The Use of Statins as an Adjunctive Periodontal Disease Treatment: Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Focused Question
2.2. Search Strategy
- –
- Periodontitis AND simvastatin/rosuvastatin/pravastatin/lovastatin/pitavastatin/atorvastatin;
- –
- Statins AND periodontal therapy;
- –
- Statins AND periodontitis;
- –
- Periodontal disease AND statins;
- –
- Adjunctive periodontal therapy AND statins.
2.3. Study Selection
- –
- Full text available in English;
- –
- Articles published in 2012 and onwards;
- –
- Randomized Controlled Trial (RCT) performed on humans;
- –
- The use of one of the following statin gels as an adjunct to non-surgical periodontal therapy: simvastatin, rosuvastatin, pravastatin, lovastatin, pitavastatin, or atorvastatin;
- –
- Assessment of at least the following clinical parameters: Clinical Attachment Level (CAL), Bleeding on Probing (BoP), Probing Depth (PD), and Plaque Index (PI).
- –
- In vivo studies;
- –
- Case reports studies;
- –
- Case series studies;
- –
- Case control studies;
- –
- Cross-section studies;
- –
- Clinical trial studies;
- –
- Patients under the systemic treatment of statins;
- –
- RCT performed on animals.
2.4. Data Collection and Synthesis
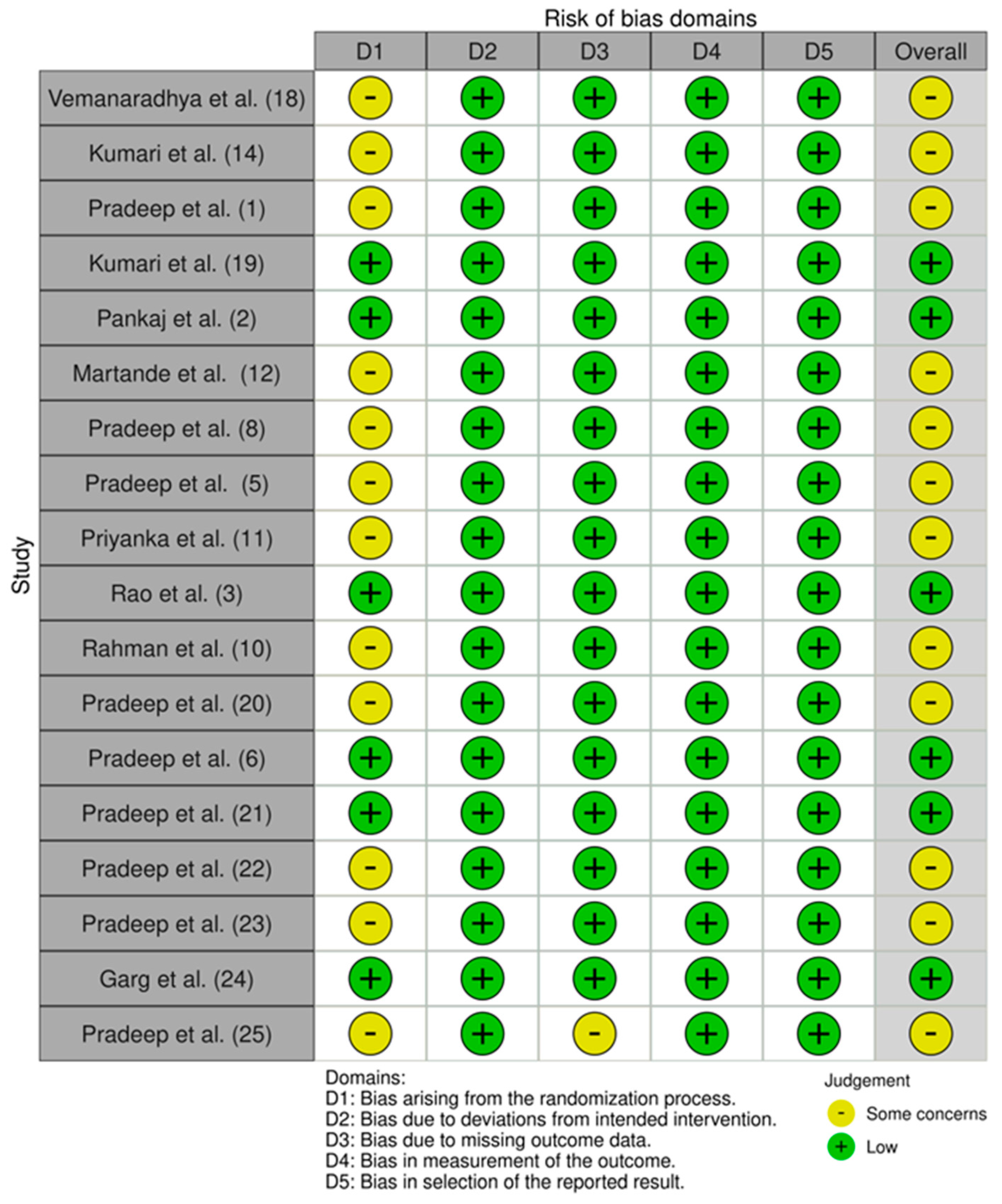
2.5. Risk of Bias Assessment
- Bias arising from the randomization process;
- Bias due to deviations from included intervention;
- Bias due to missing outcome data;
- Bias in measurement of the outcome;
- Bias in selection of the reported result.
2.6. Statistical Analysis
3. Results
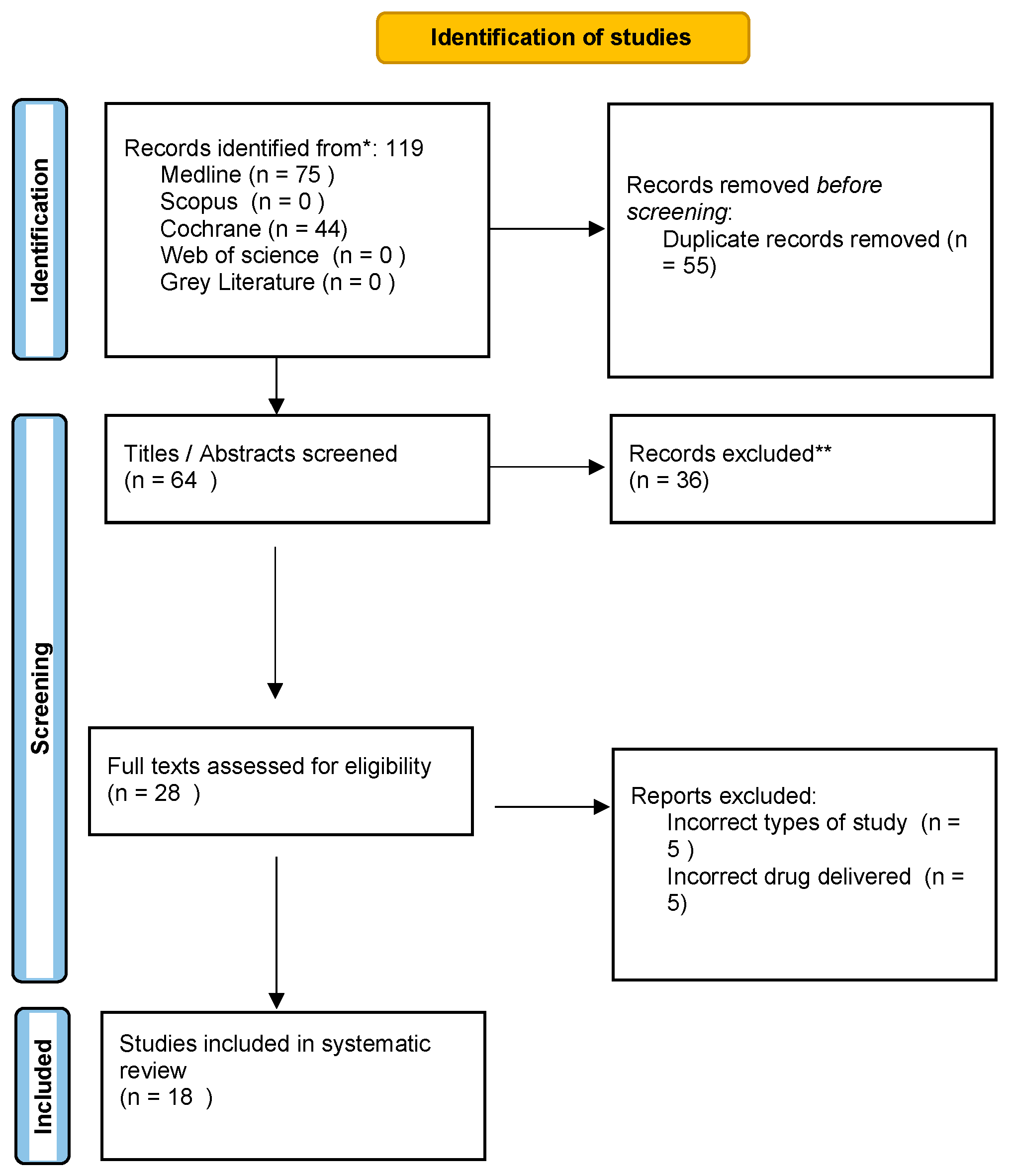
3.1. Study Selection
3.2. Study Characteristics and Descriptive Data Analysis
3.3. Statins vs. Placebo
3.3.1. Reduction in PD
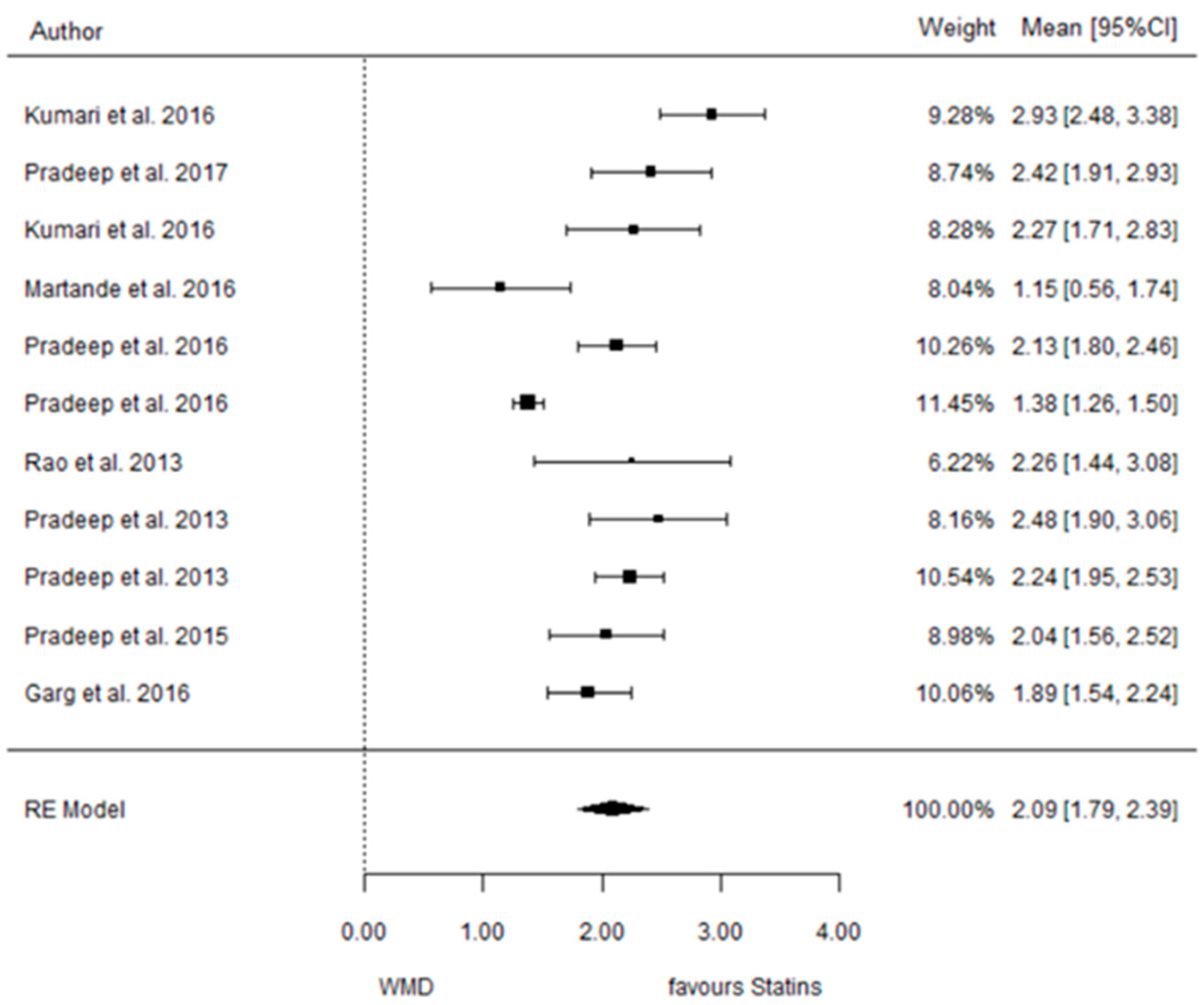
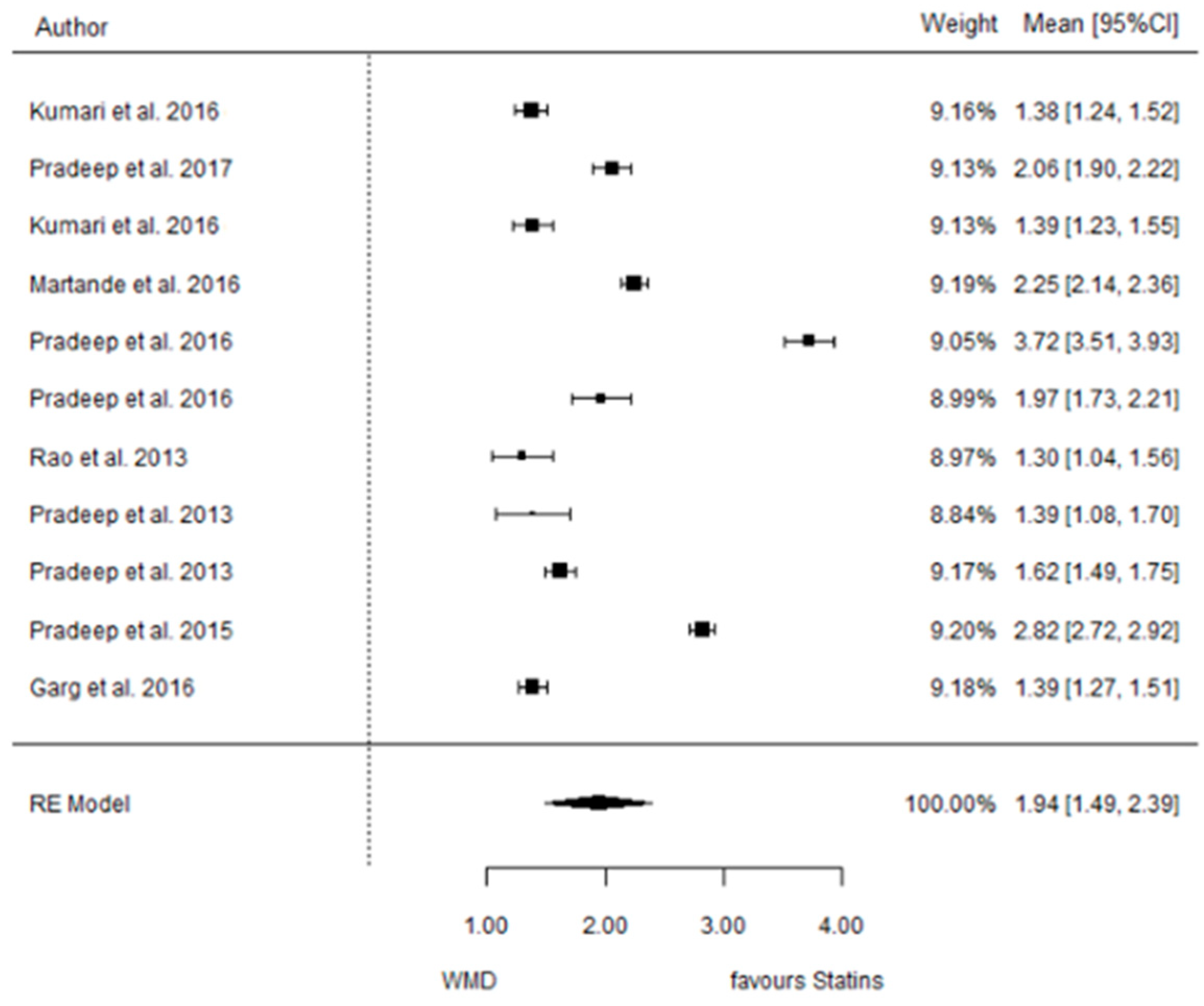
3.3.2. Reduction in CAL

3.3.3. Reduction in IBD
3.3.4. Reduction in RX Bone Depth
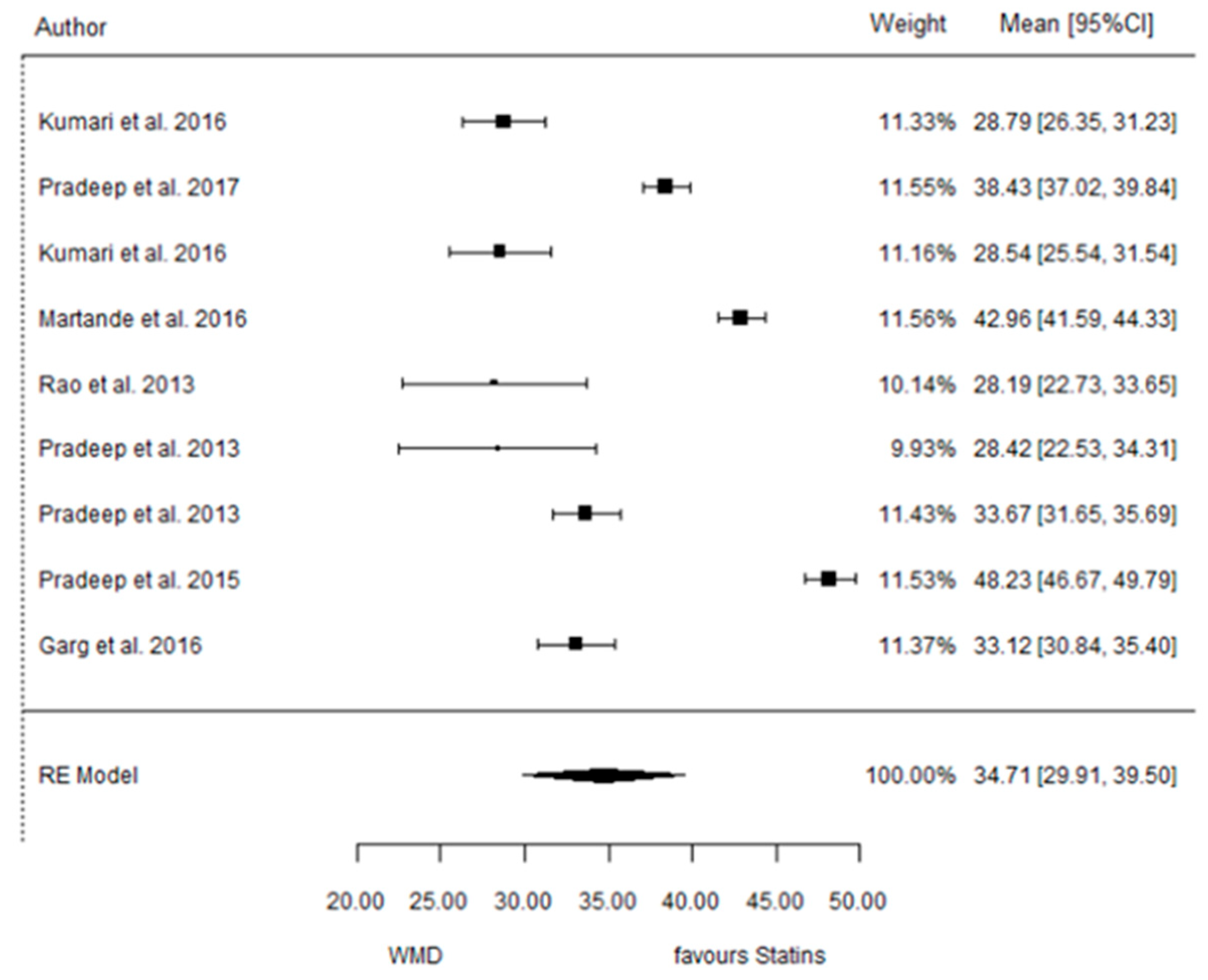
3.4. ATV vs. Placebo
3.4.1. Reduction in PD
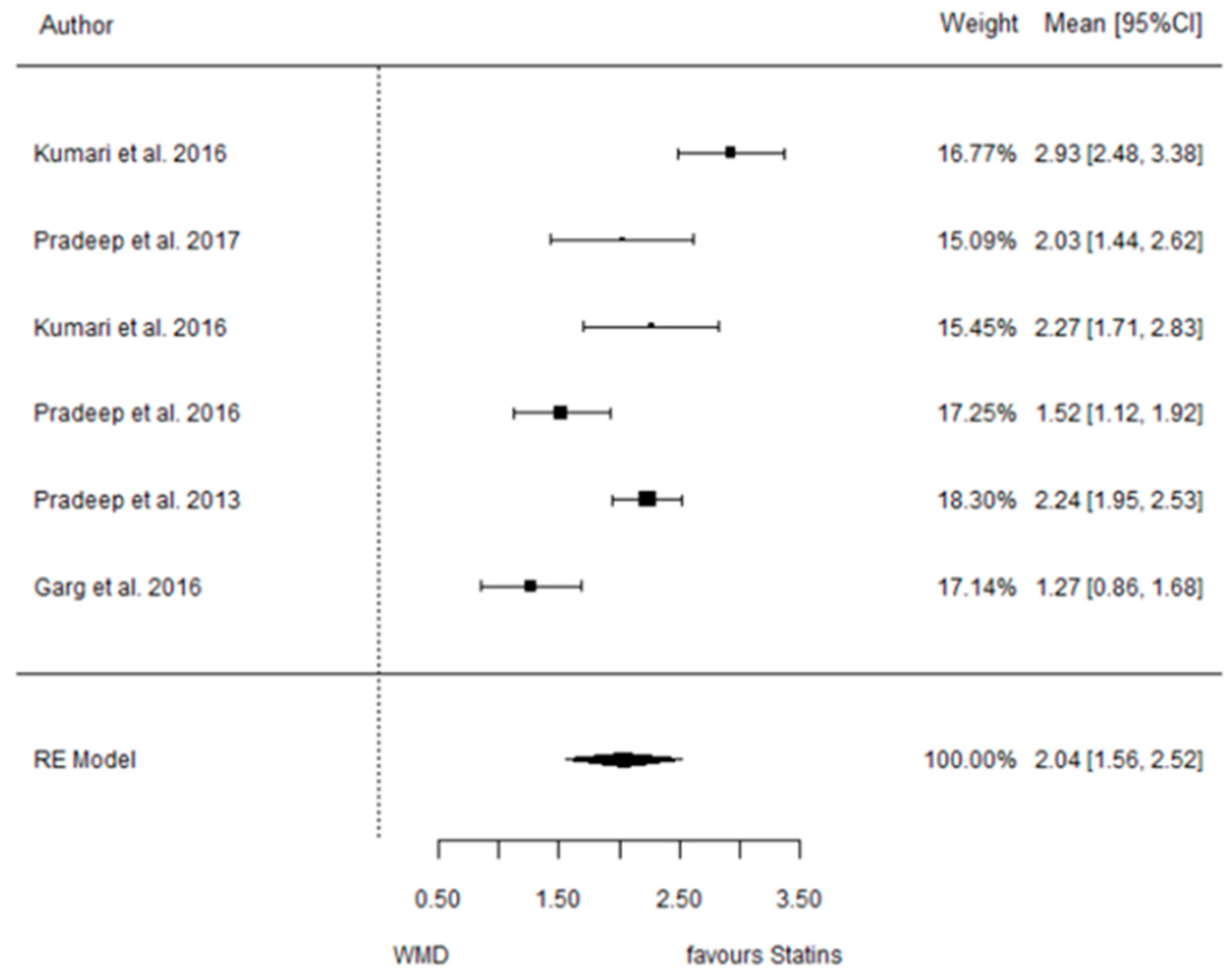
3.4.2. Reduction in CAL
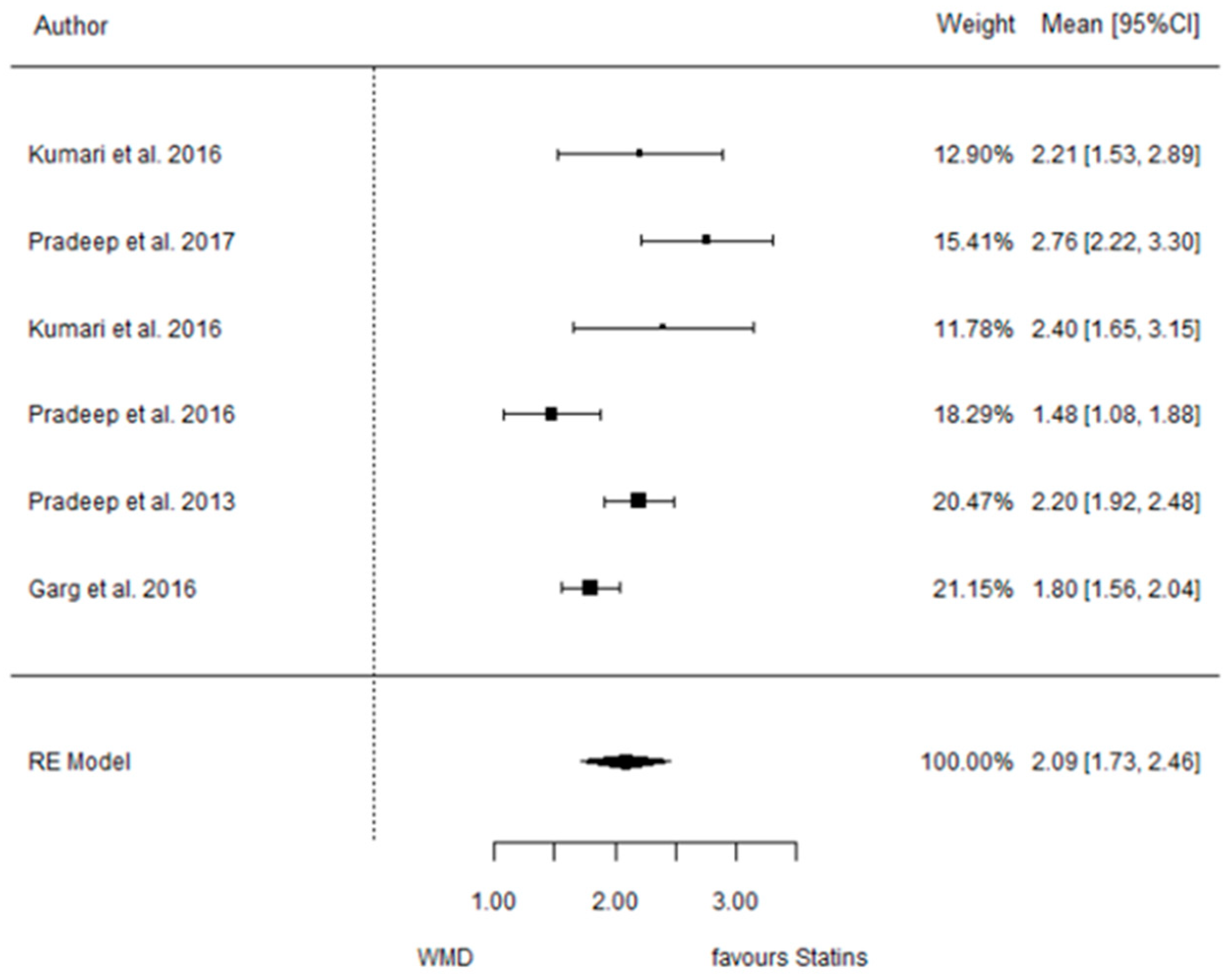
3.4.3. Reduction in IBD
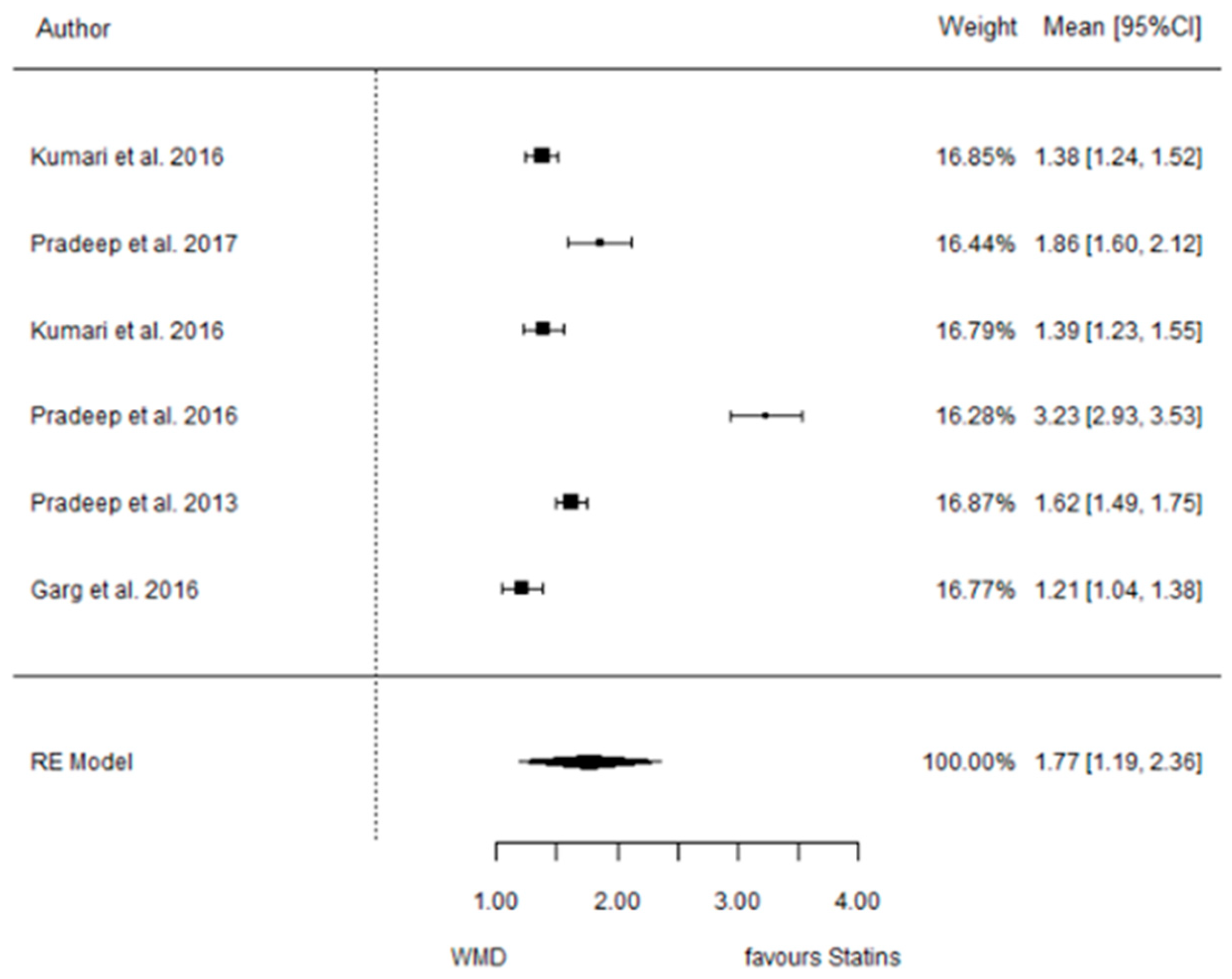
3.4.4. Reduction in RX Bone Depth
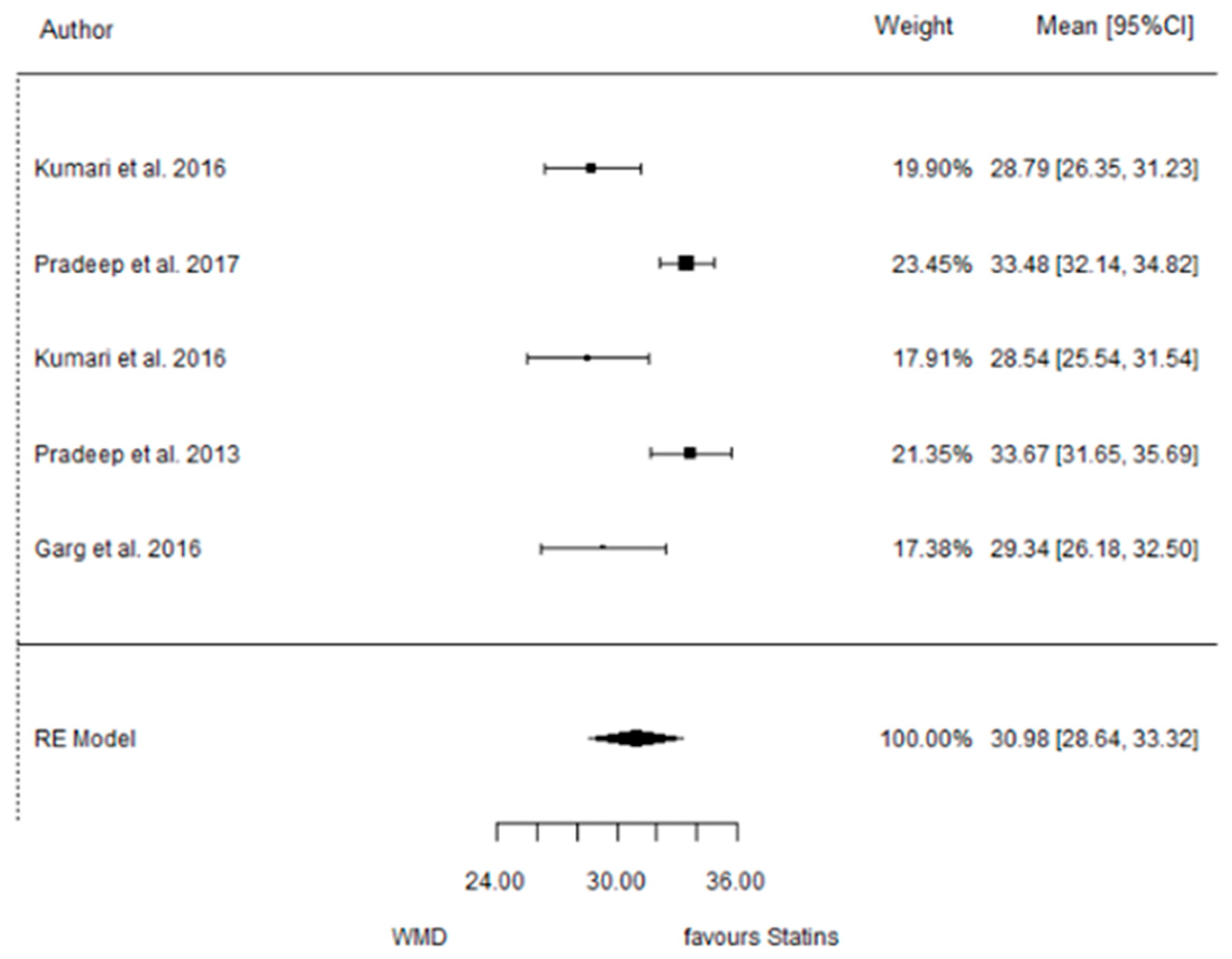
3.5. SMV vs. Placebo
3.5.1. Reduction in PD
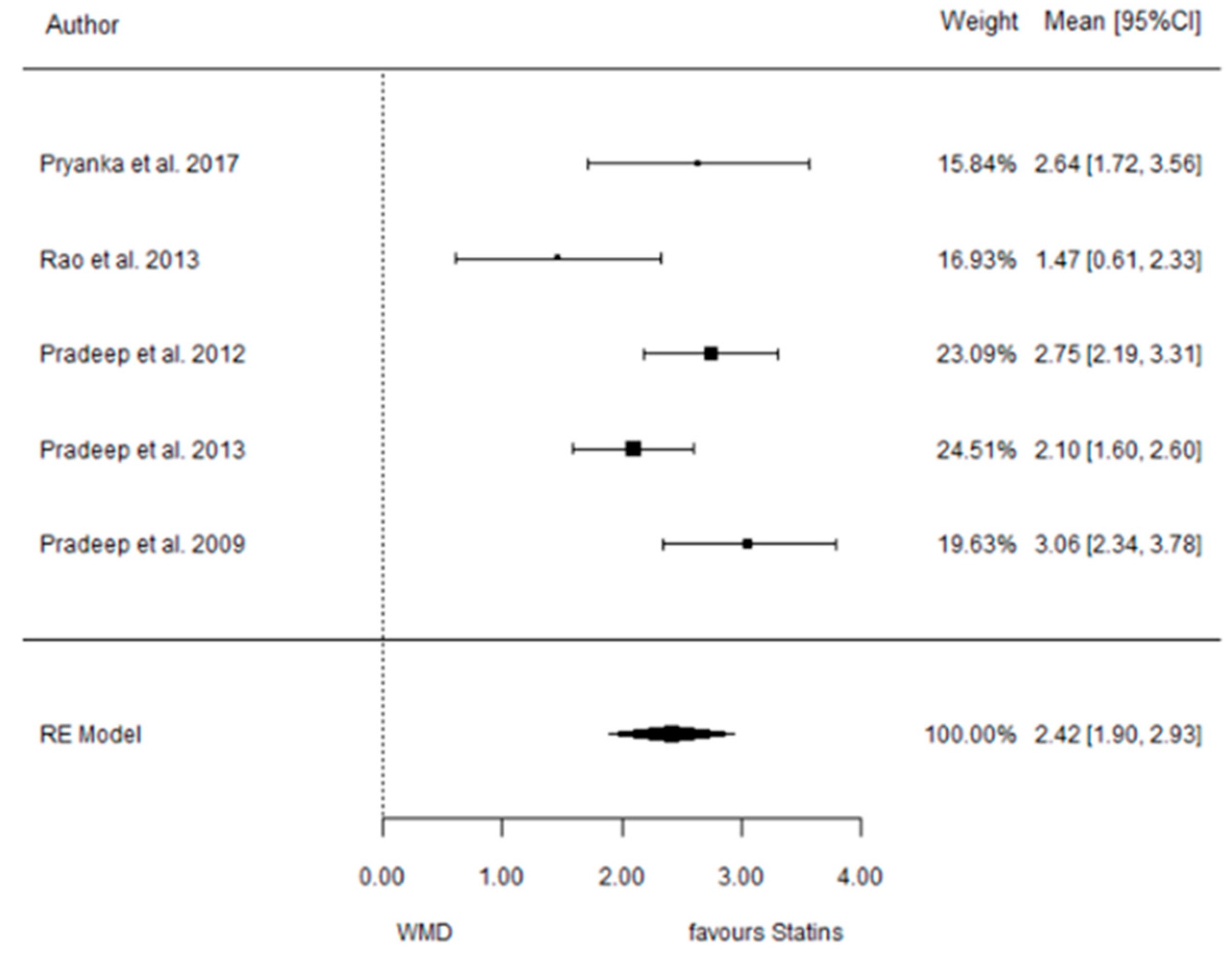
3.5.2. Reduction in CAL
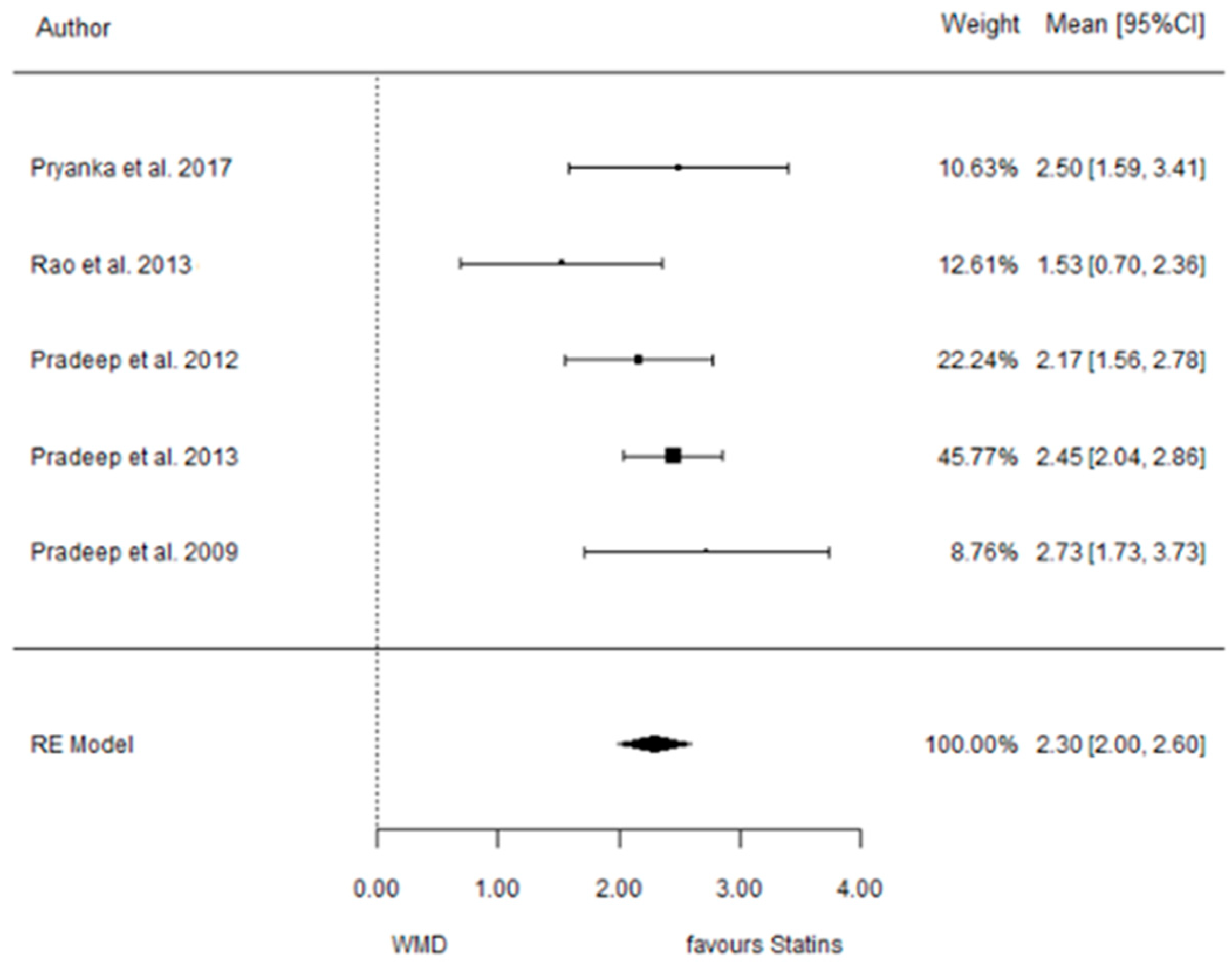
3.6. (ATV vs. SMV vs. RSV vs. Placebo): Network Meta-Analysis
3.6.1. Reduction in PD

3.6.2. Reduction in CAL

3.6.3. Reduction in IBD

3.6.4. Reduction in RX Bone Depth

4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Pradeep, A.R.; Kanoriya, D.; Singhal, S.; Garg, V.; Manohar, B.; Chatterjee, A. Comparative evaluation of subgingivally delivered 1% alendronate versus 1.2% atorvastatin gel in treatment of chronic periodontitis: A randomized placebo-controlled clinical trial. J. Investig. Clin. Dent. 2017, 8, e12215. [Google Scholar] [CrossRef] [PubMed]
