera, W.R. Exercise in the elderly: Research and clinical practice. Clin. Geriatr. Med. 2006, 22, 239–256. [Google Scholar] [CrossRef] [PubMed]
- Crocker, T.; Forster, A.; Young, J. Physical rehabilitation for older people in long-term care. Cochrane Database Syst. Rev. 2013, 2, CD004294. [Google Scholar] [CrossRef]
- Fiatarone, M.A.; O’Neill, E.F.; Ryan, N.D. Exercise training and nutritional supplementation for physical frailty in very elderly people. N. Engl. J. Med. 1994, 330, 1769–1775. [Google Scholar] [CrossRef]
- Garzòn, C.; Guerrero, J.M.; Aramburu, O. Effect of melatonin administration on sleep, behavioral disorders and hypnotic drug discontinuation in the elderly: A randomized, double-blind, placebo-controlled study. Aging Clin. Exp. Res. 2009, 21, 38–42. [Google Scholar] [CrossRef]
- Buscemi, N.; Vandermeer, B.; Hooton, N. Efficacy and safety of exogenous melatonin for secondary sleep disorders and sleep disorders accompanying sleep restriction: Meta-analysis. BMJ 2006, 332, 385–393. [Google Scholar] [CrossRef]
- Brown, G.M.; Pandi-Perumal, S.R.; Trakht, I. Melatonin and its relevance to jet lag. Travel Med. Infect. Dis. 2009, 7, 69–81. [Google Scholar] [CrossRef]
- Hardeland, R.; Cardinali, D.P.; Brown, G.M. Melatonin and brain inflammaging. Prog. Neurobiol. 2015, 127, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Chang, Q.; Cai, J. Protective Effects of Melatonin on Retinal Inflammation and Oxidative Stress in Experimental Diabetic Retinopathy. Oxid Med. Cell Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Koh, P.O. Melatonin regulates nitric oxide synthase expression in ischemic brain injury. J. Vet. Med. Sci. 2008, 70, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Negi, G.; Kumar, A.; Sharma, S.S. Melatonin modulates neuroinflammation and oxidative stress in experimental diabetic neuropathy: effects on NF-kappaB and Nrf2 cascades. J. Pineal. Res. 2011, 50, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Hong, S.W.; Zheng, H.M. Melatonin ameliorates cerulein-induced pancreatitis by the modulation of nuclear erythroid 2-related factor 2 and nuclear factor-kappaB in rats. J. Pineal. Res. 2010, 48, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.F.; Wang, D.; Reiter, R.J. Melatonin attenuates lung inflammation and airway hyperreactivity induced by inhalation of aerosolized pancreatic fluid in rats. J. Pineal. Res. 2011, 50, 46–53. [Google Scholar] [CrossRef]
- Veneroso, C.; Tuñón, M.J.; González-Gallego, J.; Collado, P.S. Melatonin reduces cardiac inflammatory injury induced by acute exercise. J. Pineal. Res. 2009, 47, 184–191. [Google Scholar] [CrossRef]
- Doolen, S.; Krause, D.N.; Dubocovich, M.L. Melatonin mediates two distinct responses in vascular smooth muscle. Eur. J. Pharmacol. 1998, 345, 67–69. [Google Scholar] [CrossRef]
- Peschke, E.; Muhlbauer, E.; Musshoff, U. Receptor (MT(1)) mediated influence of melatonin on cAMP concentration and insulin secretion of rat insulinoma cells INS-1. J. Pineal. Res. 2002, 33, 63–71. [Google Scholar] [CrossRef]
