Driving Simulator Training Is Associated with Reduced Inhibitory Workload in Older Drivers
Abstract
:1. Introduction
2. Materials and Methods
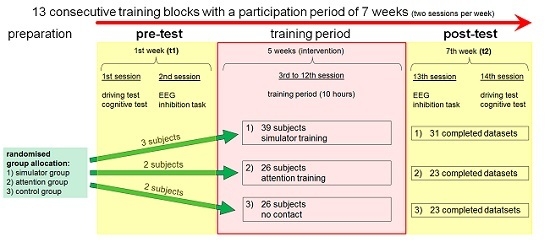
2.1. General Study Design
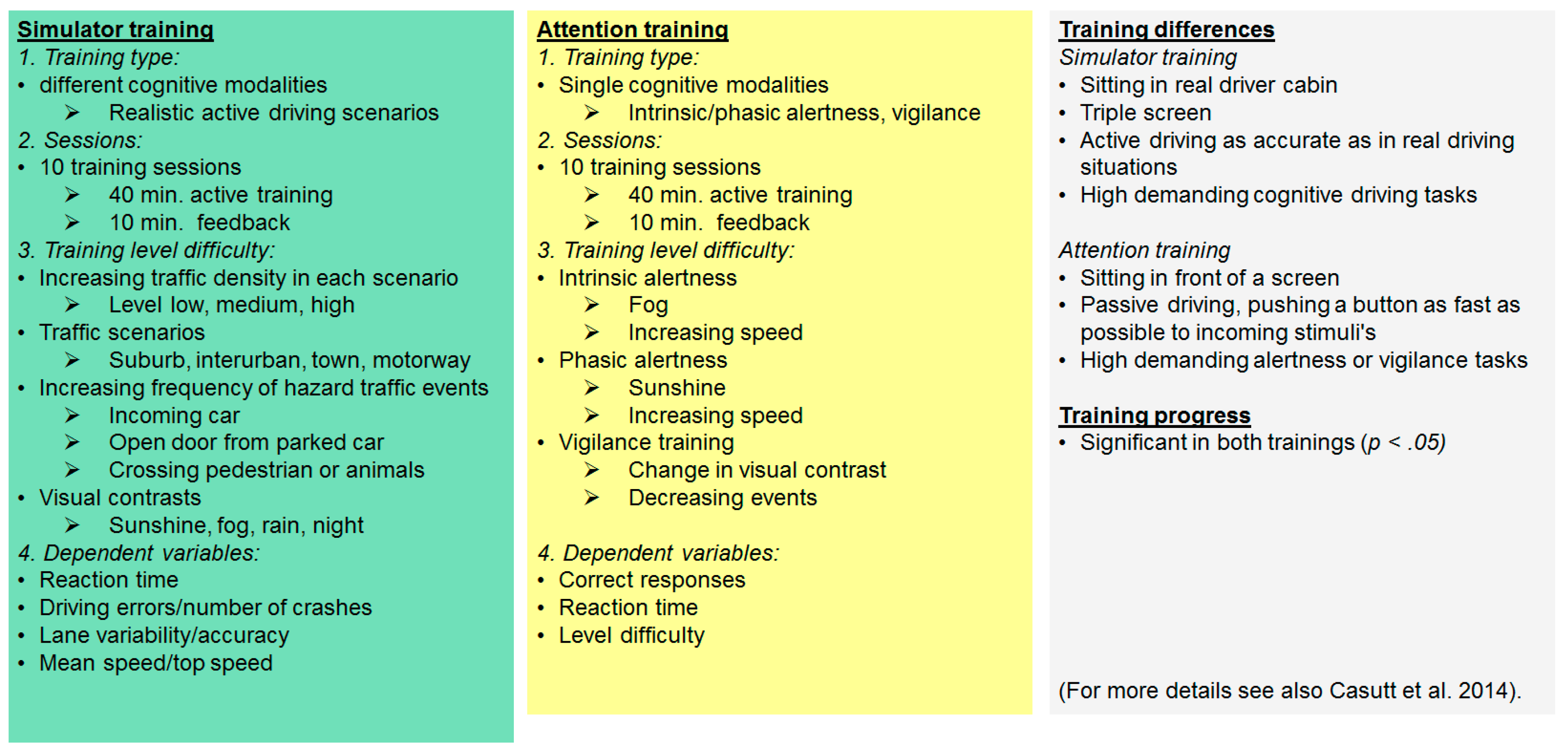
2.2. Training Interventions
2.3. Participants
2.4. EEG Recording and EEG Data Analysis
2.5. Inhibition Tasks
2.6. Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Summala, H. Accident risk and driver behaviour. Saf. Sci. 1996, 22, 103–117. [Google Scholar] [CrossRef]
- Anstey, K.J.; Wood, J. Chronological age and age-related cognitive deficits are associated with an increase in multiple types of driving errors in late life. Neuropsychology 2011, 25, 613–621. [Google Scholar] [CrossRef] [PubMed]
- OECD. Ageing and Transport: Mobility Needs and Safety Issues; Organization For Economic: Paris, France, 2001. [Google Scholar]
- Casutt, G.; Martin, M.; Jäncke, L. Alterseffekte auf die fahrsicherheit bei schweizer kraftfahrern im jahr 2010. Z. Verkehrssicher. 2013, 59, 162–169. [Google Scholar]
- Siren, A.; Meng, A. Cognitive screening of older drivers does not produce safety benefits. Accid. Anal. Prev. 2012, 45, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fraser, S.; Lindsay, J.; Clarke, K.; Mao, Y. Age-specific patterns of factors related to fatal motor vehicle traffic crashes: Focus on young and elderly drivers. Public Health 1998, 112, 289–295. [Google Scholar] [CrossRef]
- Braitman, K.A.; Kirley, B.B.; Ferguson, S.; Chaudhary, N.K. Factors leading to older drivers' intersection crashes. Traffic Inj. Prev. 2007, 8, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Owsley, C.; Ball, K.; McGwin, G., Jr.; Sloane, M.E.; Roenker, D.L.; White, M.F.; Overley, E.T. Visual processing impairment and risk of motor vehicle crash among older adults. J. Am. Med. Assoc. 1998, 279, 1083–1088. [Google Scholar] [CrossRef]
- Romoser, M.R.; Fisher, D.L. The effect of active versus passive training strategies on improving older drivers’ scanning in intersections. Hum. Factors Soc. 2009, 51, 652–668. [Google Scholar] [CrossRef]
- Rizzo, M.; McGehee, D.V.; Dawson, J.D.; Anderson, S.N. Simulated car crashes at intersections in drivers with alzheimer disease. Alzheimer Dis. Assoc. Disord. 2001, 15, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Anstey, K.J.; Wood, J.; Lord, S.; Walker, J.G. Cognitive, sensory and physical factors enabling driving safety in older adults. Clin. Psycholo. Rev. 2005, 25, 45–65. [Google Scholar] [CrossRef] [PubMed]
- Joanisse, M.; Gagnon, S.; Voloaca, M. Overly cautious and dangerous: An empirical evidence of the older driver stereotypes. Accid. Anal. Prev. 2012, 45, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Ross, L.A.; Clay, O.J.; Edwards, J.D.; Ball, K.K.; Wadley, V.G.; Vance, D.E.; Cissell, G.M.; Roenker, D.L.; Joyce, J.J. Do older drivers at-risk for crashes modify their driving over time? J. Gerontol. B Psychol. Sci. Soc. Sci. 2009, 64, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Hakamies-Blomqvist, L. Compensation in older drivers as reflected in their fatal accidents. Accid. Anal. Prev. 1994, 26, 107–112. [Google Scholar] [CrossRef]