- Pankaj, D.; Sahu, I.; Kurian, I.G.; Pradeep, A.R. Comparative evaluation of subgingivally delivered 1.2% Rosuvastatin and 1% Metformin gel in treatment of intrabony defects in chronic periodontitis: A randomized controlled clinical trial. J. Periodontol. 2018, 89, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Rao, N.; Pradeep, A.; Bajaj, P.; Kumari, M.; Naik, S. Simvastatin local drug delivery in smokers with chronic periodontitis: A randomized controlled clinical trial. Aust. Dent. J. 2013, 58, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89 (Suppl. S1), S159–S172. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Garg, V.; Kanoriya, D.; Singhal, S. Platelet-rich fibrin with 1.2% rosuvastatin for treatment of intrabony defects in chronic periodontitis: A randomized controlled clinical rial. J. Periodontol. 2016, 87, 1468–1473. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.; Rao, N.S.; Bajaj, P.; Kumari, M. Efficacy of subgingivally delivered simvastatin in the treatment of patients with type 2 diabetes and chronic periodontitis: A randomized double-masked controlled clinical trial. J. Periodontol. 2013, 84, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, S.; Emami, H.; Vucic, E.; Singh, P.; Vijayakumar, J.; Fifer, K.M.; Alon, A.; Shankar, S.S.; Farkouh, M.; Rudd, J.H.; et al. High-dose atorvastatin reduces periodontal inflammation: A novel pleiotropic effect of statins. J. Am. Coll. Cardiol. 2013, 62, 2382–2391. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.; Garg, V.; Kanoriya, D.; Singhal, S. 1.2% Rosuvastatin versus 1.2% atorvastatin gel local drug delivery and redelivery in treatment of intrabony defects in chronic periodontitis: A randomized placebo-controlled clinical trial. J. Periodontol. 2016, 87, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, D.R.; Andrade, C.X.; Chaparro, A.P.; Inostroza, C.M.; Ramirez, V.; Violant, D.; Nart, J. Short-term effects of 2% atorvastatin dentifrice as an adjunct to periodontal therapy: A randomized double-masked clinical trial. J. Periodontol. 2015, 86, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Gayathri, G.V.; Mehta, D.S. A clinico-microbiological and biochemical study evaluating the adjunctive use of antimicrobial photodynamic therapy and local drug delivery of 1.2% simvastatin gel compared to scaling and root planing alone. Photodiagn. Photodyn. Ther. 2020, 32, 102017. [Google Scholar] [CrossRef] [PubMed]
- Priyanka, N.; Abhilash, A.; Saquib, S.; Malgaonkar, N.; Kudyar, N.; Gupta, A.; Kalra, N.; Pradeep, A. Clinical efficacy of subgingivally delivered 1.2 mg simvastatin in the treatment of patients with aggressive periodontitis: A randomized controlled clinical trial. Int. J. Periodontics Restor. Dent. 2017, 37, e135–e141. [Google Scholar] [CrossRef] [PubMed]
