A Radical-Free Approach to Teeth Whitening
Abstract
:1. Introduction
2. Materials and Methods
2.1. Enamel Erosion and Hardness Tests
2.2. Bleaching Effectiveness In Vitro
3. Results
3.1. Enamel Erosion and Hardness Tests
3.2. Bleaching Effectiveness In Vitro
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fearon, J. Tooth whitening: Concepts and controversies. J. Ir. Dent. Assoc. 2007, 53, 132–140. [Google Scholar] [PubMed]
- Viscio, D.; Gaffar, A.; Fakhry-Smith, S.; Xu, T. Present and future technologies of tooth whitening. Compend. Contin. Educ. Dent. Suppl. 2000, 28, S36–S43. [Google Scholar]
- Rodríguez-Martínez, J.; Valiente, M.; Sánchez-Martín, M.J. Tooth whitening: From the established treatments to novel approaches to prevent side effects. J. Esthet. Rest. Dent. 2019, 31, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Tredwin, C.J.; Naik, S.; Lewis, N.J.; Scully, C. Hydrogen peroxide tooth-whitening (bleaching) products: Review of adverse effects and safety issues. Br. Dent. J. 2006, 200, 371–376. [Google Scholar] [CrossRef] [Green Version]
- Sulieman, M.A.M. An overview of tooth-bleaching techniques: Chemistry, safety and efficacy. Periodontology 2000 2008, 48, 148–169. [Google Scholar] [CrossRef]
- Briso, A.L.F.; Rahal, V.; Gallinari, M.O.; Soares, D.G.; de Souza Costa, C.A. Complications from the use of peroxides. In Tooth Whitening: An Evidence-Based Perspective, 1st ed.; Perdigão, J., Ed.; Springer: Cham, Switzerland, 2016; pp. 45–79. [Google Scholar]
- Jurema, A.L.B.; de Souza, M.Y.; Torres, C.R.G.; Borges, A.B.; Caneppele, T.M.F. Effect of pH on whitening efficacy of 35% hydrogen peroxide and enamel microhardness. J. Esthet. Rest. Dent. 2018, 30, E39–E44. [Google Scholar] [CrossRef]
