Potential and Prospects of Continuous Polyhydroxyalkanoate (PHA) Production
Abstract
:1. Introduction
| Risk/Challenge | Remedy |
|---|---|
| Typically mesophile organisms are implemented in large-scale biotechnological production processes. Continuous cultivation of such organisms implies a substantial risk of microbial contamination; this can endanger whole fermentation batches, and, consequently, causes extensive economic loss. | Careful handling! High operational skills of staff! Application of extremophile production strains (e.g., thermophiles, osmophiles), which are robust, and outperform the growth rate of microbial competitors! Continuous antibiotic feed (generally not recommended!) |
| Long-term stability of production strain not assured | Strain improvement by genetic engineering. Application of robust production strains, which are rigid enough to withstand the shear forces generated by peristaltic pumping. Application of microbial fermentation broth that can conveniently be pumped (impedes the application of mycelia-forming species!) |
| Coming from the bioreactor’s interior, microbes may reach the tank of sterile medium; by subsequent conversion of substrate compounds, this can change the feed composition | Taking appropriate technical precautions, such as interrupting the liquid path in tubes by air barriers in which the medium drops, or by implementing thermo-traps (heating of the medium), etc. Note: Medium quality needs to be verified after the thermo-trap: precipitation or destruction of heat labile components has to be avoided. |
| Dropping of the media into the bioreactor’s interior (cultivation broth) results in small nutrient pulses and thus locally inconsistent nutrient concentrations. The same goes for locally fluctuating pH-values by acid or hydroxide pulses to maintain the pH-value. This prevents real “steady state” conditions. | Reduction of the volume ratio droplets/fermentation broth (small droplets favorable) |
| Highly fragile microbial cells can get disrupted by agitation and aeration | If possible, no excessive agitation and aeration. Application of robust microbial strains. Application of special stirrer (impeller) systems, which generate less shear stress. |
| Cell growth on the inner walls or other surfaces (e.g., baffles, probes, etc.) of the bioreactor during long-term operation | Equipping the reactor wall with a hydrophobic surface by application of e.g., silanes. “Close clearance” of inner walls by stirrers operating close to the wall |
| If mixing does not occur completely uniform, true “steady state” conditions are not warranted | Application of advanced adapted mixing systems (encompasses stirrer, sparger, baffles) |
| Instable reactor volumes by foaming, resulting in an overflow of fermentation broth | Application of effective antifoam agents tailored for the substrate-strain combination. Mechanical foam suppression by “anti-foam centrifuge”. Integrated solution: Application of a mass controlled pump (“mass-stat regime”) in combination with foam sensors and antifoam solutions. |
| Process separated in different phases (formation of secondary bio-product PHA after autocatalytic phase of biomass formation): optimum composition of the feed stream varies for the two different phases | Switch from single- to multistage continuous processes |
2. Continuous vs. Discontinuous PHA Production
| Criterion | Benefit | References |
|---|---|---|
| Investment costs for bioreactor | Due to higher volumetric productivity in continuous processes, (fed)batch cultivation requires large bioreactor facilities to generate the same output per time; continuous production contributes to lower investment costs by resorting to smaller operation facilities | [32] |
| Time demand | No “dead time” needed for pre- and post-treatment (“re-vamping”) of bioreactor | [32] |
| Investment costs for downstream processing | Manageable quantities of PHA-rich biomass accrue continuously. Downstream processing (e.g., extraction) of crude product stream can be accomplished continuously in smaller (cheaper!) recovery facilities. | [32,42] |