- Kemp, D.M.; Ubeda, M.; Habener, J.F. Identification and functional characterization of melatonin Mel 1a receptors in pancreatic beta cells: potential role in incretin-mediated cell function by sensitization of cAMP signaling. Mol. Cell Endocrinol. 2002, 191, 157–166. [Google Scholar] [CrossRef]
- Ramracheya, R.D.; Muller, D.S.; Squires, P.E. Function and expression of melatonin receptors on human pancreatic islets. J. Pineal. Res. 2008, 44, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Soultoukis, G.A.; Partridge, L. Dietary Protein, Metabolism, and Aging. Annu. Rev. Biochem. 2016, 85, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Muscle strength: clinical and prognostic value of hand-grip dynamometry. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 465–470. [Google Scholar] [CrossRef]
- Merle, A.; Delagrange, P.H.; Renard, P.; Lesieur, D.; Cuber, J.C.; Roche, M.; Pellissier, S. Effect of melatonin on motility pattern of small intestine in rats and its inhibition by melatonin receptor antagonist S 22153. J. Pineal. Res. 2000, 29, 116–124. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Pmean ± SD | Mmean ± SD | eAAmean ± SD | eAAMmean ± SD | F | p-Value |
|---|---|---|---|---|---|---|
| General characteristics | ||||||
| Gender | Women: 31 Men: 13 | Women: 36 Men: 6 | Women: 31 Men: 9 | Women: 19 Men: 14 | X2: 8.06 | p < 0.05 |
| Age (years) | 81.86 ± 6.43 | 81.64 ± 7.04 | 80.55 ± 6.76 | 81.42 ± 8.02 | 0.28 | 0.84 |
| Mini Nutritional Assessment (Score) | 18.48 ± 3.45 | 18.31 ± 3.09 | 16.59 ± 3.79 | 17.93 ± 2.92 | 2.44 | 0.07 |
| Mini Mental State Examination (Score) | 21.43 ± 4.75 | 17.23 ± 3.38 | 21.70 ± 4.77 | 19.10 ± 6.48 | 4.92 | p < 0.05 |
| Weight (kg) | 57.20 ±10.40 | 59.72 ±11.73 | 53.24 ±8.26 | 60.34 ±12.54 | 3.45 | p < 0.05 |
| Body Mass Index (kg/m2) | 22.86 ± 0.63 | 24.04 ± 0.83 | 21.94 ± 0.72 | 24.29 ± 0.67 | 2.36 | 0.074 |
| Biochemical parameters | ||||||
| White Blood Cells (K/uL) (4.00–10.00) | 7.01 ± 2.36 | 6.72 ± 2.09 | 7.58 ± 2.14 | 6.50 ± 1.98 | 1.73 | 0.16 |
| Red Blood Cells (K/uL) (4.00–5.00) | 4.18 ± 0.58 | 4.35 ± 0.65 | 4.20 ± 0.50 | 4.29 ± 0.42 | 0.84 | 0.47 |
| Hemoglobine (g/dL) (12.0–16.0) | 12.45 ± 1.51 | 12.88 ± 1.24 | 11.92 ± 1.73 | 12.79 ± 1.48 | 3.31 | 0.02 |
| Hematrocrit (%)(36.0–46.0) | 38.06 ± 4.27 | 39.07 ± 3.71 | 36.63 ± 4.38 | 38.59 ± 4.23 | 2.53 | 0.06 |
| Iron (mcg/dL) (45–145) | 67.55 ± 27.26 | 74.49 ± 29.63 | 60.87 ± 32.39 | 64.30 ± 25.75 | 1.60 | 0.19 |
| Triglycerid (mmol/L) (<200) | 124.98 ± 57.39 | 156.51 ± 115.23 | 126.08 ± 45.48 | 98.82 ± 28.31 | 4.02 | p < 0.01 |
| Cholesterol (mmol/L) (<200) | 184.73 ± 48.74 | 211.27 ± 48.67 | 190.69 ± 52.20 | 186.82 ± 38.08 | 2.67 | 0.06 |
| Albumine (gr/dL) (4.02–4.76) | 3.61 ± 0.51 | 3.81 ± 0.40 | 3.53 ± 0.60 | 3.80 ± 0.41 | 3.20 | 0.03 |
| Creatinine (mg/dL) (0.57–1.11) | 0.87 ± 0.27 | 0.88 ± 0.28 | 0.85 ± 0.66 | 0.82 ± 0.29 | 0.20 | 0.90 |
