Algal and Cyanobacteria Cell Walls as Biosorbents for Phenolic Compounds: Comparative Performance and Sustainability Assessment of Limnospira platensis
Abstract
1. Introduction
2. Materials and Methods
2.1. Algal Cultivation and Biomass Functionalisation
2.2. Limnospira Platensis Growth and Characterization
2.3. Phenol Adsorption Experiments
- •
- Species screening—A fixed biomass dose of 0.1 g·L−1 (dry weight) was tested for all algal species with phenolic solution (30 mg·L−1) to compare uptake capacity.
- •
- Dose optimization with L. platensis—Biomass concentration was varied (0.1–1.0 g·L−1) with 30 mg·L−1 phenolic solution to evaluate adsorption as a function of biomass-to-phenol ratio. Untreated, pigment-extracted, and protein-extracted L. platensis were tested under identical conditions.
Fitting of Adsorption Data
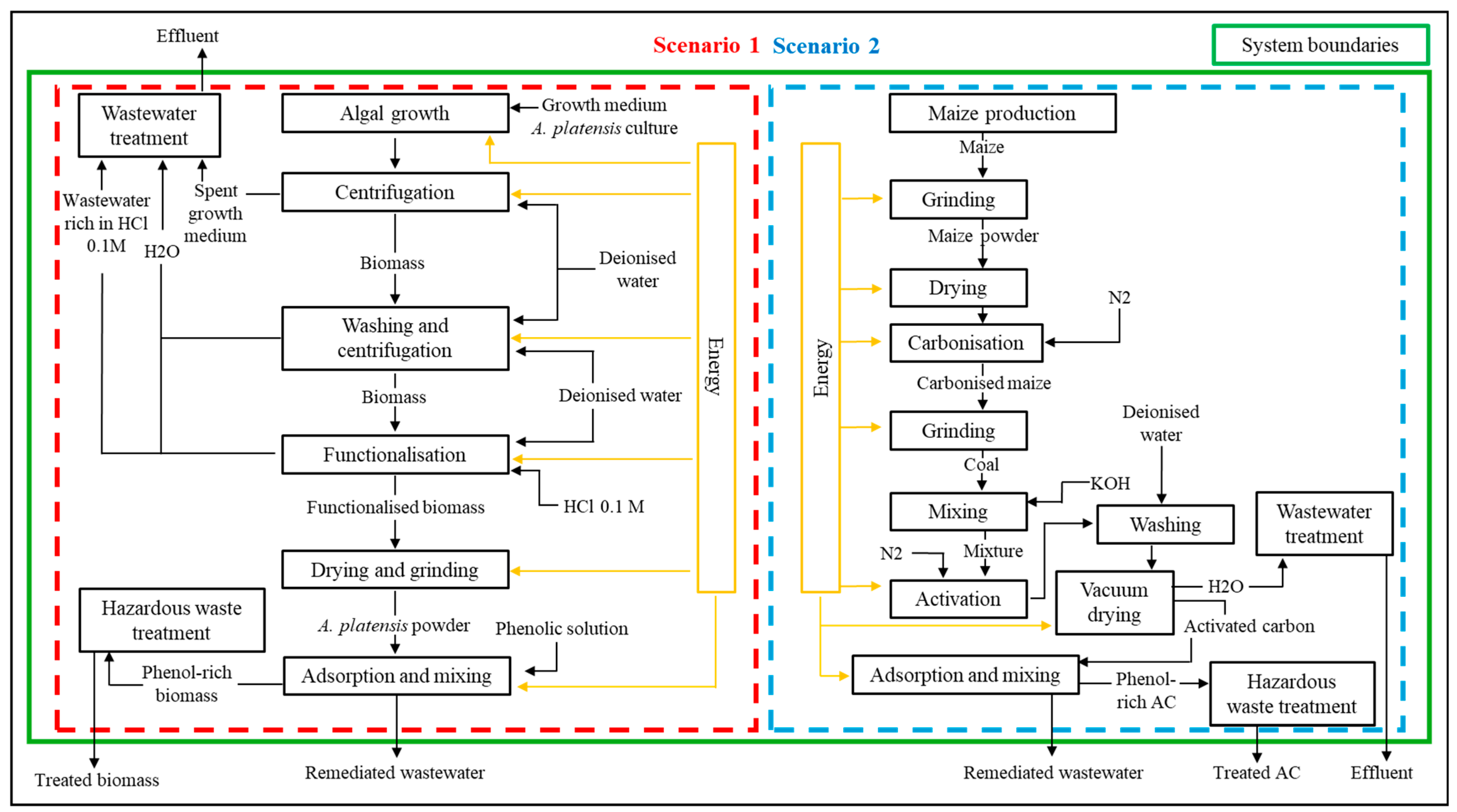
2.4. Life Cycle Assessment (LCA) Methodology
2.5. Statistical Analysis
3. Results
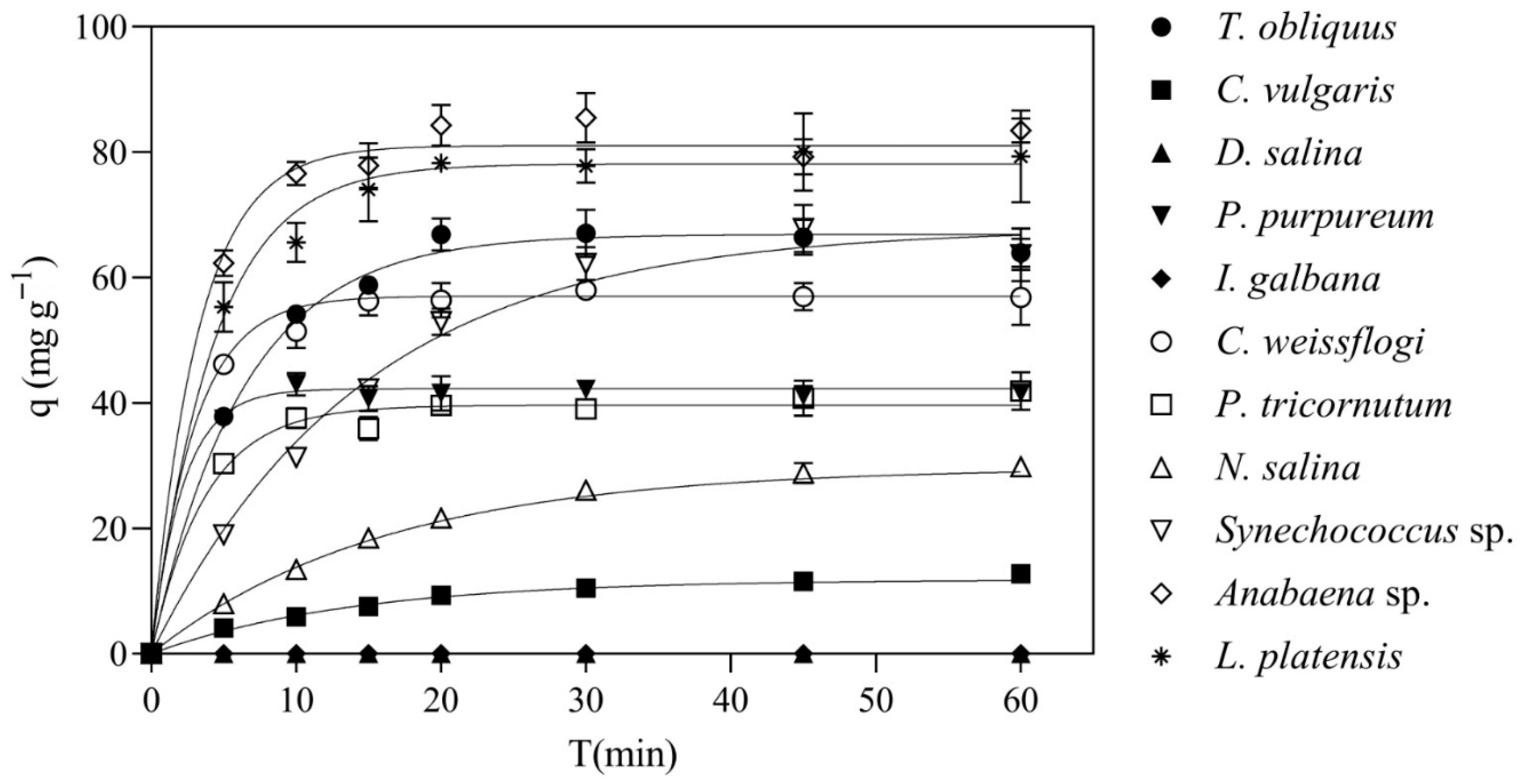
3.1. Species Screening
3.2. Optimization of Limnospira Platensis as Biosorbent
| Model | Parameter | Value | R2 | ||
|---|---|---|---|---|---|
| Langmuir | Qmax (mg g−1) | 1.38 × 107 | ± | 2.4 × 106 | 0.732 |
| KL (L mg−1) | 2.8 × 10−7 | ± | 1.2 × 10−7 | ||
| Freundlich | KF (mg g−1) | 0.746 | ± | 0.231 | 0.821 |
| 1/n | 1.55 | ± | 0.59 | ||

3.3. L. platensis as Biosorbent After Protein or Phycobilin Extraction
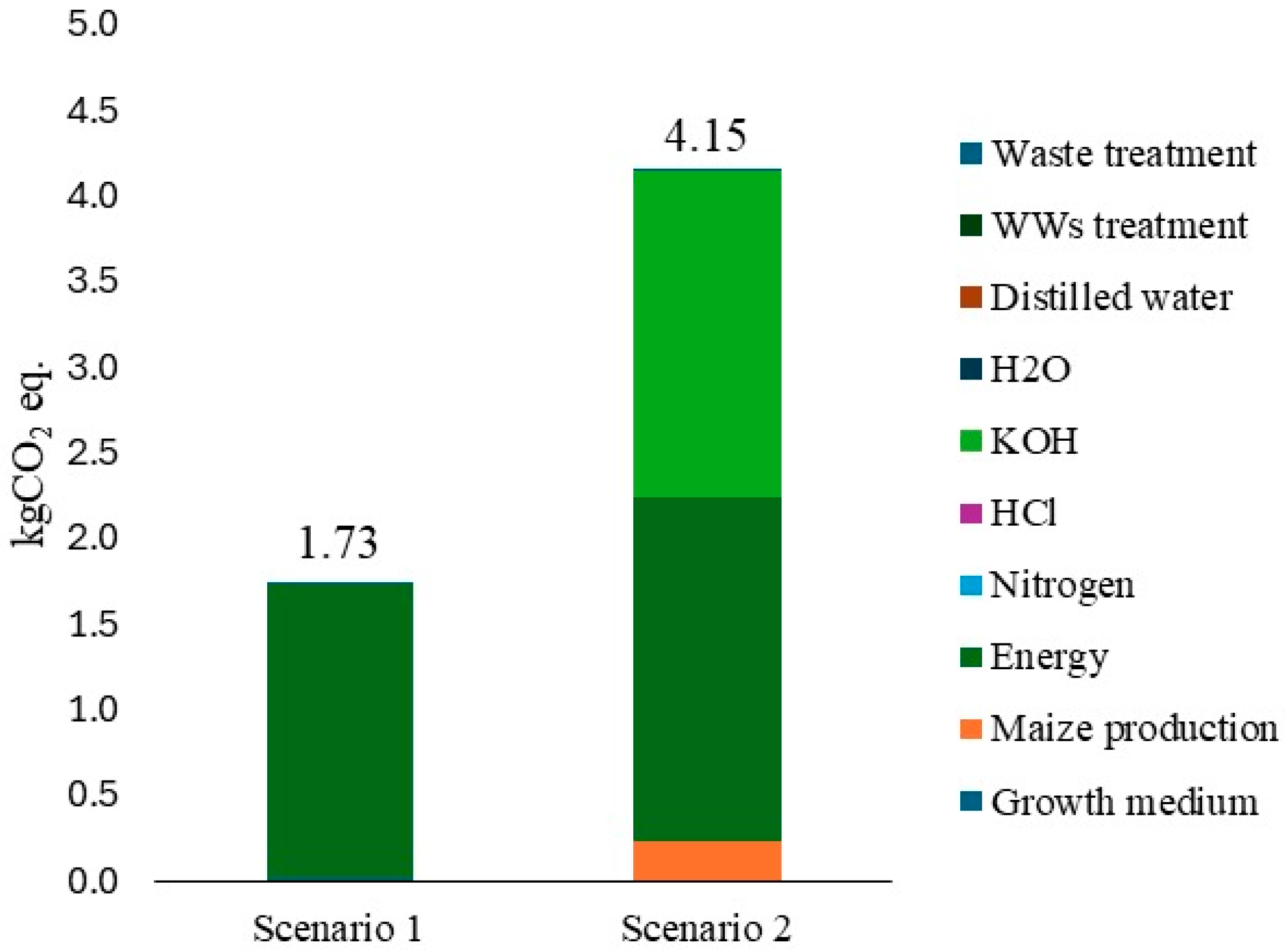
3.4. LCA Analysis of L. platensis as Biosorbent
4. Discussion
4.1. The Role of Algal Cell Wall in Adsorbing Phenols
Losses of Adsorption Capacity in L. platensis After Macromolecular Extraction
4.2. Limnospira Platensis as Biosorbent (Capacity and LCA Outcomes)
4.3. Future Perspectives
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Souilem, S.; El-Abbassi, A.; Kiai, H.; Hafidi, A.; Sayadi, S.; Galanakis, C.M. Olive oil production sector: Environmental effects and sustainability challenges. In Olive Mill Waste: Recent Advances for the Sustainable Management; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–28. [Google Scholar] [CrossRef]
- Regni, L.; Nasini, L.; Ilarioni, L.; Brunori, A.; Massaccesi, L.; Agnelli, A.; Proietti, P. Long term amendment with fresh and composted solid olive mill waste on olive grove affects carbon sequestration by prunings, fruits, and soil. Front. Plant Sci. 2017, 7, 228296. [Google Scholar] [CrossRef]
- Paraskeva, P.; Diamadopoulos, E. Technologies for olive mill wastewater (OMW) treatment: A review. J. Chem. Technol. Biotechnol. 2006, 81, 1475–1485. [Google Scholar] [CrossRef]
- Danellakis, D.; Ntaikou, I.; Kornaros, M.; Dailianis, S. Olive oil mill wastewater toxicity in the marine environment: Alterations of stress indices in tissues of mussel Mytilus galloprovincialis. Aquat. Toxicol. 2011, 101, 358–366. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Scalbert, A.; Johnson, I.T.; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr. 2005, 81, 215S–217S. [Google Scholar] [CrossRef]
- Pavlidou, A.; Anastasopoulou, E.; Dassenakis, M.; Hatzianestis, I.; Paraskevopoulou, V.; Simboura, N.; Rousselaki, E.; Drakopoulou, P. Effects of olive oil wastes on river basins and an oligotrophic coastal marine ecosystem: A case study in Greece. Sci. Total Environ. 2014, 497–498, 38–49. [Google Scholar] [CrossRef]
- Barbera, A.C.; Maucieri, C.; Cavallaro, V.; Ioppolo, A.; Spagna, G. Effects of spreading olive mill wastewater on soil properties and crops, a review. Agric. Water Manag. 2013, 119, 43–53. [Google Scholar] [CrossRef]
- Benaddi, R.; Osmane, A.; Zidan, K.; El Harfi, K.; Ouazzani, N. A review on Processes for olive mill waste water treatment. Ecol. Eng. Environ. Technol. 2023, 24, 196–207. [Google Scholar] [CrossRef]