- Stalvey, B.T.; Owsley, C. The development and efficacy of a theory-based educational curriculum to promote self-regulation among high-risk older drivers. Health Promot. Pract. 2003, 4, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Owsley, C.; McGwin, G., Jr.; Phillips, J.M.; McNeal, S.F.; Stalvey, B.T. Impact of an educational program on the safety of high-risk, visually impaired, older drivers. Am. J. Prev. Med. 2004, 26, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Ball, K.; Edwards, J.D.; Ross, L.A.; McGwin, G., Jr. Cognitive training decreases motor vehicle collision involvement of older drivers. J. Am. Geriatr. Soc. 2011, 58, 2107–2113. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.D.; Lunsman, M.; Perkins, M.; Rebok, G.W.; Roth, D.L. Driving cessation and health trajectories in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 1290–1295. [Google Scholar] [CrossRef] [PubMed]
- Lavallière, M.; Simoneau, M.; Tremblay, M.; Laurendeau, D.; Teasdale, N. Active training and driving-specific feedback improve older drivers’ visual search prior to lane changes. BMC Geriatr. 2012, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Roenker, D.L.; Cissell, G.M.; Ball, K.K.; Wadley, V.G.; Edwards, J.D. Speed-of-processing and driving simulator training result in improved driving performance. Hum. Factors Soc. 2003, 45, 218–233. [Google Scholar] [CrossRef]
- Romoser, M.R. The long-term effects of active training strategies on improving older drivers’ scanning in intersections: A two-year follow-up to romoser and fisher (2009). Hum. Factors Soc. 2013, 55, 278–284. [Google Scholar] [CrossRef]
- Basak, C.; Boot, W.R.; Voss, M.W.; Kramer, A.F. Can training in a real-time strategy video game attenuate cognitive decline in older adults? Psychol. Aging 2008, 23, 765–777. [Google Scholar] [CrossRef] [PubMed]
- O’Donnel, R.D.; Eggemeier, F.T. Cognitive processes and performance. In Handbook of Perception and Human Performance; Kaufman, B.K., Wiley, T.J., Eds.; John Wiley: New York, NY, USA, 1986; Volume 2, pp. 41–42. [Google Scholar]
- Borghini, G.; Astolfi, L.; Vecchiato, G.; Mattia, D.; Babiloni, F. Measuring neurophysiological signals in aircraft pilots and car drivers for the assessment of mental workload, fatigue and drowsiness. Neurosci. Biobehav. Rev. 2014, 44, 58–75. [Google Scholar] [CrossRef] [PubMed]
- Paxion, J.; Galy, E.; Berthelon, C. Mental workload and driving. Front. Psychol. 2014, 5, 1344. [Google Scholar] [CrossRef] [PubMed]
