Bacterial Colonization and Tissue Compatibility of Denture Base Resins
Abstract
1. Introduction
2. Materials and Methods
3. Microbiological Assessments
4. Cytology Assessments
5. Results
5.1. Microbiological Results
5.2. Cytology Results
6. Discussion
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Aas, J.A.; Paster, B.J.; Stokes, L.N.; Olsen, I.; Dewhirst, F.E. Defining the normal bacterial flora of the oral cavity. J. Clin. Microbiol. 2005, 43, 5721–5732. [Google Scholar] [CrossRef] [PubMed]
- Sachdeo, A.; Haffajee, A.D.; Socransky, S.S. Biofilms in the edentulous oral cavity. J. Prosthodont. 2008, 17, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Marsh, P.; Lewis, M.; Rogers, H.; Williams, D.; Wilson, M. Oral Microbiology, 6th ed.; Elsevier, Churchill Livingstone: London, UK, 2016. [Google Scholar]
- Majchrzak, K.; Mierzwinska-Nastalska, E.; Chmura, A.; Kwiatkowski, A.; Paczek, L.; Mlynarczyk, G.; Szymanek-Majchrzak, K. Clinical and Microbiological Evaluation of Removable Prosthetic Restorations in Patients Treated with an Organ Transplant. Transplant. Proc. 2016, 48, 1418–1422. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, L.E.; Smith, K.; Williams, C.; Nile, C.J.; Lappin, D.F.; Bradshaw, D.; Lambert, M.; Robertson, D.P.; Bagg, J.; Hannah, V.; et al. Dentures are a Reservoir for Respiratory Pathogens. J. Prosthodont. 2016, 25, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Gendreau, L.; Loewy, Z. Epidemiology and etiology of denture stomatitis. J. Prosthodont. 2011, 20, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Marinoski, J.; Bokor-Bratic, M.; Cankovic, M. Is denture stomatitis always related with candida infection? A case control study. Med. Glas. 2014, 11, 379–384. [Google Scholar]
- Pietrokovski, J.; Azuelos, J.; Tau, S.; Mostavoy, R. Oral findings in elderly nursing home residents in selected countries: Oral hygiene conditions and plaque accumulation on denture surfaces. J. Prosthet. Dent. 1995, 73, 136–141. [Google Scholar] [CrossRef]
- Pietrokovski, J.; Harfin, J.; Mostavoy, R.; Levy, F. Oral findings in elderly nursing home residents in selected countries: Quality of and satisfaction with complete dentures. J. Prosthet. Dent. 1995, 73, 132–135. [Google Scholar] [CrossRef]