- Martande, S.S.; Kumari, M.; Pradeep, A.; Singh, S.P.; Suke, D.K.; Guruprasad, C. Platelet-rich fibrin combined with 1.2% atorvastatin for treatment of intrabony defects in chronic periodontitis: A randomized controlled clinical trial. J. Periodontol. 2016, 87, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Jeger, R.; Dieterle, T. Statins: Have we found the holy grail? Swiss Med. Wkly. 2012, 3, w13515. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Martande, S.S.; Pradeep, A.R. Subgingivally delivered 1.2% atorvastatin in the treatment of chronic periodontitis among smokers: A randomized, controlled clinical trial. J. Investig. Clin. Dent. 2016, 8, 12213. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 29, n71. [Google Scholar] [CrossRef] [PubMed]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Vemanaradhya, G.G.; Emani, S.; Mehta, D.S.; Bhandari, S. Effect of 1.2% of simvastatin gel as a local drug delivery system on Gingival Crevicular Fluid interleukin-6 & interleukin-8 levels in non surgical treatment of chronic periodontitis patients. Arch. Oral Biol. 2017, 82, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Martande, S.S.; Pradeep, A.; Naik, S.B. Efficacy of subgingivally delivered 1.2% atorvastatin in the treatment of chronic periodontitis in patients with type 2 diabetes mellitus: A randomized controlled clinical trial. J. Periodontol. 2016, 87, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Priyanka, N.; Kalra, N.; Naik, S.B.; Singh, S.P.; Martande, S. Clinical efficacy of subgingivally delivered 1.2-mg Simvastatin in the treatment of individuals with Class II furcation defects: A randomized controlled clinical trial. J. Periodontol. 2012, 83, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.; Kumari, M.; Rao, N.S.; Martande, S.S.; Naik, S.B. Clinical efficacy of subgingivally delivered 1.2% atorvastatin in chronic periodontitis: A randomized controlled clinical trial. J. Periodontol. 2013, 84, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Karvekar, S.; Nagpal, K.; Patnaik, K.; Guruprasad, C.N.; Kumaraswamy, K.M. Efficacy of locally delivered 1.2% Rosuvastatin gel to non-surgical treatment of patients with chronic periodontitis: A randomized, placebo-controlled clinical trial. J. Periodontol. 2015, 86, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.; Karvekar, S.; Nagpal, K.; Patnaik, K.; Raju, A.; Singh, P. Rosuvastatin 1.2 mg in situ gel combined with 1:1 mixture of autologous platelet-rich fibrin and porous hydroxyapatite bone graft in surgical treatment of mandibular class II furcation defects: A randomized clinical control trial. J. Periodontol. 2016, 87, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Pradeep, A. 1.2% Rosuvastatin and 1.2% atorvastatin gel local drug delivery and redelivery in the treatment of class ii furcation defects: A randomized controlled clinical trial. J. Periodontol. 2017, 88, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Thorat, M.S. Clinical effect of subgingivally delivered simvastatin in the treatment of patients with chronic periodontitis: A randomized clinical trial. J. Periodontol. 2010, 81, 214–222. [Google Scholar] [CrossRef]
- Cecoro, G.; Piccirillo, A.; Martuscelli, G.; Del Fabbro, M.; Annunziata, M.; Guida, L. Efficacy of locally delivered statins as an adjunct to scaling and root planning in the treatment of periodontitis: A systematic review and meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 5737–5754. [Google Scholar] [PubMed]
