Sequential Hydrothermal Processing of Sewage Sludge to Produce Low Nitrogen Biocrude
Abstract
1. Introduction
2. Materials and Methods
2.1. Biomass and Chemicals
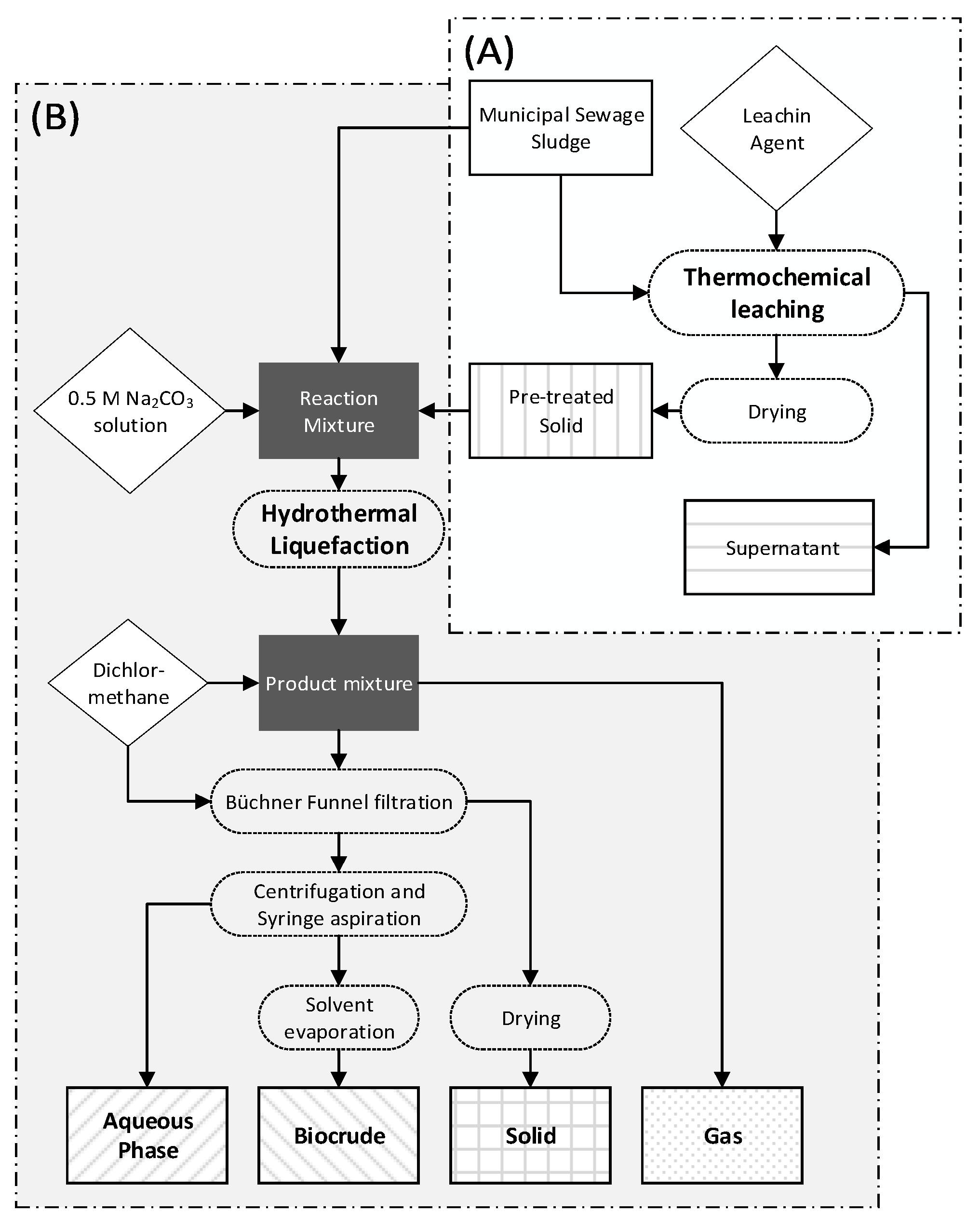
2.2. Pre-Treatment
2.3. Hydrothermal Liquefaction
2.4. Bulk Analysis of Feedstock, Pre-Treated Solids and HTL-Products
2.5. Chemical Composition of Biocrude
2.6. Calculations
3. Results
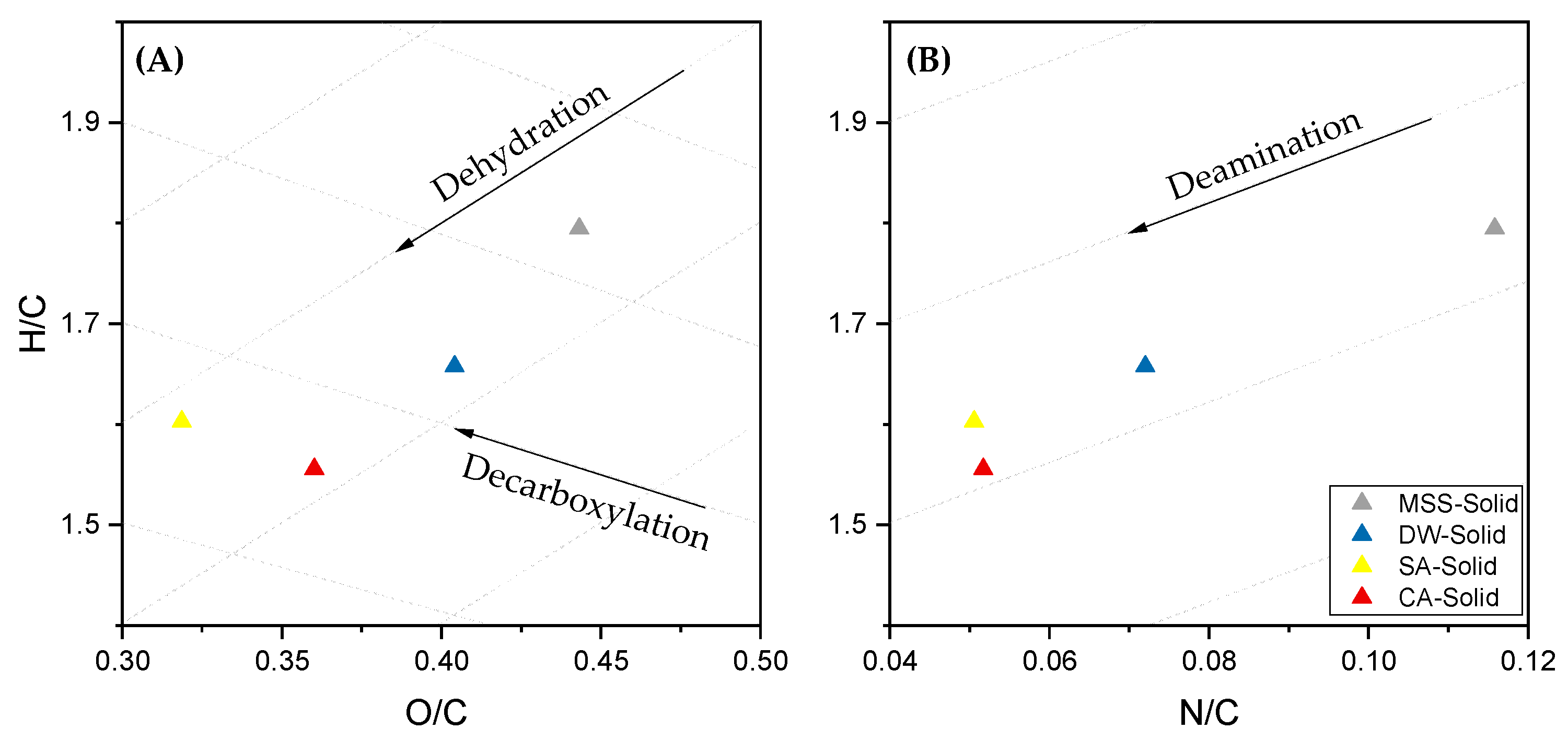
3.1. Effects of Pre-Treatment on Sewage Sludge