- Gouveia, T.H.N.; de Souza, D.F.S.; Aguiar, F.H.B.; Ambrosano, G.M.B.; Lima, D.A.N.L. Effect of ammonium acryloyldimethyltaurate copolymer on the physical and chemical properties of bleached dental enamel. Clin. Oral Investig. 2020, 24, 2701–2711. [Google Scholar] [CrossRef]
- Greenwall-Cohen, J.; Francois, P.; Silikas, N.; Greenwall, L.; Le Goff, S.; Attal, J.P. The safety and efficacy of ‘over the counter’ bleaching products in the UK. Br. Dent. J. 2019, 226, 271–276. [Google Scholar] [CrossRef]
- Watts, A.; Addy, M. Tooth discolouration and staining: A review of the literature. Br. Dent. J. 2001, 190, 309–316. [Google Scholar] [CrossRef]
- Joiner, A. The bleaching of teeth: A review of the literature. J. Dent. 2006, 34, 412–419. [Google Scholar] [CrossRef]
- Kwon, S.R.; Wertz, P.W. Review of the mechanism of tooth whitening. J. Esthet. Rest. Dent. 2015, 27, 240–257. [Google Scholar] [CrossRef]
- Bizhang, M.; Domin, J.; Danesh, G.; Zimmer, S. Effectiveness of a new non-hydrogen peroxide bleaching agent after single use—A double-blind placebo-controlled short-term study. J. Appl. Oral Sci. 2017, 25, 575–584. [Google Scholar] [CrossRef] [Green Version]
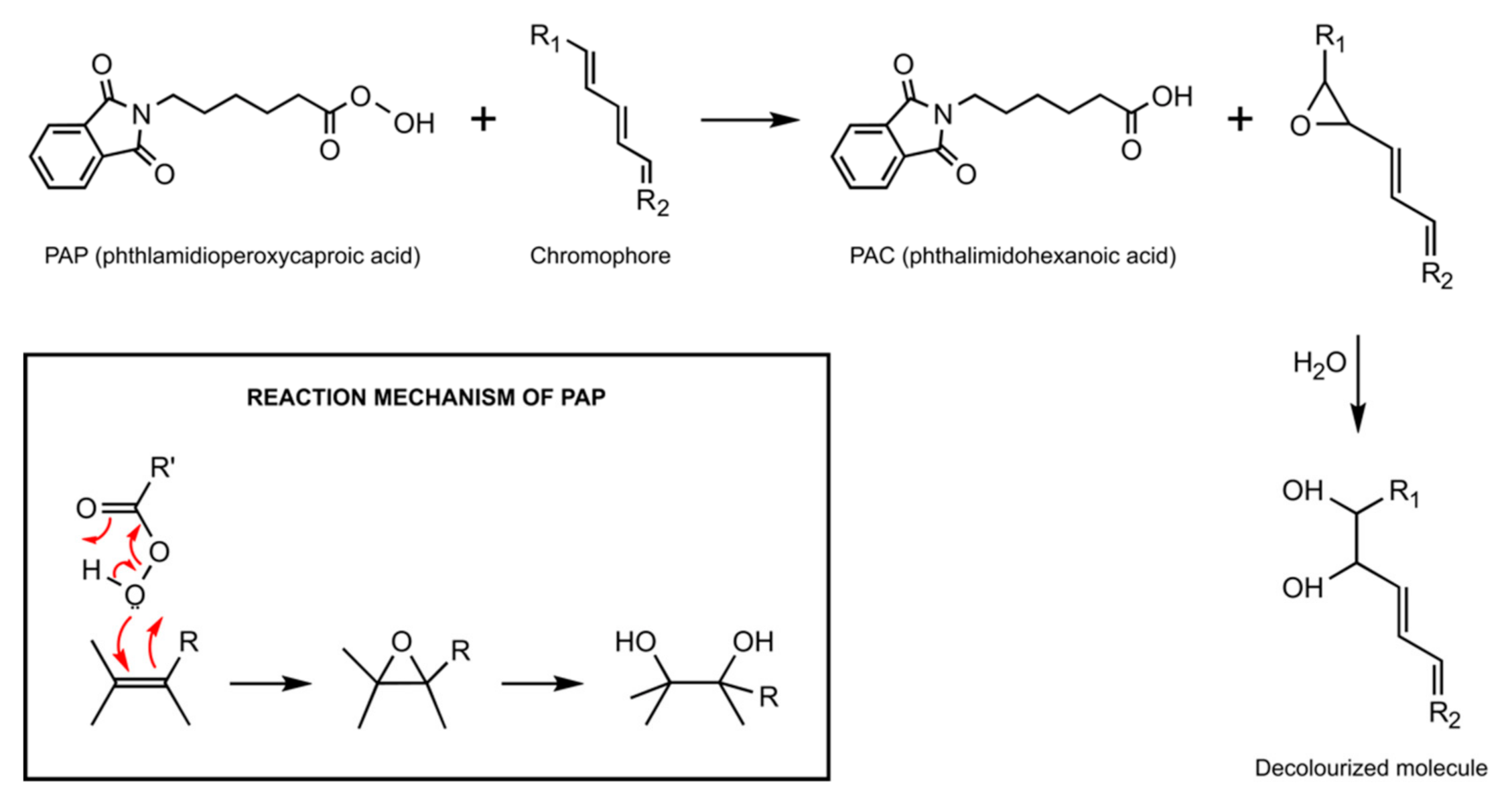
- Qin, J.; Zeng, L.; Min, W.; Tan, L.; Lv, M.; Chen, Y. A bio-safety tooth-whitening composite gels with novel phthalimide peroxy caproic acid. Compos. Commun. 2019, 13, 107–111. [Google Scholar] [CrossRef]
- Rodrigues, F.T.; Serro, A.P.; Polido, M.; Ramalho, A.; Figueiredo-Pina, C.G. Effect of bleaching teeth with hydrogen peroxide on the morphology, hydrophilicity, and mechanical and tribological properties of the enamel. Wear 2017, 374–375, 21–28. [Google Scholar] [CrossRef] [Green Version]
