Development of a Specialized Telemedicine Protocol for Cognitive Disorders: The TeleCogNition Project in Greece
Abstract
1. Introduction
2. Materials and Methods
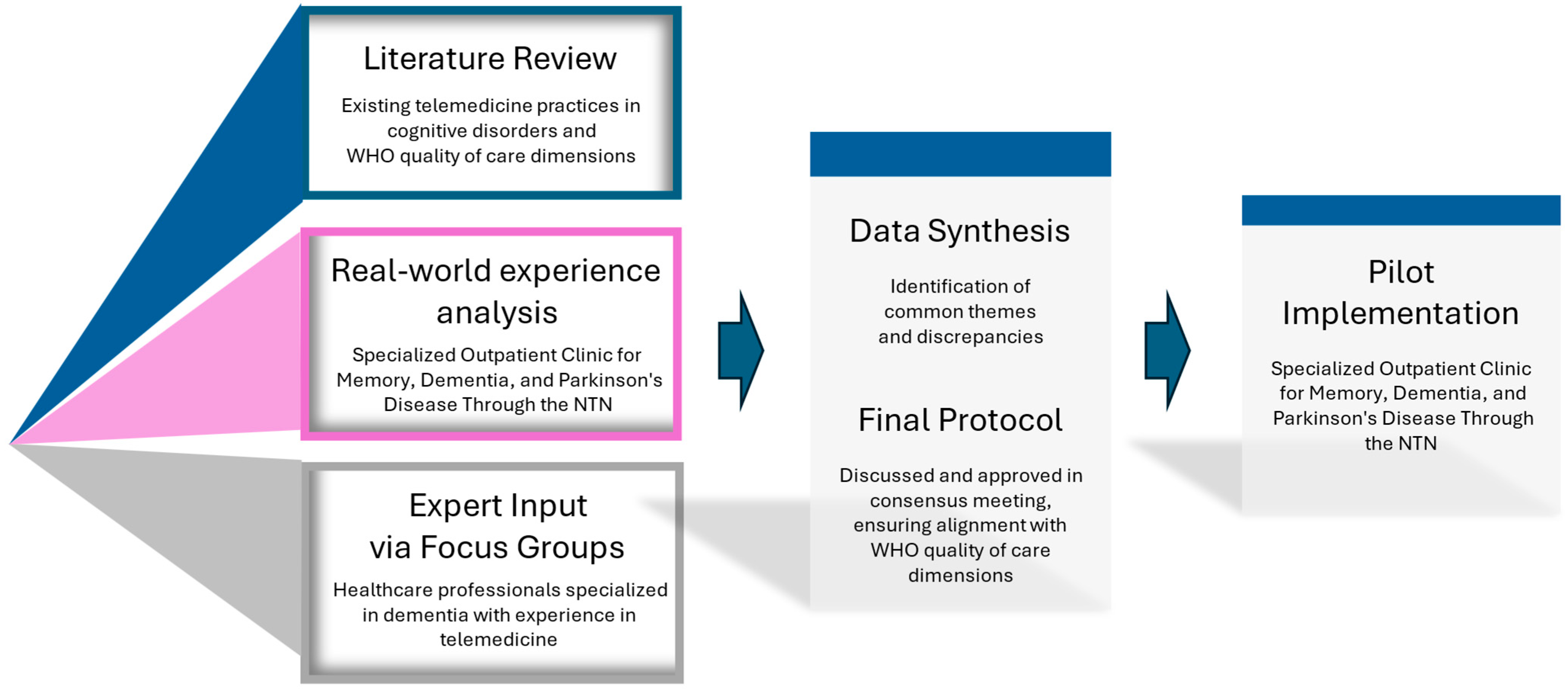
2.1. Overview
2.2. Literature Review
2.3. Experience from the “Specialized Outpatient Clinic for Memory, Dementia, and Parkinson’s Disease Through the National Telemedicine Network”
2.4. Focus Groups
2.5. Data Synthesis
2.6. Pilot Implementation Phase in the “Specialized Outpatient Clinic for Memory, Dementia, and Parkinson’s Disease Through the NTN”
3. Results
3.1. Focus Group Participants’ Characteristics
3.2. Responses to the Preliminary Questionnaires
3.3. The Final Form of the Comprehensive Telemedicine Protocol for the Assessment of Patients with Cognitive Disorders
3.3.1. Organizational Aspects
3.3.2. Technical Aspects
3.3.3. Initiation of a Telemedicine Session
3.3.4. Medical History
3.3.5. Neurological Examination
3.3.6. Neuropsychological Assessment
3.3.7. Neuropsychiatric Assessment
3.3.8. Functional Assessment
3.3.9. Dementia Staging
3.3.10. Evaluation of Laboratory and Neuroimaging Findings and Referrals for Diagnostic Work-Up
3.3.11. Ethical and Legal Considerations
3.3.12. Clinician–Patient Communication
3.3.13. Interconnection with Other Services
3.3.14. Instructions
3.3.15. Satisfaction of the Patients, Caregivers, and Healthcare Professionals
3.3.16. Training and Certification of Telemedicine Users
3.3.17. Alignment with the WHO’s Seven Dimensions of Healthcare Quality
3.4. Short Form of the Final Protocol
3.5. Pilot Implementation of the Protocol in the “Specialized Outpatient Clinic for Memory, Dementia, and Parkinson’s Disease Through the National Telemedicine Network”
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2019 Dementia Forecasting Collaborators. Estimation of the Global Prevalence of Dementia in 2019 and Forecasted Prevalence in 2050: An Analysis for the Global Burden of Disease Study 2019. Lancet Public Health 2022, 7, e105–e125. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, M.; Fiorilla, C.; Mercogliano, M.; Stilo, I.; Esposito, F.; Moccia, M.; Lavorgna, L.; Salvatore, E.; Sormani, M.P.; Majeed, A.; et al. Barriers for Access and Utilization of Dementia Care Services in Europe: A Systematic Review. BMC Geriatr. 2025, 25, 162. [Google Scholar] [CrossRef] [PubMed]
- Maresova, P.; Tomsone, S.; Lameski, P.; Madureira, J.; Mendes, A.; Zdravevski, E.; Chorbev, I.; Trajkovik, V.; Ellen, M.; Rodile, K. Technological Solutions for Older People with Alzheimer’s Disease: Review. Curr. Alzheimer Res. 2018, 15, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Pietrantonio, F.; Florczak, M.; Kuhn, S.; Kärberg, K.; Leung, T.; Said Criado, I.; Sikorski, S.; Ruggeri, M.; Signorini, A.; Rosiello, F.; et al. Applications to Augment Patient Care for Internal Medicine Specialists: A Position Paper from the EFIM Working Group on Telemedicine, Innovative Technologies & Digital Health. Front. Public Health 2024, 12, 1370555. [Google Scholar] [CrossRef]
- Geddes, M.R.; O’Connell, M.E.; Fisk, J.D.; Gauthier, S.; Camicioli, R.; Ismail, Z. Alzheimer Society of Canada Task Force on Dementia Care Best Practices for COVID-19 Remote Cognitive and Behavioral Assessment: Report of the Alzheimer Society of Canada Task Force on Dementia Care Best Practices for COVID-19. Alzheimers Dement. 2020, 12, e12111. [Google Scholar] [CrossRef]
