Ginsenoside Rg3: Potential Molecular Targets and Therapeutic Indication in Metastatic Breast Cancer
Abstract
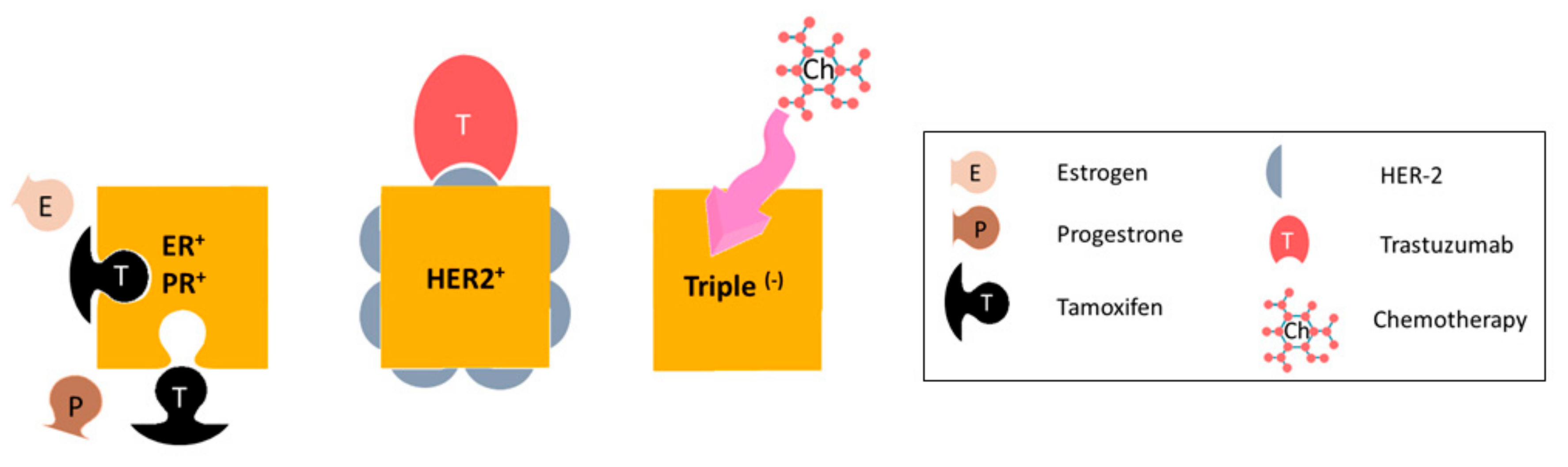
:1. Metastatic Breast Cancer
2. Ginseng—History and Medicinal Use
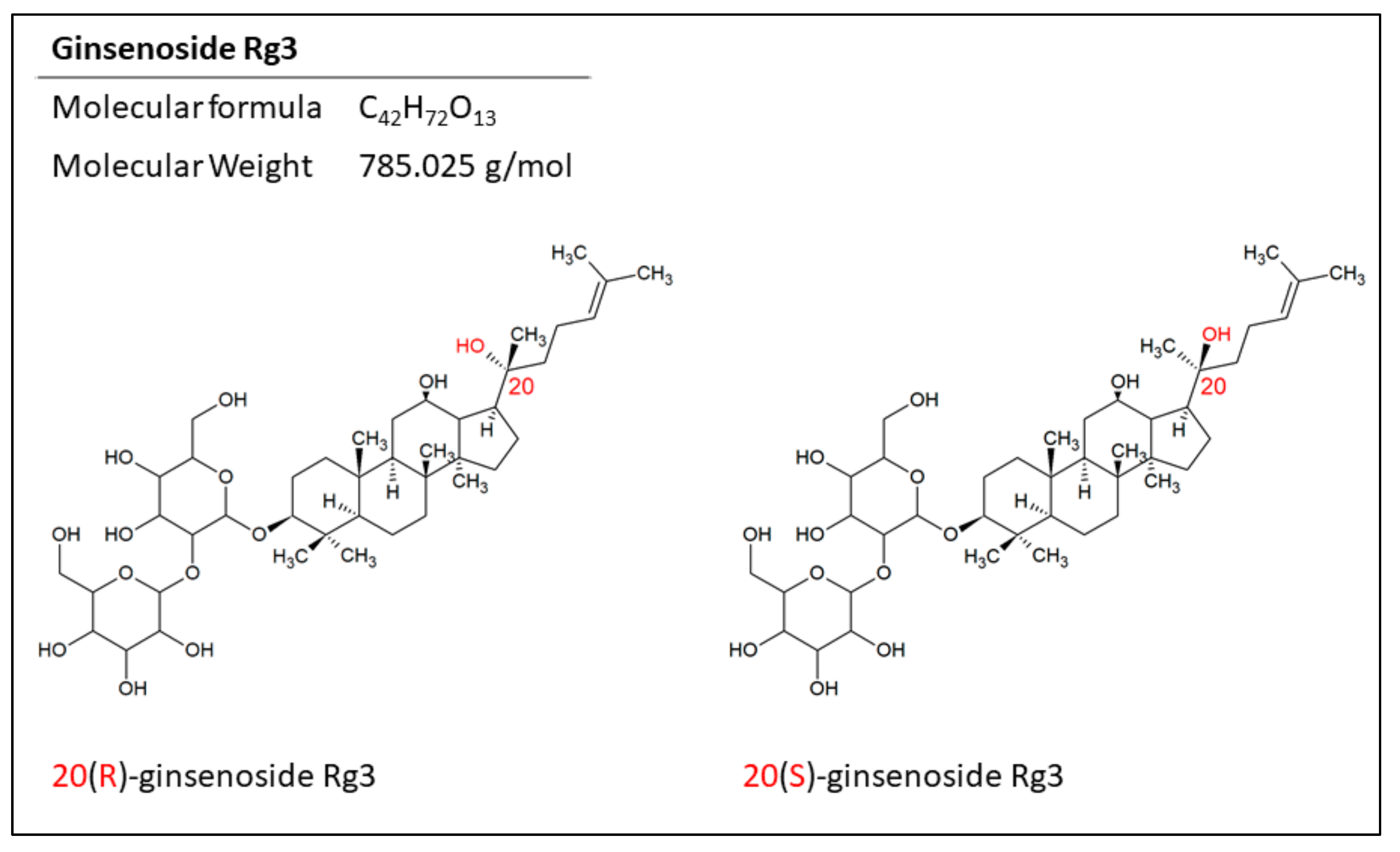
3. Epimers of Ginsenoside Rg3 in the Treatment of Cancer
4. Mechanisms of Action of Ginsenoside Rg3 in Breast Cancer
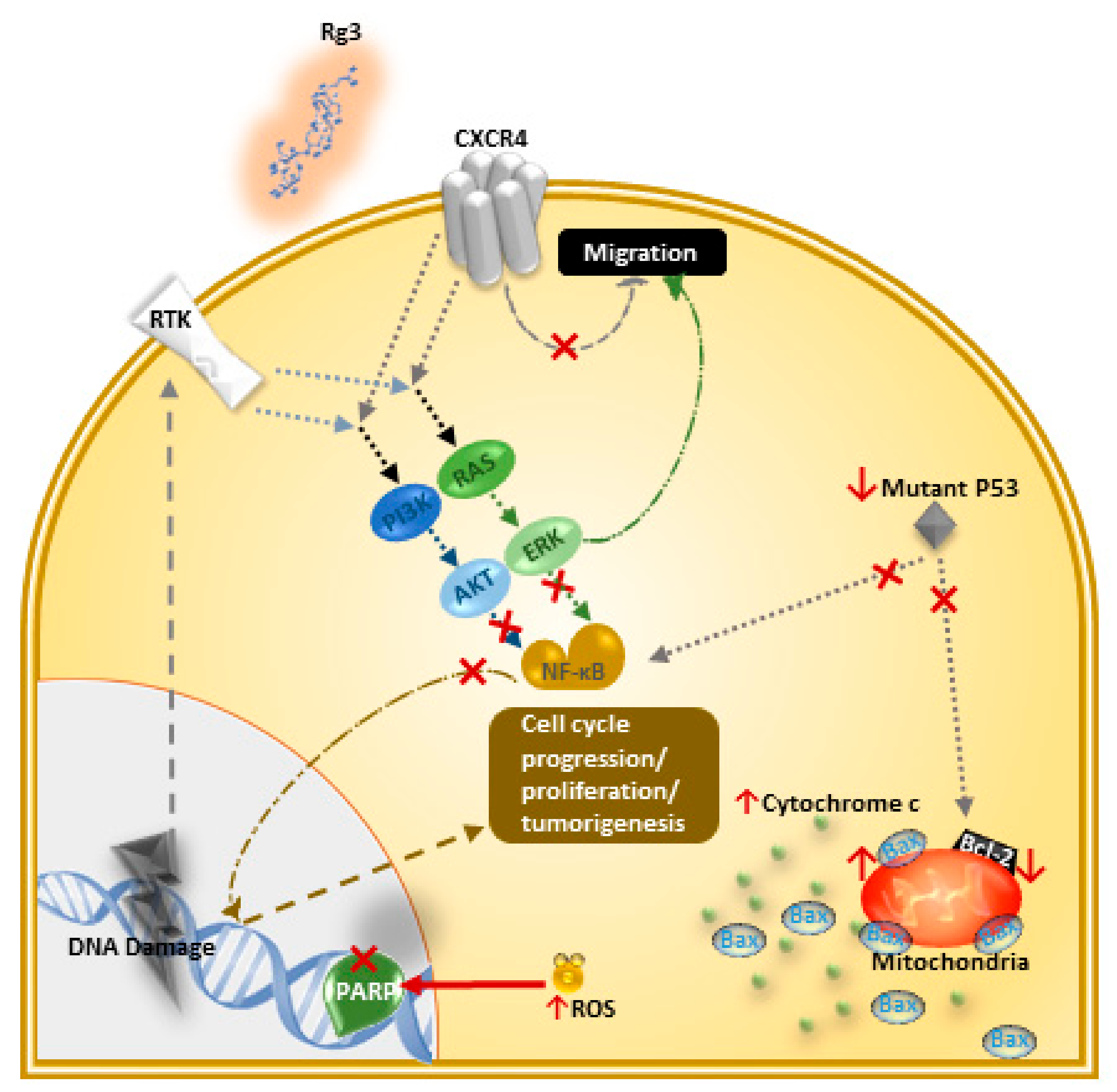
4.1. Induction of Apoptosis and Inhibition of Proliferation
4.2. Inhibition of Migration, Invasion, Angiogenesis, and Metastasis
4.3. Multidrug Resistance (MDR) and Combination Therapy
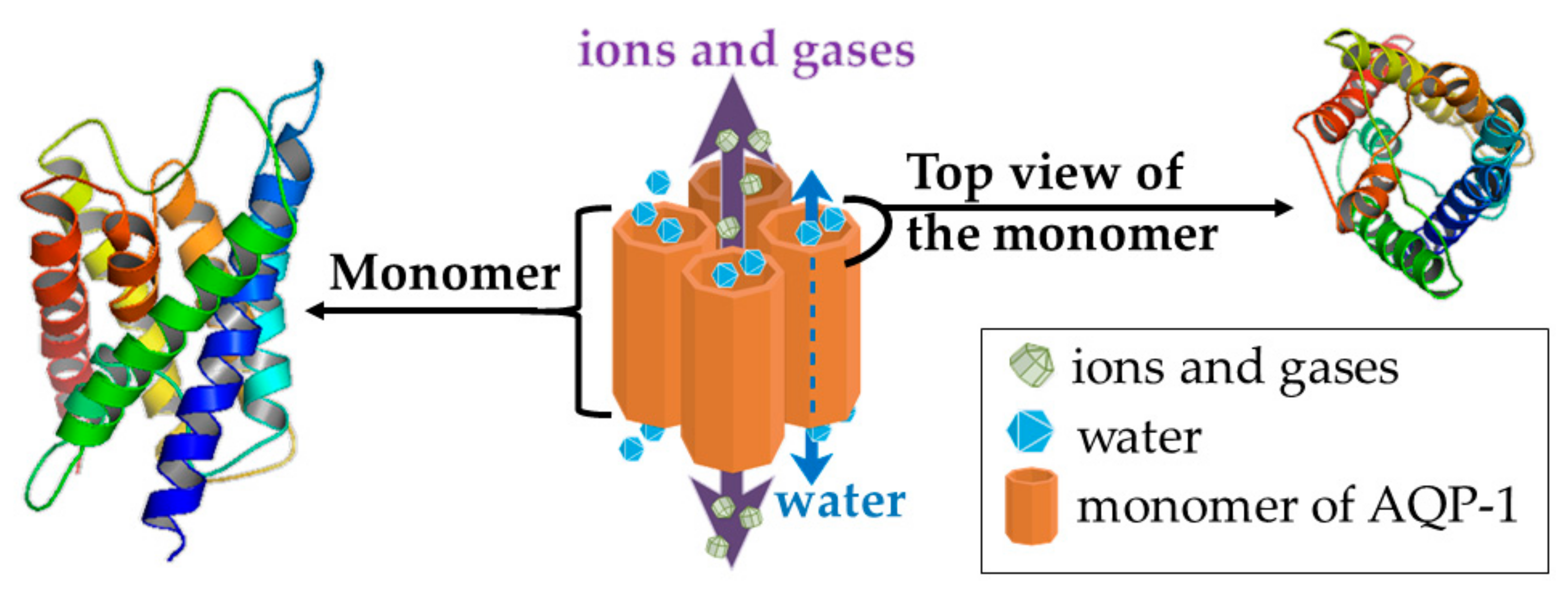
4.4. Aquaporin (AQP) 1—a Putative Target of Rg3
AQP1 and Breast Cancer
4.5. Other Suggested Mechanisms of Action
5. Metabolism and Pharmacokinetics of Rg3
6. Clinical Trials
6.1. Application and Safety of Ginseng Extract on Healthy Human Volunteers
6.2. Clinial Trials and Application of Rg3 in Cancer Patients
7. Conclusions
Funding
Conflicts of Interest
References
- Giuliano, A.E.; Connolly, J.L.; Edge, S.B.; Mittendorf, E.A.; Rugo, H.S.; Solin, L.J.; Weaver, D.L.; Winchester, D.J.; Hortobagyi, G.N. Breast cancer—Major changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J. Clin. 2017, 67, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, N. Platinum resistance in breast and ovarian cancer cell lines. J. Exp. Clin. Cancer Res. 2011, 30, 1. [Google Scholar] [CrossRef] [PubMed]
- Cronin, K.A.; Harlan, L.C.; Dodd, K.W.; Abrams, J.S.; Ballard-Barbash, R. Population-based estimate of the prevalence of HER-2 positive breast cancer tumors for early stage patients in the US. Cancer Investig. 2010, 28, 963–968. [Google Scholar] [CrossRef] [PubMed]
- Fischer, O.M.; Streit, S.; Hart, S.; Ullrich, A. Beyond herceptin and gleevec. Curr. Opin. Chem. Biol. 2003, 7, 490–495. [Google Scholar] [CrossRef]
- Elias, A.D. Triple-negative breast cancer: A short review. Am. J. Clin. Oncol. 2010, 33, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, S. Systemic Therapy. In Breast Cancer; Mahon, S.M., Ed.; Oncology Nursing Society: Pittsburgh, PA, USA, 2011. [Google Scholar]
- Anders, C.K.; Carey, L.A. Biology, metastatic patterns, and treatment of patients with triple-negative breast cancer. Clin. Breast Cancer 2009, 9, S73–S81. [Google Scholar] [CrossRef] [PubMed]
- Robert, N.; Leyland-Jones, B.; Asmar, L.; Belt, R.; Ilegbodu, D.; Loesch, D.; Raju, R.; Valentine, E.; Sayre, R.; Cobleigh, M. Randomized phase III study of trastuzumab, paclitaxel, and carboplatin compared with trastuzumab and paclitaxel in women with HER-2–overexpressing metastatic breast cancer. J. Clin. Oncol. 2006, 24, 2786–2792. [Google Scholar] [CrossRef] [PubMed]
- Attele, A.S.; Wu, J.A.; Yuan, C.-S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Park, J.D.; Rhee, D.K.; Lee, Y.H. Biological activities and chemistry of saponins from Panax ginseng CA Meyer. Phytochem. Rev. 2005, 4, 159–175. [Google Scholar] [CrossRef]
- Park, H.J.; Kim, D.H.; Park, S.J.; Kim, J.M.; Ryu, J.H. Ginseng in traditional herbal prescriptions. J. Ginseng Res. 2012, 36, 225. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.S.; Wu, M.Y. Chinese ginseng. In Nutraceuticals; Elsevier: New York City, NY, USA, 2016; pp. 693–705. [Google Scholar]
- Chang-Xiao, L.; Pei-Gen, X. Recent advances on ginseng research in China. J. Ethnopharmacol. 1992, 36, 27–38. [Google Scholar] [CrossRef]
- Choi, K.-T. Botanical characteristics, pharmacological effects and medicinal components of Korean Panax ginseng CA Meyer. Acta Pharmacol. Sin. 2008, 29, 1109. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Z.; Aung, H.H.; Ni, M.; Wu, J.-A.; Tong, R.; Wicks, S.; He, T.-C.; Yuan, C.-S. Red American ginseng: Ginsenoside constituents and antiproliferative activities of heat-processed Panax quinquefolius roots. Planta Med. 2007, 73, 669. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Wang, C.-Z.; Tong, R.; Li, X.-L.; Fishbein, A.; Wang, Q.; He, T.-C.; Du, W.; Yuan, C.-S. Effects of steaming the root of Panax notoginseng on chemical composition and anticancer activities. Food Chem. 2010, 118, 307–314. [Google Scholar] [CrossRef]
- Chang, Y.H.; Ng, P.K. Effects of Extrusion process variables on extractable ginsenosides in wheat-ginseng extrudates. J. Agric. Food Chem. 2009, 57, 2356–2362. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Chen, F. Degradation of ginsenosides in American ginseng (Panax quinquefolium) extracts during microwave and conventional heating. J. Agric. Food Chem. 1999, 47, 1501–1505. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.-I.; Lee, J.-Y.; Yang, J.-Y.; Jeong, S.M.; Lee, J.-H.; Nah, S.-Y.; Kim, Y. Evidence that the tertiary structure of 20 (S)-ginsenoside Rg 3 with tight hydrophobic packing near the chiral center is important for Na+ channel regulation. Biochem. Biophys. Res. Commun. 2005, 333, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.-W.; Wang, C.-Z.; Yuan, C.-S. Ginsenosides from American ginseng: Chemical and pharmacological diversity. Phytochemistry 2011, 72, 689–699. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.M.; Lee, J.-H.; Kim, J.-H.; Lee, B.-H.; Yoon, I.-S.; Lee, J.-H.; Kim, D.-H.; Rhim, H.; Kim, Y.; Nah, S.-Y. Stereospecificity of Ginsenoside Rg 3 Action on Ion Channels. Mol Cells 2004, 18, 383–389. [Google Scholar] [PubMed]