- Kawara, M.; Iwata, Y.; Iwasaki, M.; Komoda, Y.; Iida, T.; Asano, T.; Komiyama, O. Scratch test of thermoplastic denture base resins for non-metal clasp dentures. J. Prosthodont. Res. 2014, 58, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Hahnel, S.; Schwarz, S.; Zeman, F.; Schafer, L.; Behr, M. Prevalence of xerostomia and hyposalivation and their association with quality of life in elderly patients in dependence on dental status and prosthetic rehabilitation: A pilot study. J. Dent. 2014, 42, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, W.; Schimmel, M.; Muller, F. Oral health and dental care of elderly adults dependent on care. Swiss Dent. J. 2015, 125, 417–426. [Google Scholar] [PubMed]
- Bremer, F.; Grade, S.; Kohorst, P.; Stiesch, M. In vivo biofilm formation on different dental ceramics. Quintessence Int. 2011, 42, 565–574. [Google Scholar] [PubMed]
- Dittmer, M.P.; Hellemann, C.F.; Grade, S.; Heuer, W.; Stiesch, M.; Schwestka-Polly, R.; Demling, A.P. Comparative three-dimensional analysis of initial biofilm formation on three orthodontic bracket materials. Head Face Med. 2015, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Fuchslocher Hellemann, C.; Grade, S.; Heuer, W.; Dittmer, M.P.; Stiesch, M.; Schwestka-Polly, R.; Demling, A.P. Three-dimensional analysis of initial biofilm formation on polytetrafluoroethylene in the oral cavity. J. Orofac. Orthop. 2013, 74, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Heuer, W.; Stiesch, M.; Abraham, W.R. Microbial diversity of supra- and subgingival biofilms on freshly colonized titanium implant abutments in the human mouth. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Heuer, W.; Kettenring, A.; Demling, A.; Stumpp, S.N.; Gellermann, E.; Winkel, A.; Stiesch, M. Microbial diversity of peri-implant biofilms on implant fixed bar and telescopic double crown attachments. J. Oral Implantol. 2013, 39, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, D.; David, A.; Inquimbert, C.; Tramini, P.; Molinari, N.; Carrouel, F. Quantification of carious pathogens in the interdental microbiota of young caries-free adults. PLoS ONE 2017, 12, e0185804. [Google Scholar] [CrossRef] [PubMed]
- Klinke, T.; Guggenheim, B.; Klimm, W.; Thurnheer, T. Dental caries in rats associated with Candida albicans. Caries Res. 2011, 45, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Moalic, E.; Gestalin, A.; Quinio, D.; Gest, P.E.; Zerilli, A.; Le Flohic, A.M. The extent of oral fungal flora in 353 students and possible relationships with dental caries. Caries Res. 2001, 35, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Zaremba, M.L.; Daniluk, T.; Rozkiewicz, D.; Cylwik-Rokicka, D.; Kierklo, A.; Tokajuk, G.; Dabrowska, E.; Pawinska, M.; Klimiuk, A.; Stokowska, W.; et al. Incidence rate of Candida species in the oral cavity of middle-aged and elderly subjects. Adv. Med. Sci. 2006, 51 (Suppl. 1), 233–236. [Google Scholar] [PubMed]
