The Effect of Topology on Phase Behavior under Confinement
Abstract
:1. Introduction
2. Theory
2.1. Minkowski Functionals
3. Methods
3.1. Density Functional Theory
3.2. Geometries
3.3. Simulation Parameters
3.4. Minkowski Coefficients
4. Results
5. Discussion & Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berube, V.; Radtke, G.; Dresselhaus, M.; Chen, G. Size effects on the hydrogen storage properties of nanostructured metal hydrides: A review. Int. J. Energy Res. 2007, 31, 637–663. [Google Scholar] [CrossRef]
- He, L.; Melnichenko, Y.B.; Gallego, N.C.; Contescu, C.I.; Guo, J.; Bahadur, J. Investigation of morphology and hydrogen adsorption capacity of disordered carbons. Carbon 2014, 80, 82–90. [Google Scholar] [CrossRef] [Green Version]
- He, T.; Pachfule, P.; Wu, H.; Xu, Q.; Chen, P. Hydrogen carriers. Nat. Rev. Mater. 2016, 1, 16059. [Google Scholar] [CrossRef]
- Li, M.; Carter, R.; Douglas, A.; Oakes, L.; Pint, C.L. Sulfur vapor-infiltrated 3D carbon nanotube foam for binder-free high areal capacity lithium–sulfur battery composite cathodes. ACS Nano 2017, 11, 4877–4884. [Google Scholar] [CrossRef]
- Barsotti, E.; Tan, S.P.; Saraji, S.; Piri, M.; Chen, J.H. A review on capillary condensation in nanoporous media: Implications for hydrocarbon recovery from tight reservoirs. Fuel 2016, 184, 344–361. [Google Scholar] [CrossRef]
- Belmabkhout, Y.; Serna-Guerrero, R.; Sayari, A. Adsorption of CO2 from dry gases on MCM-41 silica at ambient temperature and high pressure. 1: Pure CO2 adsorption. Chem. Eng. Sci. 2009, 64, 3721–3728. [Google Scholar] [CrossRef]
- Yoshimoto, Y.; Hori, T.; Kinefuchi, I.; Takagi, S. Effect of capillary condensation on gas transport properties in porous media. Phys. Rev. E 2017, 96, 043112. [Google Scholar] [CrossRef] [PubMed]
- Vincent, O.; Marguet, B.; Stroock, A.D. Imbibition triggered by capillary condensation in nanopores. Langmuir 2017, 33, 1655–1661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bui, B.T.; Liu, H.H.; Chen, J.; Tutuncu, A.N. Effect of capillary condensation on gas transport in shale: A pore-scale model study. SPE J. 2016, 21, 601–612. [Google Scholar] [CrossRef]
- Melnichenko, Y.B. Supercritical Fluids in Confined Geometries. In Small-Angle Scattering from Confined and Interfacial Fluids; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2016; pp. 251–309. [Google Scholar] [CrossRef]
- Ghosh, K.; Krishnamurthy, C. Structural behavior of supercritical fluids under confinement. Phys. Rev. E 2018, 97, 012131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Douglas Frink, L.J.; Salinger, A.G. Rapid analysis of phase behavior with density functional theory. II. Capillary condensation in disordered porous media. J. Chem. Phys. 2003, 118, 7466–7476. [Google Scholar] [CrossRef]
- Coasne, B.; Galarneau, A.; Di Renzo, F.; Pellenq, R.J. Effect of morphological defects on gas adsorption in nanoporous silicas. J. Phys. Chem. C 2007, 111, 15759–15770. [Google Scholar] [CrossRef]
- Mason, G. The effect of pore space connectivity on the hysteresis of capillary condensation in adsorption—Desorption isotherms. J. Colloid Interface Sci. 1982, 88, 36–46. [Google Scholar] [CrossRef]
- Mason, G. Determination of the pore-size distributions and pore-space interconnectivity of Vycor porous glass from adsorption-desorption hysteresis capillary condensation isotherms. Proc. R. Soc. Lond. A Math. Phys. Sci. 1988, 415, 453–486. [Google Scholar] [CrossRef]
- Sarkisov, L.; Monson, P. Modeling of adsorption and desorption in pores of simple geometry using molecular dynamics. Langmuir 2001, 17, 7600–7604. [Google Scholar] [CrossRef]