- Kim, J.-H.; Lee, J.-H.; Jeong, S.M.; Lee, B.-H.; Yoon, I.-S.; Lee, J.-H.; Choi, S.-H.; Kim, D.-H.; Park, T.-K.; Kim, B.-K. Stereospecific effects of ginsenoside Rg3 epimers on swine coronary artery contractions. Biol. Pharm. Bull. 2006, 29, 365–370. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kim, H.Y.; Kang, K.S.; Lee, J.G.; Yokozawa, T.; Park, J.H. The chemical and hydroxyl radical scavenging activity changes of ginsenoside-Rb1 by heat processing. Bioorg. Med. Chem. Lett. 2008, 18, 4515–4520. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-H.; Kim, Y.-J.; Yamabe, N.; Park, S.-H.; Kim, H.-K.; Jang, H.-J.; Kim, J.H.; Cheon, G.J.; Ham, J.; Kang, K.S. Stereospecific anticancer effects of ginsenoside Rg3 epimers isolated from heat-processed American ginseng on human gastric cancer cell. J. Ginseng Res. 2014, 38, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Teng, S.; Wang, Y.; Li, P.; Liu, J.; Wei, A.; Wang, H.; Meng, X.; Pan, D.; Zhang, X. Effects of R type and S type ginsenoside Rg3 on DNA methylation in human hepatocarcinoma cells. Mol. Med. Rep. 2017, 15, 2029–2038. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Su, F.; Su, X.; Hu, T.; Hu, S. Stereospecific antioxidant effects of ginsenoside Rg3 on oxidative stress induced by cyclophosphamide in mice. Fitoterapia 2012, 83, 636–642. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Chen, J.; Su, F.; Su, X.; Hu, T.; Hu, S. Stereospecificity of ginsenoside Rg3 in promotion of the immune response to ovalbumin in mice. Int. Immunol. 2012, 24, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Ru, Q.; Chen, L.; Ma, B.; Li, C. Stereospecificity of Ginsenoside Rg3 in the Promotion of Cellular Immunity in Hepatoma H22-Bearing Mice. J. Food Sci. 2014, 79, H1430–H1435. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Choi, W.-I.; Jeon, B.-N.; Choi, K.-C.; Kim, K.; Kim, T.-J.; Ham, J.; Jang, H.J.; Kang, K.S.; Ko, H. Stereospecific effects of ginsenoside 20-Rg3 inhibits TGF-β1-induced epithelial–mesenchymal transition and suppresses lung cancer migration, invasion and anoikis resistance. Toxicology 2014, 322, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-M.; Kim, D.-H.; Park, J.-H.; Na, H.-K.; Surh, Y.-J. Ginsenoside Rg3 induces apoptosis of human breast cancer (MDA-MB-231) cells. J. Cancer Prev. 2013, 18, 177. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-M.; Kim, D.-H.; Park, J.-H.; Surh, Y.-J.; Na, H.-K. Ginsenoside Rg3 inhibits constitutive activation of NF-κB signaling in human breast cancer (MDA-MB-231) cells: ERK and Akt as potential upstream targets. J. Cancer Prev. 2014, 19, 23. [Google Scholar] [CrossRef]
- Wu, K.; Li, N.; Sun, H.; Xu, T.; Jin, F.; Nie, J. Endoplasmic reticulum stress activation mediates Ginseng Rg3-induced anti-gallbladder cancer cell activity. Biochem. Biophys. Res. Commun. 2015, 466, 369–375. [Google Scholar] [CrossRef]
- Aziz, F.; Wang, X.; Liu, J.; Yan, Q. Ginsenoside Rg3 induces FUT4-mediated apoptosis in H. pylori CagA-treated gastric cancer cells by regulating SP1 and HSF1 expressions. Toxicol. In Vitro 2016, 31, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Nah, S.Y.; Jeon, J.H.; So, I.; Kim, S.J. Transient Receptor Potential Melastatin 7 Channels are Involved in Ginsenoside Rg3-Induced Apoptosis in Gastric Cancer Cells. Basic Clin. Pharmacol. Toxicol. 2011, 109, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Zhang, P.; Zeng, H.Q.; Lou, S.F.; Wang, D.X. Ginsenoside Rg3 induces apoptosis in human multiple myeloma cells via the activation of Bcl-2-associated X protein. Mol. Med. Rep. 2015, 12, 3557–3562. [Google Scholar] [CrossRef] [PubMed]
- Shan, X.; Tian, L.L.; Zhang, Y.M.; Wang, X.Q.; Yan, Q.; Liu, J.W. Ginsenoside Rg3 suppresses FUT4 expression through inhibiting NF-κB/p65 signaling pathway to promote melanoma cell death. Int. J. Oncol. 2015, 47, 701–709. [Google Scholar] [CrossRef]
- Choi, Y.J.; Lee, H.J.; Kang, D.W.; Han, I.H.; Choi, B.K.; Cho, W.H. Ginsenoside Rg3 induces apoptosis in the U87MG human glioblastoma cell line through the MEK signaling pathway and reactive oxygen species. Oncol. Rep. 2013, 30, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Joo, E.J.; Chun, J.; Ha, Y.W.; Ko, H.J.; Xu, M.-Y.; Kim, Y.S. Novel roles of ginsenoside Rg3 in apoptosis through downregulation of epidermal growth factor receptor. Chem. Biol. Interact. 2015, 233, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-W.; Chen, X.-M.; Chen, X.-H.; Zheng, S.-S. Ginsenoside Rg3 inhibit hepatocellular carcinoma growth via intrinsic apoptotic pathway. World J. Gastroenterol. 2011, 17, 3605. [Google Scholar] [CrossRef]
- Xie, Q.; Wen, H.; Zhang, Q.; Zhou, W.; Lin, X.; Xie, D.; Liu, Y. Inhibiting PI3K-AKt signaling pathway is involved in antitumor effects of ginsenoside Rg3 in lung cancer cell. Biomed. Pharmacother. 2017, 85, 16–21. [Google Scholar] [CrossRef]
- Sun, H.Y.; Lee, J.H.; Han, Y.-S.; Yoon, Y.M.; Yun, C.W.; Kim, J.H.; Song, Y.S.; Lee, S.H. Pivotal roles of ginsenoside Rg3 in tumor apoptosis through regulation of reactive oxygen species. Anticancer Res. 2016, 36, 4647–4654. [Google Scholar] [CrossRef]
- Li, Y.; Yang, T.; Li, J.; Hao, H.L.; Wang, S.Y.; Yang, J.; Luo, J.M. Inhibition of multiple myeloma cell proliferation by ginsenoside Rg3 via reduction in the secretion of IGF-1. Mol. Med. Rep. 2016, 14, 2222–2230. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Jung, K.H.; Morgan, M.J.; Kang, Y.-R.; Lee, H.-S.; Koo, G.-B.; Hong, S.-S.; Kwon, S.W.; Kim, Y.-S. Sensitization of TRAIL-induced cell death by 20 (S)-ginsenoside Rg3 via CHOP-mediated DR5 upregulation in human hepatocellular carcinoma cells. Mol. Cancer Ther. 2013, 12, 274–285. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, L.; Yu, Y.; Chen, B.; Tang, C.; Li, X. Antitumor effects of ginsenoside Rg3 on human hepatocellular carcinoma cells. Mol. Med. Rep. 2012, 5, 1295–1298. [Google Scholar] [PubMed]
- Yuan, H.-D.; Quan, H.-Y.; ZHanG, Y.; KiM, S.H.; Chung, S.-H. 20 (S)-Ginsenoside Rg3-induced apoptosis in HT-29 colon cancer cells is associated with AMPK signaling pathway. Mol. Med. Rep. 2010, 3, 825–831. [Google Scholar] [PubMed]