- Christiansen, D.L.; Killeen, A.C.; Ramer-Tait, A.; Hattervig, R.L.; Nawshad, A.; Beede, K.; Samson, K.K.; Reinhardt, R.A. Local simvastatin and inflammation during periodontal mini-flap wound healing: Exploratory results. J. Periodontol. 2022, 94, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Hasan, F.; Ikram, R.; Simjee, S.U.; Iftakhar, K.; Asadullah, K. Effectiveness of Simvastatin 1% oral gel and mouthwash used as an adjunct treatment of scaling and root planning in the treatment of periodontal diseases. Pak. J. Pharm. Sci. 2019, 32, 2673–2677. [Google Scholar] [PubMed]
- Amato, M.; Di Spirito, F.; D’ambrosio, F.; Boccia, G.; Moccia, G.; De Caro, F. Probiotics in Periodontal and Peri-Implant Health Management: Biofilm Control, Dysbiosis Reversal, and Host Modulation. Microorganisms 2022, 10, 2289. [Google Scholar] [CrossRef]
- Di Spirito, F.; Schiavo, L.; Pilone, V.; Lanza, A.; Sbordone, L.; D’ambrosio, F. Periodontal and Peri-Implant Diseases and Systemically Administered Statins: A Systematic Review. Dent. J. 2021, 9, 100. [Google Scholar] [CrossRef]
| Aim of Study | Type of Drug and Dose | Follow-Ups | Sample | Outcomes | Conclusions | |
|---|---|---|---|---|---|---|
| Author Year Journal Reference | Sample Number Sample Ages Other Sample Characteristics | Patients That Finished the Study Adverse Effects of the Drug Clinical Parameters Radiographic Parameters | ||||
| Vemanaradhya et al., 2017 Archives of Oral Biology [18] | Evaluate the efficacy of 1.2% simvastatin gel as an LDD in adjunct SRP on GCF IL-6 and IL-8 levels in chronic periodontitis patients and correlate their values with clinical parameters | Simvastatin 1.2% | 45 days | 46 35–60 years old 18 males and 28 females | Acceptable statin toleration Reduction in all clinical parameters in all groups SS higher reduction in statin group for PI, GI, and SBI No SS differences in PPD and CAL between groups | Simvastatin 1.2% has an effective role in controlling the inflammation of the periodontium. |
| Kumari et al., 2016 Journal of Investigative and Clinical Dentistry [14] | Evaluate the efficacy of a 1.2% ATV local drug delivery as an adjunct to SRP for the treatment of IBD (intrabody defects) in smokers with CP in comparison with placebo gel. | Atorvastatin 1.2% | 3–6–9 months | 71 smokers 30–50 years old | 66 Acceptable statin toleration SS higher mSBI and PD reduction and CAL gain in statin group SS higher reduction in IBD in statin group | Significant improvement in clinical parameters compared to placebo gel as an adjunct to SRP. |
| Pradeep et al., 2016 Journal of Investigative and Clinical Dentistry [1] | Evaluate and compare the efficacy of 1% ALN and 1.2% ATV gel as a local drug delivery system in adjunct to scaling and root planing (SRP) for treatment for intrabony defects in CP patients. | Atorvastatin 1.2% Alendronate 1% | 3–6–9 months | 104 30–50 years old 53 males and 51 females | 90 Acceptable statin toleration No SS differences in PI between groups. ALD and ATV groups showed SS in all parameters. ALN showed SS differences in PD, CAL, and DDR% when compared to ATV. | Both ATV and ALN can be used as an effective mode of treatment for CP patients. However, ALN was comparatively better than ATV. |