- Angelopoulou, E.; Koros, C.; Stanitsa, E.; Stamelos, I.; Kontaxopoulou, D.; Fragkiadaki, S.; Papatriantafyllou, J.D.; Smaragdaki, E.; Vourou, K.; Pavlou, D.; et al. Neurological Examination via Telemedicine: An Updated Review Focusing on Movement Disorders. Medicina 2024, 60, 958. [Google Scholar] [CrossRef] [PubMed]
- McCleery, J.; Laverty, J.; Quinn, T.J. Diagnostic Test Accuracy of Telehealth Assessment for Dementia and Mild Cognitive Impairment. Cochrane Database Syst. Rev. 2021, 7, CD013786. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, P.; Hamelin, L.; Lagarde, J.; Hahn, V.; Guichart-Gomez, E.; Roué-Jagot, C.; Sarazin, M. Characterization of the Initial Complaint and Care Pathways Prior to Diagnosis in Very Young Sporadic Alzheimer’s Disease. Alzheimers Res. Ther. 2021, 13, 90. [Google Scholar] [CrossRef] [PubMed]
- Angelopoulou, E.; Papachristou, N.; Bougea, A.; Stanitsa, E.; Kontaxopoulou, D.; Fragkiadaki, S.; Pavlou, D.; Koros, C.; Değirmenci, Y.; Papatriantafyllou, J.; et al. How Telemedicine Can Improve the Quality of Care for Patients with Alzheimer’s Disease and Related Dementias? A Narrative Review. Medicina 2022, 58, 1705. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.N.; Shaw, C.A.; Perkhounkova, Y.; Hein, M.; Coleman, C.K. Satisfaction, Utilization, and Feasibility of a Telehealth Intervention for in-Home Dementia Care Support: A Mixed Methods Study. Dementia 2021, 20, 1565–1585. [Google Scholar] [CrossRef] [PubMed]
- WHO. Handbook for National Quality Policy and Strategy: A Practical Approach for Developing Policy and Strategy to Improve Quality of Care; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- US Department of Veterans Affairs. Clinician’s Guide to Telehealth for Older Adults: Dementia Care; US Department of Veterans Affairs: Washington, DC, USA, 2023.
- Tarolli, C.G.; Biernot, J.M.; Creigh, P.D.; Moukheiber, E.; Salas, R.M.E.; Dorsey, E.R.; Cohen, A.B. Practicing in a Pandemic: A Clinician’s Guide to Remote Neurologic Care. Neurol. Clin. Pract. 2021, 11, e179–e188. [Google Scholar] [CrossRef] [PubMed]
- Angelopoulou, E.; Kontaxopoulou, D.; Fragkiadaki, S.; Stanitsa, E.; Pavlou, D.; Papatriantafyllou, J.; Koros, C.; Dimovski, V.; Šemrov, D.; Papageorgiou, S.G. Perceptions of Patients, Caregivers, and Healthcare Professionals toward Telemedicine Use for Cognitive and Movement Disorders in the Aegean Islands, Greece: A Pilot Study of the SI4CARE European Project. Geriatrics 2024, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Politis, A.; Vorvolakos, T.; Kontogianni, E.; Alexaki, M.; Georgiou, E.-Z.E.; Aggeletaki, E.; Gkampra, M.; Delatola, M.; Delatolas, A.; Efkarpidis, A.; et al. Old-Age Mental Telehealth Services at Primary Healthcare Centers in Low- Resource Areas in Greece: Design, Iterative Development and Single-Site Pilot Study Findings. BMC Health Serv. Res. 2023, 23, 626. [Google Scholar] [CrossRef] [PubMed]
- Angelopoulou, E.; Papageorgiou, S.G. Telemedicine in Alzheimer’s Disease and Other Dementias: Where We Are? J. Alzheimers Dis. 2025, 103, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Binoy, S.; Montaser-Kouhsari, L.; Ponger, P.; Saban, W. Remote Assessment of Cognition in Parkinson’s Disease and Cerebellar Ataxia: The MoCA Test in English and Hebrew. Front. Hum. Neurosci. 2023, 17, 1325215. [Google Scholar] [CrossRef]