- Lee, S.Y.; Kim, G.T.; Roh, S.H.; Song, J.-S.; Kim, H.-J.; Hong, S.-S.; Kwon, S.W.; Park, J.H. Proteomic analysis of the anti-cancer effect of 20S-ginsenoside Rg3 in human colon cancer cell lines. Biosci. Biotechnol. Biochem. 2009, 73, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Li, M.; Wu, X.; Hu, Y.; Cao, Y.; Wang, X.A.; Xiang, S.; Li, H.; Jiang, L.; Tan, Z. 20 (S)-ginsenoside Rg3 promotes senescence and apoptosis in gallbladder cancer cells via the p53 pathway. Drug Des. Devel. Ther. 2015, 9, 3969. [Google Scholar] [PubMed]
- Qiu, X.-M.; Bai, X.; Jiang, H.-F.; He, P.; Wang, J.-H. 20-(s)-ginsenoside Rg3 induces apoptotic cell death in human leukemic U937 and HL-60 cells through PI3K/Akt pathways. Anticancer Drugs 2014, 25, 1072–1080. [Google Scholar] [CrossRef]
- Park, H.-M.; Kim, S.-J.; Kim, J.-S.; Kang, H.-S. Reactive oxygen species mediated ginsenoside Rg3-and Rh2-induced apoptosis in hepatoma cells through mitochondrial signaling pathways. Food Chem. Toxicol. 2012, 50, 2736–2741. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Peng, H.; Ou-Yang, X.; He, X. Research on the antitumor effect of ginsenoside Rg3 in B16 melanoma cells. Melanoma Res. 2008, 18, 322–329. [Google Scholar] [CrossRef]
- Li, J.; Liu, T.; Zhao, L.; Chen, W.; Hou, H.; Ye, Z.; Li, X. Ginsenoside 20 (S)-Rg3 inhibits the Warburg effect through STAT3 pathways in ovarian cancer cells. Int. J. Oncol. 2015, 46, 775–781. [Google Scholar] [CrossRef]
- Wang, J.-H.; Nao, J.-F.; Zhang, M.; He, P. 20 (s)-ginsenoside Rg3 promotes apoptosis in human ovarian cancer HO-8910 cells through PI3K/Akt and XIAP pathways. Tumour Biol. 2014, 35, 11985–11994. [Google Scholar] [CrossRef]
- Zheng, X.; Chen, W.; Hou, H.; Li, J.; Li, H.; Sun, X.; Zhao, L.; Li, X. Ginsenoside 20 (S)-Rg3 induced autophagy to inhibit migration and invasion of ovarian cancer. Biomed. Pharmacother. 2017, 85, 620–626. [Google Scholar] [CrossRef] [PubMed]
- He, B.-C.; Gao, J.-L.; Luo, X.; Luo, J.; Shen, J.; Wang, L.; Zhou, Q.; Wang, Y.-T.; Luu, H.H.; Haydon, R.C. Ginsenoside Rg3 inhibits colorectal tumor growth through the down-regulation of Wnt/ss-catenin signaling. Int. J. Oncol. 2011, 38, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.-J.; Zhang, M.-Z.; Wang, L.-X. Gensenoside Rg3 inhibits hypoxia-induced VEGF expression in human cancer cells. Cell. Physiol. Biochem. 2010, 26, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Jung, S.-Y.; Kwon, Y.-H.; Lee, J.-H.; Lee, Y.M.; Lee, B.-Y.; Kwon, S.-M. Ginsenoside Rg3 attenuates tumor angiogenesis via inhibiting bioactivities of endothelial progenitor cells. Cancer Biol. Ther. 2012, 13, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Z.; Aung, H.H.; Zhang, B.; Sun, S.; Li, X.-L.; He, H.; Xie, J.-T.; He, T.-C.; Du, W.; Yuan, C.-S. Chemopreventive effects of heat-processed Panax quinquefolius root on human breast cancer cells. Anticancer Res. 2008, 28, 2545–2551. [Google Scholar] [PubMed]
- Luo, X.; Wang, C.-Z.; Chen, J.; Song, W.-X.; Luo, J.; Tang, N.; He, B.-C.; Kang, Q.; Wang, Y.; Du, W. Characterization of gene expression regulated by American ginseng and ginsenoside Rg3 in human colorectal cancer cells. Int. J. Oncol. 2008, 32, 975–983. [Google Scholar] [CrossRef]
- Sin, S.; Kim, S.Y.; Kim, S.S. Chronic treatment with ginsenoside Rg3 induces Akt-dependent senescence in human glioma cells. Int. J. Oncol. 2012, 41, 1669–1674. [Google Scholar] [CrossRef]
- Kim, H.-S.; Lee, E.-H.; Ko, S.-R.; Choi, K.-J.; Park, J.-H.; Im, D.-S. Effects of ginsenosides Rg 3 and Rh 2 on the proliferation of prostate cancer cells. Arch. Pharm. Res. 2004, 27, 429. [Google Scholar] [CrossRef]
- Shan, X.; Aziz, F.; Tian, L.L.; Wang, X.Q.; Yan, Q.; Liu, J.W. Ginsenoside Rg3-induced EGFR/MAPK pathway deactivation inhibits melanoma cell proliferation by decreasing FUT4/LeY expression. Int. J. Oncol. 2015, 46, 1667–1676. [Google Scholar] [CrossRef]
- Tian, L.; Shen, D.; Li, X.; Shan, X.; Wang, X.; Yan, Q.; Liu, J. Ginsenoside Rg3 inhibits epithelial-mesenchymal transition (EMT) and invasion of lung cancer by down-regulating FUT4. Oncotarget 2016, 7, 1619. [Google Scholar] [CrossRef]
- Shan, X.; Fu, Y.-S.; Aziz, F.; Wang, X.-Q.; Yan, Q.; Liu, J.-W. Ginsenoside Rg3 inhibits melanoma cell proliferation through down-regulation of histone deacetylase 3 (HDAC3) and increase of p53 acetylation. PLoS ONE 2014, 9, e115401. [Google Scholar] [CrossRef] [PubMed]
- Junmin, S.; Hongxiang, L.; Zhen, L.; Chao, Y.; Chaojie, W. Ginsenoside Rg3 inhibits colon cancer cell migration by suppressing nuclear factor kappa B activity. J. Tradit. Chin. Med. 2015, 35, 440–444. [Google Scholar] [CrossRef]
- Shinkai, K.; Akedo, H.; Mukai, M.; Imamura, F.; Isoai, A.; Kobayashi, M.; Kitagawa, I. Inhibition of in vitro tumor cell invasion by ginsenoside Rg3. Cancer Sci. 1996, 87, 357–362. [Google Scholar]
- Tm, X.; Man-hua, C.; Xin, Y.; Lp, G. Inhibitory effect of ginsenoside Rg3 on ovarian cancer metastasis. Chin. Med. J. 2008, 121, 1394–1397. [Google Scholar]
- Lee, S.G.; Kang, Y.J.; Nam, J.-O. Anti-metastasis effects of ginsenoside Rg3 in B16F10 cells. J. Microbiol. Biotechnol. 2015, 25, 1997–2006. [Google Scholar] [CrossRef]
- Liu, T.; Zhao, L.; Zhang, Y.; Chen, W.; Liu, D.; Hou, H.; Ding, L.; Li, X. Ginsenoside 20 (S)-Rg3 targets HIF-1α to block hypoxia-induced epithelial-mesenchymal transition in ovarian cancer cells. PLoS ONE 2014, 9, e103887. [Google Scholar] [CrossRef]