| Labor intensity | Higher for (fed)batch processes; not too much effort needed from staff during continuous operation as soon as steady-state conditions are reached. | [32] |
| Product quality | Higher consistency and uniformity of product quality (molar mass distribution, distribution of monomeric building blocks, thermal properties) | [43,44,45,46] |
| Triggering of polyester composition | Easier in continuous processes by possibility to exactly trigger the ratio between main- and co-substrates in the continuous feed stream. In multi-stage continuous processes: Possibility to design blocky structured polyesters consisting of soft- and hard segments. | [43,44,46,47,48] |
| Triggering molar masses | The applied dilution rate D significantly impacts the molar mass of PHA (molar mass direct proportional to D) | [49] |
| Making toxic substrates better accessible to the production strain | Toxic substrates can be continuously supplied exactly in accordance to their conversion by the cells. Thus, inhibiting concentration are never reached, actual zero concentration in cultivation medium. In addition, one can even profit from dual nutrient limited (DNL) growth conditions. Note: The acceptable substrate concentration depends on the strain properties (substrate affinity, Ks) and the specific growth rate µ (or D at steady-state, respectively) at which the strain is cultured. Excessively increasing D can cause a wash-out of the culture because of the increased substrate toxicity which by itself reduces the specific growth rate! | [33,43,44,50,51] |
| Convenient method for medium development and optimization | Fast reaction of steady-state culture kinetics to changing process parameters such as substrate concentrations triggered by pulse, shift or transient changes, temperature, pH-value, etc. | [41,52] |
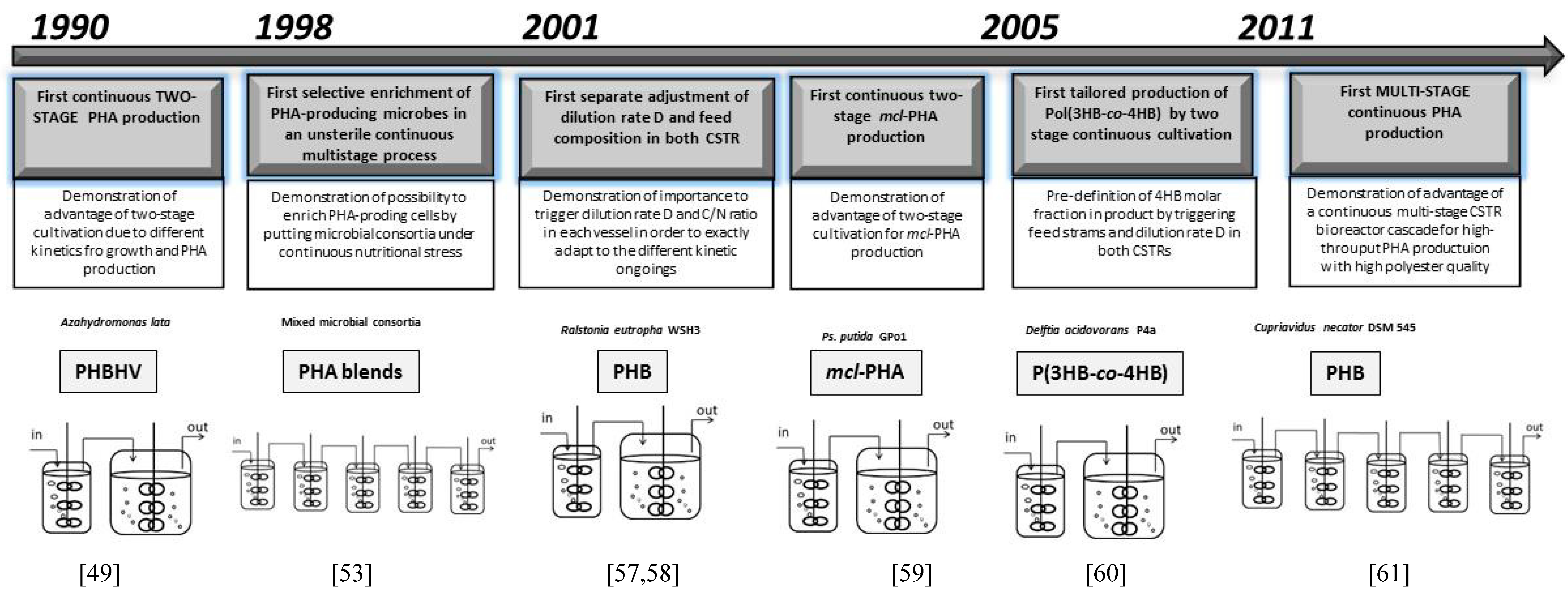
| Year | Strain | Aim | PHA Produced | Significance of the Work for the Scientific Field | Reference |
|---|---|---|---|---|---|
| 1972 | Azotobacter beijerinkii NCIB 9067 | Investigating the impact of oxygen limitation on PHA synthesis | PHB | First reported continuous PHA production | [53] |
| 1986 | Hyphomicrobium ZV620 | Investigating the impact of carbon to nitrogen ratio and D on activity of NH4+-assimilating enzymes, and on cellular composition (PHB content in biomass) | PHB | Confirmation of significance of carbon to nitrogen ratio to PHA mass fraction in biomass, confirmation of impact of D on cellular composition | [54] |
| 1990 | Cupriavidus necator DSM 545 | Increase of PHA productivity and intracellular PHA fraction by continuous operation | PHBHV | First continuous PHA production to enhance product output First continuous copolyester production | [47] |
| 1990 | Haloferax mediterranei DSM 1411 | Monoseptic continuous cultivation of osmophilic strain under unsterile conditions (“septic process”) for high-throughput PHA production | PHBHV | First continuous PHA production under unsterile conditions using extremophiles | [40] |
| 1991 | Ps. putida GPo1 | Continuous production of mcl-PHA from fatty acids and octane | mcl-PHA | First continuous mcl-PHA production | [52,55] |
| 1995 | Cupriavidus necator DSM 545 | Investigating the impact of D on polyester composition and productivity | PHBHV | First insights on impact of D on PHA production (quality and quantity) | [49] |
| 2000 | Ps. putida GPo1 | Continuous production of mcl-PHA from fatty acids under de facto C- and N-limitation | mcl-PHA | First Dual Nutrient Limited (DNL) continuous PHA production | [50] |
| 2005 | Cupriavidus necator DSM 545 | Continuous production of scl-PHA copolyesters with pre-determined monomeric composition | PHBHV | First triggering of scl-PHA copolyester composition on the nonomeric level by varying the composition of the continuous feed stream | [43] |
| 2004 | Ps. putida GPo1 | Continuous production of mcl-PHA copolyesters with pre-determined monomeric composition | mcl-PHA with aromatic building blocks | First triggering of mcl-PHA copolyester composition on the nonomeric level by varying the composition of the continuous feed stream First continuous production of an aromatic mcl-PHA | [56] |