- Marra, D.E.; Hamlet, K.M.; Bauer, R.M.; Bowers, D. Validity of Teleneuropsychology for Older Adults in Response to COVID-19: A Systematic and Critical Review. Clin. Neuropsychol. 2020, 34, 1411–1452. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-H.; Kwak, M.-J. Performance of the Geriatric Depression Scale-15 with Older Adults Aged over 65 Years: An Updated Review 2000–2019. Clin. Gerontol. 2021, 44, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Adams, N.; Hamilton, N.; Nelson, E.-L.; Smith, C.E. Using Telemedicine to Identify Depressive Symptomatology Rating Scale in a Home Parenteral Nutrition Population. J. Technol. Behav. Sci. 2017, 2, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Capozzo, R.; Zoccolella, S.; Frisullo, M.E.; Barone, R.; Dell’Abate, M.T.; Barulli, M.R.; Musio, M.; Accogli, M.; Logroscino, G. Telemedicine for Delivery of Care in Frontotemporal Lobar Degeneration During COVID-19 Pandemic: Results from Southern Italy. J. Alzheimers Dis. 2020, 76, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, S.G.; Voskou, P.; Economou, A.; Beratis, I.; Douzenis, A. Testamentary Capacity Assessment Tool (TCAT): A Brief Instrument for Patients with Dementia. J. Alzheimers Dis. 2018, 61, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Haimi, M.; Gesser-Edelsburg, A. Application and Implementation of Telehealth Services Designed for the Elderly Population during the COVID-19 Pandemic: A Systematic Review. Health Inform. J. 2022, 28, 14604582221075561. [Google Scholar] [CrossRef] [PubMed]
- Kivipelto, M.; Mangialasche, F.; Ngandu, T. Lifestyle Interventions to Prevent Cognitive Impairment, Dementia and Alzheimer Disease. Nat. Rev. Neurol. 2018, 14, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Mason, A.N. The Most Important Telemedicine Patient Satisfaction Dimension: Patient-Centered Care. Telemed. J. E-Health Off. J. Am. Telemed. Assoc. 2022, 28, 1206–1214. [Google Scholar] [CrossRef] [PubMed]
- Haleem, A.; Javaid, M.; Singh, R.P.; Suman, R. Telemedicine for Healthcare: Capabilities, Features, Barriers, and Applications. Sens. Int. 2021, 2, 100117. [Google Scholar] [CrossRef] [PubMed]
- Worman, S. Diversity, Equity and Inclusiveness in Healthcare: A Primary Care Perspective. J. Eval. Clin. Pract. 2024, 30, 1539–1542. [Google Scholar] [CrossRef] [PubMed]
- Price, J.C.; Simpson, D.C. Telemedicine and Health Disparities. Clin. Liver Dis. 2022, 19, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Wärn, E.; Andersson, L.; Berginström, N. Remote Neuropsychological Testing as an Alternative to Traditional Methods-a Convergent Validity Study. Arch. Clin. Neuropsychol. Off. J. Natl. Acad. Neuropsychol. 2025, acaf013. [Google Scholar] [CrossRef] [PubMed]
- Andias, R.; Martins, A.I.; Pais, J.; Cruz, V.T.; Silva, A.G.; Rocha, N.P. Validity and Reliability of a Digital Solution for Cognitive Assessment: The Brain on Track®. Digit. Health 2024, 10, 20552076241287371. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Kang, S.J.; Hong, S.-K.; Ma, H.-I.; Lee, U.; Kim, Y.J. A Validation Study of a Smartphone-Based Finger Tapping Application for Quantitative Assessment of Bradykinesia in Parkinson’s Disease. PLoS ONE 2016, 11, e0158852. [Google Scholar] [CrossRef] [PubMed]