- Bloching, M.; Reich, W.; Schubert, J.; Grummt, T.; Sandner, A. Micronucleus rate of buccal mucosal epithelial cells in relation to oral hygiene and dental factors. Oral Oncol. 2008, 44, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Kirsch-Volders, M.; Sofuni, T.; Aardema, M.; Albertini, S.; Eastmond, D.; Fenech, M.; Ishidate, M., Jr.; Kirchner, S.; Lorge, E.; Morita, T.; et al. Report from the in vitro micronucleus assay working group. Mutat. Res. 2003, 540, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Tadin, A.; Galic, N.; Marovic, D.; Gavic, L.; Klaric, E.; Pejda, S.; Ugrin, K.; Zeljzic, D. Cytogenetic damage in exfoliated oral buccal cells by dental composites. Am. J. Dent. 2016, 29, 219–222. [Google Scholar] [PubMed]
- Bigatti, M.P.; Lamberti, L.; Rizzi, F.P.; Cannas, M.; Allasia, G. In vitro micronucleus induction by polymethyl methacrylate bone cement in cultured human lymphocytes. Mutat. Res. 1994, 321, 133–137. [Google Scholar] [CrossRef]
- Rickman, L.J.; Padipatvuthikul, P.; Satterthwaite, J.D. Contemporary denture base resins: Part 1. Dent. Update 2012, 39, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Aeran, H.; Kumar, N.; Gupta, N. Flexible thermoplastic denture base materials for aesthetical removable partial denture framework. J. Clin. Diagn. Res. 2013, 7, 2372–2373. [Google Scholar] [CrossRef] [PubMed]
- Abuzar, M.A.; Bellur, S.; Duong, N.; Kim, B.B.; Lu, P.; Palfreyman, N.; Surendran, D.; Tran, V.T. Evaluating surface roughness of a polyamide denture base material in comparison with poly(methy methacrylate). J. Oral Sci. 2010, 52, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Kedjarune, U.; Charoenworaluk, N.; Koontongkaew, S. Release of methyl methacrylate from heat-cured and autopolymerized resins: Cytotoxicity testing related to residual monomer. Aust. Dent. J. 1999, 44, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Yu, W.-H.; Izard, J.; Baranova, O.V.; Lakshmanan, A.; Dewhirst, F.E. The Human Oral Microbiome Database: A web accessible resource for investigating oral microbe taxonomic and genomic information. Database (Oxford) 2010, baq013. [Google Scholar] [CrossRef] [PubMed]