- Anguera, J.A.; Boccanfuso, J.; Rintoul, J.L.; Al-Hashimi, O.; Faraji, F.; Janowich, J.; Kong, E.; Larraburo, Y.; Rolle, C.; Johnston, E.; et al. Video game training enhances cognitive control in older adults. Nature 2013, 501, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Lustig, C.; Shah, P.; Seidler, R.; Reuter-Lorenz, P.A. Aging, training, and the brain: A review and future directions. Neuropsychol. Rev. 2009, 19, 504–522. [Google Scholar] [CrossRef] [PubMed]
- Zelinski, E.M. Far transfer in cognitive training of older adults. Restor. Neurol. Neurosci. 2009, 27, 455–471. [Google Scholar] [PubMed]
- Bélanger, A.; Gagnon, S.; Yamin, S. Capturing the serial nature of older drivers’ responses towards challenging events: A simulator study. Accid. Anal. Prev. 2010, 42, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Hakamies-Blomqvist, L.; Mynttinen, S.; Backman, M.; Mikkonen, V. Age-related differences in driving: Are older drivers more serial? Int. J. Behav. Dev. 1999, 23, 575–589. [Google Scholar] [CrossRef]
- Arien, C.; Jongen, E.M.; Brijs, K.; Brijs, T.; Daniels, S.; Wets, G. A simulator study on the impact of traffic calming measures in urban areas on driving behavior and workload. Accid. Anal. Prev. 2013, 61, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, J.; Johansson, E.; Ostlund, J. Effects of visual and cognitive load in real and simulated motorway driving. Transp. Res. F Traffic Psychol. Behav. 2005, 8, 97–120. [Google Scholar] [CrossRef]
- Lavallière, M.; Laurendeau, D.; Simoneau, M.; Teasdale, N. Changing lanes in a simulator: Effects of aging on the control of the vehicle and visual inspection of mirrors and blind spot. Traffic Inj. Prev. 2011, 12, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Recarte, M.A.; Nunes, L.M. Mental workload while driving: Effects on visual search, discrimination, and decision making. J. Exp. Psychol. Appl. 2003, 9, 119–137. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Cameron, D.; Lee, A.H. Assessing the driving performance of older adult drivers: On-road versus simulated driving. Accid. Anal. Prev. 2003, 35, 797–803. [Google Scholar] [CrossRef]
- Casutt, G.; Martin, M.; Keller, M.; Jäncke, L. The relation between performance in on-road driving, cognitive screening and driving simulator in older healthy drivers. Transp. Res. F Traffic Psychol. Behav. 2014, 22, 232–244. [Google Scholar] [CrossRef]
- Cantin, V.; Lavalliere, M.; Simoneau, M.; Teasdale, N. Mental workload when driving in a simulator: Effects of age and driving complexity. Accid. Anal. Prev. 2009, 41, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Radvansky, G.A.; Zacks, R.T.; Hasher, L. Age and inhibition: The retrieval of situation models. J. Gerontol. B Psychol. Sci. Soc. Sci. 2005, 60, 276–278. [Google Scholar] [CrossRef]
