Pediatric Thermal Burns and Treatment: A Review of Progress and Future Prospects
Abstract
:1. Introduction
2. Background
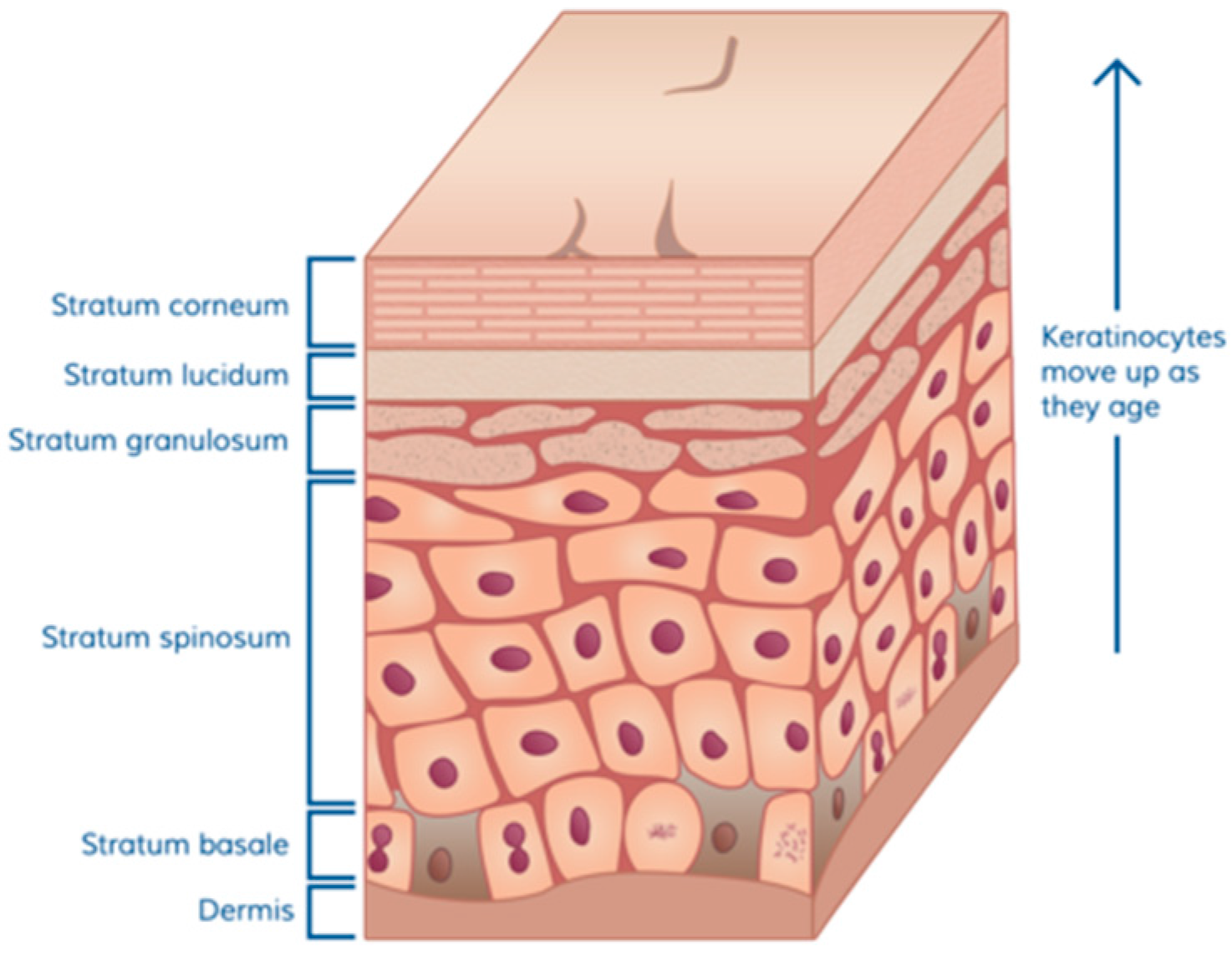
2.1. Skin Function
2.2. Overview of Pathophysiology of Burn Wounds in Pediatric Patients
3. Current Management of Complex Skin Defects—Autograft
4. Clinical Need and Rationale
5. Summary of Currently Approved Products
6. Conclusions and Future Prospects
Author Contributions
Conflicts of Interest
References
- National Burn Repository Report of Data 2006–2015; Version 12.0; American Burn Association: Chicago, IL, USA, 2016; Available online: http://alt.ameriburn.org/2016ABANBR_FINAL_42816.pdf (accessed on 14 September 2017).