| Glycemia (mg/dL) (70–110) | 100.38 ± 28.68 | 112.88 ± 47.33 | 97.58 ± 27.54 | 102.39 ± 29.22 | 1.51 | 0.22 |
| Eritrocyte Sedimentation rate (mm/h) <15 | 49.78 ± 30.85 | 37.68 ± 25.51 | 45.94 ± 29.21 | 31.06 ± 24.86 | 3.30 | p < 0.01 |
| Reactive C-protein (mg/dL) (<0.50) | 1.47 ± 3.18 | 0.48 ± 0.91 | 1.43 ± 2.50 | 0.88 ± 1.76 | 1.69 | 0.17 |
| Homocysetine (micromol/L) (5–12) | 19.13 ± 8.49 | 18.72 ± 7.39 | 17.33 ± 7.63 | 21.72 ± 16.37 | 0.82 | 0.49 |
| DXA measurements | ||||||
| Fat-free arm mass (grams) | 4161.43 ± 133.57 | 3833.17 ± 178.25 | 3977.85 ± 153.06 | 4103.81 ± 142.38 | 0.85 | 0.47 |
| Fat-free leg mass (grams) | 11,827.98 ± 326.76 | 11,879.43 ± 436.07 | 11,922.22 ± 374.44 | 13,297.76 ± 348.31 | 4.07 | 0.008 |
| Fat-free Mass (grams) | 39,540.35 ± 813.94 | 37,775.79 ± 1086.21 | 39,074.55 ± 932.71 | 41,570.33 ± 867.63 | 2.76 | 0.044 |
| Gynoid Fat mass (%) | 36.45 ± 1.25 | 35.08 ± 1.67 | 33.61 ± 1.43 | 34.27 ± 1.33 | 0.86 | 0.46 |
| Android Fat mass (%) | 35.09 ± 1.82 | 36.83 ± 2.44 | 30.72 ± 2.09 | 30.98 ± 1.95 | 2.00 | 0.12 |
| Fat Mass (grams) | 18,272.27 ± 1317.45 | 19,517.44 ± 1758.14 | 14,916.64 ± 1509.68 | 17,418.56 ± 1404.34 | 1.53 | 0.21 |
| Variable | Group | ∆ Change within-Group Pre Post Intervention | p-Value Within-Group | ∆ Change between Groups Pre Post Intervention | p-Value between Groups |
|---|---|---|---|---|---|
| Albumin (grams) | M vs. P: −0.39 (−0.67; 0.10) | <0.01 | |||
| P | −0.12 (−0.31; 0.07) | 0.19 | eAA vs. P: 0.08 (−0.22; 0.38) | 0.59 | |
| M | −0.44 (−0.67; −0.20) | <0.001 | eAAM vs. P: −0.22 (−0.51; 0.06) | 0.12 | |
| eAA | 0.15 (−0.16; 0.47) | 0.29 | eAA vs. M: 0.47 (0.11; 0.82) | <0.01 | |
| eAAM | −0.34 (−0.63; −0.05) | <0.05 | eAAM vs. M: 0.16 (−0.14; 0.47) | 0.29 | |
| eAAM vs. eAA: −0.30 (−0.65; 0.05) | 0.09 | ||||
| Reactive C−protein (mg/dL) | M vs. P: 0.94 (−0.83; 2.71) | 0.29 | |||
| P | −0.20 (−0.98; 0.58) | 0.6 | eAA vs. P: 0.72 (−1.19; 2.64) | 0.46 | |
| M | 0.80 (0.88; 2.49) | 0.33 | eAAM vs. P: 0.75 (−1.12; 2.63) | 0.43 | |
| eAA | 0.63 (−0.47; 1.72) | 0.22 | eAA vs. M: −0.22 (−2.29; 1.86) | 0.84 | |
| eAAM | 0.65 (−0.83; 2.12) | 0.35 | eAAM vs. M: 0.16 (−0.14; 0.47) | 0.84 | |
| eAAM vs. eAA: −0.30 (−0.65; 0.05) | 0.98 | ||||
| Fat−free arm mass (grams) | M vs. P: 333.83 (−113.85; 781.52) | 0.14 | |||
| P | 0.34 (−0.46; 1.15) | 0.38 | eAA vs. P: 397.80 (−73.99; 869.60) | 0.1 | |
| M | −0.67 (−1.47; 0.24) | 0.09 | eAAM vs. P: 424.26 (−41.08; 889.59) | 0.07 | |
| eAA | 0.45 (−0.18; 1.08) | 0.12 | eAA vs. M: 63.97 (−445.36; 573.31) | 0.8 | |
| eAAM | 0.29 (−0.15; 0.72) | 0.17 | eAAM vs. M: 90.42 (−388.49; 569.33) | 0.71 | |
| eAAM vs. eAA: 26.45 (−495.81; 548.71) | 0.92 | ||||
| Fat−free leg mass (grams) | M vs. P: −230.59 (−972.52; 511.33) | 0.54 | |||
| P | 0.14 (−0.35; 0.64) | 0.55 | eAA vs. P: 393.99 (−373.81; 11171.79) | 0.31 | |
| M | −0.06 (−0.56; 0.44) | 0.81 | eAAM vs. P: 194.67 (−578.33; 967.67) | 0.62 | |