- Potočnik, I.; Kosec, L.; Gašperšič, D. Effect of 10% carbamide peroxide bleaching gel on enamel microhardness, microstructure, and mineral content. J. Endod. 2000, 26, 203–206. [Google Scholar] [CrossRef]
- Pinto, C.F.; de Oliveira, R.; Cavalli, V.; Giannini, M. Peroxide bleaching agent effects on enamel surface microhardness, roughness and morphology. Braz. Oral Res. 2004, 18, 306–311. [Google Scholar] [CrossRef]
- Redha, O.; Strange, A.; Maeva, A.; Sambrook, R.; Mordan, N.; McDonald, A.; Bozec, L. Impact of a carbamide peroxide whitening agent dentinal collagen. J. Dent. Res. 2019, 98, 443–449. [Google Scholar] [CrossRef]
- Wijetunga, C.L.; Otsuki, M.; Abdou, A.; Luong, M.N.; Qi, F.; Tagami, J. The effect of in-office bleaching materials with different pH on the surface topography of bovine enamel. Dent. Mater. J. 2021, 40, 1345–1351. [Google Scholar] [CrossRef]
- Ebadifar, A.; Nomani, M.; Fatemi, S.A. Effect of nano-hydroxyapatite toothpaste on microhardness ofartificial carious lesions created on extracted teeth. J. Dent. Res. Dent. Clin. Dent. Prospect. 2017, 11, 14–17. [Google Scholar] [CrossRef] [Green Version]
- Esteves-Oliveira, M.; Santos, N.M.; Meyer-Lueckel, H.; Wierichs, R.J.; Rodrigues, J.A. Caries-preventing effect of anti-erosivive and nano-hydroxyapatite-containing toothpaste in vitro. Clin. Oral Investig. 2017, 21, 291–300. [Google Scholar] [CrossRef]
- Sudradjat, H.; Meyer, F.; Loza, K.; Epple, M.; Enax, J. In Vivo Effects of a Hydroxyapatite-Based Oral Care Gel on the Calcium and Phosphorus Levels of Dental Plaque. Eur. J. Dent. 2020, 14, 206–211. [Google Scholar] [CrossRef] [Green Version]

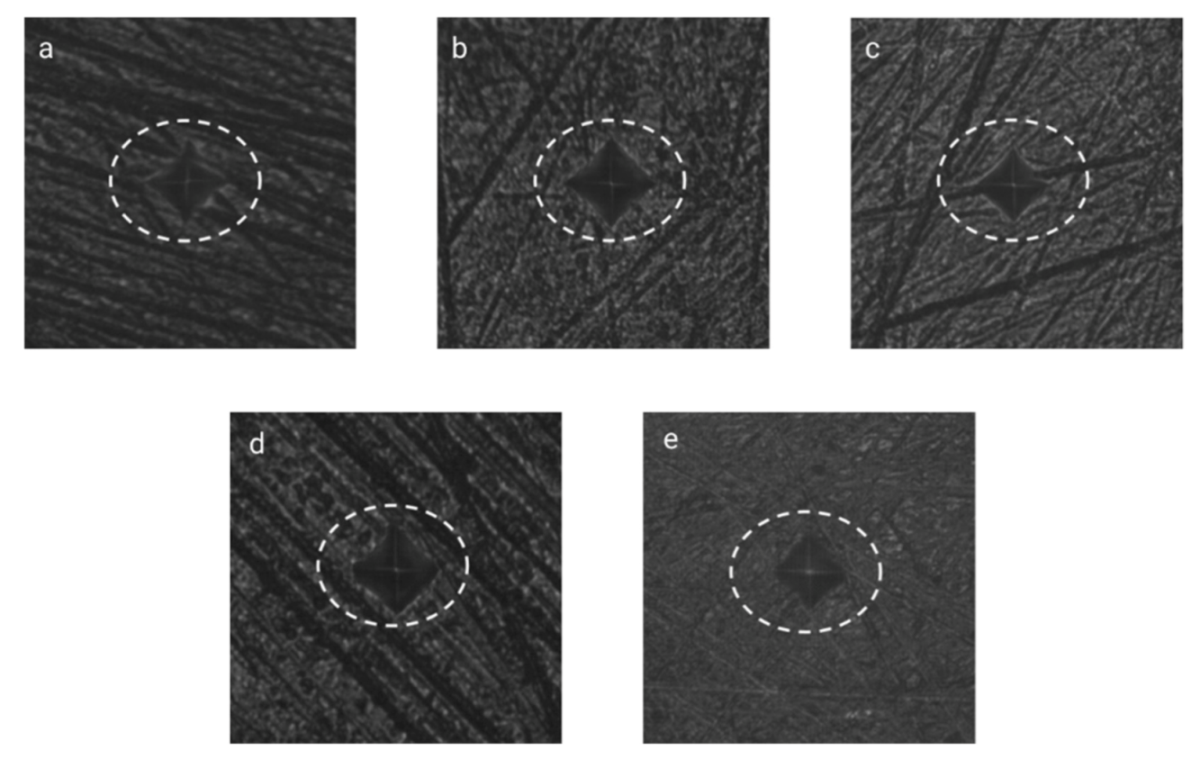

| Treatment | Enamel Erosion (mm) | Change in SMH | Mean Enamel Erosion (mm) | Mean Change in SMH | SMH SD |
|---|---|---|---|---|---|
| 6% HP | 0.183 | −42.67 | 0.114 | −62.22 | 19.52 |