- Soleimani, T.; Evans, T.A.; Sood, R.; Hartman, B.C.; Hadad, I.; Tholpady, S.S. Pediatric burns: Kids’ Inpatient Database vs. the National Burn Repository. J. Surg. Res. 2016, 201, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Child Safety and Injury Prevention: Burn Prevention; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2016. Available online: https://www.cdc.gov/safechild/burns/index.html (accessed on 8 August 2017).
- Centers for Disease Control and Prevention. Web-Based Injury Statistics Query and Reporting System (WISQARS™) [Online]; National Center for Injury Prevention and Control, Centers for Disease Control and Prevention (Producer): Atlanta, GA, USA, 2003. Available online: https://www.cdc.gov/injury/wisqars/index.html (accessed on 9 August 2017).
- National Safety Council. Injury Facts; National Safety Council: Itasca, IL, USA, 2017; pp. 32–143. [Google Scholar]
- Lee, J.; Norbury, W.; Herndon, D.N. Special Considerations of age: The pediatric burned patient. In Total Burn Care, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 405–414. [Google Scholar]
- A National Action Plan for Child Injury Prevention: Reducing Fire and Burn Injuries; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2016. Available online: https://www.cdc.gov/safechild/pdf/nap_burns_injuries-a.pdf (accessed on 9 August 2017).
- Krishnamoorthy, V.; Ramaiah, R.; Bhananker, S.M. Pediatric burn injuries. Int. J. Crit. Illn. Inj. Sci. 2012, 2, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn Wound Infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.; Balaji, S.; Keswani, S.G. Biology and function of fetal and pediatric skin. Facial Plast. Surg. Clin. N. Am. 2013, 21, 1–6. [Google Scholar] [CrossRef]
- Layton, D. Curious to learn more about the layers of skin? The Stratum Corneum: Protector of Your Internal Organs. Skin Sci. Blogs 2016. Available online: https://skinsaverapp.com/blog/2016/8/25/the-stratum-corneum-protector-of-your-internal-organs (accessed on 26 August 2017).
- Maibach, H.I. Evidence Based Dermatology, 2nd ed.; People’s Media Publishing House: Shelton, CT, USA, 2011; p. 998. ISBN 978-1-60795-039-4. [Google Scholar]
- Blanpain, C.; Fuchs, E. Epidermal homeostasis: A balancing act of stem cells in the skin. Nat. Rev. Mol. Cell Biol. 2009, 10, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef] [PubMed]
- Stamatas, G.N.; Nikolovski, J.; Luedtke, M.A.; Kollias, N.; Wiegand, B.C. Infant skin microstructure assessed in vivo differs from adult skin in organization at the cellular level. Pediatr. Dermatol. 2010, 27, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Derraik, J.G.B.; Rademaker, M.; Cutfield, W.S.; Pinto, T.E.; Tregurtha, S.; Faherty, A.; Peart, J.M.; Drury, P.L.; Hofman, P.L. Effects of age, gender, BMI, and anatomical site on skin thickness in children and adults with diabetes. PLoS ONE 2014, 9, E86637. [Google Scholar] [CrossRef] [PubMed]
- Fenlon, S.; Nene, S. Burns in children. Contin. Educ. Anaesth. Crit. Care Pain 2007, 7, 76–80. [Google Scholar] [CrossRef]
- Vallez, L.J.; Plourde, B.D.; Wentz, J.E.; Nelson-Cheeseman, B.B.; Abraham, J.P. A Review of Scald Burn Injuries. Intern. Med. Rev. 2017, 3, 1–18. [Google Scholar]
- Sharma, R.K.; Parashar, A. Special considerations in paediatric burn patients. Indian J. Plast. Surg. 2010, 43, S43–S50. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, J.A.; Schraga, E.D. Emergent management of thermal burns. Medscape 2014. Available online: http://emedicine.medscape.com/article/769193-overview (accessed on 10 July 2015).