| 2009 | Ps. putida GPo1 | Production of non-amorphous (crystalline) mcl-PHA on a relevant scale | mcl-PHA with aromatic building blocks | First continuous production of a crystalline mcl-PHA | [45] |
3. Single-Stage Continuous Systems with Pure Cultures
3.1. Scl-PHA Production by Single-Stage Continuous Cultures
3.2. Mcl-PHA Production by Single-Stage Continuous Cultures
4. Two-Stage Continuous Systems with Pure Cultures
4.1. Scl-PHA Production by Two-Stage Continuous Cultures
4.2. Mcl-PHA Production by Two-Stage Continuous Cultures
5. Multistage Process with Pure Cultures
6. Strategies for Continuous Enrichment of PHA-Accumulating Organisms
7. Unsterile and Open Continuous Processes for PHA Production
8. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Li, T.; Chen, X.B.; Chen, J.C.; Wu, Q.; Chen, G.Q. Open and continuous fermentation: Products, conditions and bioprocess economy. Biotechnol. J. 2014, 9, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Bankar, S.B.; Survase, S.A.; Singhal, R.S.; Granström, T. Continuous two stage acetone-butanol-ethanol fermentation with integrated solvent removal using Clostridium acetobutylicum B 5313. Bioresour. Technol. 2012, 106, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Raganati, F.; Olivieri, G.; Procentese, A.; Russo, M.E.; Salatino, P.; Marzocchella, A. Butanol production by bioconversion of cheese whey in a continuous packed bed reactor. Bioresour. Technol. 2013, 138, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Patakova, P.; Linhova, M.; Rychtera, M.; Paulova, L.; Melzoch, K. Novel and neglected issues of acetone-butanol-ethanol (ABE) fermentation by clostridia: Clostridium metabolic diversity, tools for process mapping and continuous fermentation systems. Biotechnol. Adv. 2013, 31, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Survase, S.A.; van Heiningen, A.; Granström, T. Continuous bio-catalytic conversion of sugar mixture to acetone-butanol-ethanol by immobilized Clostridium acetobutylicum DSM 792. Appl. Microbiol. Biotechnol. 2012, 93, 2309–2316. [Google Scholar] [CrossRef] [PubMed]
- Bouallagui, H.; Touhami, Y.; Hanafi, N.; Ghariani, A.; Hamdi, M. Performances comparison between three technologies for continuous ethanol production from molasses. Biomass Bioenergy 2013, 48, 25–32. [Google Scholar] [CrossRef]
- Crespo, C.F.; Badshah, M.; Alvarez, M.T.; Mattiasson, B. Ethanol production by continuous fermentation of D-(+)-cellobiose, D-(+)-xylose and sugarcane bagasse hydrolysate using the thermoanaerobe Caloramator boliviensis. Bioresour. Technol. 2012, 103, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Gabardo, S.; Rech, R.; Rosa, C.A.; Ayub, M.A.Z. Dynamics of ethanol production from whey and whey permeate by immobilized strains of Kluyveromyces marxianus in batch and continuous bioreactors. Renew. Energy 2014, 69, 89–96. [Google Scholar] [CrossRef]
- Saha, B.C.; Cotta, M.A. Continuous ethanol production from wheat straw hydrolysate by recombinant ethanologenic Escherichia coli strain FBR5. Appl. Microbiol. Biotechnol. 2011, 90, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Dey, P.; Pal, P. Direct production of l (+) lactic acid in a continuous and fully membrane-integrated hybrid reactor system under non-neutralizing conditions. J. Membr. Sci. 2012, 389, 355–362. [Google Scholar] [CrossRef]
- Roy, D.; Goulet, J.; Le Duy, A. Continuous production of lactic acid from whey permeate by free and calcium alginate entrapped Lactobacillus helveticus. J. Dairy Sci. 1987, 70, 506–513. [Google Scholar] [CrossRef]
- Silva, E.M.; Yang, S.T. Kinetics and stability of a fibrous-bed bioreactor for continuous production of lactic acid from unsupplemented acid whey. J. Biotechnol. 1995, 41, 59–70. [Google Scholar] [CrossRef]
- Zhang, D.X.; Cheryan, M. Starch to lactic acid in a continuous membrane bioreactor. Process. Biochem. 1994, 29, 145–150. [Google Scholar] [CrossRef]