- Libby, B.; Monson, P. Adsorption/desorption hysteresis in inkbottle pores: A density functional theory and Monte Carlo simulation study. Langmuir 2004, 20, 4289–4294. [Google Scholar] [CrossRef]
- Coasne, B.; Galarneau, A.; Pellenq, R.J.; Di Renzo, F. Adsorption, intrusion and freezing in porous silica: The view from the nanoscale. Chem. Soc. Rev. 2013, 42, 4141–4171. [Google Scholar] [CrossRef] [PubMed]
- Mecke, K.R. Integral geometry in statistical physics. Int. J. Mod. Phys. B 1998, 12, 861–899. [Google Scholar] [CrossRef]
- Bañados, M.; Teitelboim, C.; Zanelli, J. Black hole entropy and the dimensional continuation of the Gauss-Bonnet theorem. Phys. Rev. Lett. 1994, 72, 957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmalzing, J.; Górski, K.M. Minkowski functionals used in the morphological analysis of cosmic microwave background anisotropy maps. Mon. Not. R. Astron. Soc. 1998, 297, 355–365. [Google Scholar] [CrossRef] [Green Version]
- Mecke, K.; Wagner, H. Euler characteristic and related measures for random geometric sets. J. Stat. Phys. 1991, 64, 843–850. [Google Scholar] [CrossRef]
- Mecke, K.R.; Sofonea, V. Morphology of spinodal decomposition. Phys. Rev. E 1997, 56, R3761. [Google Scholar] [CrossRef] [Green Version]
- Scheel, M.; Seemann, R.; Brinkmann, M.; Di Michiel, M.; Sheppard, A.; Breidenbach, B.; Herminghaus, S. Morphological clues to wet granular pile stability. Nat. Mater. 2008, 7, 189. [Google Scholar] [CrossRef]
- Saadatfar, M.; Takeuchi, H.; Robins, V.; Francois, N.; Hiraoka, Y. Pore configuration landscape of granular crystallization. Nat. Commun. 2017, 8, 15082. [Google Scholar] [CrossRef]
- Vogel, H.J. Topological characterization of porous media. In Morphology of Condensed Matter; Springer: Berlin/Heidelberg, Germany, 2002; pp. 75–92. [Google Scholar] [CrossRef]
- Schlüter, S.; Berg, S.; Rücker, M.; Armstrong, R.; Vogel, H.J.; Hilfer, R.; Wildenschild, D. Pore-scale displacement mechanisms as a source of hysteresis for two-phase flow in porous media. Water Resour. Res. 2016, 52, 2194–2205. [Google Scholar] [CrossRef] [Green Version]
- Mecke, K.R. Additivity, convexity, and beyond: Applications of Minkowski Functionals in statistical physics. In Statistical Physics and Spatial Statistics; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2000; pp. 111–184. [Google Scholar] [CrossRef]
- Hadwiger, H. Vorlesungen über Inhalt, Oberfläche und Isoperimetrie; Springer: Berlin, Germany, 1957; Volume 93. [Google Scholar] [CrossRef]
- König, P.M.; Roth, R.; Mecke, K. Morphological thermodynamics of fluids: Shape dependence of free energies. Phys. Rev. Lett. 2004, 93, 160601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simeski, F.; Boelens, A.M.; Ihme, M. Modeling Adsorption in Silica Pores via Minkowski Functionals and Molecular Electrostatic Moments. Energies 2020, 13, 5976. [Google Scholar] [CrossRef]
- Evans, R. The nature of the liquid-vapour interface and other topics in the statistical mechanics of non-uniform, classical fluids. Adv. Phys. 1979, 28, 143–200. [Google Scholar] [CrossRef]
- Kierlik, E.; Monson, P.; Rosinberg, M.; Tarjus, G. Adsorption hysteresis and capillary condensation in disordered porous solids: A density functional study. J. Phys. Condens. Matter 2002, 14, 9295. [Google Scholar] [CrossRef]
- Ustinov, E.; Do, D.; Jaroniec, M. Adsorption of argon and nitrogen in cylindrical pores of MCM-41 materials: Application of density functional theory. Appl. Surf. Sci. 2005, 252, 1013–1028. [Google Scholar] [CrossRef]