- Herndon, D.N. Total Burn Care, 3rd ed.; Saunders Elsevier: Chatswood, New South Wales, Australia, 2007; ISBN 9781416032748. [Google Scholar]
- Kagan, R.J.; Peck, M.D.; Ahrenholz, D.H.; Hickerson, W.L.; Holmes, J.H.; Korentager, R.A.; Kraatz, J.J.; Kotoski, G.M. White Paper: Surgical Management of the Burn Wound and Use of Skin Substitutes; American Burn Association: Chicago, IL, USA, 2009; pp. 1–44. [Google Scholar]
- Wolf, S.E.; Herndon, D.N. Burn Care; Landes Bioscience: Austin, TX, USA, 1999. [Google Scholar]
- Shah, A.R.; Liao, L.F. Pediatric burn care: Unique considerations in management. Clin. Plast. Surg. 2017, 44, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Ohgi, S.; Gu, S. Pediatric burn rehabilitation: Philosophy and strategies. Burns Trauma 2013, 1, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Birchall, M.A.; Varma, S.; Milward, T.M. The Moriarty sign: An appraisal. Br. J. Plast. Surg. 1991, 44, 149–150. [Google Scholar] [CrossRef]
- Bayat, A.; Ramaiah, R.; Bhananker, S.M. Analgesia and sedation for children undergoing burn wound care. Exp. Rev. Neurother. 2011, 10, 1747–1759. [Google Scholar] [CrossRef] [PubMed]
- Fuzaylov, G.; Fidkowski, C.W. Anesthetic considerations for major burn injury in pediatric patients. Paediatr. Anaesth. 2009, 19, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Burn Incidence and Treatment in the United States. American Burn Association Web Site. 2016. Available online: http://www.ameriburn.org/who-we-are/media/burn-incidence-fact-sheet/ (accessed on 7 July 2017).
- CDC Injury Prevention. Burns; Department of Health and Human Services: Washington, DC, USA. Available online: http://www.cdc.gov/masstrauma/factsheets/public/burns.pdf (accessed on 7 July 2017).
- Akan, M.; Misirlioglu, A.; Yildirim, S.; Cakir, B.; Taylan, G.; Akoz, T. Ice application to minimize pain in the split-thickness skin graft donor site. Aesthet. Plast. Surg. 2003, 27, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Janzekovic, Z. Once upon a time how west discovered east. J. Plast. Reconstr. Aesthet. Surg. 2008, 61, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Pruitt, B.A., Jr.; Levine, N.S. Characteristics and uses of biologic dressings and skin substitutes. Arch. Surg. 1984, 119, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, I.S.; Prowse, S.; Potokar, T.S. A critical evaluation of the use of Biobrane as a biologic skin substitute: A versatile tool for the plastic and reconstructive surgeon. Ann. Plast. Surg. 2008, 60, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Lesher, A.P.; Curry, R.H.; Evans, J.; Smith, V.A.; Fitzgerald, M.T.; Cina, R.A.; Streck, C.J.; Hebra, A.V. Effectiveness of Biobrane for treatment of partial-thickness burns in children. J. Pediatr. Surg. 2011, 46, 1759–1763. [Google Scholar] [CrossRef] [PubMed]
- Barret, J.P.; Dziewulski, P.; Ramzy, P.I.; Wolf, S.E.; Desai, M.H.; Herndon, D.N. Biobrane versus 1% silver sulfadiazine in second-degree pediatric burns. Plast. Reconstr. Surg. 2000, 105, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Branski, L.K.; Herndon, D.N.; Pereira, C.; Mlcak, R.P.; Celis, M.M.; Lee, J.O.; Sanford, A.P.; Norbury, W.B.; Zhang, X.J.; Jeschke, M.G. Longitudinal assessment of Integra in primary burn management: A randomized pediatric clinical trial. Crit. Care Med. 2007, 35, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Ghazi, B.H.; Williams, J.K. Use of Integra in complex pediatric wounds. Ann. Plast. Surg. 2011, 66, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Shahrokhi, S.; Anna, A.; Jeschke, M.G. The Use of Dermal Substitutes in Burn Surgery: Acute Phase. Wound Repair Regen. 2014, 22. [Google Scholar] [CrossRef] [PubMed]