- Wiese, L.A.K.; Gibson, A.; Guest, M.A.; Nelson, A.R.; Weaver, R.; Gupta, A.; Carmichael, O.; Lewis, J.P.; Lindauer, A.; Loi, S.; et al. Global Rural Health Disparities in Alzheimer’s Disease and Related Dementias: State of the Science. Alzheimers Dement. J. Alzheimers Assoc. 2023, 19, 4204–4225. [Google Scholar] [CrossRef] [PubMed]
- Meeuwsen, E.J.; Melis, R.J.F.; Van Der Aa, G.C.H.M.; Golüke-Willemse, G.A.M.; De Leest, B.J.M.; Van Raak, F.H.J.M.; Schölzel-Dorenbos, C.J.M.; Verheijen, D.C.M.; Verhey, F.R.J.; Visser, M.C.; et al. Effectiveness of Dementia Follow-up Care by Memory Clinics or General Practitioners: Randomised Controlled Trial. BMJ 2012, 344, e3086. [Google Scholar] [CrossRef] [PubMed]
- Kampouraki, M.; Paraskevopoulos, P.; Fachouri, E.; Terzoudis, S.; Karanasios, D. Building Strong Primary Healthcare Systems in Greece. Cureus 2023, 15, e41333. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, X.; Yuan, S. A Scoping Review of National Policies for Dementia Prevention and Control in Mainland China. Health Res. Policy Syst. 2025, 23, 42. [Google Scholar] [CrossRef] [PubMed]
| Telemedicine Visit Sub-Area | Questions for the Semi-Structured Interview Guide for Focus Groups |
|---|---|
| Organizational and technical aspects | 1. How do you think the teleconference with the patient should begin? |
| 2. In your opinion, what should be the essential requirements in the patient’s examination room? | |
| Neurological assessment (neurological examination, neuropsychiatric and functional assessment) | 3. Which parts of the neurological examination do you consider essential and which could be omitted when assessing a patient with cognitive complaints via telemedicine, and why? |
| 4. Do you use mobility assessment scales (e.g., UPDRS III, Timed Up and Go test), and which do you consider most suitable for telemedicine evaluation? | |
| 5. What is your experience and opinion regarding the use of neuropsychiatric symptom scales for patients with cognitive disorders through telemedicine? | |
| 6. What is your experience and opinion regarding the use of functionality scales for patients with cognitive disorders through telemedicine? | |
| Neuropsychological assessment | 7. Which neuropsychological tests do you consider most suitable for a comprehensive neuropsychological assessment protocol via telemedicine? |
| 8. What is your experience and opinion regarding the use of new technologies, such as specific software, during neuropsychological assessment via telemedicine? | |
| Ethical and legal aspects | 9. Do you take a separate history from the caregiver without the patient’s presence during telemedicine assessment for patients with cognitive disorders, and how do you think this should be completed? |
| 10. What ethical and legal issues do you believe arise when examining patients with cognitive disorders via telemedicine, and how do you address them? | |
| Instructions and linkage with other services | 11. How would you provide instructions at the end of the telemedicine visit? Via email, and, if yes, to whom, verbally to the caregiver, or verbally to the healthcare professional present, if any? |