- Angumeenal, A.R.; Venkappayya, D. An overview of citric acid production. Food Sci. Technol.-LEB 2013, 50, 367–370. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, C.B. Continuous production of citric acid from sugarcane molasses using a combination of submerged immobilized and surface stabilized cultures of Aspergillus niger KCU 520. Biotechnol. Lett. 1994, 16, 599–604. [Google Scholar] [CrossRef]
- Wieczorek, S.; Brauer, H. Continuous production of citric acid with recirculation of the fermentation broth after product recovery. Bioprocess Eng. 1997, 18, 1–5. [Google Scholar] [CrossRef]
- Phillips, K.R.; Pik, J.; Lawford, H.G.; Lavers, B.; Kligerman, A.; Lawford, G.R. Production of curdlan-type polysaccharide by Alcaligenes faecalis in batch and continuous culture. Can. J. Microbiol. 1983, 29, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.Y.; Yousef, A.E.; Yang, S.T. Continuous production of pediocin by immobilized Pediococcus acidilactici PO2 in a packed-bed bioreactor. Appl. Microbiol. Biotechnol. 1996, 45, 589–594. [Google Scholar] [CrossRef]
- Digan, M.E.; Lair, S.V.; Brierley, R.A.; Siegel, R.S.; Williams, M.E.; Ellis, S.B.; Kellaris, P.A.; Provow, S.A.; Craig, W.S.; Veliçelebi, G.; et al. Continuous production of a novel lysozyme via secretion from the yeast, Pichia pastoris. Nat. Biotechnol. 1989, 7, 160–164. [Google Scholar]
- Wan, J.; Hickey, M.W.; Coventry, M.J. Continuous production of bacteriocins, brevicin, nisin and pediocin, using calcium alginate-immobilized bacteria. J. Appl. Microbiol. 1995, 79, 671–676. [Google Scholar] [CrossRef]
- Almeida, C.; Brányik, T.; Moradas-Ferreira, P.; Teixeira, J. Continuous production of pectinase by immobilized yeast cells on spent grains. J. Biosci. Bioeng. 2003, 96, 513–518. [Google Scholar] [CrossRef]
- Domingues, L.; Lima, N.; Teixeira, J.A. Aspergillus niger β-galactosidase production by yeast in a continuous high cell density reactor. Process Biochem. 2005, 40, 1151–1154. [Google Scholar] [CrossRef]
- Yang, H.; Shao, P.; Lu, T.; Shen, J.; Wang, D.; Xu, Z.; Yuan, X. Continuous bio-hydrogen production from citric acid wastewater via facultative anaerobic bacteria. Int. J. Hydrog. Energy 2006, 31, 1306–1313. [Google Scholar]
- Koller, M.; Salerno, A.; Dias, M.; Reiterer, A.; Braunegg, G. Modern biotechnological polymer synthesis: A review. Food Technol. Biotechnol. 2010, 48, 255–269. [Google Scholar]
- Castilho, L.R.; Mitchell, D.A.; Freire, D.M. Production of polyhydroxyalkanoates (PHAs) from waste materials and by-products by submerged and solid-state fermentation. Bioresour. Technol. 2009, 100, 5996–6009. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Muhr, A. Continuous production mode as a viable process-engineering tool for efficient poly(hydroxyalkanoate) (PHA) bio-production. Chem. Biochem. Eng. Q. 2014, 28, 65–77. [Google Scholar]
- Koller, M.; Hesse, P.; Salerno, A.; Reiterer, A.; Braunegg, G. A viable antibiotic strategy against microbial contamination in biotechnological production of polyhydroxyalkanoates from surplus whey. Biomass Bioenergy 2011, 35, 748–753. [Google Scholar] [CrossRef]
- Horvat, P.; Špoljarić, I.V.; Lopar, M.; Atlić, A.; Koller, M.; Braunegg, G. Mathematical modelling and process optimization of a continuous 5-stage bioreactor cascade for production of poly[-(R)-3-hydroxybutyrate] by Cupriavidus necator. Bioprocess Biosyst. Eng. 2013, 36, 1235–1250. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Salerno, A.; Braunegg, G. Polyhydroxyalkanoates: Basics, production and applications of microbial biopolyesters. In Bio-Based Plastics: Materials and Applications, 1st ed.; Wiley: West Sussex, UK, 2013; pp. 137–170. [Google Scholar]
- Tan, G.Y.A.; Chen, C.L.; Li, L.; Ge, L.; Wang, L.; Razaad, I.M.N.; Li, Y.; Zhao, L.; Mo, Y.; Wang, J.Y. Start a research on biopolymer polyhydroxyalkanoate (PHA): A review. Polymers 2014, 6, 706–754. [Google Scholar] [CrossRef]
- Nonato, R.; Mantelatto, P.; Rossell, C. Integrated production of biodegradable plastic, sugar and ethanol. Appl. Microbiol. Biotechonl. 2001, 57, 1–5. [Google Scholar]
- Braunegg, G.; Lefebvre, G.; Renner, G.; Zeiser, A.; Haage, G.; Loidl-Lanthaler, K. Kinetics as a tool for polyhydroxyalkanoate production optimization. Can. J. Microbiol. 1995, 41, 239–248. [Google Scholar] [CrossRef]