- Chen, X.-P.; Qian, L.-L.; Jiang, H.; Chen, J.-H. Ginsenoside Rg3 inhibits CXCR 4 expression and related migrations in a breast cancer cell line. Int. J. Clin. Oncol. 2011, 16, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, M.; Yoo, Y.; Matsuzawa, K.; Sato, K.; Saiki, I.; Tonooka, S.; Samukawa, K.; Azuma, I. Inhibitory effect of tumor metastasis in mice by saponins, ginsenoside-Rb2, 20 (R)-and 20 (S)-ginsenoside-Rg3, of red ginseng. Biol. Pharm. Bull. 1995, 18, 1197–1202. [Google Scholar] [CrossRef]
- Pan, X.-Y.; Guo, H.; Han, J.; Hao, F.; An, Y.; Xu, Y.; Xiaokaiti, Y.; Pan, Y.; Li, X.-J. Ginsenoside Rg3 attenuates cell migration via inhibition of aquaporin 1 expression in PC-3M prostate cancer cells. Eur. J. Pharmacol. 2012, 683, 27–34. [Google Scholar] [CrossRef]
- Park, D.; Bae, D.-K.; Jeon, J.H.; Lee, J.; Oh, N.; Yang, G.; Yang, Y.-H.; Kim, T.K.; Song, J.; Lee, S.H. Immunopotentiation and antitumor effects of a ginsenoside Rg3-fortified red ginseng preparation in mice bearing H460 lung cancer cells. Environ. Toxicol. Pharmacol. 2011, 31, 397–405. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Song, Y.M.; Wang, B.; Zhang, F.R.; Yang, R.; Wang, H.Q.; Zhang, G.J. Ginsenoside Rg3 sensitizes human non-small cell lung cancer cells to γ-radiation by targeting the nuclear factor-κB pathway. Mol. Med. Rep. 2015, 12, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.-Y.; Kim, E.-H.; Kim, S.-W.; Kim, S.-N.; Park, J.-D.; Rhee, D.-K. Selective toxicity of ginsenoside Rg 3 on multidrug resistant cells by membrane fluidity modulation. Arch. Pharm. Res. 2008, 31, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.; Li, H.-D.; Li, B.; Jiang, S.-D.; Jiang, L.-S. Ginsenoside Rg3 induces DNA damage in human osteosarcoma cells and reduces MNNG-induced DNA damage and apoptosis in normal human cells. Oncol. Rep. 2014, 31, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.-Q.; Zheng, Q.-H.; Chen, H.; Chen, L.; Xu, J.-B.; Chen, M.-Y.; Lu, D.; Wang, Z.-H.; Tong, H.-F.; Lin, S. Ginsenoside Rg3 inhibition of vasculogenic mimicry in pancreatic cancer through downregulation of VE-cadherin/EphA2/MMP9/MMP2 expression. Int. J. Oncol. 2014, 45, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Yue, P.Y.; Wong, D.Y.; Wu, P.; Leung, P.; Mak, N.; Yeung, H.; Liu, L.; Cai, Z.; Jiang, Z.-H.; Fan, T. The angiosuppressive effects of 20 (R)-ginsenoside Rg3. Biochem. Pharmacol. 2006, 72, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Ren, Y.; Zhang, J.; Ma, S.; Gao, F.; Wu, Y. Correlation of insulin-like growth factor-1 (IGF-1) to angiogenesis of breast cancer in IGF-1-deficient mice. Ai Zheng 2007, 26, 1215–1220. [Google Scholar] [PubMed]
- Yang, L.Q.; Wang, B.; Gan, H.; Fu, S.T.; Zhu, X.X.; Wu, Z.N.; Zhan, D.W.; Gu, R.L.; Dou, G.F.; Meng, Z.Y. Enhanced oral bioavailability and anti-tumour effect of paclitaxel by 20 (s)-ginsenoside Rg3 in vivo. Biopharm. Drug Dispos. 2012, 33, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-W.; Kwon, H.-Y.; Chi, D.-W.; Shim, J.-H.; Park, J.-D.; Lee, Y.-H.; Pyo, S.; Rhee, D.-K. Reversal of P-glycoprotein-mediated multidrug resistance by ginsenoside Rg3. Biochem. Pharmacol. 2003, 65, 75–82. [Google Scholar] [CrossRef]
- Zhang, Q.; Kang, X.; Yang, B.; Wang, J.; Yang, F. Antiangiogenic effect of capecitabine combined with ginsenoside Rg3 on breast cancer in mice. Cancer Biother. Radiopharm. 2008, 23, 647–654. [Google Scholar] [CrossRef]
- Zhang, Q.; Kang, X.; Zhao, W. Antiangiogenic effect of low-dose cyclophosphamide combined with ginsenoside Rg3 on Lewis lung carcinoma. Biochem. Biophys. Res. Commun. 2006, 342, 824–828. [Google Scholar] [CrossRef]
- Zhang, Q.H.; Wu, C.F.; Duan, L.; Yang, J.Y. Protective effects of ginsenoside Rg 3 against cyclophosphamide-induced DNA damage and cell apoptosis in mice. Arch. Toxicol. 2008, 82, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-G.; Huang, Y.; Cui, D.-D.; Huang, X.-B.; Mao, S.-H.; Ji, L.-L.; Song, H.-B.; Yi, C. Inhibitory effect of ginsenoside Rg3 combined with gemcitabine on angiogenesis and growth of lung cancer in mice. BMC Cancer 2009, 9, 250. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Yu, Y.; Wang, L.; Wu, B.; Xia, L.; Feng, F.; Ling, Z.; Wang, S. Additive antiangiogenesis effect of ginsenoside Rg3 with low-dose metronomic temozolomide on rat glioma cells both in vivo and in vitro. J. Exp. Clin. Cancer Res. 2016, 35, 32. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.K.; Park, K.-K.; Chung, A.-S.; Chung, W.-Y. Ginsenoside Rg3 enhances the chemosensitivity of tumors to cisplatin by reducing the basal level of nuclear factor erythroid 2-related factor 2-mediated heme oxygenase-1/NAD (P) H quinone oxidoreductase-1 and prevents normal tissue damage by scavenging cisplatin-induced intracellular reactive oxygen species. Food Chem. Toxicol. 2012, 50, 2565–2574. [Google Scholar] [PubMed]
- Lee, Y.J.; Lee, S.; Ho, J.N.; Byun, S.-S.; Hong, S.K.; Lee, S.E.; Lee, E. Synergistic antitumor effect of ginsenoside Rg3 and cisplatin in cisplatin-resistant bladder tumor cell line. Oncol. Rep. 2014, 32, 1803–1808. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Huo, B.; Lv, Y.; Wang, Y.; Liu, W. Ginsenoside Rg3 enhances the inhibitory effects of chemotherapy on esophageal squamous cell carcinoma in mice. Mol. Clin. Oncol. 2014, 2, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Lee, S.Y.; Yuk, D.Y.; Moon, D.C.; Choi, S.S.; Kim, Y.; Han, S.B.; Oh, K.-W.; Hong, J.T. Inhibition of NF-κB by ginsenoside Rg3 enhances the susceptibility of colon cancer cells to docetaxel. Arch. Pharm. Res. 2009, 32, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Lee, S.Y.; Cho, J.S.; Son, S.M.; Choi, S.S.; Yun, Y.P.; Yoo, H.S.; Oh, K.-W.; Han, S.B.; Hong, J.T. Combination of ginsenoside Rg3 with docetaxel enhances the susceptibility of prostate cancer cells via inhibition of NF-κB. Eur. J. Pharmacol. 2010, 631, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-G.; Jung, K.H.; Lee, D.-G.; Yoon, J.-H.; Choi, K.S.; Kwon, S.W.; Shen, H.-M.; Morgan, M.J.; Hong, S.-S.; Kim, Y.-S. 20 (S)-Ginsenoside Rg3 is a novel inhibitor of autophagy and sensitizes hepatocellular carcinoma to doxorubicin. Oncotarget 2014, 5, 4438. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, L.; Wang, T.; Jiang, X.; Zhang, H.; Li, P.; Lv, B.; Gao, X. Ginsenoside Rg3 antagonizes adriamycin-induced cardiotoxicity by improving endothelial dysfunction from oxidative stress via upregulating the Nrf2-ARE pathway through the activation of akt. Phytomedicine 2015, 22, 875–884. [Google Scholar] [CrossRef]