- Al-Hmoud, L.; Al-Saida, B. Olive Mill Wastewater Treatment: A Recent Review. Jordanian J. Eng. Chem. Ind. 2020, 3, 91–106. [Google Scholar] [CrossRef]
- Kontos, S.S.; Koutsoukos, P.G.; Paraskeva, C.A. Application of combined physicochemical techniques for the efficient treatment of olive mill wastewaters. Desalination Water Treat. 2016, 57, 17051–17060. [Google Scholar] [CrossRef]
- Moglie, M.; Biancini, G.; Cioccolanti, L. Environmental and economic analysis of an olive mill wastewater treatment system integrated with microalgae production. Int. J. Life Cycle Assess. 2024, 29, 1000–1020. [Google Scholar] [CrossRef]
- Enaime, G.; Dababat, S.; Wichern, M.; Lübken, M. Olive mill wastes: From wastes to resources. Environ. Sci. Pollut. Res. 2024, 31, 20853–20880. [Google Scholar] [CrossRef]
- Vaz, T.; Quina, M.M.J.; Martins, R.C.; Gomes, J. Olive mill wastewater treatment strategies to obtain quality water for irrigation: A review. Sci. Total Environ. 2024, 931, 172676. [Google Scholar] [CrossRef]
- Hodaifa, G.; Malvis, A.; Maaitah, M.; Sánchez, S. Combination of physicochemical operations and algal culture as a new bioprocess for olive mill wastewater treatment. Biomass Bioenergy 2020, 138, 105603. [Google Scholar] [CrossRef]
- Hachicha, R.; Elleuch, J.; Dubessay, P.; Hachicha, R.; Abdelkafi, S.; Michaud, P.; Fendri, I. Integrated processes for olive mill wastewater treatment and its revalorization for microalgae culture. Int. Microbiol. 2025, 28, 1071–1085. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.; Iqbal, H.M.N.; Vishal, G.; Lee, H.S.; Nagra, S. Algal biorefinery: A sustainable approach to valorize algal-based biomass towards multiple product recovery. Bioresour. Technol. 2019, 278, 346–359. [Google Scholar] [CrossRef] [PubMed]
- Vuppaladadiyam, A.K.; Prinsen, P.; Raheem, A.; Luque, R.; Zhao, M. Microalgae cultivation and metabolites production: A comprehensive review. Biofuels Bioprod. Biorefining 2018, 12, 304–324. [Google Scholar] [CrossRef]
- Kaplan, D. Absorption and Adsorption of Heavy Metals by Microalgae. In Handbook of Microalgal Culture: Applied Phycology and Biotechnology, 2nd ed.; Wiley: Hoboken, NJ, USA, 2013; pp. 602–611. [Google Scholar] [CrossRef]
- Schmitt, D.; Müller, A.; Csögör, Z.; Frimmel, F.H.; Posten, C. The adsorption kinetics of metal ions onto different microalgae and siliceous earth. Water Res. 2001, 35, 779–785. [Google Scholar] [CrossRef]
- Genduso, M.G.; Guagliano, M.; Finocchio, E.; Cristiani, C.; Dotelli, G.; Santomauro, G. Adsorption of Heavy Metals from Low Concentration Solutions onto Dried Chlamydomonas reinhardtii. Appl. Sci. 2024, 14, 11057. [Google Scholar] [CrossRef]
- Aragaw, T.A.; Bogale, F.M. Biomass-Based Adsorbents for Removal of Dyes From Wastewater: A Review. Front. Environ. Sci. 2021, 9, 764958. [Google Scholar] [CrossRef]
- Hariskos, I.; Posten, C. Biorefinery of microalgae—Opportunities and constraints for different production scenarios. Biotechnol. J. 2014, 9, 739–752. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Goswami, S. Microalgae—A green multi-product biorefinery for future industrial prospects. Biocatal. Agric. Biotechnol. 2020, 25, 101580. [Google Scholar] [CrossRef]
- Lindner, A.V.; Pleissner, D. Utilization of phenolic compounds by microalgae. Algal Res. 2019, 42, 101602. [Google Scholar] [CrossRef]
- Ramesh, B.; Saravanan, A.; Kumar, P.S.; Yaashikaa, P.R.; Thamarai, P.; Shaji, A.; Rangasamy, G. A review on algae biosorption for the removal of hazardous pollutants from wastewater: Limiting factors, prospects and recommendations. Environ. Pollut. 2023, 327, 121572. [Google Scholar] [CrossRef]
- Johnson, B.B. Effect of pH, Temperature, and Concentration on the Adsorption of Cadmium on Goethite. Environ. Sci. Technol. 1990, 24, 112–118. [Google Scholar] [CrossRef]