- Durner, R.; Zinn, M.; Witholt, B.; Egli, T. Accumulation of poly[(R)-3-hydroxyalkanoates] in Pseudomonas oleovorans during growth in batch and chemostat culture with different carbon sources. Biotechnol. Bioeng. 2001, 72, 278–288. [Google Scholar] [CrossRef]
- Rai, R.; Keshavarz, T.; Roether, J.A.; Boccaccini, A.R.; Roy, I. Medium chain length polyhydroxyalkanoates, promising new biomedical materials for the future. Mater. Sci. Eng. R 2011, 72, 29–47. [Google Scholar] [CrossRef]
- Xi, J.; Wu, Q.; Yan, Y.; Zhang, Z.; Yu, P.H.F.; Cheung, M.K.; Zhang, R.; Chen, G.Q. Hyperproduction of polyesters consisting of medium-chain-length hydroxyalkanoate monomers by strain Pseudomonas stutzeri 1317. Antonie van Leeuwenhoek 2000, 78, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Muhr, A.; Rechberger, E.M.; Salerno, A.; Reiterer, A.; Schiller, M.; Kwiecień, M.; Adamus, G.; Kowalczuk, M.; Strohmeier, K.; Schober, S.; et al. Biodegradable latexes from animal-derived waste: Biosynthesis and characterization of mcl-PHA accumulated by Ps. citronellolis. React. Funct. Polym. 2013, 73, 1391–1398. [Google Scholar]
- Muhr, A.; Rechberger, E.M.; Salerno, A.; Reiterer, A.; Malli, K.; Strohmeier, K.; Schober, S.; Mittelbach, M.; Koller, M. Novel description of mcl-PHA biosynthesis by Pseudomonas chlororaphis from animal-derived waste. J. Biotechnol. 2013, 165, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Zinn, M. Tailor-made synthesis of polyhydroxyalkanoate. Eur. Cell Mater. 2003, 5, 38–39. [Google Scholar]
- Sun, Z.; Ramsay, J.A.; Guay, M.; Ramsay, B.A. Fermentation process development for the production of medium-chain-length poly-3-hydroxyalkanoates. Appl. Microbiol. Biotechnol. 2007, 75, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Lillo, J.G.; Rodriguez-Valera, F. Effects of culture conditions on poly(β-hydroxybutyric acid) production by Haloferax mediterranei. Appl. Environ. Microbiol. 1990, 56, 2517–2521. [Google Scholar] [PubMed]
- Tsuchiya, Y.; Nishio, N.; Nagai, S. Medium optimization for a methanol utilizing bacterium based on chemostat theory. Eur. J. Appl. Microbiol. Biotechnol. 1980, 9, 121–127. [Google Scholar] [CrossRef]
- Villano, M.; Valentino, F.; Barbetta, A.; Martino, L.; Scandola, M.; Majone, M. Polyhydroxyalkanoates production with mixed microbial cultures: From culture selection to polymer recovery in a high-rate continuous process. New Biotechnol. 2014, 31, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Zinn, M.; Weilenmann, H.U.; Hany, R.; Schmid, M.; Egli, T.H. Tailored Synthesis of Poly([R]-3-hydroxybutyrate-co-3-hydroxyvalerate) (PHB/HV) in Ralstonia eutropha DSM 428. Acta Biotechnol. 2003, 23, 309–316. [Google Scholar] [CrossRef]
- Hany, R.; Böhlen, C.; Geiger, T.; Schmid, M.; Zinn, M. Toward non-toxic antifouling: Synthesis of hydroxy-, cinnamic acid-, sulfate-, and zosteric acid-labeled poly[3-hydroxyalkanoates]. Biomacromolecules 2004, 5, 1452–1456. [Google Scholar] [CrossRef] [PubMed]
- Hany, R.; Brinkmann, M.; Ferri, D.; Hartmann, R.; Pletscher, E.; Rentsch, D.; Zinn, M. Crystallization of an Aromatic Biopolyester. Macromolecules 2009, 42, 6322–6326. [Google Scholar] [CrossRef]
- Jung, K.; Hazenberg, W.; Prieto, M.; Witholt, B. Two-stage continuous process development for the production of medium-chain-length poly(3-hydroxyalkanoates). Biotechnol. Bioeng. 2001, 72, 19–24. [Google Scholar] [CrossRef]
- Ramsay, B.A.; Lomaliza, K.; Chavarie, C.; Dube, B.; Bataille, P.; Ramsay, J.A. Production of poly-(beta-hydroxybutyric-co-β-hydroxyvaleric) acids. Appl. Environ. Microbiol. 1990, 56, 2093–2098. [Google Scholar] [PubMed]
- Yu, S.T.; Lin, C.C.; Too, J.R. PHBV production by Ralstonia eutropha in a continuous stirred tank reactor. Process Biochem. 2005, 40, 2729–2734. [Google Scholar] [CrossRef]
- Koyama, N.; Doi, Y. Continuous production of poly (3-hydroxybutyrate-co-3-hyhroxyvalerate) by Alcaligenes eutrophus. Biotechnol. Lett. 1995, 17, 281–284. [Google Scholar]
- Durner, R.; Witholt, B.; Egli, T. Accumulation of poly[(R)-3-hydroxyalkanoates] in Pseudomonas oleovorans during growth with octanoate in continuous culture at different dilution rates. Appl. Environ. Microbiol. 2000, 66, 3408–3414. [Google Scholar] [CrossRef] [PubMed]