- Remmerbach, T.W.; Pomjanski, N.; Bauer, U.; Neumann, H. Liquid-based versus conventional cytology of oral brush biopsies: A split-sample pilot study. Clin. Oral Investig. 2017, 21, 2493–2498. [Google Scholar] [CrossRef] [PubMed]
- Samanta, S.; Dey, P. Micronucleus and its applications. Diagn. Cytopathol. 2012, 40, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Belien, J.A.M.; Copper, M.P.; Braakhuis, B.J.M. Standardization of counting micronuclei: Definition of a protocol to measure genotoxic damage in human exfoliated cells. Carcinogenesis 1995, 16, 2395–2400. [Google Scholar] [CrossRef] [PubMed]
- Ambroise, M.M.; Balasundaram, K.; Phansalkar, M. Predictive value of micronucleus count in cervical intraepithelial neoplasia and carcinoma. Turk Patoloji Derg. 2013, 29, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Grover, S.; Mujib, A.B.R.; Jahagirdar, A.; Telagi, N.; Kulkarni, P.G. A comparative study for selectivity of micronuclei in oral exfoliated epithelial cells. J. Cytol. 2012, 29, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Ayyad, S.B.A.; Israel, E.; El-Setouhy, M.; Nasr, G.R.; Mohamed, M.K.; Loffredo, C.A. Evaluation of Papanicolaou Stain for Studying Micronuclei in Buccal Cells Under Field Conditions. Acta Cytol. 2006, 50, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.M.D.; Alves, G.R.; Avanço, G.T.; Parizi, J.L.S.; Nai, G.A. Assessment of methyl methacrylate genotoxicity by the micronucleus test. Braz. Oral Res. 2013, 27, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, R. (Ed.) Oral Cytology. A Concise Guide; The Development of a Novel Oral Cytologic Grading System; Springer: New York, NY, USA; Heidelberg, Germany; Dordrecht, The Netherlands; London, UK, 2012; pp. 73–90. [Google Scholar]
- Groß, G.; Olms, C. Einsatz einer intraoralen Testmethode zur Biokompatibilitätsprüfung von zahnärztlichen Materialien—eine Pilotstudie. 13. Research Festival for Life Science. 2014. Available online: http://resfest.uniklinikum-leipzig.de/docs/2014_Abstract_Band.pdf (accessed on 13 June 2018).
- Lindel, I.D.; Elter, C.; Heuer, W.; Heidenblut, T.; Stiesch, M.; Schwestka-Polly, R.; Demling, A.P. Comparative analysis of long-term biofilm formation on metal and ceramic brackets. Angle Orthod. 2011, 81, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Olms, C.; Hix, N.; Neumann, H.; Yahiaoui-Doktor, M.; RemmCach, T.W. Clinical comparison of liquid-based and conventional cytology of oral brush biopsies: A randomized controlled trial. Head Face Med. 2018, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Hayama, F.H.; Motta, A.C.F.; Silva, P.A.; Migliari, D.A. Liquid-based preparations versus conventional cytology: Specimen adequacy and diagnostic agreement in oral lesions. Med. Oral Patol. Oral Cir. Bucal 2005, 10, 115–122. [Google Scholar] [PubMed]