3.2. Effect of Pre-Treatment on the Yield and the Elemental Composition of HTL-Products
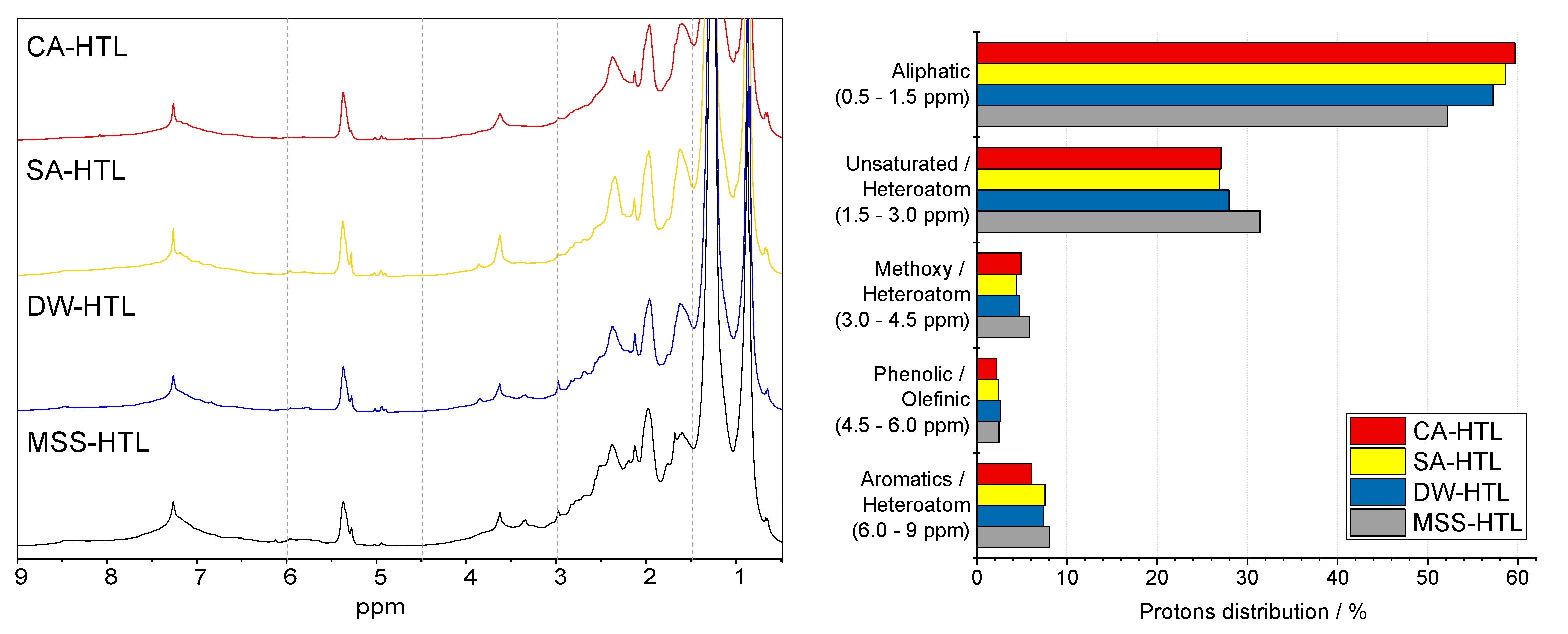
3.3. Effect of Pre-Treatment on the Chemical Composition of HTL-Biocrude
4. Conclusions
- (1)
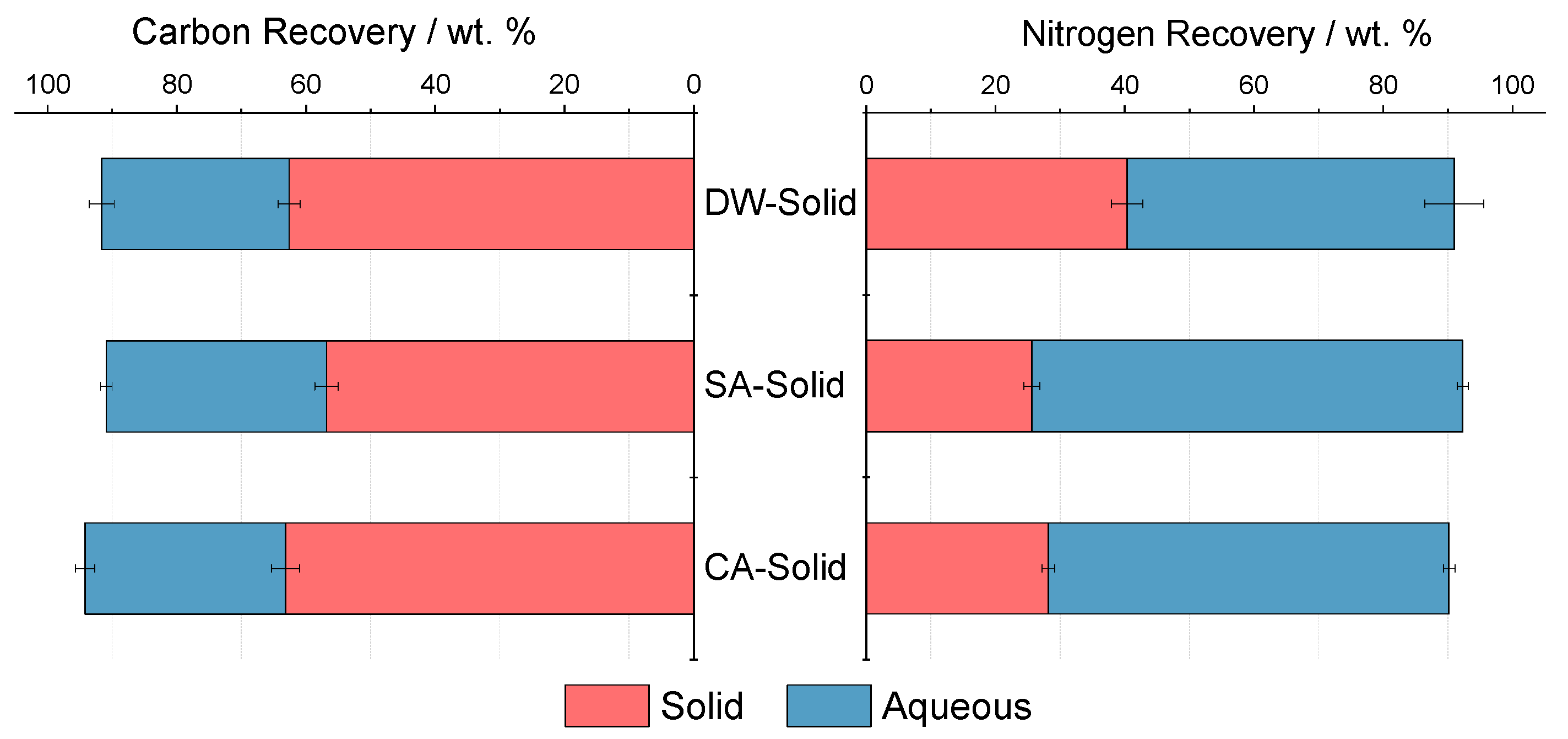
- Bulk analysis of the products from pre-treatment shows a reduction in nitrogen due to solubilization of proteins in the aqueous supernatant, while crude lipids are retained. With the addition of an acidic agent, the solubilization of nitrogen is enhanced.