- Che, J.-B.; Liu, Z.-H.; Ma, H.-B.; Li, Y.; Zhao, H.; Li, X.-H.; Liu, W.-C.; Shi, G.-N. Influence of As2O3 combined with ginsenosides Rg3 on inhibition of lung cancer NCI-H1299 cells and on subsistence of nude mice bearing hepatoma. Asian Pac. J. Trop. Med. 2014, 7, 772–775. [Google Scholar] [CrossRef]
- Yool, A.J.; Brown, E.A.; Flynn, G.A. Roles for novel pharmacological blockers of aquaporins in the treatment of brain oedema and cancer. Clin. Exp. Pharmacol. Physiol. 2010, 37, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Yool, A.J. Functional domains of aquaporin-1: Keys to physiology, and targets for drug discovery. Curr. Pharm. Des. 2007, 13, 3212–3221. [Google Scholar] [CrossRef] [PubMed]
- Dorward, H.S.; Du, A.; Bruhn, M.A.; Wrin, J.; Pei, J.V.; Evdokiou, A.; Price, T.J.; Yool, A.J.; Hardingham, J.E. Pharmacological blockade of aquaporin-1 water channel by AqB013 restricts migration and invasiveness of colon cancer cells and prevents endothelial tube formation in vitro. J. Exp. Clin. Cancer Res. 2016, 35, 36. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, M.; Saadoun, S.; Verkman, A. Aquaporins and cell migration. Pflüg. Arch 2008, 456, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Verkman, A.; Hu, J.; Verkman, A. Increased migration and metastatic potential of tumor cells expressing aquaporin water channels. FASEB J. 2006, 20, 1892–1894. [Google Scholar] [CrossRef] [PubMed]
- Wragg, J.W.; Heath, V.L.; Bicknell, R. Sunitinib treatment enhances metastasis of innately drug resistant breast tumors. Cancer Res. 2016, 77, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Verkman, A.; Hara-Chikuma, M.; Papadopoulos, M.C. Aquaporins—New players in cancer biology. J. Mol. Med. 2008, 86, 523–529. [Google Scholar] [CrossRef]
- Agre, P.; Preston, G.M.; Smith, B.L.; Jung, J.S.; Raina, S.; Moon, C.; Guggino, W.B.; Nielsen, S. Aquaporin CHIP: The archetypal molecular water channel. Am. J. Physiol. Ren. Physiol. 1993, 265, F463–F476. [Google Scholar] [CrossRef]
- Yool, A.J.; Campbell, E.M. Structure, function and translational relevance of aquaporin dual water and ion channels. Mol. Asp. Med. 2012, 33, 553–561. [Google Scholar] [CrossRef]
- De Ieso, M.L.; Yool, A.J. Mechanisms of Aquaporin-Facilitated Cancer Invasion and Metastasis. Front. Chem. 2018, 6, 135. [Google Scholar] [CrossRef] [PubMed]
- Campbell, E.M.; Birdsell, D.N.; Yool, A.J. The activity of human aquaporin 1 as a cGMP-gated cation channel is regulated by tyrosine phosphorylation in the carboxyl terminal domain. Mol. Pharmacol. 2011, 81, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Baetz, N.W.; Stamer, W.D.; Yool, A.J. Stimulation of aquaporin-mediated fluid transport by cyclic GMP in human retinal pigment epithelium in vitro. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2127–2132. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Zhang, H.; Shao, Y.; Liu, X.; Yang, L.; Huang, Y.; Fu, L.; Gu, F.; Ma, Y. Expression of aquaporin1, a water channel protein, in cytoplasm is negatively correlated with prognosis of breast cancer patients. Oncotarget 2016, 7, 8143. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.-B.; Shi, S.; Zhang, R.-J.; Wang, T.-T.; Tan, Y.-J.; Zhang, D.; Fei, X.-Y.; Ding, G.-L.; Gao, Q.; Chen, C. Aquaporin-1 plays a crucial role in estrogen-induced tubulogenesis of vascular endothelial cells. J. Clin. Endocrinol. Metab. 2013, 98, E672–E682. [Google Scholar] [CrossRef] [PubMed]
- Endo, M.; Jain, R.K.; Witwer, B.; Brown, D. Water channel (aquaporin 1) expression and distribution in mammary carcinomas and glioblastomas. Microvasc. Res. 1999, 58, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Saadoun, S.; Papadopoulos, M.C.; Hara-Chikuma, M.; Verkman, A. Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature 2005, 434, 786. [Google Scholar] [CrossRef]
- Esteva-Font, C.; Jin, B.-J.; Verkman, A. Aquaporin-1 gene deletion reduces breast tumor growth and lung metastasis in tumor-producing MMTV-PyVT mice. FASEB J. 2014, 28, 1446–1453. [Google Scholar] [CrossRef]
- Mobasheri, A.; Shakibaei, M.; Marples, D. Immunohistochemical localization of aquaporin 10 in the apical membranes of the human ileum: A potential pathway for luminal water and small solute absorption. Histochem. Cell Biol. 2004, 121, 463–471. [Google Scholar] [CrossRef]
- Mobasheri, A.; Airley, R.; Hewitt, S.M.; Marples, D. Heterogeneous expression of the aquaporin 1 (AQP1) water channel in tumors of the prostate, breast, ovary, colon and lung: A study using high density multiple human tumor tissue microarrays. Int. J. Oncol. 2005, 26, 1149–1158. [Google Scholar] [CrossRef]