- Neimark, A.V.; Lin, Y.; Ravikovitch, P.I.; Thommes, M. Quenched solid density functional theory and pore size analysis of micro-mesoporous carbons. Carbon 2009, 47, 1617–1628. [Google Scholar] [CrossRef]
- Yatsyshin, P.; Durán-Olivencia, M.; Kalliadasis, S. Microscopic aspects of wetting using classical density functional theory. J. Phys. Condens. Matter 2018, 30, 274003. [Google Scholar] [CrossRef] [Green Version]
- Minkowski, H. Volumen und Oberfläche. Math. Ann. 1903, 57, 447–495. [Google Scholar] [CrossRef] [Green Version]
- Schneider, R. Convex Bodies: The Brunn–Minkowski Theory; Cambridge University Press: Cambridge, UK, 2013; Volume 151. [Google Scholar] [CrossRef]
- Legland, D.; Kiêu, K.; Devaux, M.F. Computation of Minkowski measures on 2D and 3D binary images. Image Anal. Stereol. 2011, 26, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Kierlik, E.; Monson, P.; Rosinberg, M.; Sarkisov, L.; Tarjus, G. Capillary condensation in disordered porous materials: Hysteresis versus equilibrium behavior. Phys. Rev. Lett. 2001, 87, 055701. [Google Scholar] [CrossRef] [Green Version]
- Ravikovitch, P.I.; Neimark, A.V. Density functional theory model of adsorption on amorphous and microporous silica materials. Langmuir 2006, 22, 11171–11179. [Google Scholar] [CrossRef]
- Mecke, K.; Arns, C. Fluids in porous media: A morphometric approach. J. Phys. Condens. Matter 2005, 17, S503. [Google Scholar] [CrossRef]
- Salinger, A.G.; Douglas Frink, L.J. Rapid analysis of phase behavior with density functional theory. I. Novel numerical methods. J. Chem. Phys. 2003, 118, 7457–7465. [Google Scholar] [CrossRef]
- Evans, R.; Marini Bettolo Marconi, U. Phase equilibria and solvation forces for fluids confined between parallel walls. J. Chem. Phys. 1987, 86, 7138–7148. [Google Scholar] [CrossRef]
- Evans, R. Fluids adsorbed in narrow pores: Phase equilibria and structure. J. Phys. Condens. Matter 1990, 2, 8989. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous electron gas. Phys. Rev. 1964, 136, B864. [Google Scholar] [CrossRef] [Green Version]
- Heroux, M.A.; Salinger, A.G.; Frink, L.J. Parallel segregated Schur complement methods for fluid density functional theories. SIAM J. Sci. Comput. 2007, 29, 2059–2077. [Google Scholar] [CrossRef] [Green Version]
- Frink, L.J.D.; Salinger, A.G. Two-and three-dimensional nonlocal density functional theory for inhomogeneous fluids: I. Algorithms and parallelization. J. Comput. Phys. 2000, 159, 407–424. [Google Scholar] [CrossRef]
- Frink, L.J.D.; Salinger, A.G. Two-and three-dimensional nonlocal density functional theory for inhomogeneous fluids: II. Solvated polymers as a benchmark problem. J. Comput. Phys. 2000, 159, 425–439. [Google Scholar] [CrossRef]
- Weeks, J.D.; Chandler, D.; Andersen, H.C. Role of repulsive forces in determining the equilibrium structure of simple liquids. J. Chem. Phys. 1971, 54, 5237–5247. [Google Scholar] [CrossRef]
- Hansen-Goos, H.; Roth, R. Density functional theory for hard-sphere mixtures: The White Bear version mark II. J. Phys. Condens. Matter 2006, 18, 8413. [Google Scholar] [CrossRef]
- Keller, H.B. Numerical solution of bifurcation and nonlinear eigenvalue problems. In Applications of Bifurcation Theory; Rabinowitz, P., Ed.; Academic Press: New York, NY, USA, 1977; pp. 359–384. [Google Scholar]
- Salinger, A.G.; Bou-Rabee, N.M.; Pawlowski, R.P.; Wilkes, E.D.; Burroughs, E.A.; Lehoucq, R.B.; Romero, L.A. LOCA 1.0 Library of Continuation Algorithms: Theory and Implementation Manual; SAND2002-0396; Sandia National Laboratories: Albuquerque, NM, USA, 2002.