- (2)
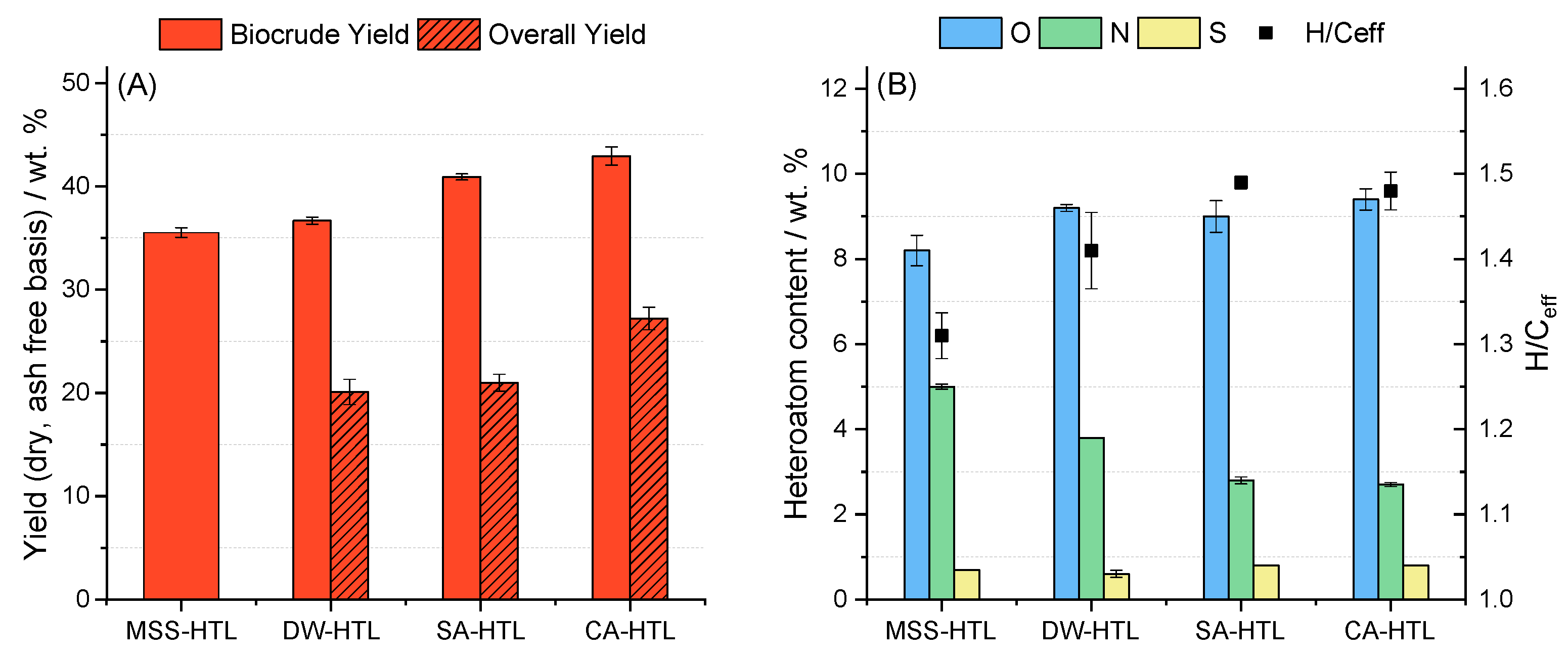
- The yield of hydrothermal liquefaction can be increased due to the hydrothermal acid pre-treatment, under significant concomitant solubilization of organic matter in the pre-treatment. This is reflected in the overall yield after the combined process. An in-depth discussion on potential valorization or recovery of the solubilized organics of the pre-treatment is out of scope of this study, but will be an issue of further studies.