| Kumari et al., 2016 Journal of Periodontology [19] | Evaluate the effectiveness of 1.2 ATV gel, as an adjunct to SRP in the treatment of infrabony defects in chronic periodontitis in subjects with DM2 | 1.2% Atorvastatin | 3–6–9 months | 75 individuals 40–50 years old. 38 males and 37 females | 60 Acceptable statin toleration SS greater mSBI and PD reduction, RAL gain, and IBD reduction in statin group. | Local delivery of 1.2% ATV into periodontal pockets of type 2 DM patients stimulated a significant improvement in clinical and rx parameters as compared to placebo gel |
| Pankaj et al., 2018 Journal of Periodontology [2] | Investigate the effectiveness (both clinical and rx) of locally delivered 1.2% RSV gel, 1% MF gel, and placebo as an adjunct to SRP in the treatment of infrabony defects. | 1.2% Rosuvastatin 1% Metrformin | 6–12 months | 90 patients 25 to 45 years old 44 males and 46 females | Acceptable statin toleration Both groups displayed improvement in PI and mSBI, but no SS difference in PI between groups. RSV and MF higher decrease in mSBI, PD, and CAL values and also greater depth reduction (radiographically) and higher DDR% Comparing RSV and MF, RSV showed statistically significant improvement in PD and CAL and also greater DDR% | The study shows that both drugs are used as an affective mode of treatment. However, RSV was comparatively better than MF. |
| Martande et al., 2016 Journal of Periodontology [12] | Evaluate the combined efficacy of PRF and 1.2% ATV gel with OFD in treatment of infrabony defects in CP individuals RCCT | 1.2% Atorvastatin Platelet Rich Fibrin | 9 months | 96 patients mean age of 37.6 48 males and 48 females | 90 Acceptable statin toleration SS reduction in PI and mSBI in all three groups Greater PD reduction and RAL gain in PRF and PRF + ATV sites with no SS differences between them. | There was a greater improvement in clinical parameters in PRF + ATV and PRF as compared to the control group. 1.2% ATV failed to increase the regenerative potential by PRF alone due to similar outcomes between the trial groups. |
| Pradeep et al., 2016 Journal of Periodontology [8] | Investigate the clinical and rx effects of LDD and re-delivery of 1.2% RSV and 1.2% ATV gels with scaling and root planing in the treatment of 2/3-welled IBD in CP patients. | 1.2% Rosuvastatin 1.2% Atrovastatin | 6–9 months | 90 patients aged 25 to 45 45 males and 45 females | 81 Acceptable statin toleration Reduction in PI, mSBI, and PD as well as CA gain was observed in all groups, except PI reduction in group 1. DDR was SS in the statin groups. No SS difference in the mean PI and mSBI reduction between both control groups between baseline at 3, 6, and 9. However, PI reduction between 6 and 9 m, PD reduction, CA level gain, and DDR were significantly greater in RSV than ATV from BL-6 and 6–9. | The administration of statins is superior to mechanical periodontal therapy alone, with LDD of 1.2% rosuvastatin resulting in significantly greater clinic-radiographic improvements compared to 1.2% ATV. |