- McGlamery, M.D.; Slife, F.W. The Adsorption and Desorption of Atrazine as Affected by pH, Temperature, and Concentration. Weeds 1966, 14, 237–239. [Google Scholar] [CrossRef]
- Mehta, S.K.; Tripathi, B.N.; Gaur, J.P. Influence of pH, temperature, culture age and cations on adsorption and uptake of Ni by Chlorella vulgaris. Eur. J. Protistol. 2000, 36, 443–450. [Google Scholar] [CrossRef]
- Tang, Y.Z.; Gin, K.Y.H.; Aziz, M.A. The relationship between pH and heavy metal ion sorption by algal biomass. Adsorpt. Sci. Technol. 2003, 21, 525–538. [Google Scholar] [CrossRef]
- Ouasfi, N.; Zbair, M.; Bouzikri, S.; Anfar, Z.; Bensitel, M.; Ahsaine, H.A.; Sabbar, E.; Khamliche, L. Selected pharmaceuticals removal using algae derived porous carbon: Experimental, modeling and DFT theoretical insights. RSC Adv. 2019, 9, 9792–9808. [Google Scholar] [CrossRef]
- Lin, Z.; Li, J.; Luan, Y.; Dai, W. Application of algae for heavy metal adsorption: A 20-year meta-analysis. Ecotoxicol. Environ. Saf. 2020, 190, 110089. [Google Scholar] [CrossRef] [PubMed]
- Aravindhan, R.; Rao, J.R.; Nair, B.U. Application of a chemically modified green macro alga as a biosorbent for phenol removal. J. Environ. Manag. 2009, 90, 1877–1883. [Google Scholar] [CrossRef] [PubMed]
- Navarro, A.E.; Lazo, J.C.; Cuizano, N.A.; Sun-Kou, M.R.; Llanos, B.P. Insights into Removal of Phenol from Aqueous Solutions by Low Cost Adsorbents: Clays Versus Algae. Sep. Sci. Technol. 2009, 44, 2491–2509. [Google Scholar] [CrossRef]
- Ali, M.E.M.; El-Aty, A.M.A.; Badawy, M.I.; Ali, R.K. Removal of pharmaceutical pollutants from synthetic wastewater using chemically modified biomass of green alga Scenedesmus obliquus. Ecotoxicol. Environ. Saf. 2018, 151, 144–152. [Google Scholar] [CrossRef]
- Xu, S.; Yang, J.; Marrakchi, F.; Wei, M.; Liu, Y.; Xiao, Y.; Li, C.; Wang, S. Macro- and micro-algae-based carbon composite for pharmaceutical wastewater treatment: Batch adsorption and mechanism study. Process Saf. Environ. Prot. 2023, 176, 641–652. [Google Scholar] [CrossRef]
- Feng, D.; Aldrich, C. Adsorption of heavy metals by biomaterials derived from the marine alga Ecklonia maxima. Hydrometallurgy 2004, 73, 1–10. [Google Scholar] [CrossRef]
- Popper, Z.A.; Michel, G.; Hervé, C.; Domozych, D.S.; Willats, W.G.T.; Tuohy, M.G.; Kloareg, B.; Stengel, D.B. Evolution and diversity of plant cell walls: From algae to flowering plants. Annu. Rev. Plant Biol. 2011, 62, 567–590. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Saxena, R.C. An Introduction to Microalga. In Handbook of Marine Microalgae; Elsevier: Amsterdam, The Netherlands, 2015; pp. 11–24. [Google Scholar] [CrossRef]
- Alhattab, M.; Kermanshahi-Pour, A.; Brooks, M.S.-L. Microalgae disruption techniques for product recovery: Influence of cell wall composition. J. Appl. Phycol. 2019, 31, 61–88. [Google Scholar] [CrossRef]
- de Almeida, C.; Figueredo, C.C. What we really know about the composition and function of microalgae cell coverings?—An overview. Acta Bot. Brasilica 2020, 34, 599–614. [Google Scholar] [CrossRef]
- Spain, O.; Funk, C. Detailed Characterization of the Cell Wall Structure and Composition of Nordic Green Microalgae. J. Agric. Food Chem. 2022, 70, 9711–9721. [Google Scholar] [CrossRef]
- De Tommasi, E.; Gielis, J.; Rogato, A. Diatom Frustule Morphogenesis and Function: A Multidisciplinary Survey. Mar. Genom. 2017, 35, 1–18. [Google Scholar] [CrossRef]
- Allaf, M.M.; Peerhossaini, H. Cyanobacteria: Model Microorganisms and Beyond. Microorganisms 2022, 10, 696. [Google Scholar] [CrossRef]
- Chen, H.; Jiang, J. Osmotic responses of Dunaliella to the changes of salinity. J. Cell. Physiol. 2009, 219, 251–258. [Google Scholar] [CrossRef]