- Shi, Z.; Zhang, T.; Luo, L.; Zhao, H.; Cheng, J.; Xiang, J.; Zhao, C. Aquaporins in human breast cancer: Identification and involvement in carcinogenesis of breast cancer. J. Surg. Oncol. 2012, 106, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Otterbach, F.; Callies, R.; Adamzik, M.; Kimmig, R.; Siffert, W.; Schmid, K.W.; Bankfalvi, A. Aquaporin 1 (AQP1) expression is a novel characteristic feature of a particularly aggressive subgroup of basal-like breast carcinomas. Breast Cancer Res. Treat. 2010, 120, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Liu, F.; Ma, Y.; Gu, F. Cytoplasmic expression of aquaporin-1 in breast cancer cells and its relationship with clinicopathological characteristics and prognosis. Zhonghua Zhong Liu Za Zhi 2013, 35, 904–909. [Google Scholar] [PubMed]
- Kanaoka, M. Metabolism of ginseng saponins, ginsenosides, by human intestinal bacteria. J. Trad. Med. 1994, 11, 241–245. [Google Scholar]
- Karikura, M.; Miyase, T.; Tanizawa, H.; Taniyama, T.; Takino, Y. Studies on absorption, distribution, excretion and metabolism of ginseng saponins. VII. Comparison of the decomposition modes of ginsenoside-Rb1 and-Rb2 in the digestive tract of rats. Chem. Pharm. Bull. 1991, 39, 2357–2361. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, E.; Kim, D.; Lee, J.; Yoo, J.; Koh, B. Studies on absorption, distribution and metabolism of ginseng in humans after oral administration. J. Ethnopharmacol. 2009, 122, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.; Zhao, Y.; Chen, P.; Huang, H.; Liu, H.; Jiang, H.; Zhang, R.; Wang, H. Structure-activity relationship and substrate-dependent phenomena in effects of ginsenosides on activities of drug-metabolizing P450 enzymes. PLoS ONE 2008, 3, e2697. [Google Scholar] [CrossRef]
- Bae, E.-A.; Han, M.J.; Choo, M.-K.; Park, S.-Y.; Kim, D.-H. Metabolism of 20 (S)-and 20 (R)-ginsenoside Rg3 by human intestinal bacteria and its relation to in vitro biological activities. Biol. Pharm. Bull. 2002, 25, 58–63. [Google Scholar] [CrossRef]
- Lu, J.-M.; Yao, Q.; Chen, C. Ginseng compounds: An update on their molecular mechanisms and medical applications. Curr. Vasc. Pharmacol. 2009, 7, 293–302. [Google Scholar] [CrossRef]
- Li, K.; Chen, X.; Xu, J.; Li, X.; Zhong, D. Liquid chromatography/tandem mass spectrometry for pharmacokinetic studies of 20 (R)-ginsenoside Rg3 in dog. Rapid Commun. Mass Spectrom. 2005, 19, 813–817. [Google Scholar] [CrossRef]
- Qian, T.; Cai, Z.; Wong, R.N.; Mak, N.K.; Jiang, Z.-H. In vivo rat metabolism and pharmacokinetic studies of ginsenoside Rg3. J. Chromatogr. B 2005, 816, 223–232. [Google Scholar] [CrossRef]
- Cai, Z.; Qian, T.; Wong, R.N.; Jiang, Z.-H. Liquid chromatography–electrospray ionization mass spectrometry for metabolism and pharmacokinetic studies of ginsenoside Rg3. Anal. Chim. Acta 2003, 492, 283–293. [Google Scholar] [CrossRef]
- Xie, H.-T.; Wang, G.-J.; Sun, J.-G.; Tucker, I.; Zhao, X.-C.; Xie, Y.-Y.; Li, H.; Jiang, X.-L.; Wang, R.; Xu, M.-J. High performance liquid chromatographic–mass spectrometric determination of ginsenoside Rg3 and its metabolites in rat plasma using solid-phase extraction for pharmacokinetic studies. J. Chromatogr. B 2005, 818, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zheng, X.; Jiang, J.; Zhou, H.; Hu, P. Determination of ginsenoside Rg3 in human plasma and urine by high performance liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2010, 878, 2266–2273. [Google Scholar] [CrossRef]
- Huan, P.; Hailin, W.; Li, F.; Chengye, S. Pharmacokinetics of 20 (R)-Ginsenoside Rg3 in Human Volunteers. JCPS 2001, 10, 140–143. [Google Scholar]
- Bae, S.H.; Park, J.B.; Zheng, Y.F.; Jang, M.J.; Kim, S.O.; Kim, J.Y.; Yoo, Y.H.; Yoon, K.D.; Oh, E.; Bae, S.K. Pharmacokinetics and tissue distribution of ginsenoside Rh2 and Rg3 epimers after oral administration of BST204, a purified ginseng dry extract, in rats. Xenobiotica 2014, 44, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zou, H.; Kong, L.; Zhang, Y.; Pang, H.; Su, C.; Liu, G.; Hui, M.; Fu, L. Determination of ginsenoside Rg3 in plasma by solid-phase extraction and high-performance liquid chromatography for pharmacokinetic study. J. Chromatogr. B Biomed. Sci. Appl. 1999, 731, 403–409. [Google Scholar] [CrossRef]
- Kim, H.-G.; Yoo, S.-R.; Park, H.-J.; Lee, N.-H.; Shin, J.-W.; Sathyanath, R.; Cho, J.-H.; Son, C.-G. Antioxidant effects of Panax ginseng CA Meyer in healthy subjects: A randomized, placebo-controlled clinical trial. Food Chem. Toxicol. 2011, 49, 2229–2235. [Google Scholar] [CrossRef]
- Seo, S.K.; Hong, Y.; Yun, B.H.; Chon, S.J.; Jung, Y.S.; Park, J.H.; Cho, S.; Choi, Y.S.; Lee, B.S. Antioxidative effects of Korean red ginseng in postmenopausal women: A double-blind randomized controlled trial. J. Ethnopharmacol. 2014, 154, 753–757. [Google Scholar] [CrossRef]
- Lee, N.-H.; Yoo, S.-R.; Kim, H.-G.; Cho, J.-H.; Son, C.G. Safety and tolerability of Panax ginseng root extract: A randomized, placebo-controlled, clinical trial in healthy Korean volunteers. J. Altern. Complement. Med. 2012, 18, 1061–1069. [Google Scholar] [CrossRef]
- Lu, P.; Su, W.; Miao, Z.-H.; Niu, H.-R.; Liu, J.; Hua, Q.-L. Effect and mechanism of ginsenoside Rg3 on postoperative life span of patients with non-small cell lung cancer. Chin. J. Integr. Med. 2008, 14, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, Y.; Niu, K.; Chen, X.; Xia, L.; Lu, D.; Kong, R.; Chen, Z.; Duan, Y.; Sun, J. Clinical benefit from EGFR-TKI plus ginsenoside Rg3 in patients with advanced non-small cell lung cancer harboring EGFR active mutation. Oncotarget 2016, 7, 70535. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Yan, Z.; Liu, R.; Shi, P.; Qian, S.; Qu, X.; Zhu, L.; Zhang, W.; Wang, J. Prospective study of transcatheter arterial chemoembolization (TACE) with ginsenoside Rg3 versus TACE alone for the treatment of patients with advanced hepatocellular carcinoma. Radiology 2016, 280, 630–639. [Google Scholar] [CrossRef] [PubMed]