| 12. Which non-pharmacological interventions would you recommend for patients with cognitive disorders whom you assess via telemedicine (e.g., psychoeducation, caregiver support, physical and cognitive training, social services)? Do you think there should be a specific plan for linking with other entities, and if so, with whom? |
| Question | Physicians | Psychologists/ Neuropsychologists |
|---|---|---|
| 1. Do you believe that developing a specialized assessment telemedicine protocol for cognitive disorders would be useful? | ||
| Yes | 100% | 100% |
| No | 0% | 0% |
| 2. In your daily practice, do you obtain the patient’s and/or caregiver’s consent before the remote assessment? | ||
| Yes, verbal consent | 75% | 14.3% |
| Yes, written consent | 25% | 85.7% |
| No | ||
| 3. Before the remote assessment, do you require the presence of a caregiver during the session? | ||
| Yes | 100% | 28.6% |
| Depending on the situation | 0% | 42.9% |
| No | 0% | 28.6% |
| 4. Do you conduct a separate interview with the caregiver (without the patient being present) through telemedicine? | ||
| Yes, systematically | 25% | 26.6% |
| Depending on the situation | 75% | 42.9% |
| No | 0% | 28.6% |
| 5. In patients with cognitive impairment/dementia, do you systematically assess their mobility through telemedicine? | ||
| Yes, systematically | 50% | 28.6% |
| Sometimes | 50% | 28.6% |
| Never | 42.9% | |
| 6. For patients with suspected Parkinsonism or movement disorders, do you use the UPDRS III scale (excluding muscle tone and postural reflex testing) via telemedicine? | ||
| Yes, systematically | 25% | 14.3% |
| Sometimes | 25% | 42.9% |
| Never | 50% | 42.9% |
| 7. Do you systematically use neuropsychiatric assessment scales (e.g., NPI, FBI) in the telemedicine visit? | ||
| Yes, systematically | 50% | 57.1% |
| Sometimes | 50% | 42.9% |
| Never | 0% | 0% |
| 8. Do you systematically use self-assessment or informant-based questionnaires for cognitive impairment/dementia (e.g., IQCODE) via telemedicine? | ||
| Yes, systematically | 25% | 0% |
| Sometimes | 50% | 57.1% |
| Never | 25% | 42.9% |
| 9. Do you systematically use functional assessment scales (e.g., FAQ, Lawton IADL) via telemedicine? | ||
| Yes, systematically | 75% | 85.7% |
| Sometimes | 0% | 14.3% |
| Never | 25% | 0% |
| 10. Which neuropsychological tool do you use as a screening test via telemedicine? | ||
| MMSE | 50% | 57.1% |
| ACE–R | 25% | 14.3% |
| MoCA | 25% | 28.6% |
| 11. Do you personally prescribe medications to patients that you assess via telemedicine? | ||
| Yes | 100% | 0% |
| No | 0% | 100% |
| 12. Do you provide medical certificates (for example, for disability or other purposes)? | ||
| Yes | 75% | 0% |
| No | 25% | 100% |
| 13. Do you have a structured referral plan to other services (e.g., cognitive training, psychoeducation/support for caregivers, social services, etc.) for patients you assess through telemedicine and caregivers? | ||
| Yes | 75% | 85.7% |
| No | 25% | 14.3% |
| 14. Do you provide a satisfaction questionnaire to patients and caregivers after a telemedicine consultation? | ||
| Yes | 100% | 14.3% |
| No | 0% | 85.7% |
| 15. For patients with cognitive impairment/dementia whom you have assessed via telemedicine, what percentage do you believe might have received a different diagnosis if assessed in person? | ||
| <20% | 75% | 85.7% |
| 20–40% | 0% | 14.3% |
| 40–60% | 0% | 0% |
| >60% | 25% | 0% |