- (3)
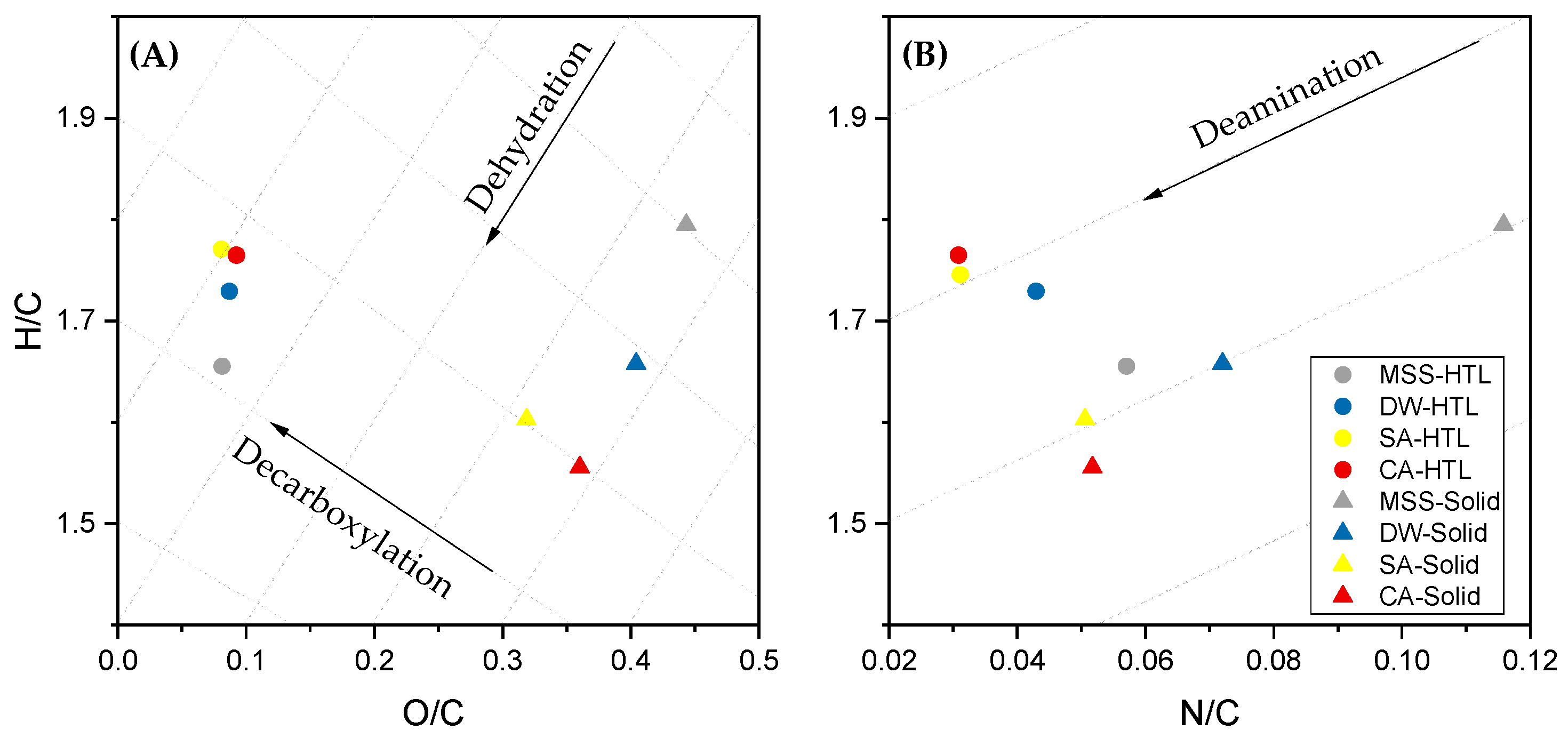
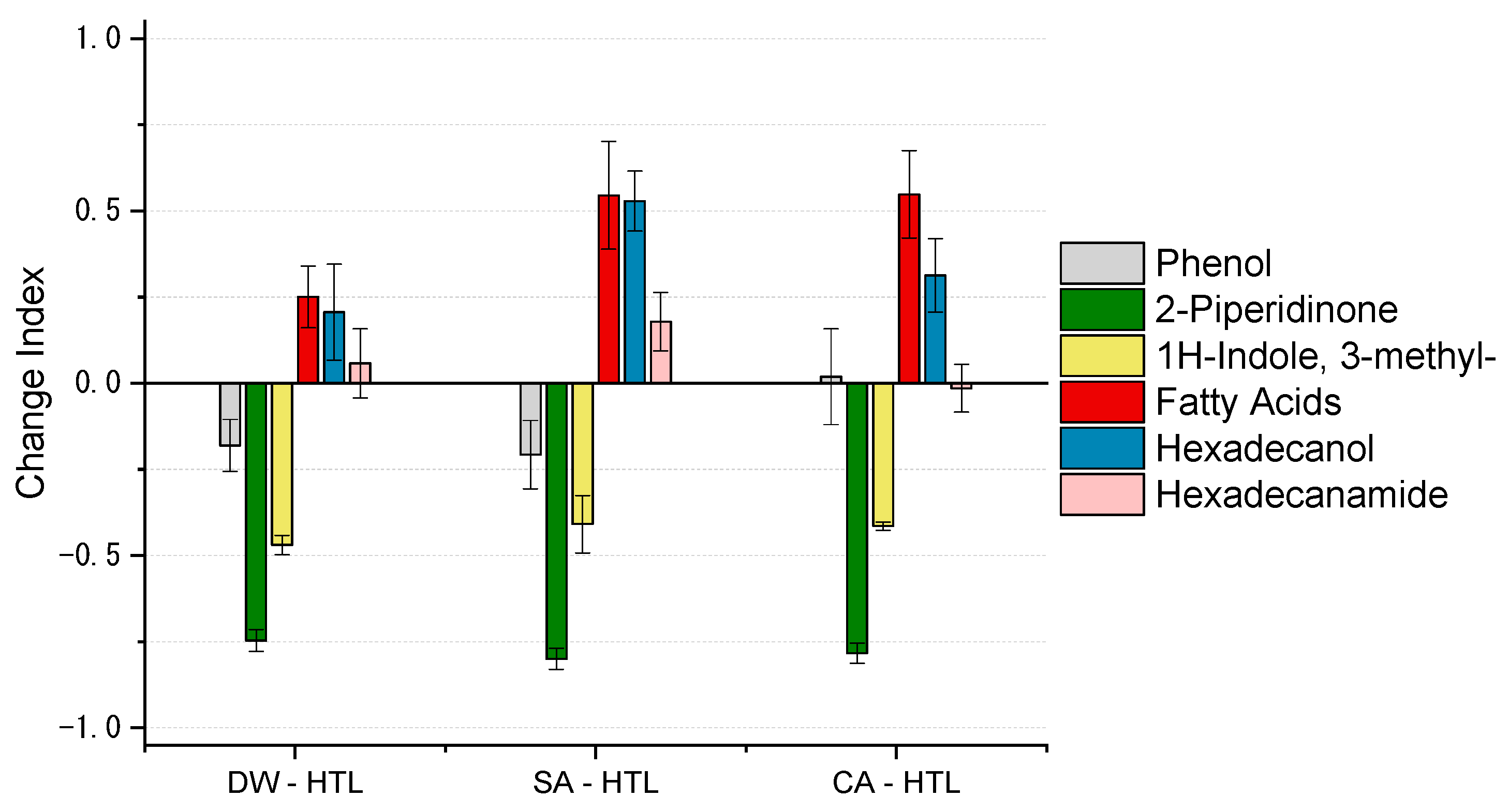
- Biocrude quality was increased due to a pre-treatment as the N-heteroatom content is reduced, H/Ceff ratio is increased. Furthermore, long-chain aliphatic structures are accumulated and N-heteroatoms are primarily in the form of fatty acid amides. Directly, hydrothermal liquefied sewage sludge suffers from N-heteroatoms bound in a cyclic or even aromatic structure which can affect further upgrading strategies.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eurostat. Sewage Sludge Production and Disposal in the EU. 2020. Available online: http://appsso.eurostat.ec.europa.eu/nui/submitViewTableAction.do (accessed on 8 August 2020).
- Collivignarelli, M.C.; Abbà, A.; Miino, M.C.; Torretta, V. What advanced treatments can be used to minimize the production of sewage sludge in WWTPs? Appl. Sci. 2019, 9, 2650. [Google Scholar] [CrossRef]
- Kelessidis, A.; Stasinakis, A.S. Comparative study of the methods used for treatment and final disposal of sewage sludge in European countries. Waste Manag. 2012, 32, 1186–1195. [Google Scholar] [CrossRef]
- European Parliament; Council of the European Union. Directive (EU) 2018/851 of the European Parliament and of the Council of 30 May 2018 amending Directive 2008/98/EC on waste. Off. J. Eur. Union L-150/109–140 2018, 150, 109–140. [Google Scholar]
- Collivignarelli, M.C.; Abbà, A.; Frattarola, A.; Carnevale Miino, M.; Padovani, S.; Katsoyiannis, I.; Torretta, V. Legislation for the reuse of biosolids on agricultural land in Europe: Overview. Sustainability 2019, 11, 6015. [Google Scholar] [CrossRef]
- Mahon, A.M.; O’Connell, B.; Healy, M.G.; O’Connor, I.; Officer, R.; Nash, R.; Morrison, L. Microplastics in Sewage Sludge: Effects of treatment. Environ. Sci. Technol. 2017, 51, 810–818. [Google Scholar] [CrossRef]
- Verlicchi, P.; Zambello, E. Pharmaceuticals and personal care products in untreated and treated sewage sludge: Occurrence and environmental risk in the case of application on soil—A critical review. Sci. Total Environ. 2015, 538, 750–767. [Google Scholar] [CrossRef]
- Thomsen, M.; Seghetta, M.; Mikkelsen, M.H.; Gyldenkærne, S.; Becker, T.; Caro, D.; Frederiksen, P. Comparative life cycle assessment of biowaste to resource management systems–A Danish case study. J. Clean Prod. 2017, 142, 4050–4058. [Google Scholar] [CrossRef]
- Hao, X.; Chen, Q.; van Loosdrecht, M.C.; Li, J.; Jiang, H. Sustainable disposal of excess sludge: Incineration without anaerobic digestion. Water Res. 2020, 170. [Google Scholar] [CrossRef]
- Manara, P.; Zabaniotou, A. Towards sewage sludge based biofuels via thermochemical conversion–A review. Renew. Sustain. Energy Rev. 2012, 16, 2566–2582. [Google Scholar] [CrossRef]
- Dimitriadis, A.; Bezergianni, S. Hydrothermal liquefaction of various biomass and waste feedstocks for biocrude production: A state of the art review. Renew. Sustain. Energy Rev. 2017, 68, 113–125. [Google Scholar] [CrossRef]
- Yin, X.; Han, P.; Lu, X.; Wang, Y. A review on the dewaterability of bio-sludge and ultrasound pretreatment. Ultrason Sonochem. 2004, 11, 337–348. [Google Scholar] [CrossRef]
- Kruse, A.; Dahmen, N. Water–A magic solvent for biomass conversion. J. Supercrit Fluids 2015, 96, 36–45. [Google Scholar] [CrossRef]
- Kruse, A.; Dinjus, E. Hot compressed water as reaction medium and reactant. J. Supercrit Fluids 2007, 39, 362–380. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342. [Google Scholar] [CrossRef]
- Munir, M.T.; Mansouri, S.S.; Udugama, I.A.; Baroutian, S.; Gernaey, K.V.; Young, B.R. Resource recovery from organic solid waste using hydrothermal processing: Opportunities and challenges. Renew. Sustain. Energy Rev. 2018, 96, 64–75. [Google Scholar] [CrossRef]