- Zeraatkar, A.K.; Ahmadzadeh, H.; Talebi, A.F.; Moheimani, N.R.; McHenry, M.P. Potential use of algae for heavy metal bioremediation, a critical review. J. Environ. Manage. 2016, 181, 817–831. [Google Scholar] [CrossRef]
- Gu, S.; Lan, C.Q. Lipid-extraction algal biomass for biosorption of bivalent lead and cadmium ions: Kinetics and isotherm. Chem. Eng. Sci. 2023, 276, 118778. [Google Scholar] [CrossRef]
- Faruque, M.O.; Uddin, S.; Hossain, M.M.; Hossain, S.M.Z.; Shafiquzzaman, M.; Razzak, S.A. A comprehensive review on microalgae-driven heavy metals removal from industrial wastewater using living and non living microalgae. J. Hazard. Mater. Adv. 2024, 16, 100492. [Google Scholar] [CrossRef]
- Braud, L.; McDonnell, K.; Murphy, F. Environmental life cycle assessment of algae systems: Critical review of modelling approaches. Renew. Sustain. Energy Rev. 2023, 179, 113218. [Google Scholar] [CrossRef]
- Ubando, A.T.; Ng, E.A.S.; Chen, W.H.; Culaba, A.B.; Kwon, E.E. Life cycle assessment of microalgal biorefinery: A state-of-the-art review. Bioresour. Technol. 2022, 360, 127615. [Google Scholar] [CrossRef] [PubMed]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Pennesi, C.; Totti, C.; Beolchini, F. Removal of Vanadium(III) and Molybdenum(V) from Wastewater Using Posidonia oceanica (Tracheophyta) Biomass. PLoS ONE 2013, 8, e76870. [Google Scholar] [CrossRef] [PubMed]
- Zarrouk, C. Contribution à L’étude d’une Cyanophycée. Influence de Divers Facteurs Physiques et Chimiques sur la Croissance et la Photosynthèse de Spirulina maxima (Setch. et Gardner) Geitler; University of Paris: Paris, France, 1966. [Google Scholar]
- Mollo, L.; Drigo, F.; Moglie, M.; Norici, A. Screening for tolerance to natural phenols of different algal species: Toward the phycoremediation of olive mill wastewater. Algal Res. 2023, 75, 103256. [Google Scholar] [CrossRef]
- Singh, D.K.; Pathak, J.; Pandey, A.; Rajneesh; Singh, V.; Sinha, R.P. Purification, characterization and assessment of stability, reactive oxygen species scavenging and antioxidative potentials of mycosporine-like amino acids (MAAs) isolated from cyanobacteria. J. Appl. Phycol. 2022, 34, 3157–3175. [Google Scholar] [CrossRef]
- Singh, S.P.; Singh, P. Effect of temperature and light on the growth of algae species: A review. Renew. Sustain. Energy Rev. 2015, 50, 431–444. [Google Scholar] [CrossRef]
- Peterson, G.L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 1977, 83, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Deeb, A.A.; Fayyad, M.K.; Alawi, M.A. Separation of Polyphenols from Jordanian Olive Oil Mill Wastewater. Chromatogr. Res. Int. 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Chandra, S.; Khan, S.; Avula, B.; Lata, H.; Yang, M.H.; ElSohly, M.A.; Khan, I.A. Assessment of Total Phenolic and Flavonoid Content, Antioxidant Properties, and Yield of Aeroponically and Conventionally Grown Leafy Vegetables and Fruit Crops: A Comparative Study. Evid.-Based Complement. Altern. Med. 2014, 2014, 253875. [Google Scholar] [CrossRef] [PubMed]
- Lagergreen, S. Zur Theorie der sogenannten Adsorption gelöster Stoffe. Zeitschr Chem. Ind. Kolloide 1899, 24, 15. [Google Scholar] [CrossRef]
- Marquardt, D.W. An Algorithm for Least-Squares Estimation of Nonlinear Parameters on JSTOR. Soc. Ind. Appl. Math. 1963, 11, 431–441. Available online: https://www.jstor.org/stable/2098941 (accessed on 28 August 2025). [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über die Adsorption in Lösungen. Z. Für Phys. Chem. 1907, 57U, 385–470. [Google Scholar] [CrossRef]
- ISO 14040:2021; Gestione Ambientale—Valutazione del Ciclo di Vita—Principi e Quadro di Riferimento. Ente Italiano di Unificazione (UNI): Milano, Italy, 2021.