| Element of Neurological Examination | Required in All Cases | Required in All Cases If a Local Trained Healthcare Professional Is Present |
|---|---|---|
| Posture and gait assessment |
|
|
| Oculomotor examination (cranial nerves III, IV, VI) |
|
|
| Assessment of dysarthria (cranial nerves IX, X) | As in-person | |
| Assessment of bradykinesia |
| |
| Tremor examination |
| |
| Muscle tone examination | - |
|
| Mobility scales |
|
|
| Sub-Area of the Telemedicine Visit | Key Components |
|---|---|
| Organizational aspects | Adequate space for gait assessment Well-lit private, quiet room Available pen and A4 piece of paper Prefilled sent form with medical history and medications No calendars and mobile phones close to the patient |
| Technical aspects | High-definition camera and screen, noise-canceling microphone, safe and stable Internet connection, screensharing, direct access to technical support, alternative communication mode in case of connectivity disruption, documentation in encrypted electronic database that is shared between local and remote healthcare professionals |
| Initiation of the telemedicine session | Greet the participants, ask if they can hear you Introduce yourself and the other team members Verify patient’s name, date of birth Ask and record other attendees’ names Keep the patient’s address and phone number in case of an emergency Ask about any vision/hearing problems, encourage the use of eyeglasses and/or hearing aids Ensure all the participants are within the camera’s field of view |
| Medical history | Obtain focused, detailed medical history as in-person, first from the patient Obtain family history With the patient’s permission, obtain history from the family member/caregiver either at the same session or separately |
| Neurological examination | Gait assessment: ask the patient to walk freely in front of the camera (ca. 5 steps in both directions) Perform smooth pursuit (use your finger) and saccadic eye movements examination Examine dysarthria as in-person Examine bradykinesia, tremor as in-person (ensure that the body part of interest is fully captured by the camera) Perform modified UPDRS III in case of parkinsonism (excluding muscle tone and postural reflex testing) If a local trained healthcare professional is present and/or depending on the clinical scenario: tandem walking, muscle tone, postural instability, coordination evaluation, Romberg testing, assessment of the rest of cranial nerves, reflexes, frontal release signs, Barre and Mingazzini signs, sensory examination (including stereognosis, graphesthesia, two-point discrimination) |
| Neuropsychological assessment | MoCA as a screening tool For a detailed assessment when needed: semantic and phonemic verbal fluency, CDT, JLO, Digit Span (Forward and Backward), O-TMT (A and B), HVLT-R, BNT, apraxia testing |
| Neuropsychiatric assessment | NPI, GDS-15 in patients ≥ 65 years PHQ-9 in patients ≤ 65 years MBI-C in subjective cognitive decline or MCI Short Anxiety Screening Test in suspected anxiety EQ-5D |
| Functional assessment | Lawton IADL |
| Dementia staging | CDR, CDR-FTLD |
| Evaluation of laboratory and neuroimaging findings and referrals for diagnostic work-up | Referrals for diagnostic work-up based on the up-to-date guidelines Blood or CSF samples sent for analysis (AD biomarkers, genetic testing, other tests) Referrals for hospital admission for further evaluation when needed |