- Mecke, K.R.; Stoyan, D. Morphology of Condensed Matter: Physics and Geometry of Spatially Complex Systems; Lecture Notes in Physics; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2008; Volume 600. [Google Scholar] [CrossRef]
- Scholz, C.; Wirner, F.; Götz, J.; Rüde, U.; Schröder-Turk, G.E.; Mecke, K.; Bechinger, C. Permeability of porous materials determined from the Euler characteristic. Phys. Rev. Lett. 2012, 109, 264504. [Google Scholar] [CrossRef] [Green Version]
- Brewer, D.; Champeney, D. Sorption of helium and nitrogen on Vycor porous glass. Proc. Phys. Soc. 1962, 79, 855. [Google Scholar] [CrossRef]
- Levitz, P.; Ehret, G.; Sinha, S.; Drake, J. Porous Vycor glass: The microstructure as probed by electron microscopy, direct energy transfer, small-angle scattering, and molecular adsorption. J. Chem. Phys. 1991, 95, 6151–6161. [Google Scholar] [CrossRef]
- Boher, C.; Martin, I.; Lorente, S.; Frizon, F. Experimental investigation of gas diffusion through monomodal materials. Application to geopolymers and Vycor® glasses. Microporous Mesoporous Mater. 2014, 184, 28–36. [Google Scholar] [CrossRef]
- Ravikovitch, P.I.; Haller, G.L.; Neimark, A.V. Density functional theory model for calculating pore size distributions: Pore structure of nanoporous catalysts. Adv. Colloid Interface Sci. 1998, 76, 203–226. [Google Scholar] [CrossRef]
- Gelb, L.D.; Gubbins, K. Characterization of porous glasses: Simulation models, adsorption isotherms, and the Brunauer-Emmett-Teller analysis method. Langmuir 1998, 14, 2097–2111. [Google Scholar] [CrossRef]
- Jaroniec, M.; Kruk, M.; Olivier, J.P. Standard nitrogen adsorption data for characterization of nanoporous silicas. Langmuir 1999, 15, 5410–5413. [Google Scholar] [CrossRef]