- Rennert, R.C.; Rodrigues, M.; Wong, V.W.; Duscher, D.; Hu, M.; Maan, Z.; Sorkin, M.; Gurtner, G.C.; Longaker, M.T. Biological therapies for the treatment of cutaneous wounds: Phase III and launched therapies. Expert Opin. Biol. Ther. 2013, 13, 1523–1541. [Google Scholar] [CrossRef] [PubMed]
- Parcells, A.L.; Karcich, J.; Granick, M.S.; Marano, M.A. The Use of Fetal Bovine Dermal Scaffold (PriMatrix) in the Management of Full-Thickness Hand Burns. Eplasty 2014, 14, e36. [Google Scholar] [PubMed]
- Rennert, R.C.; Sorkin, M.; Garg, R.K.; Januszyk, M.; Gurtner, G.C. Cellular response to a novel fetal acellular collagen matrix: Implications for tissue regeneration. Int. J. Biomater. 2013, 2013, 527–957. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. Recall of Human Tissue Products-LifeCell Corp 30 September 2005; Alloderm Recall; Food and Drug Administration: Silver Spring, MD, USA, 2005. Available online: http://www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/Recalls/ucm053666.htm (accessed on 4 August 2017).
- Food and Drug Administration. Enforcement Report for 2 April 2008; Alloderm Recall; Food and Drug Administration: Silver Spring, MD, USA, 2008. Available online: http://www.fda.gov/Safety/Recalls/EnforcementReports/2008/ucm120500.htm (accessed on 15 August 2017).
- Food and Drug Administration. Enforcement Report for 16 April 2008; Alloderm Recall; Food and Drug Administration: Silver Spring, MD, USA, 2008. Available online: http://www.fda.gov/Safety/Recalls/EnforcementReports/2008/ucm120502.htm (accessed on 15 August 2017).
- Wainwright, D.; Madden, M.; Luterman, A.; Hunt, J.; Monafo, W.; Heimbach, D.; Kagan, R.; Sittig, K.; Dimick, A.; Herndon, D. Clinical evaluation of an acellular allograft dermal matrix in fullthickness burns. J. Burn Care Rehabil. 1996, 17, 124–136. [Google Scholar] [CrossRef] [PubMed]
- Lukish, J.R.; Eichelberger, M.R.; Newman, K.D.; Pao, M.; Nobuhara, K.; Keating, M.; Golonka, N.; Pratsch, G.; Misra, V.; Valladares, E.; et al. The use of a bioactive skin substitute decreases length of stay for pediatric burn patients. J. Pediatr. Surg. 2001, 36, 1118–1121. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.J.; Kimble, R.M.; Boots, R.; Pegg, S.P. Treatment of partial-thickness burns: A prospective, randomized trial using Transcyte. ANZ J. Surg. 2004, 74, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Rab, M.; Koller, R.; Ruzicka, M.; Burda, G.; Kamolz, L.P.; Bierochs, B.; Meissl, G.; Frey, M. Should dermal scald burns in children be covered with autologous skin grafts or with allogeneic cultivated keratinocytes—“The Viennese concept”. Burns 2005, 31, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Hickerson, W.; McKeen, T.; Fisher, J.; van Bemden, B. Cultured Epidermal Autografts (CEA) for Coverage of Large Burn Wounds in Pediatric and Adult Patients, 1989–2015. In Proceedings of the 2017 Annual Meeting of the American Burn Association, Philadelphia, PA, USA, 23 March 2017; Available online: http://www.abstractsonline.com/pp8/#!/4303/presentation/88 (accessed on 20 September 2017).