| eAA | 0.35 (0.03; 0.67) | <0.05 | eAA vs. M: 629.58 (−200.42; 1459.59) | 0.14 | |
| eAAM | 0.04 (−0.99; 1.06) | 0.94 | eAAM vs. M: 425.26 (−353.71; 1201.23) | 0.28 | |
| eAAM vs. eAA: −204.32 (−1056.06; 647.42) | 0.64 | ||||
| Fat−free Mass (grams) | P | −233.73 (−1081.62; 614.16) | 0.58 | M vs. P: 82.16 (−1461.35; 1625.68) | 0.92 |
| 807.72 (−210.07; 1825.52) | 0.11 | eAA vs. P: 2618.49 (978.23; 4258.74) | <0.05 | ||
| M | 1794.17 (783.29; 2805.05) | <0.01 | eAAM vs. P:2190.01 (555.16; 3824.87) | <0.01 | |
| 1015.45 (139.65; 1891.25) | <0.05 | eAA vs. M: 2536.33 (766.13; 4306.53) | <0.01 | ||
| eAA | eAAM vs. M: 2107.85 (436.45; 3779.25) | <0.05 | |||
| eAAM vs. eAA: −428.48 (−2275.11; 1418.16) | 0.65 | ||||
| eAAM | |||||
| Gynoid Fat mass (%) | M vs. P: −1.60 (−3.78; 0.59) | 0.15 | |||
| P | 0.01 (−1.87; 1.89) | 0.99 | eAA vs. P: −2.23 (−4.49; 0.04) | <0.05 | |
| M | −1.03 (−2.05; −0.01) | <0.05 | eAAM vs. P: −0.76 (−3.03; 1.51) | 0.51 | |
| eAA | −1.54 (−3.51; 0.42) | 0.11 | eAA vs. M: −0.63 (−3.08; 1.82) | 0.61 | |
| eAAM | −0.33 (−1.85; 1.20) | 0.66 | eAAM vs. M: 0.84 (−1.53; 3.20) | 0.48 | |
| eAAM vs. eAA: 1.47 (−1.09; 4.02) | 0.26 | ||||
| Android Fat mass (%) | M vs. P: 0.53 (−2.20; 3.27) | 0.7 | |||
| P | −1.29 (−3.84; 1.25) | 0.3 | eAA vs. P: −1.51 (−4.39; 1.36) | 0.3 | |
| M | −1.07 (−2.60; 0.46) | 0.16 | eAAM vs. P: 0.42 (−2.48; 3.33) | 0.77 | |
| eAA | −2.72 (−4.77; −0.68) | <0.01 | eAA vs. M: −2.04 (−5.16; 1.08) | 0.2 | |
| eAAM | −0.04 (−2.23; 2.15) | 0.97 | eAAM vs. M: −0.11 (−3.07; 2.85) | 0.94 | |
| eAAM vs. eAA: 1.93 (−1.33; 5.19) | 0.24 | ||||
| Handgrip Strength (Kg) | M vs. P: −0.37 (−2.52; 1.77) | 0.73 | |||
| P | −0.11 (−1.40; 1.40) | 1 | eAA vs. P: 0.98 (−1.37; 3.33) | 0.41 | |
| M | 0.09 (−1.77; 1.95) | 0.92 | eAAM vs. P: 0.04 (−2.07; 2.15) | 0.97 | |
| eAA | 0.15 (−1.36; 1.66) | 0.82 | eAA vs. M: 1.36 (−0.97; 3.68) | 0.25 | |
| eAAM | −0.06 (−1.54; 1.43) | 0.94 | eAAM vs. M: 0.42 (−1.61; 2.44) | 0.68 | |
| eAAM vs. eAA: −0.94 (−3.27; 1.39) | 0.42 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rondanelli, M.; Peroni, G.; Gasparri, C.; Infantino, V.; Nichetti, M.; Cuzzoni, G.; Spadaccini, D.; Perna, S. Is a Combination of Melatonin and Amino Acids Useful to Sarcopenic Elderly Patients? A Randomized Trial. Geriatrics 2019, 4, 4. https://doi.org/10.3390/geriatrics4010004
Rondanelli M, Peroni G, Gasparri C, Infantino V, Nichetti M, Cuzzoni G, Spadaccini D, Perna S. Is a Combination of Melatonin and Amino Acids Useful to Sarcopenic Elderly Patients? A Randomized Trial. Geriatrics. 2019; 4(1):4. https://doi.org/10.3390/geriatrics4010004
Chicago/Turabian StyleRondanelli, Mariangela, Gabriella Peroni, Clara Gasparri, Vittoria Infantino, Mara Nichetti, Giovanni Cuzzoni, Daniele Spadaccini, and Simone Perna. 2019. "Is a Combination of Melatonin and Amino Acids Useful to Sarcopenic Elderly Patients? A Randomized Trial" Geriatrics 4, no. 1: 4. https://doi.org/10.3390/geriatrics4010004