| Ethical and legal considerations | Written informed consent GDPR-compliant platform, encrypted access to the electronic database, data security Privacy in the room TCAT when needed |
| Clinician–patient communication | Look frequently at the camera Show empathy, use gestures Speak clearly and slowly |
| Interconnection with other services | Structured plan for referrals to cognitive rehabilitation, tele-exercise, speech therapy, caregiver support groups or social services, tele-psychoeducation |
| Instructions | Provision of a written medical report to the referring physician Advice on non-pharmacological interventions, safety measures at home, driving and family planning, and genetic counseling in accordance with the current guidelines Provision of online informational materials on dementia Clear instructions for the re-evaluation and follow-up |
| Satisfaction questionnaires | Ask the patients, family members/caregivers, and local healthcare professionals to complete satisfaction questionnaires regarding their experience of the telemedicine visit |
| Training of the local and remote healthcare professionals | Training of the local and remote healthcare professionals on the telemedicine system (initial and regular sessions) Regular training of the team on the telemedicine protocol |
| Alignment with the WHO’s seven dimensions of healthcare quality | The key elements of the protocol are aligned with the WHO’s seven dimensions of healthcare quality: effectiveness, safety, people-centeredness, timeliness, equitability, efficiency, and integrated care |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Angelopoulou, E.; Stamelos, I.; Smaragdaki, E.; Vourou, K.; Stanitsa, E.; Kontaxopoulou, D.; Koros, C.; Papatriantafyllou, J.; Zilidou, V.; Romanopoulou, E.; et al. Development of a Specialized Telemedicine Protocol for Cognitive Disorders: The TeleCogNition Project in Greece. Geriatrics 2025, 10, 94. https://doi.org/10.3390/geriatrics10040094
Angelopoulou E, Stamelos I, Smaragdaki E, Vourou K, Stanitsa E, Kontaxopoulou D, Koros C, Papatriantafyllou J, Zilidou V, Romanopoulou E, et al. Development of a Specialized Telemedicine Protocol for Cognitive Disorders: The TeleCogNition Project in Greece. Geriatrics. 2025; 10(4):94. https://doi.org/10.3390/geriatrics10040094
Chicago/Turabian StyleAngelopoulou, Efthalia, Ioannis Stamelos, Evangelia Smaragdaki, Kalliopi Vourou, Evangelia Stanitsa, Dionysia Kontaxopoulou, Christos Koros, John Papatriantafyllou, Vasiliki Zilidou, Evangelia Romanopoulou, and et al. 2025. "Development of a Specialized Telemedicine Protocol for Cognitive Disorders: The TeleCogNition Project in Greece" Geriatrics 10, no. 4: 94. https://doi.org/10.3390/geriatrics10040094
APA StyleAngelopoulou, E., Stamelos, I., Smaragdaki, E., Vourou, K., Stanitsa, E., Kontaxopoulou, D., Koros, C., Papatriantafyllou, J., Zilidou, V., Romanopoulou, E., Georgopoulou, E.-M., Sakka, P., Karanikas, H., Stefanis, L., Bamidis, P., & Papageorgiou, S. (2025). Development of a Specialized Telemedicine Protocol for Cognitive Disorders: The TeleCogNition Project in Greece. Geriatrics, 10(4), 94. https://doi.org/10.3390/geriatrics10040094