- ISO 14044:2021; Gestione Ambientale—Valutazione del Ciclo di Vita—Requisiti e Linee Guida. Ente Italiano di Unificazione (UNI): Milano, Italy, 2021.
- ISO 14067:2018; Greenhouse Gases—Carbon Footprint of Products—Requirements and Guidelines for Quantification. International Organization for Standardization: Geneva, Switzerland, 2018.
- Zampori, L.; Pant, R. Suggestions for Updating the Product Environmental Footprint (PEF) Method; Publications Office of the European Union: Luxembourg, 2019. [Google Scholar] [CrossRef]
- LArashiro, T.; Josa, I.; Ferrer, I.; Van Hulle, S.W.H.; Rousseau, D.P.L.; Garfí, M. Life cycle assessment of microalgae systems for wastewater treatment and bioproducts recovery: Natural pigments, biofertilizer and biogas. Sci. Total Environ. 2022, 847, 157615. [Google Scholar] [CrossRef]
- Park, K.H.; Balathanigaimani, M.S.; Shim, W.G.; Lee, J.W.; Moon, H. Adsorption characteristics of phenol on novel corn grain-based activated carbons. Microporous Mesoporous Mater. 2010, 127, 1–8. [Google Scholar] [CrossRef]
- Da, A.; Podkościelny, P.; Hubicki, Z.; Barczak, M. Adsorption of phenolic compounds by activated carbon—A critical review. Chemosphere 2005, 58, 1049–1070. [Google Scholar] [CrossRef] [PubMed]
- Chunzhuk, E.A.; Grigorenko, A.V.; Chernova, N.I.; Kiseleva, S.V.; Ryndin, K.G.; Popel, O.S.; Malaniy, S.Y.; Slavkina, O.V.; de Farias Neves, F.; Leng, L.; et al. Direct Study of CO2 Capture Efficiency during Microalgae Arthrospira platensis Cultivation at High CO2 Concentrations. Energies 2023, 16, 822. [Google Scholar] [CrossRef]
- Tzachor, A.; Smidt-Jensen, A.; Ramel, A.; Geirsdóttir, M. Environmental Impacts of Large-Scale Spirulina (Arthrospira platensis) Production in Hellisheidi Geothermal Park Iceland: Life Cycle Assessment. Mar. Biotechnol. 2022, 24, 991–1001. [Google Scholar] [CrossRef]
- Kishi, M.; Yamada, Y.; Katayama, T.; Matsuyama, T.; Toda, T. Carbon Mass Balance in Arthrospira platensis Culture with Medium Recycle and High CO2 Supply. Appl. Sci. 2019, 10, 228. [Google Scholar] [CrossRef]
- Razzaq, R. Phytoremediation: An Environmental Friendly Technique—A Review. J. Environ. Anal. Chem. 2017, 4, 2380–2391. [Google Scholar] [CrossRef]
- Chugh, M.; Kumar, L.; Shah, M.P.; Bharadvaja, N. Algal Bioremediation of heavy metals: An insight into removal mechanisms, recovery of by-products, challenges, and future opportunities. Energy Nexus 2022, 7, 100129. [Google Scholar] [CrossRef]
- Mustafa, S.; Bhatti, H.N.; Maqbool, M.; Iqbal, M. Microalgae biosorption, bioaccumulation and biodegradation efficiency for the remediation of wastewater and carbon dioxide mitigation: Prospects, challenges and opportunities. J. Water Process Eng. 2021, 41, 102009. [Google Scholar] [CrossRef]
- Long, S.; Hamilton, P.B.; Wang, C.; Li, C.; Xue, X.; Zhao, Z.; Wu, P.; Gu, E.; Uddin, M.M.; Li, B.; et al. Bioadsorption, bioaccumulation and biodegradation of antibiotics by algae and their association with algal physiological state and antibiotic physicochemical properties. J. Hazard. Mater. 2024, 468, 133787. [Google Scholar] [CrossRef] [PubMed]
- González, F.; Romera, E.; Ballester, A.; Blázquez, M.L.; Muñoz, J.Á.; García-Balboa, C. Algal Biosorption and Biosorbents. In Microbial Biosorption of Metals; Springer: Dordrecht, The Netherlands, 2011; pp. 159–178. [Google Scholar] [CrossRef]
- Romera, E.; González, F.; Ballester, A.; Blázquez, M.L.; Muñoz, J.A. Biosorption with Algae: A Statistical Review. Crit. Rev. Biotechnol. 2006, 26, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Du, Y.; Zheng, F.; Wang, J.; Wang, Z.; Ji, R.; Chen, J. Bioaccumulation and elimination of bisphenol a (BPA) in the alga Chlorella pyrenoidosa and the potential for trophic transfer to the rotifer Brachionus calyciflorus. Environ. Pollut. 2017, 227, 460–467. [Google Scholar] [CrossRef] [PubMed]
- Doshi, H.; Seth, C.; Ray, A.; Kothari, I.L. Bioaccumulation of heavy metals by green algae. Curr. Microbiol. 2008, 56, 246–255. [Google Scholar] [CrossRef]
- Radziff, S.B.M.; Ahmad, S.A.; Shaharuddin, N.A.; Merican, F.; Kok, Y.Y.; Zulkharnain, A.; Gomez-Fuentes, C.; Wong, C.Y. Potential Application of Algae in Biodegradation of Phenol: A Review and Bibliometric Study. Plants 2021, 10, 2677. [Google Scholar] [CrossRef]
- Touliabah, H.E.S.; El-Sheekh, M.M.; Ismail, M.M.; El-Kassas, H. A Review of Microalgae- and Cyanobacteria-Based Biodegradation of Organic Pollutants. Molecules 2022, 27, 1141. [Google Scholar] [CrossRef]
- Lindner, A.V.; Pleissner, D. Removal of Phenolic Compounds from Olive Mill Wastewater by Microalgae Grown Under Dark and Light Conditions. Waste Biomass Valorization 2022, 13, 525–534. [Google Scholar] [CrossRef]
- Singh, S. Biosorption of Heavy Metals by Cyanobacteria: Potential of Live and Dead Cells in Bioremediation. In Microbial Bioremediation & Biodegradation; Springer: Singapore, 2020; pp. 409–423. [Google Scholar] [CrossRef]
- Doshi, H.; Ray, A.; Kothari, I.L. Biosorption of Cadmium by Live and Dead Spirulina: IR Spectroscopic, Kinetics, and SEM Studies. Curr. Microbiol. 2007, 54, 213–218. [Google Scholar] [CrossRef]
- Suresh, R.; Rajendran, S.; Chen, W.H.; Soto-Moscoso, M.; Sundaram, T.; Jalil, A.A.; Sekar, S.K.R. A review on algae-mediated adsorption and catalytic processes for organic water pollution remediation. Front. Mater. 2024, 11, 1432704. [Google Scholar] [CrossRef]
- Spain, O.; Plöhn, M.; Funk, C. The cell wall of green microalgae and its role in heavy metal removal. Physiol. Plant. 2021, 173, 526–535. [Google Scholar] [CrossRef]