- Zacks, R.T.; Hasher, L. Directed Ignoring: Inhibitory Regulation of Working Memory. In Inhibitory Processes in Attention, Memory, and Language, 1st ed.; Dagenbach, D., Carr, T.H., Eds.; Academic Press: San Diego, CA, USA, 1994; pp. 241–264. [Google Scholar]
- Lustig, C.; Hasher, L.; Zacks, R.T. Inhibitory deficit theory: Recent developments in a “new view”. In The Place of Inhibition in Cognition; Gorfein, D.S., MacLeod, C.M., Eds.; American Psychological Association: Washington, DC, USA, 2007; pp. 145–162. [Google Scholar]
- Hasher, L.; Zacks, R.T.; Rahhal, T.A. Timing, instructions, and inhibitory control: Some missing factors in the age and memory debate. Gerontology 1999, 45, 355–357. [Google Scholar] [CrossRef] [PubMed]
- Daigneault, G.; Joly, P.; Frigon, J.Y. Executive functions in the evaluation of accident risk of older drivers. J. Clin. Exp. Neuropsychol. 2002, 24, 221–238. [Google Scholar] [PubMed]
- Wild-Wall, N.; Falkenstein, M.; Hohnsbein, J. Flanker interference in young and older participants as reflected in event-related potentials. Brain Res. 2008, 1211, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Chmielewski, W.X.; Muckschel, M.; Stock, A.K.; Beste, C. The impact of mental workload on inhibitory control subprocesses. NeuroImage 2015, 112, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.; Smith, M.E. Neurophysiological measures of working memory and individual differences in cognitive ability and cognitive style. Cereb. Cortex 2000, 10, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.; Smith, M.E.; Leong, H.; McEvoy, L.; Whitfield, S.; Du, R.; Rush, G. Monitoring working memory load during computer-based tasks with EEG pattern recognition methods. Hum. Factors Soc. 1998, 40, 79–91. [Google Scholar] [CrossRef]
- Gevins, A.; Smith, M.E.; McEvoy, L.; Yu, D. High-resolution EEG mapping of cortical activation related to working memory: Effects of task difficulty, type of processing, and practice. Cereb. Cortex 1997, 7, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.; Smith, M.E.; McEvoy, L.K.; Ilan, A.B.; Chan, C.S.; Jiang, A.; Sam-Vargas, L.; Abraham, G. A cognitive and neurophysiological test of change from an individual’s baseline. Clin. Neurophysiol. 2011, 122, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Holm, A.; Lukander, K.; Korpela, J.; Sallinen, M.; Muller, K.M. Estimating brain load from the EEG. Sci. World J. 2009, 9, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.M. Synchronous neural oscillations and cognitive processes. Trends Cogn. Sci. 2003, 7, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Borghini, G.; Vecchiato, G.; Toppi, J.; Astolfi, L.; Maglione, A.; Isabella, R.; Caltagirone, C.; Kong, W.; Wei, D.; Zhou, Z.; et al. Assessment of mental fatigue during car driving by using high resolution EEG activity and neurophysiologic indices. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; Volume 2012, pp. 6442–6445.