- Egli, T.; Zinn, M. The concept of multiple-nutrient-limited growth of microorganisms and its application in biotechnological processes. Biotechnol. Adv. 2003, 22, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Zinn, M.; Durner, R.; Zinn, H.; Ren, Q.; Egli, T.; Witholt, B. Growth and accumulation dynamics of poly (3-hydroxyalkanoate)(PHA) in Pseudomonas putida GPo1 cultivated in continuous culture under transient feed conditions. Biotechnol. J. 2011, 6, 1240–1252. [Google Scholar] [CrossRef] [PubMed]
- Senior, P.J.; Beech, G.A.; Ritchie, G.A.F.; Dawes, E.A. The role of oxygen limitation in the formation of poly-β-hydroxybutyrate during batch and continuous culture of Azotobacter beijerinckii. Biochem. J. 1972, 128, 1193–1201. [Google Scholar] [PubMed]
- Gräzer-Lampart, S.D.; Egli, T.; Hamer, G. Growth of Hyphomicrobium ZV620 in the chemostat: Regulation of NH4+-assimilating enzymes and cellular composition. J. Gen. Microbiol. 1986, 132, 3337–3347. [Google Scholar]
- Ramsay, B.A.; Saracovan, I.; Ramsay, J.A.; Marchessault, R.H. Continuous production of long-side-chain poly-β-hydroxyalkanoates by Pseudomonas oleovorans. Appl. Environ. Microbiol. 1991, 57, 625–629. [Google Scholar] [PubMed]
- Hartmann, R.; Hany, R.; Geiger, T.; Egli, T.; Witholt, B.; Zinn, M. Tailored biosynthesis of olefinic medium-chain-length Poly[(R)-3-hydroxyalkanoates] in Pseudomonas putida GPo1 with improved thermal properties. Macromolecules 2004, 37, 6780–6785. [Google Scholar] [CrossRef]
- Du, G.; Chen, J.; Yu, J.; Lun, S. Continuous production of poly-3-hydroxybutyrate by Ralstonia eutropha in a two-stage culture system. J. Biotechnol. 2001, 88, 59–65. [Google Scholar] [CrossRef]
- Du, G.; Chen, J.; Yu, J.; Lun, S. Kinetic studies on poly-3-hydroxybutyrate formation by Ralstonia eutropha in a two-stage continuous culture system. Process Biochem. 2001, 37, 219–227. [Google Scholar] [CrossRef]
- Preusting, H.; Kingma, J.; Witholt, B. Physiology and polyester formation of Pseudomonas oleovorans in continuous two-liquid-phase cultures. Enzym. Microb. Technol. 1991, 13, 770–780. [Google Scholar] [CrossRef]
- Mothes, G.; Ackermann, J.U. Synthesis of poly(3-hydroxybutyrate-co-4-hydroxybutyrate) with a target mole fraction of 4-hydroxybutyric acid units by two-stage continuous cultivation of Delftia acidovorans P4a. Eng. Life Sci. 2005, 5, 58–62. [Google Scholar]
- Atlić, A.; Koller, M.; Scherzer, D.; Kutschera, C.; Grillo-Fernandes, E.; Horvat, P.; Chiellini, E.; Braunegg, G. Continuous production of poly ([R]-3-hydroxybutyrate) by Cupriavidus necator in a multistage bioreactor cascade. Appl. Microbiol. Biotechnol. 2011, 91, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Kollmer, A.; Witholt, B. Effects of biosurfactant and emulsification on two-liquid phase Pseudomonas oleovorans cultures and cell-free emulsions containing n-decane. Enzym. Microb. Technol. 1998, 22, 487–493. [Google Scholar] [CrossRef]
- Ramsay, B.A.; Saracovan, I.; Ramsay, J.A.; Marchessault, R.H. Effect of nitrogen limitation on long-side-chain poly-β-hydroxyalkanoate synthesis by Pseudomonas resinovorans. Appl. Environ. Microbiol. 1992, 58, 744–746. [Google Scholar] [PubMed]
- Hazenberg, W.; Witholt, B. Efficient production of medium-chain-length poly(3-hydroxyalkanoates) from octane by Pseudomonas oleovorans: Economic considerations. Appl. Microbiol. Biotechnol. 1997, 48, 588–596. [Google Scholar] [CrossRef]
- Follonier, S.; Henes, B.; Panke, S.; Zinn, M. Putting cells under pressure: A simple and efficient way to enhance the productivity of medium-chain-length polyhydroxyalkanoate in processes with Pseudomonas putida KT2440. Biotechnol. Bioeng. 2012, 109, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Kedia, G.; Passanha, P.; Dinsdale, R.M.; Guwy, A.J.; Esteves, S.R. Evaluation of feeding regimes to enhance PHA production using acetic and butyric acids by a pure culture of Cupriavidus necator. Biotechnol. Bioprocess Eng. 2014, 19, 989–995. [Google Scholar] [CrossRef]