| Pradeep et al., 2016 Journal of Periodontology [5] | Investigate the clinico-rx effects of OFD, OFD + PRF, and OFD + PRF + 1.2% RSV in the treatment of 2/3-walled IBD in CP patients. | 1.2% Rosuvastatin Platelet Rich Fibrin | 9 months | 90 patients 25–45 years old | 90 Acceptable statin toleration All groups showed significant improvements in all periodontal outcomes from BL-9 months. No SS differences between the 3 groups for mSBI and PI PD and IBD depth reductions as well as CA level gain were significantly greater in control groups. Significantly greater improvements in these parameters were found in group 3 over 2. | The use of RSV has a positive effect on the parameters. |
| Priyanka et al., 2017 The International Journal of Periodontics & Restorative Dentistry [11] | Investigate the effectiveness of 1.2 mg SMV as a local drug delivery system and as an adjunct to scaling and root planing in the treatment of aggressive periodontitis | 1.2% Simvastatin | 3–6 months | 24 patients 30 to 50 years old 14 males and 10 females | 21 Acceptable statin toleration PI, both groups maintained comparable levels of oral hygiene. mSBI presented a greater reduction in the SMV group PD, greater reduction in the SMV group CAL, greater gain in group 2. Bone fill is also greater in group 2. | This clinical trial demonstrates that local delivery of 1.2 mg of simvastatin into the periodontal pocket in the group 2 patients stimulated a significant increase in PD reduction and CAL gain, and improved bone fill compared to group 1 patients. |
| Rao et al., 2013 Australian Dental Journal [3] | Evaluate the efficacy of SMV 1% as local drug delivery as an adjunct to SRP for the treatment of smokers with CP in comparison with placebo gel | 1.2% Simvastatin | 3–6–9 months | 40 male smokers 30–50 years old. | 35 Acceptable statin toleration Both groups presented an improvement in PI but no SS in both parameters at any visit. Significant decrease in mSBI in the SMV group compared to placebo. SV greater CAL gain and PD decrease in the SMV group. SMV group showed SS IBD mean reduction. SS greater vertical bone defect fill in SMV group. | LDD of 1.2 5 SMV in smokers stimulated a significant increase in PD reduction, CAL gain, and improved bone fill as compared to placebo gel as an adjunct to SRP. |