- Lei, S.; Roetting, M. Influence of task combination on EEG spectrum modulation for driver workload estimation. Hum. Factors Soc. 2011, 53, 168–179. [Google Scholar] [CrossRef]
- Casutt, G.; Theill, N.; Martin, M.; Keller, M.; Jäncke, L. The drive-wise project: Driving simulator training increases real driving performance in healthy older drivers. Front. Aging Neurosci. 2014, 6, 85. [Google Scholar] [CrossRef] [PubMed]
- Hauke, J.; Fimm, B.; Sturm, W. Efficacy of alertness training in a case of brainstem encephalitis: Clinical and theoretical implications. Neuropsychol. Rehabil. 2011, 21, 164–182. [Google Scholar] [CrossRef] [PubMed]
- Schuhfried. Cogniplus. Available online: http://www.schuhfried.com/cogniplus-cps/the-training-programs (accessed on 14 August 2016).
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643–662. [Google Scholar] [CrossRef]
- MacLeod, C.M. Half a century of research on the stroop effect: An integrative review. Psychol. Bull. 1991, 109, 163–203. [Google Scholar] [CrossRef] [PubMed]
- Hanslmayr, S.; Pastotter, B.; Bauml, K.H.; Gruber, S.; Wimber, M.; Klimesch, W. The electrophysiological dynamics of interference during the stroop task. J. Cogn. Neurosci. 2008, 20, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Andrés, P.; Guerrini, C.; Phillips, L.H.; Perfect, T.J. Differential effects of aging on executive and automatic inhibition. Dev. Neuropsychol. 2008, 33, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, B.A.; Eriksen, C.W. Effects of noise letters upon identification of a target letter in a non-search task. Percep. Psychophys. 1974, 16, 143–149. [Google Scholar] [CrossRef]
- Salthouse, T.A. Is flanker-based inhibition related to age? Identifying specific influences of individual differences on neurocognitive variables. Brain Cogn. 2010, 73, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Pedhazur, E.J. Multiple regrssion. Explanation and prediction. In Behavioral Research; Holt, Rinehart & Winston: New York, NY, USA, 1982. [Google Scholar]
- Krauth, J. Distribution-Free Statistics. An Application-Oriented Approach; Elsevier: Amsterdam, The Netherland, 1988. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Earlbaum Associates: Hillsdale, MI, USA, 1988. [Google Scholar]
- Morris, S.B.; DeShon, R.P. Combining effect size estimates in meta-analysis with repeated measures and independent-groups designs. Psychol. Methods 2002, 7, 105–125. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Kawashima, R. Effects of processing speed training on cognitive functions and neural systems. Rev. Neurosci. 2012, 23, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Shriram, R.; Sundhararajan, M.; Daimiwal, N. EEG based cognitive workload assessment for maximum efficiency. IOSR J. Electron. Commun. Eng. 2014, 7, 34–38. [Google Scholar]