- Hamada, Y.; Koga, K.; Tanaka, H. Phase equilibria and interfacial tension of fluids confined in narrow pores. J. Chem. Phys. 2007, 127, 084908. [Google Scholar] [CrossRef] [Green Version]
- Neimark, A.V.; Ravikovitch, P.I.; Vishnyakov, A. Bridging scales from molecular simulations to classical thermodynamics: Density functional theory of capillary condensation in nanopores. J. Phys. Condens. Matter 2003, 15, 347. [Google Scholar] [CrossRef]
- McClure, J.E.; Armstrong, R.T.; Berrill, M.A.; Schlüter, S.; Berg, S.; Gray, W.G.; Miller, C.T. Geometric state function for two-fluid flow in porous media. Phys. Rev. Fluids 2018, 3, 084306. [Google Scholar] [CrossRef]
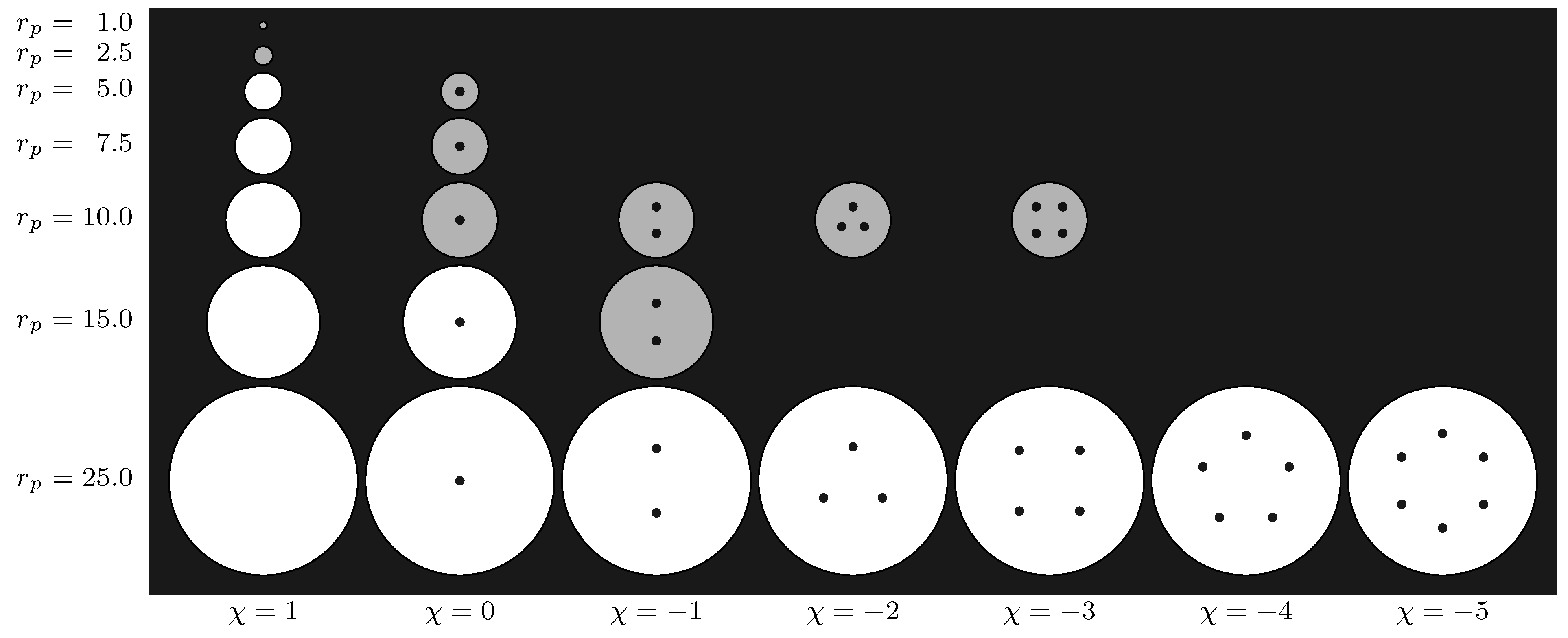









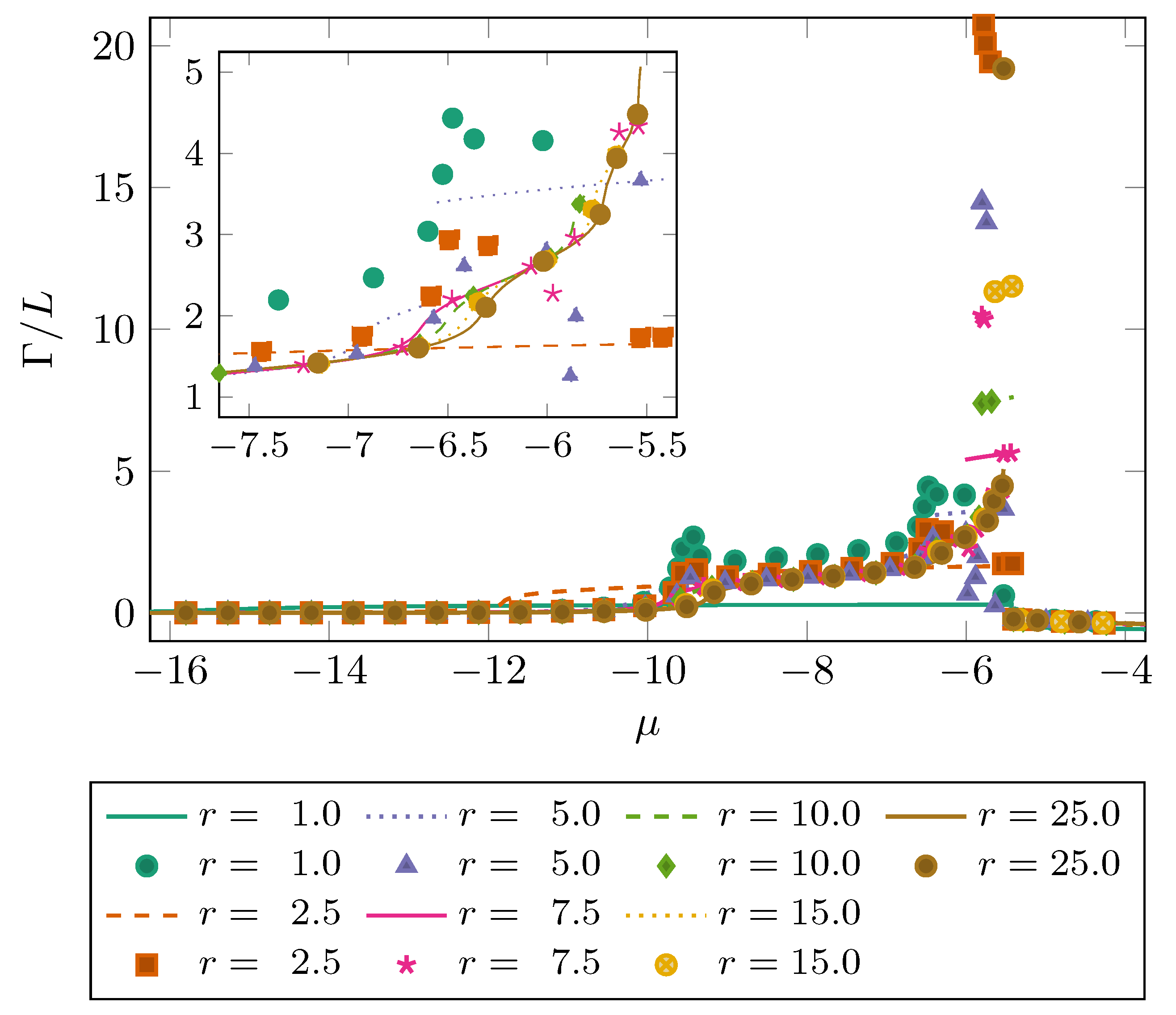

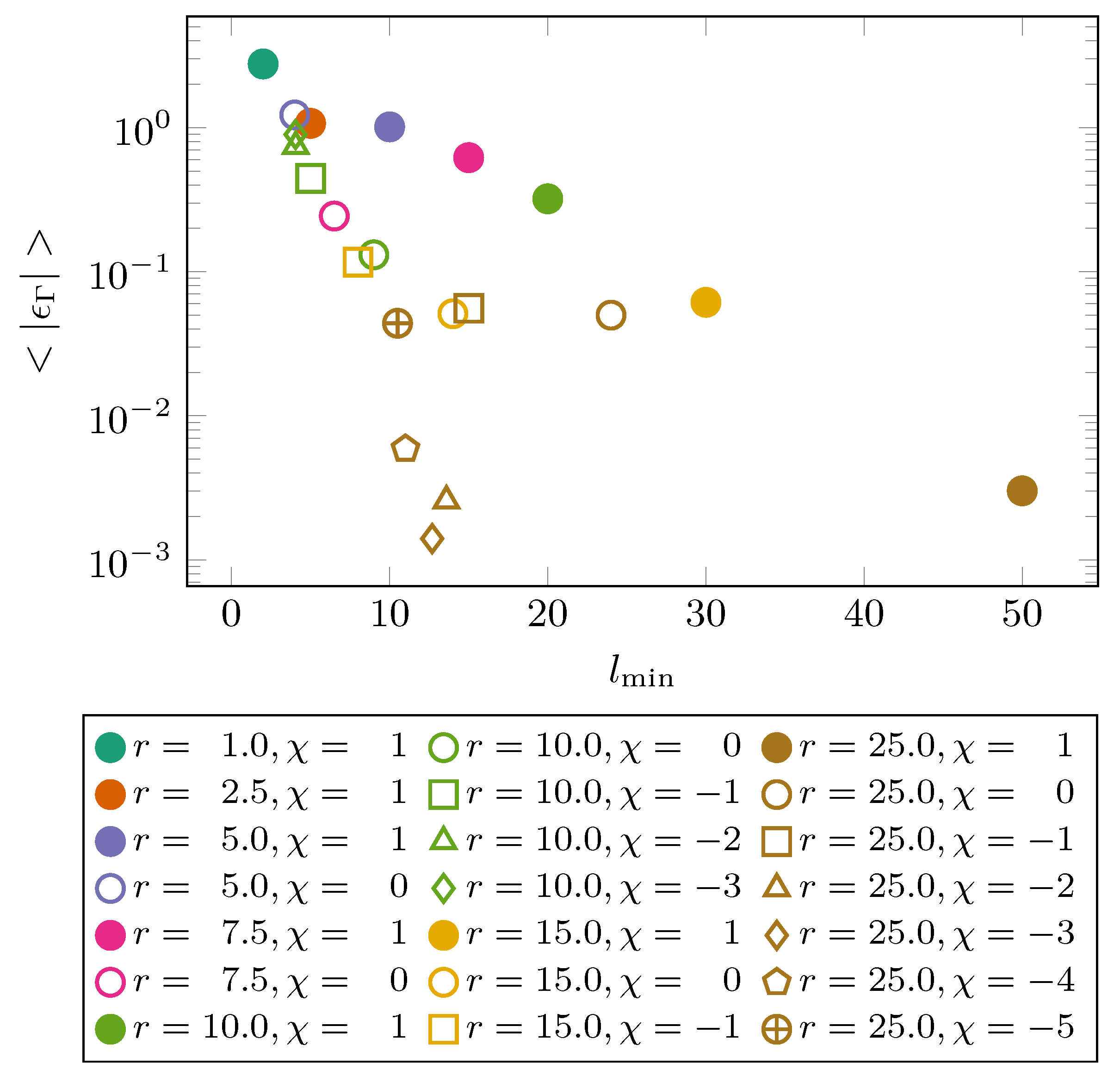
| N2 | 94.45 | 0.3575 | 0.3575 | 147.3 | 0.317 |
| Description | Symbol | Dimensionless Definition |
|---|---|---|
| Distance | ||
| Density | ||
| Grand potential | ||
| Excess adsorption | ||
| Chemical potential | ||
| Pressure | p | |
| Surface tension | ||
| Bending rigidity | ||
| Pseudo pressure | ||
| Pseudo surface tension |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boelens, A.M.P.; Tchelepi, H.A. The Effect of Topology on Phase Behavior under Confinement. Processes 2021, 9, 1220. https://doi.org/10.3390/pr9071220
Boelens AMP, Tchelepi HA. The Effect of Topology on Phase Behavior under Confinement. Processes. 2021; 9(7):1220. https://doi.org/10.3390/pr9071220
Chicago/Turabian StyleBoelens, Arnout M. P., and Hamdi A. Tchelepi. 2021. "The Effect of Topology on Phase Behavior under Confinement" Processes 9, no. 7: 1220. https://doi.org/10.3390/pr9071220
APA StyleBoelens, A. M. P., & Tchelepi, H. A. (2021). The Effect of Topology on Phase Behavior under Confinement. Processes, 9(7), 1220. https://doi.org/10.3390/pr9071220