- Pederson, E.N.; McChalicher, C.W.; Srienc, F. Bacterial synthesis of PHA block copolymers. Biomacromolecules 2006, 7, 1904–1911. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Chung, A.L.; Wu, L.P.; Zhang, X.; Wu, Q.; Chen, J.C.; Chen, G.Q. Biosynthesis and characterization of polyhydroxyalkanoate block copolymer P3HB-b-P4HB. Biomacromolecules 2011, 12, 3166–3173. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, L.; Wu, L.P.; Chen, J.; Chen, G.Q. Synthesis of Diblock copolymer poly-3-hydroxybutyrate-block-poly-3-hydroxyhexanoate [PHB-b-PHHx] by a β-oxidation weakened Pseudomonas putida KT2442. Microb. Cell Factories 2012, 11, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, L.; Wu, L.P.; Meng, D.; Chen, J.; Chen, G.Q. Biosynthesis and characterization of diblock copolymer of P (3-hydroxypropionate)-block-P (4-hydroxybutyrate) from recombinant Escherichia coli. Biomacromolecules. 2013, 14, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, L.; Wu, L.P.; Dechuan, M.; Chen, J.; Wu, Q.; Chen, G.Q. Pseudomonas putida KT2442 as a platform for the biosynthesis of polyhydroxyalkanoates with adjustable monomer contents and compositions. Bioresour. Technol. 2013, 142, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Lopar, M.; Špoljarić, I.V.; Atlić, A.; Koller, M.; Braunegg, G.; Horvat, P. Five-step continuous production of PHB analyzed by elementary flux, modes, yield space analysis and high structured metabolic model. Biochem. Eng. J. 2013, 79, 57–70. [Google Scholar]
- Johnson, K.; Jiang, Y.; Kleerebezem, R.; Muyzer, G.; van Loosdrecht, M.C. Enrichment of a mixed bacterial culture with a high polyhydroxyalkanoate storage capacity. Biomacromolecules 2009, 10, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Moralejo-Gárate, H.; Mar’atusalihat, E.; Kleerebezem, R.; van Loosdrecht, M.C. Microbial community engineering for biopolymer production from glycerol. Appl. Microbiol. Biotechnol. 2011, 92, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Moralejo-Gárate, H.; Palmeiro-Sánchez, T.; Kleerebezem, R.; Mosquera-Corral, A.; Campos, J.L.; van Loosdrecht, M. Influence of the cycle length on the production of PHA and polyglucose from glycerol by bacterial enrichments in sequencing batch reactors. Biotechnol. Bioeng. 2013, 110, 3148–3155. [Google Scholar] [CrossRef] [PubMed]
- Renner, G.; Schellauf, F.; Braunegg, G. Selective enrichment of bacteria accumulating polyhydroxyalkanoates in multistage continuous culture. Food Technol. Biotechnol. 1998, 36, 203–208. [Google Scholar]
- Yin, J.; Chen, J.C.; Wu, Q.; Chen, G.Q. Halophiles, coming stars for industrial biotechnology. Biotechnol. Adv. 2014. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; di Donato, P.; Abbamondi, G.R.; Nicolaus, B. Synthesis, production, and biotechnological applications of exopolysaccharides and polyhydroxyalkanoates by archaea. Archaea 2011, 2011. Article ID 693253. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Potential of various archae-and eubacterial strains as industrial polyhydroxyalkanoate producers from whey. Macromol. Biosci. 2007, 7, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Quillaguamán, J.; Guzmán, H.; van-Thuoc, D.; Hatti-Kaul, R. Synthesis and production of polyhydroxyalkanoates by halophiles: Current potential and future prospects. Appl. Microbiol. Biotechnol. 2010, 85, 1687–1696. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Biosynthesis of high quality polyhydroxyalkanoate co-and terpolyesters for potential medical application by the archaeon Haloferax mediterranei. Macromol. Symp. 2007, 253, 33–39. [Google Scholar] [CrossRef]
- Chen, C.W.; Don, T.M.; Yen, H.F. Enzymatic extruded starch as a carbon source for the production of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. Process Biochem. 2006, 41, 2289–2296. [Google Scholar] [CrossRef]