- Wild-Wall, N.; Falkenstein, M.; Gajewski, P.D. Neural correlates of changes in a visual search task due to cognitive training in seniors. Neural. Plast. 2012, 2012, 529057. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, E.; Nyberg, L.; Backman, L.; Neely, A.S. Plasticity of executive functioning in young and older adults: Immediate training gains, transfer, and long-term maintenance. Psychol. Aging 2008, 23, 720–730. [Google Scholar] [CrossRef] [PubMed]
- Reuter-Lorenz, P.A.; Cappell, K.A. Neurocognitive aging and the compensation hypothesis. Curr. Dir. Psychol. Sci. 2008, 17, 177–182. [Google Scholar] [CrossRef]
- Davis, S.W.; Dennis, N.A.; Daselaar, S.M.; Fleck, M.S.; Cabeza, R. Que pasa? The posterior-anterior shift in aging. Cereb. Cortex 2008, 18, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- West, R.; Baylis, G.C. Effects of increased response dominance and contextual disintegration on the stroop interference effect in older adults. Psychol. Aging 1998, 13, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Zeef, E.J.; Sonke, C.J.; Kok, A.; Buiten, M.M.; Kenemans, J.L. Perceptual factors affecting age-related differences in focused attention: Performance and psychophysiological analyses. Psychophysiology 1996, 33, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Salthouse, T.A. The processing-speed theory of adult age differences in cognition. Psychol. Rev. 1996, 103, 403–428. [Google Scholar] [CrossRef] [PubMed]
- Rush, B.K.; Barch, D.M.; Braver, T.S. Accounting for cognitive aging: Context processing, inhibition or processing speed? Neuropsychol. Dev. Cogn. B Aging Neuropsychol. Cogn. 2006, 13, 588–610. [Google Scholar] [CrossRef] [PubMed]
- Braver, T.S.; Paxton, J.L.; Locke, H.S.; Barch, D.M. Flexible neural mechanisms of cognitive control within human prefrontal cortex. Proc. Nati. Acad. Sci. USA 2009, 106, 7351–7356. [Google Scholar] [CrossRef] [PubMed]
- Puccioni, O.; Vallesi, A. Conflict resolution and adaptation in normal aging: The role of verbal intelligence and cognitive reserve. Psychol. Aging 2012, 27, 1018–1026. [Google Scholar] [CrossRef] [PubMed]