- Samuel, H.S.; Okino, I.A.; Okibe, G.; Etim, E.E. Sustainable Wastewater Treatment: Recent Progress in the use of Bio-Waste-Derived Adsorbents for Organic Dye Removal. Asian J. Environ. Res. 2024, 1, 137–151. [Google Scholar] [CrossRef]
- Alsharif, M.A. Understanding Adsorption: Theories, Techniques, and Applications. In Adsorption—Fundamental Mechanisms and Applications; IntechOpen: Rijeka, Croatia, 2025. [Google Scholar] [CrossRef]
- Rajasekar, P.; Palanisamy, S.; Anjali, R.; Vinosha, M.; Elakkiya, M.; Marudhupandi, T.; Tabarsa, M.; You, S.G.; Prabhu, N.M. Isolation and structural characterization of sulfated polysaccharide from Spirulina platensis and its bioactive potential: In vitro antioxidant, antibacterial activity and Zebrafish growth and reproductive performance. Int. J. Biol. Macromol. 2019, 141, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Mouhim, R.F.; Cornet, J.-F.; Fontane, T.; Fournet, B.; Dubertret, G. Production, isolation and preliminary characterization of the exopolysaccharide of the cyanobacterium Spirulina platensis. Biotechnol. Lett. 1993, 15, 567–572. [Google Scholar] [CrossRef]
- Abo-Shady, A.M.; Mohamed, Y.A.; Lasheen, T. Chemical composition of the cell wall in some green algae species. Biol. Plant. 1993, 35, 629–632. [Google Scholar] [CrossRef]
- Domozych, D.S.; Ciancia, M.; Fangel, J.U.; Mikkelsen, M.D.; Ulvskov, P.; Willats, W.G.T. The cell walls of green algae: A journey through evolution and diversity. Front. Plant Sci. 2012, 3, 25249. [Google Scholar] [CrossRef]
- Allard, B.; Templier, J. Comparison of neutral lipid profile of various trilaminar outer cell wall (TLS)-containing microalgae with emphasis on algaenan occurrence. Phytochemistry 2000, 54, 369–380. [Google Scholar] [CrossRef]
- Kodner, R.B.; Summons, R.E.; Knoll, A.H. Phylogenetic investigation of the aliphatic, non-hydrolyzable biopolymer algaenan, with a focus on green algae. Org. Geochem. 2009, 40, 854–862. [Google Scholar] [CrossRef]
- Saoud, H.A.A.L.; Sprynskyy, M.; Pashaei, R.; Kawalec, M.; Pomastowski, P.; Buszewski, B. Diatom biosilica: Source, physical-chemical characterization, modification, and application. J. Sep. Sci. 2022, 45, 3362–3376. [Google Scholar] [CrossRef]
- Annenkov, V.V.; Danilovtseva, E.N.; Gordon, R. Steps of Silicic Acid Transformation to Siliceous Frustules: Main Hypotheses and Discoveries. In Diatom Morphogenesis; Scrivener Publishing LLC : Beverly, MA, USA, 2021; pp. 301–347. [Google Scholar] [CrossRef]
- Lim, G.W.; Lim, J.K.; Ahmad, A.L.; Chan, D.J.C. Influences of diatom frustule morphologies on protein adsorption behavior. J. Appl. Phycol. 2015, 27, 763–775. [Google Scholar] [CrossRef]
- Savio, S.; Farrotti, S.; Di Giulio, A.; De Santis, S.; Ellwood, N.T.W.; Ceschin, S.; Congestri, R. Functionalization of Frustules of the Diatom Staurosirella pinnata for Nickel (Ni) Adsorption from Contaminated Aqueous Solutions. Front. Mar. Sci. 2022, 9, 889832. [Google Scholar] [CrossRef]
- Kalita, N.; Baruah, P.P. Cyanobacteria as a potent platform for heavy metals biosorption: Uptake, responses and removal mechanisms. J. Hazard. Mater. Adv. 2023, 11, 100349. [Google Scholar] [CrossRef]
- Dotto, G.L.; Gonçalves, J.O.; Cadaval, T.R.S.; Pinto, L.A.A. Biosorption of phenol onto bionanoparticles from Spirulina sp. LEB 18. J. Colloid Interface Sci. 2013, 407, 450–456. [Google Scholar] [CrossRef]
- Afsharnia, M.; Saeidi, M.; Zarei, A.; Narooie, M.R.; Biglari, H. Phenol Removal from Aqueous Environment by Adsorption onto Pomegranate Peel Carbon. Electron. Physician 2016, 8, 3248. [Google Scholar] [CrossRef]
- Chen, W.; Xu, J.; Yu, Q.; Yuan, Z.; Kong, X.; Sun, Y.; Wang, Z.; Zhuang, X.; Zhang, Y.; Guo, Y. Structural insights reveal the effective Spirulina platensis cell wall dissociation methods for multi-output recovery. Bioresour. Technol. 2020, 300, 122628. [Google Scholar] [CrossRef] [PubMed]
- Van Eykelenburg, C. On the morphology and ultrastructure of the cell wall of Spirulina platensis. Antonie Van. Leeuwenhoek 1977, 43, 89–99. [Google Scholar] [CrossRef]
- Priatni, S.; Ratnaningrum, D.; Warya, S.; Audina, E. Phycobiliproteins production and heavy metals reduction ability of Porphyridium sp. IOP Conf. Ser. Earth Environ. Sci. 2018, 160, 012006. [Google Scholar] [CrossRef]
- Bellamy-Carter, J.; Sound, J.K.; Leney, A.C. Probing heavy metal binding to phycobiliproteins. FEBS J. 2022, 289, 4646–4656. [Google Scholar] [CrossRef] [PubMed]
- Kola, H.; Laglera, L.M.; Parthasarathy, N.; Wilkinson, K.J. Cadmium Adsorption by Chlamydomonas reinhardtii and its Interaction with the Cell Wall Proteins. Environ. Chem. 2004, 1, 172–179. [Google Scholar] [CrossRef]
- Quignard, F.; Di Renzo, F.; Guibal, E. From Natural Polysaccharides to Materials for Catalysis, Adsorption, and Remediation. Top. Curr. Chem. 2010, 294, 165–197. [Google Scholar] [CrossRef]
- Siemińska-Kuczer, A.; Szymańska-Chargot, M.; Zdunek, A. Recent advances in interactions between polyphenols and plant cell wall polysaccharides as studied using an adsorption technique. Food Chem. 2022, 373, 131487. [Google Scholar] [CrossRef]
- Michalak, I.; Mironiuk, M.; Godlewska, K.; Trynda, J.; Marycz, K. Arthrospira (Spirulina) platensis: An effective biosorbent for nutrients. Process Biochem. 2020, 88, 129–137. [Google Scholar] [CrossRef]