- Szewczyk, G.; Rak, J.; Ruth, J.H. Inflammatory Mediators of Angiogenesis. Med. Inflamm. 2013, 2013, 610543. [Google Scholar] [CrossRef] [PubMed]
- Naldini, A.; Carraro, F. Role of inflammatory mediators in angiogenesis. Curr. Drug Targets Inflamm. Allergy 2005, 4, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Jiang, H.; Zhu, X.; Liu, X.; Li, J. Ginsenoside Rg3 promotes cytotoxicity of Paclitaxel through inhibiting NF-κB signaling and regulating Bax/Bcl-2 expression on triple-negative breast cancer. Biomed. Pharmacother. 2017, 89, 227–232. [Google Scholar] [CrossRef] [PubMed]
| Cancer | Mechanism of Action | Reference | |
|---|---|---|---|
| Ovary | IA | Downregulation of PI3K/Akt and the proteins of the IAP family * Activation of caspases -3 and -9 * | [52] |
| Inhibition of Warburg effect by inactivation of Stat3 * | [51] | ||
| IP | Suppression of the Warburg effect and modulating the Stat3/HK2 pathway | [51] | |
| Colon | IA | Activation of AMPK * Increased DNA fragmentation, cleavage of PARP * Downregulation of Bcl-2 * Upregulation of p53, Bax, release of cytochrome c and caspase-3 and -9 * | [45,46] |
| IP | Inhibiting the function of β-catenin and the ß-catenin/Tcf signalling Inhibits cell proliferation | [54] | |
| Reduced mitosis-related proteins * Reduced DNA-repair proteins * | [46] | ||
| Changes in the Eph/ephrin signalling axis * | [58] | ||
| Lung | IA | Activation of the intrinsic and extrinsic pathways Regulation of apoptosis-associated proteins such as BCL2, BAX, PARP-1 Cleaving caspase-3 Inhibition of EGFR, Stat3, Akt and PI3/Akt signalling | [38,40,41] |
| IP | Decreasing the expression of FUT4 and biosynthesis of LeY ◊ Decreasing the activation of EGFR and its downstream signaling ◊ | [38,62] | |
| Suppression of some of the cell cycle proteins such as cyclin D1 and E, CDK-2 and -4 Suppression of some of the MAPK-associated growth proteins such as JNK, ERK and P38 | [41] | ||
| Liver | IA | Activation of the intrinsic and extrinsic pathways through increasing Bax, caspase-3, release of cytochrome c, decreasing Bcl-2, Bcl-xL | [39,44,49] |
| Sensitizing liver cancer cells to TRAIL-induced cell death Promoting TRAIL-induced caspase-dependent apoptosis (via DR5 upregulation and induction of CHOP) | [43] | ||
| Multiple myeloma | IA | Increasing the activity of caspase-3 and expression of Bax | [35] |
| IP | Inhibiting the secretion of IGF-1 Affecting the Akt/mTOR signalling and their proliferation | [42] | |
| Leukaemia | IA | Activating caspases -3 and -9 Downregulating PI3K/Akt family proteins | [48] |
| Gallbladder | IA | Increasing caspase-12 (an endoplasmic reticulum stress-mediated apoptosis) | [32] |
| Activating p53 pathway and intrinsic apoptosis pathway * Inducing cell senescence * | [47] | ||
| Gastric | IA | Blocking TRMP7 Upregulation of caspase-3, -8, -9, Bax and downregulation of Bcl2 | [24,34] |
| Inhibiting the expression of FUT4 (via regulation of SP1 and HSD1) Activation of caspase-3, -8 and -9 | [24,33] | ||
| Melanoma | IA | Preventing the binding of NF-κB to the FUT4 promoter Activating intrinsic and extrinsic apoptosis pathways | [36] |
| Increasing the expression of caspase and Bcl-2 * | [50] | ||
| IP | Decreasing the levels of active Akt * Dysregulating the PI3K/Akt pathway, hence affecting the cell cycle * | [50] | |
| Inducing a G0/G1 cell cycle arrest ◊ Decreasing the HDAC3 ◊ Increasing the acetylation and stability of p53 ◊ | [63] | ||
| Reducing FUT4 and LeY ◊ Inhibiting the EGFR/MAPK signalling pathway ◊ | [61] | ||
| Glioblastoma multiforme | IA | Suppressing the MEK/MAPK signalling pathway and activating ROS by the antioxidant enzyme system, leading to apoptosis | [37] |
| Prostate | IP | Inhibition of DNA synthesis * Affecting the MAPK activity through ERKs, p38 and JNK * | [60] |
| Glioma | IP | Activating Akt and p53/p21 dependent signalling pathways causing cell senescence * | [59] |
| Cancer | Mechanism | Reference |
|---|---|---|
| Ovary | Inhibition of angiogenesis and cell invasion Decreased expression of MMP-9 | [66] |
| Blocking the EMT * Reducing HIF-1α expression * | [68] | |
| Colon | Suppressing NF-κB and its products (c-Myc, COX-2, MMP-9) | [64] |
| Prostate | Decreasing the expression of AQP1 * | [71] |
| Melanoma | Inhibiting the expression of MMP-13 Reducing cell adhesion, invasion and angiogenesis * | [50,67,70] |
| Decreasing the expression of HDAC3 ◊ | [63] | |
| Lung | Inhibiting TGF-β1 Inactivating proteins involved in EMT (MMP-2, p38 MAPK and Smad2) ◊ | [29] |
| Downregulating FUT4 and EGFR mediated migration (through MAPK and NF-κB) ◊ | [62] | |
| Endothelial progenitor cells | Decreasing the activation of the VEGF dependent p38/ERK signalling | [56] |
| Esophageal and renal | Decreasing the expression of VEGF Inhibiting other signalling pathways of HIF-1α, COX-2, NF-κB, STAT3 and MAPKs | [55] |
| Studied Model | Drug Combination | Effects | Reference |
|---|---|---|---|
| Lewis lung cancer mouse model | Rg3 + cyclophosphamide (continuous low-dose) | Less toxicity induced by capecitabine Longer animal survival Reduced susceptibility to drug resistance Increased anti-angiogenic activity | [82] |
| Mouse model | 20(S)-Rg3 + cyclophosphamide | Inhibiting cyclophosphamide-induced DNA damages in the peripheral lymphocyte cells and bone marrow cells Reducing number of apoptotic cells of mice and improving the anti-oxidative markers in mice (such as SOD, MDA and GPX) | [83] |
| Mouse bearing hepatocellular carcinoma model | Rg3 + cyclophosphamide | Alteration of the expression of Bcl-2 family and induction of intrinsic pathway of apoptosis Prolonging mouse survival | [39] |