- Berlingeri, M.; Danelli, L.; Bottini, G.; Sberna, M.; Paulesu, E. Reassessing the harold model: Is the hemispheric asymmetry reduction in older adults a special case of compensatory-related utilisation of neural circuits? Exp. Brain Res. 2013, 224, 393–410. [Google Scholar] [CrossRef] [PubMed]
- Reuter-Lorenz, P. New visions of the aging mind and brain. Trends Cogn. Sci. 2002, 6, 394–400. [Google Scholar] [CrossRef]
- Feyereisen, P.; Charlot, V. Are there uniform age-related changes across tasks involving inhibitory control through access, deletion, and restraint functions? A preliminary investigation. Exp. Aging Res. 2008, 34, 392–418. [Google Scholar] [CrossRef] [PubMed]
- Tisch, S.; Silberstein, P.; Limousin-Dowsey, P.; Jahanshahi, M. The basal ganglia: Anatomy, physiology, and pharmacology. Psychiatr. Clin. N. Am. 2004, 27, 757–799. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.D.; Myers, C.; Ross, L.A.; Roenker, D.L.; Cissell, G.M.; McLaughlin, A.M.; Ball, K.K. The longitudinal impact of cognitive speed of processing training on driving mobility. Gerontologist 2009, 49, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Berka, C.; Levendowski, D.J.; Lumicao, M.N.; Yau, A.; Davis, G.; Zivkovic, V.T.; Olmstead, R.E.; Tremoulet, P.D.; Craven, P.L. EEG correlates of task engagement and mental workload in vigilance, learning, and memory tasks. Aviat. Space Environ. Med. 2007, 78, B231–B244. [Google Scholar] [PubMed]
- Stikic, M.; Berka, C.; Levendowski, D.J.; Rubio, R.F.; Tan, V.; Korszen, S.; Barba, D.; Wurzer, D. Modeling temporal sequences of cognitive state changes based on a combination of EEG-engagement, EEG-workload, and heart rate metrics. Front. Neurosci. 2014, 8, 342. [Google Scholar] [CrossRef] [PubMed]
- Dijksterhuis, C.; de Waard, D.; Brookhuis, K.A.; Mulder, B.L.; de Jong, R. Classifying visuomotor workload in a driving simulator using subject specific spatial brain patterns. Front. Neurosci. 2013, 7, 149. [Google Scholar] [CrossRef] [PubMed]
- Nigg, J.T. On inhibition/disinhibition in developmental psychopathology: Views from cognitive and personality psychology and a working inhibition taxonomy. Psychol. Bull. 2000, 126, 220–246. [Google Scholar] [CrossRef] [PubMed]

| Performance Measure | Variable | Simulator Training Group (# N = 31; * N = 30) | d | Attention Training Group (N = 23) | d | Control Group (N = 23) | d | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre-Test | Post-Test | Pre-Test | Post-Test | Pre-Test | Post-Test | |||||
| M (SD) | M (SD) | M (SD) | M (SD) | M (SD) | M (SD) | |||||
| Inhibition performance a | Stroop: RT congruent trials in s | 0.899 (0.130) | 0.853 (0.147) | 0.926 (0.124) | 0.850 (0.131) | 0.893 (0.130) | 0.864 (0.118) | |||