- Conti, F.; Toor, S.S.; Pedersen, T.H.; Seehar, T.H.; Nielsen, A.H.; Rosendahl, L.A. Valorization of animal and human wastes through hydrothermal liquefaction for biocrude production and simultaneous recovery of nutrients. Energy Convers. Manag. 2020, 216, 112925. [Google Scholar] [CrossRef]
- Leng, L.; Zhang, W.; Peng, H.; Li, H.; Jiang, S.; Huang, H. Nitrogen in bio-oil produced from hydrothermal liquefaction of biomass: A review. Chem. Eng. J. 2020, 401, 126030. [Google Scholar] [CrossRef]
- Speight, J.G. Handbook of Petroleum Product Analysis; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Obeid, F.; Chu Van, T.; Brown, R.; Rainey, T. Nitrogen and sulphur in algal biocrude: A review of the HTL process, upgrading, engine performance and emissions. Energy Convers. Manag. 2019, 181, 105–119. [Google Scholar] [CrossRef]
- Akhtar, J.; Amin, N.A.S. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew. Sustain. Energy Rev. 2011, 15, 1615–1624. [Google Scholar] [CrossRef]
- Gu, X.; Yu, L.; Pang, N.; Martinez-Fernandez, J.S.; Fu, X.; Chen, S. Comparative techno-economic analysis of algal biofuel production via hydrothermal liquefaction: One stage versus two stages. Appl. Energy 2020, 259, 114115. [Google Scholar] [CrossRef]
- Barber, W.P.F. Thermal hydrolysis for sewage treatment: A critical review. Water Res. 2016, 104, 53–71. [Google Scholar] [CrossRef]
- Zhuang, X.; Huang, Y.; Song, Y.; Zhan, H.; Yin, X.; Wu, C. The transformation pathways of nitrogen in sewage sludge during hydrothermal treatment. Bioresour. Technol. 2017, 245, 463–470. [Google Scholar] [CrossRef]
- Zhao, P.; Chen, H.; Ge, S.; Yoshikawa, K. Effect of the hydrothermal pretreatment for the reduction of NO emission from sewage sludge combustion. Appl. Energy 2013, 111, 199–205. [Google Scholar] [CrossRef]
- Yoshida, H.; Tavakoli, O. Sub-critical water hydrolysis treatment for waste squid entrails and production of amino acids, organic acids, and fatty acids. J. Chem. Eng. JPN 2004, 37, 253–260. [Google Scholar] [CrossRef]
- Zhu, X.; Zhu, C.; Zhao, L.; Cheng, H. Amino acids production from fish proteins hydrolysis in subcritical water. Chin. J. Chem. Eng. 2008, 16, 456–460. [Google Scholar] [CrossRef]
- Huang, Z.; Wufuer, A.; Wang, Y.; Dai, L. Hydrothermal liquefaction of pretreated low-lipid microalgae for the production of bio-oil with low heteroatom content. Process Biochem. 2018, 69, 136–143. [Google Scholar] [CrossRef]
- Jazrawi, C.; Biller, P.; He, Y.; Montoya, A.; Ross, A.B.; Maschmeyer, T.; Haynes, B.S. Two-stage hydrothermal liquefaction of a high-protein microalga. Algal. Res. 2015, 8, 15–22. [Google Scholar] [CrossRef]
- Guo, B.; Yang, B.; Silve, A.; Akaberi, S.; Scherer, D.; Papachristou, I.; Frey, W.; Hornung, U.; Dahmen, N. Hydrothermal liquefaction of residual microalgae biomass after pulsed electric field-assisted valuables extraction. Algal. Res. 2019, 43, 101650. [Google Scholar] [CrossRef]
- Fan, Y.; Hornung, U.; Raffelt, K.; Dahmen, N. The influence of lipids on the fate of nitrogen during hydrothermal liquefaction of protein-containing biomass. J. Anal. Appl. Pyrolysis 2020, 147, 104798. [Google Scholar] [CrossRef]
- Techne SBL-1, SBL-2, SBL-2D Fluidised Baths; Operator’s Manual. 2003. Available online: http://www.techne-calibration.com/adminimages/Techne_SBL1_2_2D.pdf (accessed on 8 August 2020).
- VDI ev. VDI Heat Atlas; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Olkiewicz, M.; Caporgno, M.P.; Fortuny, A.; Stüber, F.; Fabregat, A.; Font, J.; Bengoa, C. Direct liquid–liquid extraction of lipid from municipal sewage sludge for biodiesel production. Fuel Process Technol. 2014, 128, 331–338. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 152/2009 of 27 January 2009 Laying down the Methods of Sampling and Analysis for the Official Control of Feed. Off. J. Eur. Union. 2009, 54, 1–130. [Google Scholar]