| Rahman et al., 2017 Photodiagnosis and Photodynamic Therapy [10] | Evaluate and compare the efficacy of antimicrobial photodynamic therapy and the LDD of 1.2% SMV gel as an adjunct to SRP versus SRP alone in the treatment of periodontitis using clinical, microbiological, and biochemical parameters | 1.2% Simvastatin | 3 months | 15 8 males and females 35–60 years old | Acceptable statin toleration Significant reduction in API, PBI, PPD, and RAL in all three groups. The mean PPD and RAL score reduction was found to be higher in group 3 (SMV), with no SS. | All three treatment groups showed similar efficacy in improving clinical parameters and reducing p-gingivalis and GCF RANKL levels with no SS difference in outcomes. Between both adjunct treatments used, neither has been proven to have greater potential than the other. |
| Pradeep et al., 2012 Journal of Periodontology [20] | Investigate the clinical and radiographic efficacy (bone fill) of 1.2 mg SMV as an adjunct to SRP in the treatment of mandibular buccal class II furcation defects. | 1.2% Simvastatin | 3–6 months | 72 38 males and 34 females 30–50 years old | Acceptable statin toleration. Decrease in mSBI was greater in the SMV group PD: there were statistical differences in the decrease, being greater in group 2. RVAL and RHAL: greater in group of SMV Bone fill: also greater in group 2 | There was a greater decrease in gingival index and PD and more RVAL and RHAL gain with significant bone fill with locally delivered SMV in class II furcation defects |
| Pradeep et al., 2012 Journal of Periodontology [6] | Evaluate the efficacy of 1.2% SMV as an LDD in adjunct to SRP for the treatment of patients with DM2 and CP compared to placebo gel. | 1.2% Simvastatin | 6–9 months | 38 20 males and 18 females 30–50 years old | 35 Acceptable statin toleration. No SS difference in PI. SS decreased in the SMV group for mSBI. SMV group presented greater PD reduction, CAL gain, IBD, and vertical defect fill. | The local delivery of 1.2% SMV into periodontal pockets of patients with type 2 diabetes stimulated a significant increase in PD reduction, CAL gain, and improved bone fill compared to placebo gel as an adjunct to SRP. |
| Pradeep et al., 2013 Journal of Periodontology [21] | Evaluate the efficacy of 1.2% ATV as local drug delivery in comparison with placebo gel in adjunct to SRP for the treatment of intrabony defects in individuals with CP. | 1.2% Atorvastatin | 3–6–9 months | 67 35 males and 32 females 30–50 years old | 60 Both groups showed improvement in PI. Decrease in mSBI, PD reduction, and CAL gain in the ATV group. ATV group showed significantly greater vertical radiographic defect fill. | The local delivery of 1.2% ATV in individuals with CP stimulates a significant increase in PD reduction, CAL gain, and improved bone fill in adjunct to SRP compared to placebo gel. |
| Pradeep et al., 2015 Journal of Periodontology [22] | Investigate the clinical and radiographic effects of locally delivered 1.2% RSV gel as an adjunct to the non-surgical treatment in CP patients when compared to placebo gel. | 1.2% Rosuvastatin | 1–3–4 and 6 months | 70 individuals 25–55 years old | 65 Acceptable statin toleration. Higher decrease in mSBI in RSV group RSV. CAL and PD SS RSV group. No SS in PI. Greater bone fills in RSV group | The local delivery of RSV reduced inflammation and induced bone formation. |