- Kujan, O.; Desai, M.; Sargent, A.; Bailey, A.; Turner, A.; Sloan, P. Potential applications of oral brush cytology with liquid-based technology: Results from a cohort of normal oral mucosa. Oral Oncol. 2006, 42, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Goodson, M.L.; Sloan, P.; Wadehra, V.; Johnson, S.J.; Robinson, C.M.; Aubourg, G.; Thomson, P.J. Orcellex® Brush Biopsy and Liquid-Based Cytology—Assessment of a New Diagnostic Technique in Oral Potentially Malignant Disorder Management. J. Oral Maxillofac. Surg. 2014, 117, 224–231. [Google Scholar]
- Reichardt, T.; Remmerbach, T.W. Tumoren in der Mundhöhle—Zahnärztliche Aspekte der Früherkennung. Zahnmed. up2date 2016, 10, 67–83. [Google Scholar] [CrossRef]
- Gendron, R.; Grenier, D.; Maheu-Robert, L. The oral cavity as a reservoir of bacterial pathogens for focal infections. Microbes Infect. 2000, 2, 897–906. [Google Scholar] [CrossRef]
- Hahnel, S.; Rosentritt, M.; Handel, G.; Burgers, R. In vitro evaluation of artificial ageing on surface properties and early Candida albicans adhesion to prosthetic resins. J. Mater. Sci. Mater. Med. 2009, 20, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Koch, C.; Burgers, R.; Hahnel, S. Candida albicans adherence and proliferation on the surface of denture base materials. Gerodontology 2013, 30, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Vojdani, M.; Giti, R. Polyamide as a Denture Base Material: A Literature Review. J. Dent. 2015, 16, 1–9. [Google Scholar]
- Waltimo, T.; Vallittu, P.; Haapasalo, M. Adherence of Candida species to newly polymerized and water-stored denture base polymers. Int. J. Prosthodont. 2001, 14, 457–460. [Google Scholar] [PubMed]
- Susewind, S.; Lang, R.; Hahnel, S. Biofilm formation and Candida albicans morphology on the surface of denture base materials. Mycoses 2015, 58, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Freitas Fernandes, F.; Pereira-Cenci, T.; da Silva, W.J.; Filho, A.P.R.; Straioto, F.G.; Del Bel Cury, A.A. Efficacy of denture cleansers on Candida spp. biofilm formed on polyamide and polymethyl methacrylate resins. J. Prosthet. Dent. 2011, 105, 51–58. [Google Scholar] [CrossRef]
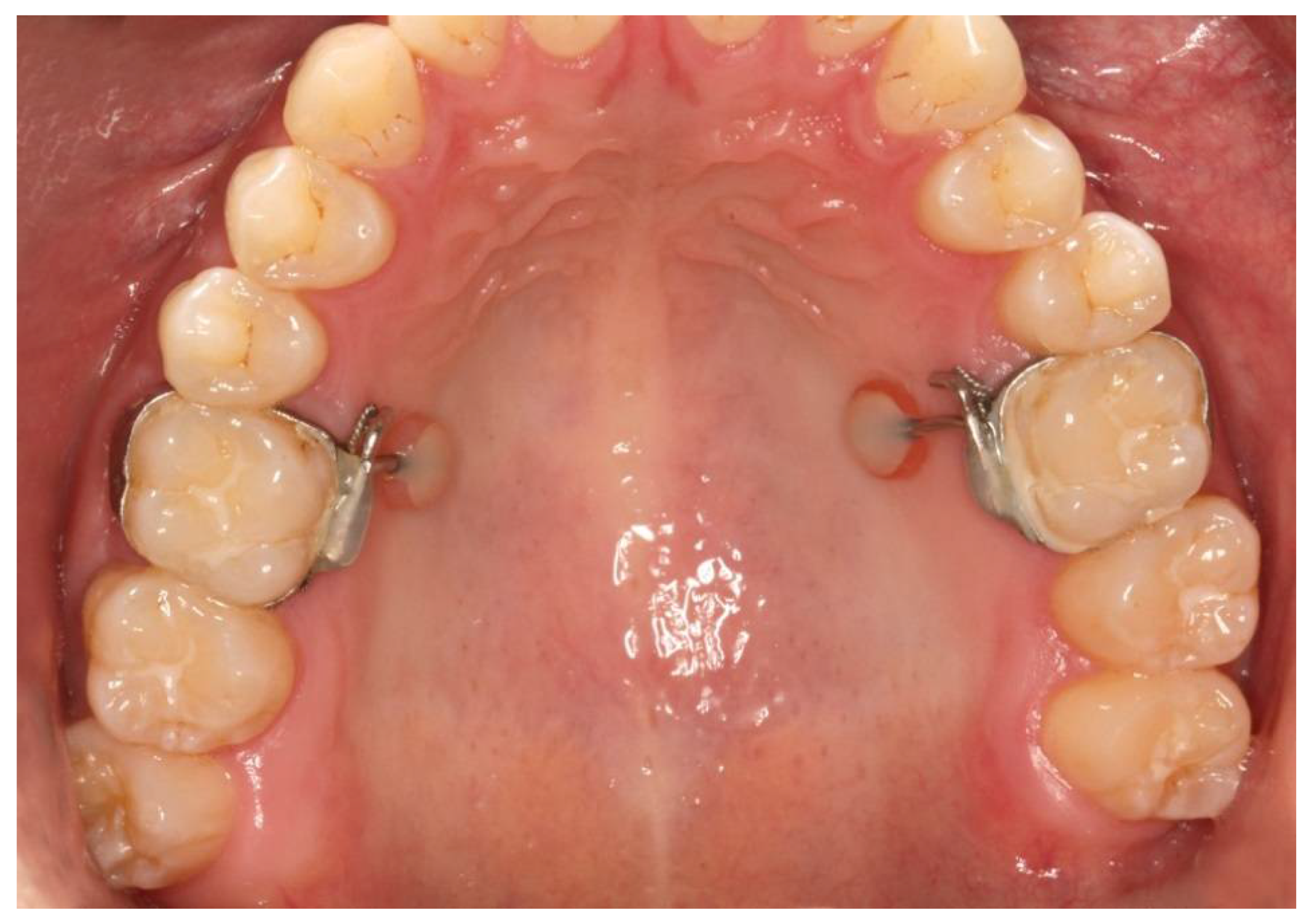
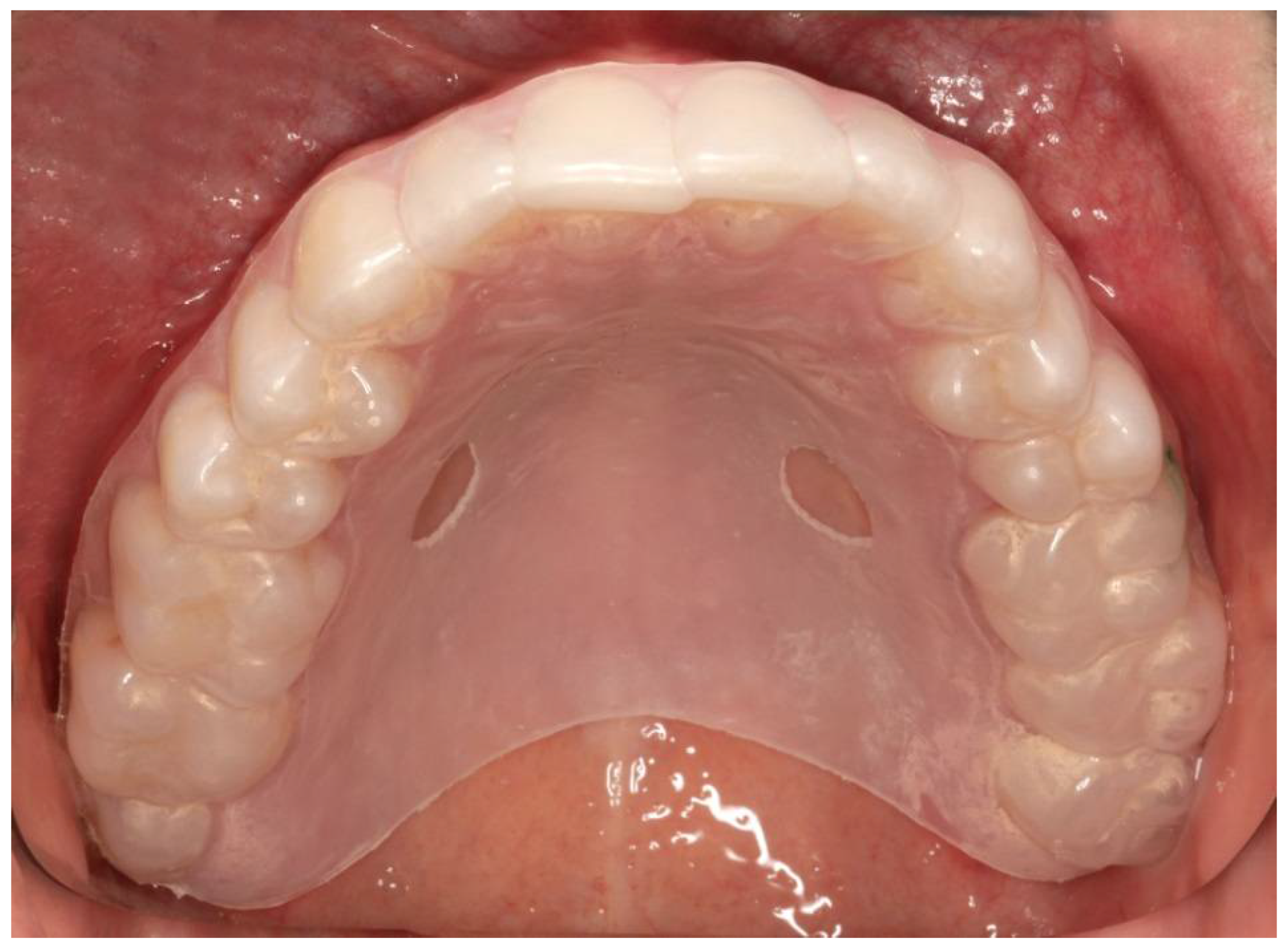