- Bai, L.; Carlton, D.D.; Schug, K.A. Complex mixture quantification without calibration using gas chromatography and a comprehensive carbon reactor in conjunction with flame ionization detection. J. Sep. Sci. 2018, 41, 4031–4037. [Google Scholar] [CrossRef]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar] [CrossRef]
- Mullen, C.A.; Strahan, G.D.; Boateng, A.A. Characterization of various fast-pyrolysis bio-oils by NMR spectroscopy. Energy Fuels 2009, 23, 2707–2718. [Google Scholar] [CrossRef]
- Fulmer, G.R.; Miller, A.J.; Sherden, N.H.; Gottlieb, H.E.; Nudelman, A.; Stoltz, B.M.; Becarw, J.E.; Goldberg, K.I. NMR Chemical shifts of trace impurities: Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 2010, 29, 2176–2179. [Google Scholar] [CrossRef]
- Huber, G.W.; Corma, A. Synergies between Bio- and oil refineries for the production of fuels from biomass. Angew. Chemie Int. Ed. 2007, 46, 7184–7201. [Google Scholar] [CrossRef]
- Park, M.; Kim, N.; Lee, S.; Park, M.; Kim, N.; Lee, S.; Yeon, S.; Seo, J.H.; Park, D. A study of solubilization of sewage sludge by hydrothermal treatment. J. Environ. Manag. 2019, 250, 109490. [Google Scholar] [CrossRef]
- Neyens, E.; Baeyens, J.; Weemaes, M.; De Heyder, B. Hot acid hydrolysis as a potential treatment of thickened sewage sludge. J. Hazard Mater. 2003, 98, 275–293. [Google Scholar] [CrossRef]
- Chen, S.; Dong, B.; Dai, X.; Wang, H.; Li, N.; Yang, D. Effects of thermal hydrolysis on the metabolism of amino acids in sewage sludge in anaerobic digestion. Waste Manag. 2019, 88, 309–318. [Google Scholar] [CrossRef]
- Bougrier, C.; Delgenès, J.P.; Carrère, H. Effects of thermal treatments on five different waste activated sludge samples solubilisation, physical properties and anaerobic digestion. Chem. Eng. J. 2008, 139, 236–244. [Google Scholar] [CrossRef]
- Wilson, C.A.; Novak, J.T. Hydrolysis of macromolecular components of primary and secondary wastewater sludge by thermal hydrolytic pretreatment. Water Res. 2009, 43, 4489–4498. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Liu, H.; Chen, S.; Dichtl, N.; Dai, X.; Li, N. Effects of thermal hydrolysis on organic matter solubilization and anaerobic digestion of high solid sludge. Chem. Eng. J. 2015, 264, 174–180. [Google Scholar] [CrossRef]
- Yuan, T.; Cheng, Y.; Zhang, Z.; Lei, Z.; Shimizu, K. Comparative study on hydrothermal treatment as pre- and post-treatment of anaerobic digestion of primary sludge: Focus on energy balance, resources transformation and sludge dewaterability. Appl. Energy 2019, 239, 171–180. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Amino acids, peptides, proteins. In Food Chemistry; Springer: Berlin/Heidelberg, Germany, 2018; pp. 8–92. [Google Scholar]
- Brunner, G. Processing of biomass with hydrothermal and supercritical water. In Hydrothermal and Supercritical Water Processes; Elsevier: Amsterdam, The Netherlands, 2014; pp. 395–509. [Google Scholar]
- Andersson, E.; Holm, N.G. The stability of some selected amino acids under attempted redox constrained hydrothermal conditions. Orig. Life Evol. Biosph. 2000, 30, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoez, W.; Yoshida, H.; Nakahasi, T. Pathways of amino acid transformation and decomposition in saturated subcritical water conditions. Int. J. Chem. React. Eng. 2010, 8. [Google Scholar] [CrossRef]
- Martins, S.I.F.S.; Van Boekel, M.A.J.S. A kinetic model for the glucose/glycine Maillard reaction pathways. Food Chem. 2005, 90, 257–269. [Google Scholar] [CrossRef]
- Hoffmann, V.; Jung, D.; Zimmermann, J.; Rodriguez Correa, C.; Elleuch, A.; Halouani, K.; Kruse, A. Conductive carbon materials from the hydrothermal carbonization of vineyard residues for the application in electrochemical double-layer capacitors (EDLCs) and direct carbon fuel cells (DCFCs). Materials (Basel) 2019, 12, 1703. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-Y.; Noike, T. Upgrading of anaerobic digestion of waste activated sludge by thermal pretreatment. Water Sci. Technol. 1992, 26, 857–866. [Google Scholar] [CrossRef]