| Pradeep et al., 2016 Journal of Periodontology [23] | Evaluate the potency of a combination of RSV 1.2 mg in situ gel with 1:1 mixture of autologous PRF and HA bone graft in the surgical treatment of mandibular degree II furcation defects when compared with autologous PRF and A bone graft placed after OFD | 1.2% Rosuvastatin Autologous platelet-rich fibrin Porus hydroxyappatite bone graft. | 9 months | 110 60 males and 50 females 25–55 years old | 105 Acceptable statin toleration SS in RSV group when looking at mSBI and PI SS in the group’s parameters at follow-up. The groups with RSV presented SS better changes in all parameters (PD, RVAL, RHAL, and IBD). | The addition of RSV to the PRF and HA improved the regenerative effect on the bone defects. |
| Garg et al., 2017 Journal of Periodontology [24] | Explore the efficacy of 1.2% RSV and 1.2% ATV gel as a local drug delivery and redelivery system as an adjunct to SRP for treatment of class II furcation defects. | 1.2% Rosuvastain 1.2% Atorvastatin | 6–9 months | 90 (55 males and 60 females) | 90 Acceptable statin toleration SS differences in mSBI in RSV in comparison to ATV PD, RHCAL, and RVCAL, SS higher changes in the statin groups, although it was higher in RSV. Bone depth reduction is greater in statin groups. | RSV has greater anti-inflammatory effects due to more effective CRP levels |
| Pradeep et al., 2010 Journal of Peritology [25] | Investigate the clinical and radiologic efficacy of SMV, 1.2 mg as an adjunct to SRP in the treatment of CP. | 1.2% Simvastatin | 1–2–4–6 months | 64 | 60 Acceptable statin toleration. mSBI decrease in both groups, although greater in SMV group. PD, CAL, and IBS were SS in SMV group. | Greater decrease in clinical and radiographic parameters in Simvastatin patients. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greethurst, A.R.; Galletti, C.; Lo Giudice, R.; Nart, J.; Vallés, C.; Real-Voltas, F.; Gay-Escoda, C.; Marchetti, E. The Use of Statins as an Adjunctive Periodontal Disease Treatment: Systematic Review and Meta-Analysis. Dent. J. 2024, 12, 150. https://doi.org/10.3390/dj12060150
Greethurst AR, Galletti C, Lo Giudice R, Nart J, Vallés C, Real-Voltas F, Gay-Escoda C, Marchetti E. The Use of Statins as an Adjunctive Periodontal Disease Treatment: Systematic Review and Meta-Analysis. Dentistry Journal. 2024; 12(6):150. https://doi.org/10.3390/dj12060150
Chicago/Turabian StyleGreethurst, Alice Rose, Cosimo Galletti, Roberto Lo Giudice, José Nart, Cristina Vallés, Francisco Real-Voltas, Cosme Gay-Escoda, and Enrico Marchetti. 2024. "The Use of Statins as an Adjunctive Periodontal Disease Treatment: Systematic Review and Meta-Analysis" Dentistry Journal 12, no. 6: 150. https://doi.org/10.3390/dj12060150
APA StyleGreethurst, A. R., Galletti, C., Lo Giudice, R., Nart, J., Vallés, C., Real-Voltas, F., Gay-Escoda, C., & Marchetti, E. (2024). The Use of Statins as an Adjunctive Periodontal Disease Treatment: Systematic Review and Meta-Analysis. Dentistry Journal, 12(6), 150. https://doi.org/10.3390/dj12060150