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Age between 20 and 30 years | Smoker |
| Healthy | Pregnancy |
| No signs of gingivitis or periodontitis | Edentulousness |
| No caries lesions | Fixed partial dentures |
| Fully dentate | Removable partial dentures |
| good mouth hygiene | Insufficient fillings |
| good compliance | Composite fillings more than 5 per person and more than ten years old |
| Amalgam fillings | |
| Known intolerance or allergies to the materials to be used | |
| Oral mucosal diseases (e.g., lichen planus, leukoplakia) | |
| Systemic diseases (e.g., diabetes mellitus) | |
| Taking immunosuppressants | |
| Antibiotic therapy in the last 6 weeks | |
| Non-consent by the subject |
| Proband | 1 (m) | 2 (m) | 3 (f) | 4 (f) | 5 (f) | 6 (m) | 7 (m) | 8 (m) | 9 (m) | 10 (m) |
|---|---|---|---|---|---|---|---|---|---|---|
| Number of different isolated bacteria per sample | ||||||||||
| PMMA | 18 | 15 | 20 | 16 | 24 | 18 | 22 | 11 | 16 | 18 |
| Polyamide | 19 | 9 | 15 | 19 | 18 | 18 | 18 | 14 | 21 | 22 |
| Most frequent isolated bacteria (cfu/mL) | ||||||||||
| PMMA | ||||||||||
| Actinomyces naeslundii | - | 1,000,000 | 2,000,000 | 100,000 | - | 200,000 | 1,010,000 | 100,000 | - | 100,000 |
| Actinomyces odontolyticus | 120,000 | 1,210,000 | 1,000,000 | 1,300,000 | 4,000,000 | - | - | 340,000 | - | 500,000 |
| Atopobium parvulum | 10,000 | 100,000 | 1,000,000 | 1,000,000 | 2,000,000 | 1,000,000 | - | 100,000 | 200,000 | 1,000,000 |
| Capnocythophaga gingivalis | 1,020,000 | 100,000 | - | 200,000 | 1,000,000 | - | - | 220,000 | 100,000 | - |
| Neisseria mucosa | - | - | - | 100,000 | 1,000,000 | 100,000 | 10,000 | 100,000 | 200,000 | 10,000 |
| Prevotella melaninogenica | 10,000 | 200,000 | 1,000,000 | 400,000 | - | 1,110,000 | - | - | - | 100,000 |
| Streptococcus gordonii | 100,000 | - | - | - | 100,000 | - | - | 10,000 | 410,000 | 2,000,000 |
| Streptococcus mitis Gruppe | - | 1,210,000 | 3,100,000 | - | 1,000,000 | 1,300,000 | 10,000 | 100,000 | 1,300,000 | - |
| Streptococcus parasanguinis | - | 10,000 | - | 200,000 | - | 100,000 | 20,000 | - | - | 1,700,000 |
| Streptococcus sanguinis | - | - | - | 100,000 | 2,000,000 | - | 100,000 | 400,000 | 500,000 | 100,000 |
| Veillonella parvula | 1,110,000 | 1,400,000 | - | 2,000,000 | 2,000,000 | 110,000 | 1,100,000 | 300,000 | 200,000 | 1,300,000 |
| Polyamide | ||||||||||
| Actinomyces naeslundii | 1,100,000 | 10,000 | 1,100,000 | 1,500,000 | - | 1,210,000 | 1,330,000 | - | - | - |