- Zhu, Z.; Toor, S.S.; Rosendahl, L.; Yu, D.; Chen, G. Influence of alkali catalyst on product yield and properties via hydrothermal liquefaction of barley straw. Energy 2015, 80, 284–292. [Google Scholar] [CrossRef]
- Yin, S.; Tan, Z. Hydrothermal liquefaction of cellulose to bio-oil under acidic, neutral and alkaline conditions. Appl. Energy 2012, 92, 234–239. [Google Scholar] [CrossRef]
- Demirbaş, A. Mechanisms of liquefaction and pyrolysis reactions of biomass. Energy Convers. Manag. 2000, 41, 633–646. [Google Scholar] [CrossRef]
- Chornet, E.; Overend, R.P. Biomass liquefaction: An overview. In Fundamentals of Thermochemical Biomass Conversion; Springer: Dordrecht, The Netherlands, 1985; pp. 967–1002. [Google Scholar]
- Chen, W.T.; Qian, W.; Zhang, Y.; Mazur, Z.; Kuo, C.T.; Scheppe, K.; Schideman, L.C.; Sharma, B.K. Effect of ash on hydrothermal liquefaction of high-ash content algal biomass. Algal. Res. 2017, 25, 297–306. [Google Scholar] [CrossRef]
- Ross, A.B.; Biller, P.; Kubacki, M.L.; Lea-Langton, A.; Jones, J.M. Hydrothermal processing of microalgae using alkali and organic acids. Fuel 2010, 89, 2234–2243. [Google Scholar] [CrossRef]
- Yoo, G.; Park, M.S.; Yang, J.-W.; Choi, M. Lipid content in microalgae determines the quality of biocrude and Energy Return On Investment of hydrothermal liquefaction. Appl. Energy 2015, 156, 354–361. [Google Scholar] [CrossRef]
- Shah, A.A.; Toor, S.S.; Conti, F.; Nielsen, A.H.; Rosendahl, L.A. Hydrothermal liquefaction of high ash containing sewage sludge at sub and supercritical conditions. Biomass Bioenergy 2020, 135. [Google Scholar] [CrossRef]
- Zapp, K.H.; Wostbrock, K.H.; Schäfer, M.; Sato, K.; Seiter, H.; Zwick, W.; Creutziger, R.; Leiter, H. Ammonium compounds. Ullmann’s Encycl. Ind. Chem. 2012. [Google Scholar] [CrossRef]
- Qian, L.; Wang, S.; Savage, P.E. Fast and isothermal hydrothermal liquefaction of sludge at different severities: Reaction products, pathways, and kinetics. Appl. Energy 2020, 260, 114312. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A.B. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Fan, Y.; Hornung, U.; Dahmen, N.; Kruse, A. Hydrothermal liquefaction of protein-containing biomass: Study of model compounds for Maillard reactions. Biomass Convers. Biorefinery 2018, 8, 909–923. [Google Scholar] [CrossRef]
- Pińkowska, H.; Wolak, P.; Złocińska, A. Hydrothermal decomposition of alkali lignin in sub- and supercritical water. Chem. Eng. J. 2012, 187, 410–414. [Google Scholar] [CrossRef]
- Watanabe, M.; Iida, T.; Inomata, H. Decomposition of a long chain saturated fatty acid with some additives in hot compressed water. Energy Convers. Manag. 2006, 47, 3344–3350. [Google Scholar] [CrossRef]
- Liu, X.; Yang, M.; Deng, Z.; Dasgupta, A.; Guo, Y. Mechanism and kinetic modeling. Chem. Eng. J. 2021, 407, 126332. [Google Scholar] [CrossRef]
- Garcia Alba, L.; Torri, C.; Samorì, C.; van der Spek, J.; Fabbri, D.; Kersten, S.R.; Brilman, D.W. Hydrothermal Treatment (HTT) of Microalgae: Evaluation of the process as conversion method in an algae biorefinery concept. Energy Fuels 2011, 26, 642–657. [Google Scholar] [CrossRef]
- McCalley, D.V.; Cooke, M.; Nickless, G. Effect of sewage treatment on faecal sterols. Water Res. 1981, 15, 1019–1025. [Google Scholar] [CrossRef]
- Fan, Y.; Fonseca, F.G.; Gong, M.; Hoffmann, A.; Hornung, U.; Dahmen, N. Energy valorization of integrating lipid extraction and hydrothermal liquefaction of lipid-extracted sewage sludge. J. Clean Prod. 2021, 285, 124895. [Google Scholar] [CrossRef]