| Stroop: RT incongruent trials in s | 1.060 (0.193) | 0.969 (0.184) | 1.101 (0.199) | 0.995 (0.179) | 1.079 (0.203) | 1.028 (0.172) | ||||
| Priming: RT normal trials in s | 0.660 (0.077) | 0.648 (0.094) | 0.664 (0.052) | 0.651 (0.076) | 0.696 (0.106) | 0681 (0.089) | ||||
| Priming: RT neg. prime trials in s | 0.675 (0.080) | 0.668 (0.089) | 0.683 (0.055) | 0.668 (0.079) | 0.710 (0.090) | 0.698 (0.083) | ||||
| Flanker: RT compatible trials in s | 0.626 (0.130) | 0.586 (0.112) | 0.612 (0.090) | 0.593 (0.104) | 0.623 (0.067) | 0.591 (0.066) | ||||
| Flanker: RT incomp. trials in s | 0.825 (0.280) | 0.722 (0.190) | 0.834 (0.264) | 0.751 (0.230) | 0.849 (0.222) | 0.737 (0.106) | ||||
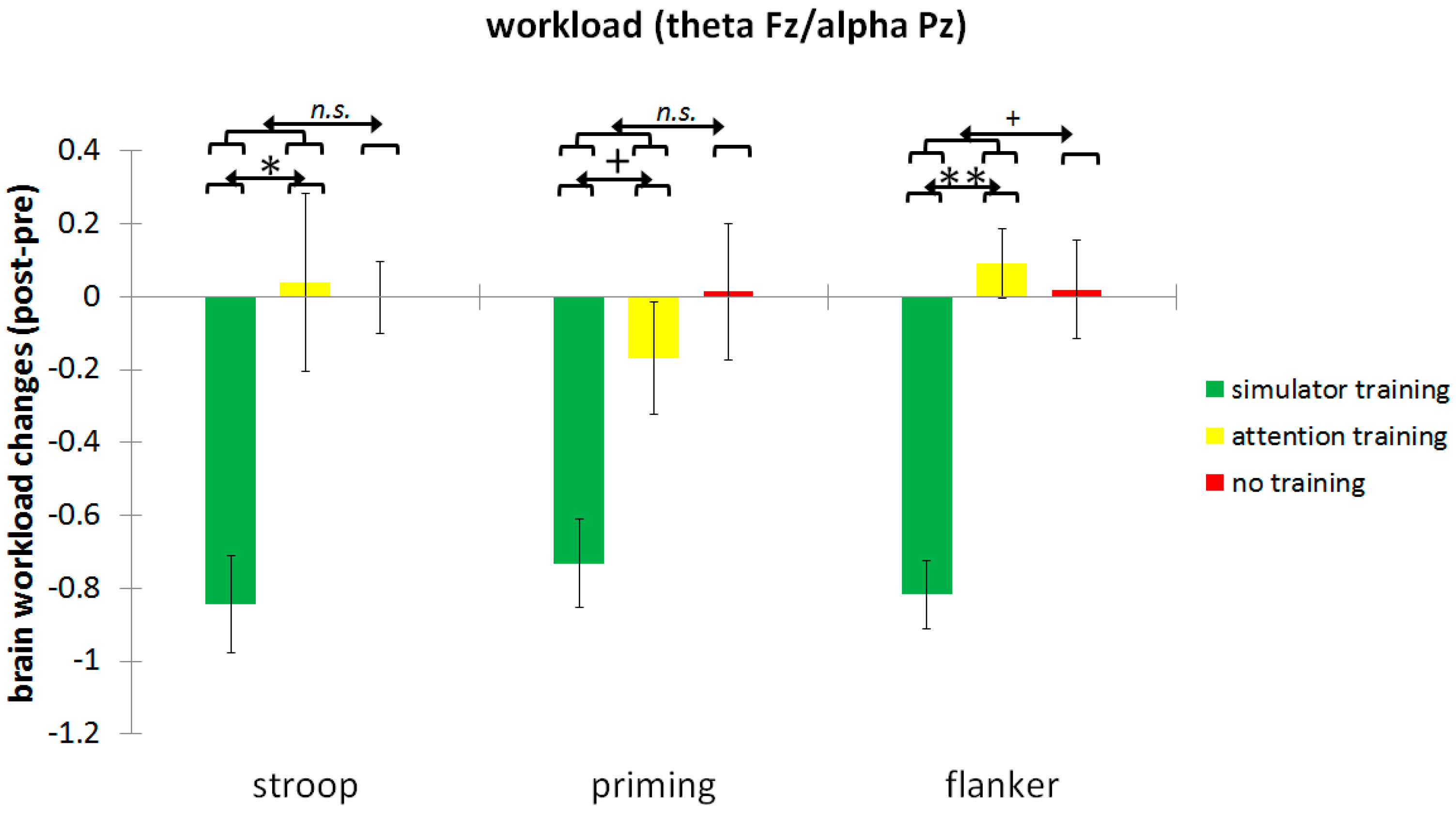
| Brain workload | Stroop | 2.06 (1.41) | 1.34 (0.67) | 0.54 | 2.03 (1.65) | 2.06 (1.52) | 0.01 | 1.41 (0.73) | 1.40 (0.98) | 0.01 |
| Priming | 2.01 (1.23) | 1.27 (0.57) | 0.61 | 1.61 (1.28) | 1.44 (0.70) | 0.15 | 1.57 (1.71) | 1.58 (1.00) | 0.01 | |
| Flanker | 1.96 (1.03) | 1.14 (0.80) | 0.67 | 1.36 (0.84) | 1.31 (0.86) | 0.05 | 1.43 (1.28) | 1.43 (1.00) | 0.00 | |
| Variable | B | SE | β |
|---|---|---|---|
| Brain workload during Stroop task | |||
| Linear interaction AB × C | −0.067 | 0.071 | −0.075 |
| Linear interaction A × B | −0.221 | 0.118 | −0.150 * |
| Brain workload during Priming task | |||
| Linear interaction AB × C | −0.077 | 0.065 | −0.094 |
| Linear interaction A × B | −0.140 | 0.108 | −0.103 + |
| Brain workload during Flanker task | |||
| Linear interaction AB × C | −0.072 | 0.048 | −0.101 + |
| Linear interaction A × B | −0.193 | 0.080 | −0.162 ** |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casutt, G.; Martin, M.; Jäncke, L. Driving Simulator Training Is Associated with Reduced Inhibitory Workload in Older Drivers. Geriatrics 2016, 1, 16. https://doi.org/10.3390/geriatrics1030016
Casutt G, Martin M, Jäncke L. Driving Simulator Training Is Associated with Reduced Inhibitory Workload in Older Drivers. Geriatrics. 2016; 1(3):16. https://doi.org/10.3390/geriatrics1030016
Chicago/Turabian StyleCasutt, Gianclaudio, Mike Martin, and Lutz Jäncke. 2016. "Driving Simulator Training Is Associated with Reduced Inhibitory Workload in Older Drivers" Geriatrics 1, no. 3: 16. https://doi.org/10.3390/geriatrics1030016
APA StyleCasutt, G., Martin, M., & Jäncke, L. (2016). Driving Simulator Training Is Associated with Reduced Inhibitory Workload in Older Drivers. Geriatrics, 1(3), 16. https://doi.org/10.3390/geriatrics1030016