- Deniz, F.; Kepekci, R.A. Equilibrium and kinetic studies of azo dye molecules biosorption on phycocyanin-extracted residual biomass of microalga Spirulina platensis. Desalination Water Treat. 2016, 57, 12257–12263. [Google Scholar] [CrossRef]
- Rose, R.K.; Shellis, R.R.; Lee, A.R. The Role of Cation Bridging in Microbial Fluoride Binding. Caries Res. 1996, 30, 458–464. [Google Scholar] [CrossRef]
- Wang, X.; Lee, S.Y.; Miller, K.; Welbourn, R.; Stocker, I.; Clarke, S.; Casford, M.; Gutfreund, P.; Skoda, M.W.A. Cation Bridging Studied by Specular Neutron Reflection. Langmuir 2013, 29, 5520–5527. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Gupta, V.K.; Rastogi, A. Biosorption of lead(II) from aqueous solutions by non-living algal biomass Oedogonium sp. and Nostoc sp.—A comparative study. Colloids Surf. B Biointerfaces 2008, 64, 170–178. [Google Scholar] [CrossRef]
- Yahya, M.A.; Al-Qodah, Z.; Ngah, C.W.Z. Agricultural bio-waste materials as potential sustainable precursors used for activated carbon production: A review. Renew. Sustain. Energy Rev. 2015, 46, 218–235. [Google Scholar] [CrossRef]
- Mahfuz, S.; Shang, Q.; Piao, X. Phenolic compounds as natural feed additives in poultry and swine diets: A review. J. Anim. Sci. Biotechnol. 2021, 12, 48. [Google Scholar] [CrossRef]
- Panzella, L.; Napolitano, A. Natural Phenol Polymers: Recent Advances in Food and Health Applications. Antioxidants 2017, 6, 30. [Google Scholar] [CrossRef]
- Branciari, R.; Galarini, R.; Miraglia, D.; Ranucci, D.; Valiani, A.; Giusepponi, D.; Servili, M.; Acuti, G.; Pauselli, M.; Trabalza-Marinucci, M. Dietary Supplementation with Olive Mill Wastewater in Dairy Sheep: Evaluation of Cheese Characteristics and Presence of Bioactive Molecules. Animals 2020, 10, 1941. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Xu, B.L.; Dong, C.M.; Sun, Y.Y. The nutritional value of Spirulina and Utilization Research. Life Res. 2023, 6, 15. [Google Scholar] [CrossRef]
- Jelínek, L.; Procházková, G.; Quintelas, C.; Beldíková, E.; Brányik, T. Chlorella vulgaris biomass enriched by biosorption of polyphenols. Algal Res. 2015, 10, 1–7. [Google Scholar] [CrossRef]
- Okeke, E.S.; Ejeromedoghene, O.; Okoye, C.O.; Ezeorba, T.P.C.; Nyaruaba, R.; Ikechukwu, C.K.; Oladipo, A.; Orege, J.I. Microalgae biorefinery: An integrated route for the sustainable production of high-value-added products. Energy Convers. Manag. X 2022, 16, 100323. [Google Scholar] [CrossRef]
- Lee, H.-C.; Lee, M.; Den, W. Spirulina maxima for Phenol Removal: Study on its Tolerance, Biodegradability and Phenol-Carbon Assimilability. Water Air Soil Pollut. 2015, 226, 395. [Google Scholar] [CrossRef]
- Zeymer, M.; Meisel, K.; Clemens, A.; Klemm, M. Technical, Economic, and Environmental Assessment of the Hydrothermal Carbonization of Green Waste. Chem. Eng. Technol. 2017, 40, 260–269. [Google Scholar] [CrossRef]
- Aruna; Bagotia, N.; Sharma, A.K.; Kumar, S. A review on modified sugarcane bagasse biosorbent for removal of dyes. Chemosphere 2021, 268, 129309. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Chitosan-based biosorbents: Modification and application for biosorption of heavy metals and radionuclides. Bioresour. Technol. 2014, 160, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Laso, J.; Espada, J.J.; Rodríguez, R.; Vicente, G.; Bautista, L.F. Novel Biorefinery Approach for Phycocyanin Extraction and Purification and Biocrude Production from Arthrospira platensis. Ind. Eng. Chem. Res. 2023, 62, 5190–5198. [Google Scholar] [CrossRef]
- Moglie, M.; Biancini, G.; Norici, A.; Mugnini, A.; Cioccolanti, L. Valorization of olive mill wastewater for Arthrospira platensis production. Cell Rep. Sustain. 2024, 1, 100017. [Google Scholar] [CrossRef]
- Ranjbar, S.; Malcata, F.X. Is Genetic Engineering a Route to Enhance Microalgae-Mediated Bioremediation of Heavy Metal-Containing Effluents? Molecules 2022, 27, 1473. [Google Scholar] [CrossRef] [PubMed]
- Chiaiese, P.; Palomba, F.; Tatino, F.; Lanzillo, C.; Pinto, G.; Pollio, A.; Filippone, E. Engineered tobacco and microalgae secreting the fungal laccase POXA1b reduce phenol content in olive oil mill wastewater. Enzym. Microb. Technol. 2011, 49, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Geissdoerfer, M.; Savaget, P.; Bocken, N.M.P.; Hultink, E.J. The Circular Economy—A new sustainability paradigm? J. Clean. Prod. 2017, 143, 757–768. [Google Scholar] [CrossRef]
| Phylum | Habitat | Algal Species | ID Code | Type of Cell Wall |
|---|---|---|---|---|
| Chlorophyta | Freshwater | Tetradesmus obliquus | CCAP 276/3A | Cellulose and algaenan |
| Freshwater | Chlorella vulgaris | CCAP 211/11b | Cellulose and algaenan | |
| Marine | Dunaliella salina | Isolated from Margherita di Savoia saltpans (Italy) | No cell wall | |
| Rhodophyta | Marine | Porphyridium purpureum | CCAP 1380/3 | No rigid cell wall, sulphated polysaccharides |
| Haptophyte | Marine | Isochrysis galbana | Roscoff RCC 1353 | No rigid cell wall, organic scales |
| Bacillariophyte | Marine | Conticribra weissflogii | DCG 0320 | Siliceous frustule |
| Marine | Phaeodactylum tricornutum | UTEX 646 | Siliceous frustule | |
| Ochrophyte | Marine | Nannochloropsis salina | CCAP 849/3 | Polysaccharides and algaenan |
| Cyanobacteria | Marine | Synechococcus sp. | UTEX LB 2380 | Peptidoglycan |
| Freshwater | Anabaena sp. | CCAP 1403/4a | Peptidoglycan | |
| Freshwater | Limnospira platensis | SAG 85.79 | Peptidoglycan |