| Actinomyces odontolyticus | 1,200,000 | - | 1,100,000 | 2,300,000 | 1,000,000 | - | 200,000 | 400,000 | 100,000 | 1,100,000 |
| Atopobium parvulum | 1,000,000 | - | - | - | - | - | - | 200,000 | 100,000 | 1,000,000 |
| Capnocythophaga gingivalis | - | - | - | - | - | - | - | - | 100,000 | - |
| Neisseria mucosa | 1,000,000 | - | - | - | 10,000 | 1,100,000 | 10,000 | 10,000 | - | - |
| Prevotella melaninogenica | - | - | - | 100,000 | - | - | - | 100,000 | - | 1,000,000 |
| Streptococcus gordonii | 1,110,000 | - | - | 100,000 | - | - | - | 700,000 | 100,000 | - |
| Streptococcus mitis Gruppe | 1,100,000 | - | - | - | 10,000 | 1,200,000 | 100,000 | 200,000 | 2,200,000 | - |
| Streptococcus parasanguinis | 210,000 | - | 100,000 | - | 1000,000 | 1,100,000 | - | - | 1,000,000 | 100,000 |
| Streptococcus sanguinis | - | - | 1,000,000 | 300,000 | 1,110,000 | - | 110,000 | 100,000 | 1,000,000 | - |
| Veillonella parvula | 1,000,000 | 100,000 | 1,000,000 | - | 1,100,000 | 1,310,000 | 1,110,000 | 100,000 | 300,000 | 2,000,000 |
| PMMA | Polyamide | |||||
|---|---|---|---|---|---|---|
| Average Number of Cells per 100 Evaluated Cells | Average Number of Cells per 100 Evaluated Cells | |||||
| Cells with Nucleus | Cells without Nucleus | MC | Cells with Nucleus | Cells without Nucleus | MC | |
| T1-I. Quadrant | 10 | 90 | 0 | 7 | 93 | 0 |
| T1-II. Quadrant | 8 | 92 | 0 | 9 | 91 | 0 |
| T1-total | 9 | 91 | 0 | 8 | 92 | 0 |
| T2-I. Quadrant | 30 | 70 | 0 | 36 | 64 | 0 |
| T2-II. Quadrant | 34 | 66 | 0 | 34 | 66 | 0 |
| T2-total | 32 | 68 | 0 | 35 | 65 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olms, C.; Yahiaoui-Doktor, M.; Remmerbach, T.W.; Stingu, C.S. Bacterial Colonization and Tissue Compatibility of Denture Base Resins. Dent. J. 2018, 6, 20. https://doi.org/10.3390/dj6020020
Olms C, Yahiaoui-Doktor M, Remmerbach TW, Stingu CS. Bacterial Colonization and Tissue Compatibility of Denture Base Resins. Dentistry Journal. 2018; 6(2):20. https://doi.org/10.3390/dj6020020
Chicago/Turabian StyleOlms, Constanze, Maryam Yahiaoui-Doktor, Torsten W. Remmerbach, and Catalina Suzana Stingu. 2018. "Bacterial Colonization and Tissue Compatibility of Denture Base Resins" Dentistry Journal 6, no. 2: 20. https://doi.org/10.3390/dj6020020
APA StyleOlms, C., Yahiaoui-Doktor, M., Remmerbach, T. W., & Stingu, C. S. (2018). Bacterial Colonization and Tissue Compatibility of Denture Base Resins. Dentistry Journal, 6(2), 20. https://doi.org/10.3390/dj6020020