- Hermann-Krauss, C.; Koller, M.; Muhr, A.; Fasl, H.; Stelzer, F.; Braunegg, G. Archaeal production of polyhydroxyalkanoate (PHA) co-and terpolyesters from biodiesel industry-derived by-products. Archaea 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Saha, J.; Haldar, S.; Bhowmic, A.; Mukhopadhyay, U.K.; Mukherjee, J. Production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei using rice-based ethanol stillage with simultaneous recovery and re-use of medium salts. Extremophiles 2014, 18, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Pramanik, A.; Maji, S.K.; Haldar, S.; Mukhopadhyay, U.K.; Mukherjee, J. Utilization of vinasse for production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei. AMB Express 2012, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Cai, L.; Liu, H.; Liu, X.; Han, J.; Zhou, J.; Xiang, H. Identification of the haloarchaeal phasin (PhaP) that functions in polyhydroxyalkanoate accumulation and granule formation in Haloferax mediterranei. Appl. Environ. Microbiol. 2012, 78, 1946–1952. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Zhang, F.; Hou, J.; Liu, X.; Li, M.; Liu, H.; Cai, L.; Zhang, B.; Chen, Y.; Zhou, J.; et al. Complete genome sequence of the metabolically versatile halophilic archaeon Haloferax mediterranei, a poly (3-hydroxybutyrate-co-3-hydroxyvalerate) producer. J. Bacteriol. 2012, 194, 4463–4464. [Google Scholar]
- Han, J.; Hou, J.; Zhang, F.; Ai, G.; Li, M.; Cai, S.; Zhao, D.; Zhou, J.; Xiang, H. Multiple propionyl coenzyme A-supplying pathways for production of the bioplastic poly (3-hydroxybutyrate-co-3-hydroxyvalerate) in Haloferax mediterranei. Appl. Environ. Microbiol. 2013, 79, 2922–2931. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Cai, L.; Wu, J.; Li, M.; Liu, H.; Han, J.; Zhou, J.; Xiang, H. Improving polyhydroxyalkanoate production by knocking out the genes involved in exopolysaccharide biosynthesis in Haloferax mediterranei. Appl. Microbiol. Biotechnol. 2013, 97, 3027–3036. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wu, L.P.; Hou, J.; Zhao, D.; Xiang, H. Biosynthesis, characterization and hemostasis potential of tailor-made Poly (3-hydroxybutyrate-co-3-hydroxyvalerate) produced by Haloferax mediterranei. Biomacromolecules 2015, 16, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Gasser, I.; Schmid, F.; Berg, G. Linking ecology with economy: Insights into polyhydroxyalkanoate-producing microorganisms. Eng. Life Sci. 2011, 11, 222–237. [Google Scholar] [CrossRef]
- Koller, M. Recycling of waste streams of the biotechnological poly(hydroxyalkanoate) production by Haloferax mediterranei on whey. Int. J. Polym. Sci. 2015, 2015. [Google Scholar] [CrossRef]
- Yue, H.; Ling, C.; Yang, T.; Chen, X.; Chen, Y.; Deng, H.; Wu, Q.; Chen, J.; Chen, G.Q. A seawater-based open and continuous process for polyhydroxyalkanoates production by recombinant Halomonas campaniensis LS21 grown in mixed substrates. Biotechnol. Biofuels 2014, 7, 108–119. [Google Scholar] [CrossRef]
- Tan, D.; Xue, Y.S.; Aibaidula, G.; Chen, G.Q. Unsterile and continuous production of polyhydroxybutyrate by Halomonas TD01. Bioresour. Technol. 2011, 102, 8130–8136. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.Z.; Tan, D.; Aibaidula, G.; Wu, Q.; Chen, J.C.; Chen, G.Q. Development of Halomonas TD01 as a host for open production of chemicals. Metab. Eng. 2014, 23, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Wu, Q.; Chen, J.C.; Chen, G.Q. Engineering Halomonas TD01 for the low-cost production of polyhydroxyalkanoates. Metab. Eng. 2014, 26, 34–47. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koller, M.; Braunegg, G. Potential and Prospects of Continuous Polyhydroxyalkanoate (PHA) Production. Bioengineering 2015, 2, 94-121. https://doi.org/10.3390/bioengineering2020094
Koller M, Braunegg G. Potential and Prospects of Continuous Polyhydroxyalkanoate (PHA) Production. Bioengineering. 2015; 2(2):94-121. https://doi.org/10.3390/bioengineering2020094
Chicago/Turabian StyleKoller, Martin, and Gerhart Braunegg. 2015. "Potential and Prospects of Continuous Polyhydroxyalkanoate (PHA) Production" Bioengineering 2, no. 2: 94-121. https://doi.org/10.3390/bioengineering2020094
APA StyleKoller, M., & Braunegg, G. (2015). Potential and Prospects of Continuous Polyhydroxyalkanoate (PHA) Production. Bioengineering, 2(2), 94-121. https://doi.org/10.3390/bioengineering2020094