| 0.00 | −54.33 | ||||
| 0.115 | −45.00 | ||||
| 0.140 | −68.67 | ||||
| 0.00 | −67.33 | ||||
| 0.245 | −95.33 | ||||
| 35% HP | 0.149 | −97.00 | 0.097 | −94.28 | 27.09 |
| 0.121 | −75.00 | ||||
| 0.00 | −80.67 | ||||
| 0.124 | −147.33 | ||||
| 0.186 | −79.00 | ||||
| 0.00 | −86.67 | ||||
| 35% CP | 0.00 | −65.7 | 0.00 | −55.3 | 24.6 |
| 0.00 | −100.7 | ||||
| 0.00 | −47.3 | ||||
| 0.00 | −35.0 | ||||
| 0.00 | −39.3 | ||||
| 0.00 | −43.7 | ||||
| PAP+ | 0.00 | −7.0 | 0.00 | 12.9 | 11.7 |
| 0.00 | 14 | ||||
| 0.00 | 7.3 | ||||
| 0.00 | 23.0 | ||||
| 0.00 | 25.0 | ||||
| 0.00 | 15.3 |
| Treatment | Timepoint | VITA Bleached Guide Shades Changes | SD |
|---|---|---|---|
| 6% HP | 1st treatment | 1.97 | 0.76 |
| PAP+ | 3.86 | 1.41 | |
| 6% HP | 2nd treatment | 2.82 | 1.09 |
| PAP+ | 5.79 | 2.20 | |
| 6% HP | 3rd treatment | 3.95 | 1.50 |
| PAP+ | 6.59 | 2.13 | |
| 6% HP | 4th treatment | 4.44 | 2.03 |
| PAP+ | 6.59 | 2.46 | |
| 6% HP | 5th treatment | 4.44 | 2.58 |
| PAP+ | 7.32 | 2.79 | |
| 6% HP | 6th treatment | 4.86 | 2.32 |
| PAP+ | 8.13 | 2.82 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pascolutti, M.; de Oliveira, D. A Radical-Free Approach to Teeth Whitening. Dent. J. 2021, 9, 148. https://doi.org/10.3390/dj9120148
Pascolutti M, de Oliveira D. A Radical-Free Approach to Teeth Whitening. Dentistry Journal. 2021; 9(12):148. https://doi.org/10.3390/dj9120148
Chicago/Turabian StylePascolutti, Mauro, and Dileusa de Oliveira. 2021. "A Radical-Free Approach to Teeth Whitening" Dentistry Journal 9, no. 12: 148. https://doi.org/10.3390/dj9120148
APA StylePascolutti, M., & de Oliveira, D. (2021). A Radical-Free Approach to Teeth Whitening. Dentistry Journal, 9(12), 148. https://doi.org/10.3390/dj9120148