- Shanti, C. Pediatric Patient with Severe Burns Treated with Epicel Cultured Epidermal Autografts (CEAs): Case Study; Fall 2012 Issue; Pediatric Experts & Innovations, DMC Children’s Hospital of Michigan: Detroit, MI, USA, 2012; Available online: http://www.childrensdmc.org/upload/docs/Newsletter/ExpertsAndInnovations/CHM-PediatricExpertsInnovations-Fall2012-Web.pdf (accessed on 9 July 2015).
- Food and Drug Administration. Executive Summary, Epicel (Cultured Epidermal Autografts) HDE# BH990200: Prepared for the 7 March 2017 Meeting of the FDA Pediatric Advisory Committee, 2017; FDA Executive Summary; Food and Drug Administration: Silver Spring, MD, USA, 2017. Available online: https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/PediatricAdvisoryCommittee/UCM543865.pdf (accessed on 11 July 2017).
- Boyce, S.T.; Simpson, P.S.; Rieman, M.T.; Warner, P.M.; Yakuboff, K.P.; Bailey, J.K.; Nelson, J.K.; Fowler, L.A.; Kagan, R.J. Randomized, paired-site comparison of autologous engineered skin substitutes and split-thickness skin graft for closure of extensive, full-thickness burns. J. Burn Care Res. 2017, 38, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Eisenbud, D.; Huang, N.F.; Luke, S.; Silberklang, M. Skin substitutes and wound healing: Current status and challenges. Wounds 2004, 16, 2–17. [Google Scholar]
- Parenteau, N.L.; Bilbo, P.; Nolte, C.J.; Mason, V.S.; Rosenberg, M. The organotypic culture of human skin keratinocytes and fibroblasts to achieve form and function. Cytotechnology 1992, 9, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Falabella, A.F.; Valencia, I.C.; Eaglstein, W.H. Tissue-engineered skin (Apligraf) in the healing of patients with epidermolysis bullosa wounds. Arch. Dermatol. 2000, 136, 1225–1230. [Google Scholar] [CrossRef] [PubMed]
- Transplant and Tissue Engineering; Novartis: East Hanover, NJ, USA, 2002; Available online: https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/n_int_050273.pdf (accessed on 1 September 2017).
- Waymack, P.; Duff, R.G.; Sabolinski, M. The effect of a tissue engineered bilayered living skin analog, over meshed split-thickness autografts on the healing of excised burn wounds. The Apligraf Burn Study Group. Burns 2000, 26, 609–619. [Google Scholar] [CrossRef]
- Hayes, D.W., Jr.; Webb, G.E.; Mandracchia, V.J.; John, K.J. Full-thickness burn of the foot: Successful treatment with Apligraf. A case report. Clin. Podiatr. Med. Surg. 2001, 18, 179–188. [Google Scholar] [PubMed]
- Ehrenreich, M.; Ruszczak, Z. Update on tissue-engineered biological dressings. Tissue Eng. 2006, 12, 2407–2424. [Google Scholar] [CrossRef] [PubMed]
- Apligraf; Organogensis Inc.: Canton, MA, USA, 2011. Available online: http://www.apligraf.com/professional/pdf/APG_Factsheet.pdf (accessed on 9 September 2017).
- Schurr, M.J.; Foster, K.N.; Centanni, J.M.; Comer, A.R.; Wicks, A.; Gibson, A.L.; Thomas-Virnig, C.L.; Schlosser, S.J.; Faucher, L.D.; Lokuta, M.A.; et al. Phase I/II clinical evaluation of StrataGraft: A consistent, pathogen-free human skin substitute. J. Trauma 2009, 66, 866–873. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mathias, E.; Srinivas Murthy, M. Pediatric Thermal Burns and Treatment: A Review of Progress and Future Prospects. Medicines 2017, 4, 91. https://doi.org/10.3390/medicines4040091
Mathias E, Srinivas Murthy M. Pediatric Thermal Burns and Treatment: A Review of Progress and Future Prospects. Medicines. 2017; 4(4):91. https://doi.org/10.3390/medicines4040091
Chicago/Turabian StyleMathias, Elton, and Madhu Srinivas Murthy. 2017. "Pediatric Thermal Burns and Treatment: A Review of Progress and Future Prospects" Medicines 4, no. 4: 91. https://doi.org/10.3390/medicines4040091