| Species | Phylum/Type | qe (mg·g−1) | k (ms−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Tetradesmus obliquus | Chlorophyta | 67.7 | ± | 8.1 | ac | 6.0 | ± | 2.0 | a |
| Chlorella vulgaris | Chlorophyta | 12.3 | ± | 1.4 | d | 2.7 | 1.5 | a | |
| Dunaliella salina | Chlorophyta | <LOD | n.a | ||||||
| Porphyridium purpureum | Rodophyta | 42.3 | ± | 26.4 | acd | 18.3 | ± | 2.9 | a |
| Isochrysis galbana | Haptophyta | <LOD | n.a | ||||||
| Conticribra weissflogii | Bacillariophyta | 56.5 | ± | 13.6 | ac | 16.0 | ± | 7.8 | a |
| Phaeodactylum tricornutum | Bacillariophyta | 39.6 | ± | 12.3 | bcd | 13.7 | ± | 11.7 | a |
| Nannochloropsis salina | Ochrophyte | 30.3 | ± | 8.1 | cd | 2.7 | ± | 0.6 | a |
| Synechococcus sp. | Cyanobacteria | 74.2 | ± | 25.8 | ba | 2.3 | ± | 0.6 | a |
| Anabaena sp. | Cyanobacteria | 81.5 | ± | 7.8 | a | 12.7 | ± | 4.9 | a |
| Limnospira platensis | Cyanobacteria | 78.7 | ± | 4.2 | a | 11.3 | ± | 7.6 | a |
| Algal Dose (g L−1) | Ce (mg L−1) | qe (mg·g−1) | Removal (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.10 | 22.3 | ± | 0.6 | 76.9 | ± | 6.0 | 25.7 | ± | 2.0 |
| 0.20 | 19.3 | ± | 0.7 | 53.3 | ± | 3.6 | 35.5 | ± | 2.4 |
| 0.33 | 13.8 | ± | 0.6 | 48.7 | ± | 1.8 | 54.1 | ± | 2.0 |
| 1.00 | 11.8 | ± | 0.3 | 18.3 | ± | 0.3 | 60.8 | ± | 1.0 |
| SCENARIO 1 | |||||
|---|---|---|---|---|---|
| INPUT | OUTPUT | ||||
| Algal Growth | |||||
| NaHCO3 | 15.1 | g | Spent growth medium with A. platensis biomass | 220 | g |
| K2HPO4 | 0.4 | g | |||
| NaNO3 | 2.25 | g | |||
| K2SO4 | 0.91 | g | |||
| NaCl | 0.91 | g | |||
| MgSO4 × 7H2O | 0.18 | g | |||
| CaCl2 | 0.04 | g | |||
| FeSO4 × 7H2O | 0.01 | g | |||
| EDTA | 0.08 | g | |||
| A5 solution | 0.01 | g | |||
| Led light | 5.04 | kWh | |||
| Distilled H2O | 220 | g | |||
| Autoclave | 0.094 | KWh | |||
| Centrifugation | |||||
| Energy | 0.14 | KWh | A. platensis biomass | 1 | g |
| H2O for balancing | 220 | g | Spent growth medium | 220 | g |
| Spent growth medium with A. platensis biomass | 220 | g | H2O | 220 | g |
| Washing and centrifugation | |||||
| A. platensis biomass | 1 | g | Washed algal biomass | 1 | g |
| H2O for biomass washing | 440 | g | H2O | 660 | g |
| H2O for balancing | 220 | g | |||
| Energy | 0.14 | kWh | |||
| Functionalisation | |||||
| Washed algal biomass | 1 | g | Functionalised algal biomass | 1 | g |
| HCI 35% | 0.09 | g | Wastewater rich in HCI | 10 | g |
| H2O for dilution | 9.91 | g | H2O | 10 | g |
| Energy | 0.185 | kWh | |||
| H2O for balancing | 10 | g | |||
| Dehydration | |||||
| Functionalised algal biomass | 1 | g | Dry algal biomass | 1 | g |
| Energy | 0.072 | kWh | |||
| Adsorption and mixing | |||||
| Energy | 0.003 | kWh | Remediated wastewater | 1 | L |
| Phenolic solution | 1 | L | Biomass rich in phenols | 1 | g |
| Dry algal biomass | 1 | g | |||
| Wastewater treatment | |||||
| Spent growth medium | 220 | g | Discharged H2O | 1.14 | kg |
| H2O | 918 | g | |||
| Wastewater rich in HCI | 0.09 | g | |||
| Waste treatment | |||||
| Biomass rich in phenols | 1 | g | Treated biomass | 1 | g |
| SCENARIO 2 | |||||
| INPUT | OUTPUT | ||||
| Grinding | |||||
| Energy | 0.011 | kWh | Maize powder | 0.25 | g |
| Maize | 0.25 | g | |||
| Dehydration | |||||
| Maize powder | 0.25 | g | Dehydrated powder | 0.25 | g |
| Energy | 5.88 | kWh | |||
| Carbonisation | |||||
| Energy | 0.22 | kWh | Carbonized powder | 0.25 | g |
| Nitrogen | 37.5 | g | |||
| Dehydrated powder | 0.25 | g | |||
| Mixing | |||||
| Biomass | 0.25 | g | Mixture | 1.25 | kg |
| Potassium hydroxide | 1 | kg | |||
| Activation | |||||
| Mixture | 1.25 | kg | Activated carbon | 1.25 | kg |
| Nitrogen | 37.5 | g | |||
| Energy | 13 | kWh | |||
| Washing | |||||
| H2O | 1 | L | Wet carbon | ||
| Activated carbon | 1.25 | kg | |||
| Vacuum drying | |||||
| Energy | 10.8 | kWh | H2O | 2 | L |
| Wet Carbon | 2.25 | kg | Dry activated carbon | 0.25 | g |
| Adsorption and mixing | |||||
| Phenolic solution | 1 | L | Remediated wastewater | 1 | L |
| Dry activated carbon | 0.25 | g | Activated carbon rich in phenols | 0.25 | g |
| Energy | 0.03 | kWh | |||
| Wastewater treatment | |||||
| H2O | 2 | L | Discharged H2O | 2 | L |
| Waste treatment | |||||
| Activated carbon rich in phenols | 0.25 | g | Treated carbon | 0.25 | g |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mollo, L.; Norici, A.; Raffaelli, L.; Amato, A. Algal and Cyanobacteria Cell Walls as Biosorbents for Phenolic Compounds: Comparative Performance and Sustainability Assessment of Limnospira platensis . Bioengineering 2026, 13, 373. https://doi.org/10.3390/bioengineering13040373
Mollo L, Norici A, Raffaelli L, Amato A. Algal and Cyanobacteria Cell Walls as Biosorbents for Phenolic Compounds: Comparative Performance and Sustainability Assessment of Limnospira platensis . Bioengineering. 2026; 13(4):373. https://doi.org/10.3390/bioengineering13040373
Chicago/Turabian StyleMollo, Lorenzo, Alessandra Norici, Linda Raffaelli, and Alessia Amato. 2026. "Algal and Cyanobacteria Cell Walls as Biosorbents for Phenolic Compounds: Comparative Performance and Sustainability Assessment of Limnospira platensis " Bioengineering 13, no. 4: 373. https://doi.org/10.3390/bioengineering13040373
APA StyleMollo, L., Norici, A., Raffaelli, L., & Amato, A. (2026). Algal and Cyanobacteria Cell Walls as Biosorbents for Phenolic Compounds: Comparative Performance and Sustainability Assessment of Limnospira platensis . Bioengineering, 13(4), 373. https://doi.org/10.3390/bioengineering13040373