| Mouse bearing lung tumor model | Rg3 + gemcitabin | Enhancing the efficacy of gemcitabine on suppressing tumor growth Increasing the quality of life Prolonging mice survival Increasing tumor’s necrosis rate Decreasing VEGF expression, microvessel density (assessed by the expression of CD31) and arterial blood flow in tumors such as peak systolic velocity | [84] |
| Glioma cell line | Rg3 + temozolomide | Inducing cell cycle arrest and apoptosis Attenuating the expression of VEGF-a and Bcl-2 | [85] |
| Glioma allograft model of mouse | Rg3 + temozolomide | Antiangiogenic effect (reduced relative cerebral blood volume, VEGF levels and microvessel density) Improving the antiangiogenic effects of temozolomide No additive effect on tumor growth | [85] |
| Mouse bearing colon tumor | Rg3 + cisplatin | Improving anti-cancer effects of cisplatin Inhibiting tumor growth Reducing the toxicities of cisplatin (decreasing the intracellular levels of ROS) | [86] |
| Kidney, liver and colon resistant cancer cells | Rg3 + cisplatin | Decreasing the high levels of etoxifying enzymes such as heme-oxygenase (HO-1) and NAD(P)H quinone oxidoreductase (NQO-1) | [86] |
| Cisplatin-resistant bladder tumor cell lines | Rg3 + cisplatin | Synergistic effect in inhibiting the proliferation (possibly through activating the intrinsic apoptosis pathway (decreased Bcl-2 and increased cytochrome c and caspase-3) and cell cycle alterations in G2/M phase) | [87] |
| Mouse bearing oesophageal squamous cell carcinoma | Rg3 + cisplatin | Enhancing the inhibitory effects of cisplatin Reducing the proliferation of cancer cells Decreasing the microvascular density of the tumors | [88] |
| Colon cancer cell lines | Rg3 + docetaxel | Sensitizing the cells to the docetaxel Improving its apoptotic effect (via inhibiting NF-κB and the expression of anti-apoptotic proteins such as Bcl-2, XIAP, and ciap-1) Increasing the expression of pro-apoptotic proteins (such as Bax, caspase-3 and -9) | [89] |
| Prostate cancer cell lines | Rg3 + docetaxel | Inhibiting cell growth Inducing apoptosis and its associated protein Arresting the cells at G0/G1 Modulating cell cycle-associated proteins Inhibiting the activity of NF-κB | [90] |
| Prostate cancer cell lines | Rg3 + docetaxel + cisplatin | More effective inhibition of the activity of NF-κB and cell growth | [90] |
| Mouse bearing hepatocellular tumor | 20(S)-Rg3 + doxorubicin | Suppressing the autophagy via regulating autophagy-associated proteins Inhibiting autophagic flux Synergistic effects in inhibiting tumor growth | [91] |
| Rat model | Rg3 + doxorubicin | Reducing doxorubicin-induced cardiotoxicity (by improving the ejection fraction, fractional shortening and left ventricular outflow) Improving the oxidative damage and apoptosis induced by doxorubicin (via the activation of Akt and the Nrf2-ARE pathway) | [92] |
| NCI-H1299 lung cancer cells | Rg3 + As2O3 | Inhibiting the proliferation of NCI-H1299 lung cancer cells | [93] |
| Mouse bearing lung tumors | Rg3 + As2O3 | Promoting apoptosis in tumor cells Prolonging the survival of the mice | [93] |
| Ginsenoside | Model | Route | Dose | Sample | Detected Rg3 | Outcomes | Reference |
|---|---|---|---|---|---|---|---|
| Rg3 | Sprague–Dawley rats | IV | 1 mg/kg | Plasma | Detected for 12 h | t1/2α: 0.12 ± 0.03 h | [125] |
| t1/2β: 2.09 ± 0.50 h | |||||||
| Oral | 10 mg/kg | Plasma | Detected for 12 h | ||||
| Healthy humans | Oral | 3.2 mg/kg | Plasma | Detected for 8 h | Cmax: 15.67 ± 6.14 ng/mL tmax: 0.66 ± 0.01 h | [129] | |
| Healthy humans | IM | 10, 30 and 60 mg | Plasma | Detected for 216 h | [126] | ||
| 20(R)-Rg3 | Sprague–Dawley rats | IV | 5 mg/kg | Plasma | Detected for 1.5 h | t1/2: 14 min | [124] |
| Urine | Not detected | ||||||
| Oral | 50 mg/kg | Urine | Not detected in 1 h | rapid GI metabolism | |||
| Plasma | Not detected in 1 h | ||||||
| Dogs | IV | 0.3 mg/kg | Plasma | Detected for 12 h | t1/2: 1.71 (± 0.11) h | [122] | |
| Oral | 2 mg/kg | Plasma | Detected for 24 h | t1/2: 5.99 (± 1.16) h | |||
| Sprague–Dawley rats | IV | 5 mg/kg, within 1 min | Plasma | Detected for 1.5 h | t1/2: 18.5 min | [123] | |
| Urine | Not detectable | N/A | |||||
| Oral | 100 mg/kg | Plasma | Not detectable | N/A | |||
| Urine | Not detectable | ||||||
| Feces | 6 different deglycosylated and oxygenated metabolites | ||||||
| Healthy humans | Oral | 3.2 mg/kg | Plasma | Detected for 8 h | tmax: 0.66 ± 0.10 h Cmax: 1± 6 ng/mL t1/2α: 0.46 ± 0.12 h t1/2β: 4.9 1.1 h t1/2(Ka): 0.28 ± 0.04 h | [127] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakhjavani, M.; Hardingham, J.E.; Palethorpe, H.M.; Tomita, Y.; Smith, E.; Price, T.J.; Townsend, A.R. Ginsenoside Rg3: Potential Molecular Targets and Therapeutic Indication in Metastatic Breast Cancer. Medicines 2019, 6, 17. https://doi.org/10.3390/medicines6010017
Nakhjavani M, Hardingham JE, Palethorpe HM, Tomita Y, Smith E, Price TJ, Townsend AR. Ginsenoside Rg3: Potential Molecular Targets and Therapeutic Indication in Metastatic Breast Cancer. Medicines. 2019; 6(1):17. https://doi.org/10.3390/medicines6010017
Chicago/Turabian StyleNakhjavani, Maryam, Jennifer E Hardingham, Helen M Palethorpe, Yoko Tomita, Eric Smith, Tim J Price, and Amanda R Townsend. 2019. "Ginsenoside Rg3: Potential Molecular Targets and Therapeutic Indication in Metastatic Breast Cancer" Medicines 6, no. 1: 17. https://doi.org/10.3390/medicines6010017
APA StyleNakhjavani, M., Hardingham, J. E., Palethorpe, H. M., Tomita, Y., Smith, E., Price, T. J., & Townsend, A. R. (2019). Ginsenoside Rg3: Potential Molecular Targets and Therapeutic Indication in Metastatic Breast Cancer. Medicines, 6(1), 17. https://doi.org/10.3390/medicines6010017