- Vardon, D.R.; Sharma, B.K.; Scott, J.; Yu, G.; Wang, Z.; Schideman, L.; Zhang, Y.; Strathmann, T.J. Chemical properties of biocrude oil from the hydrothermal liquefaction of Spirulina algae, swine manure, and digested anaerobic sludge. Bioresour. Technol. 2011, 102, 8295–8303. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.N.; Kaneko, T.; Kobayashi, K. Reaction of amino acids in a supercritical water-flow reactor simulating submarine hydrothermal systems. Bull. Chem. Soc. Jpn. 2003, 76, 1171–1178. [Google Scholar] [CrossRef]
- Moldoveanu, S.C. Analytical pyrolysis of other natural organic polymers. In Analytical Pyrolysis of Natural Organic Polymers, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 427–429. [Google Scholar]
- He, C.; Wang, K.; Yang, Y.; Amaniampong, P.N.; Wang, J.Y. Effective nitrogen removal and recovery from dewatered sewage sludge using a novel integrated system of accelerated hydrothermal deamination and air stripping. Environ. Sci. Technol. 2015, 49, 6872–6880. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Luxem, F.J.; Murugan, R.; Greenhill, J.V.; Siskin, M. Aqueous high-temperature chemistry of carbo- and heterocycles. 19. Pyrroles and indoles. Energy Fuels 1992, 6, 450–455. [Google Scholar] [CrossRef]
- Fu, X.; Liao, Y.; Glein, C.R.; Jamison, M.; Hayes, K.; Zaporski, J.; Yang, Z. Direct synthesis of amides from amines and carboxylic acids under hydrothermal conditions. ACS Earth Space Chem. 2020, 4, 722–729. [Google Scholar] [CrossRef]
- Gai, C.; Zhang, Y.; Chen, W.T.; Zhang, P.; Dong, Y. Energy and nutrient recovery efficiencies in biocrude oil produced via hydrothermal liquefaction of Chlorella pyrenoidosa. RSC Adv. 2014, 4, 16958. [Google Scholar] [CrossRef]
- Ingram, L.; Mohan, D.; Bricka, M.; Steele, P.; Strobel, D.; Crocker, D.; Mitchell, B.; Mohammed, J.; Cantrell, K.; Pittman, C.U., Jr. Pyrolysis of wood and bark in an auger reactor: Physical properties and chemical analysis of the produced bio-oils. Energy Fuels 2018, 22, 614–625. [Google Scholar] [CrossRef]
| MSS | DW-Solid | SA-Solid | CA-Solid | |
|---|---|---|---|---|
| Proximate analysis (dry basis) | ||||
| Ash [wt. %] | 30.0 (0.1) | 38.1 (0.3) | 30.5 (0.7) | 24.7 (0.1) |
| Organic a [wt. %] | 70.0 (0.1) | 61.9 (0.3) | 69.5 (0.7) | 75.3 (0.1) |
| Elemental analysis (dry, ash-free basis) | ||||
| C [wt. %] | 52.8 (0.1) | 56.3 (0.2) | 57.2 (0.5) | 59.4 (0.2) |
| H [wt. %] | 7.9 (0.1) | 7.7 (0.1) | 7.6 (0.1) | 7.7 (0.1) |
| N [wt. %] | 7.1 (0.0) | 4.7 (0.1) | 3.4 (0.1) | 3.6 (0.0) |
| S [wt. %] | 0.9 (0.0) | 0.9 (0.0) | 3.1 (0.2) | 0.8 (0.0) |
| O a [wt. %] | 31.3 (0.0) | 30.4 (0.2) | 28.6 (0.8) | 28.5 (0.1) |
| N/O | 0.26 (0.00) | 0.18 (0.00) | 0.14 (0.00) | 0.14 (0.0) |
| H/Ceff b | 0.55 (0.02) | 0.59 (0.01) | 0.66 (0.03) | 0.67 (0.02) |
| Yield and recovery (dry, ash-free basis) | ||||
| Solid yield [wt. %] | - | 54.8 (3.3) | 52.8 (2.0) | 63.4 (2.2) |
| C-recovery solid phase [wt. %] | - | 62.6 (1.7) | 56.8 (1.8) | 62.7 (2.3) |
| C-recovery aqueous phase [wt. %] | - | 29.0 (1.9) | 34.1 (0.9) | 31.0 (1.5) |
| N-recovery solid phase [wt. %] | - | 40.3 (2.4) | 25.6 (1.2) | 28.2 (1.0) |
| N-recovery aqueous phase [wt. %] | - | 50.6 (4.5) | 66.7 (0.9) | 62.0 (0.7) |
| Organic composition (dry, ash-free basis) | ||||
| Hydrolytic amino acids [wt. %] | 21.8 | 13.5 | 10.0 | 9.3 |
| Lipids c [wt. %] | 21.7 (1.1) | 24.4 (1.1) | 29.9 (1.4) | 28.6 (0.6) |
| Ash analysis (dry, organic-free basis) | ||||
| Al [wt. %] | 2.5 | 2.6 | 2.3 | 2.4 |
| Ca [wt. %] | 7.6 | 7.6 | 5.6 | 2.8 |
| Fe [wt. %] | 18.0 | 18.1 | 4.5 | 5.7 |
| Mg [wt. %] | 0.9 | 0.8 | 0.3 | 0.8 |
| P [wt. %] | 8.0 | 8.1 | 3.4 | 2.8 |
| MSS-HTL | DW-HTL | SA-HTL | CA-HTL | |
|---|---|---|---|---|
| Biocrude | ||||
| Yield [wt. %] | 35.5 (0.5) | 36.6 (0.2) | 39.4 (0.2) | 42.9 (0.7) |
| C [wt. %] | 75.6 (0.2) | 75.5 (0.2) | 76.1 (0.2) | 75.9 (0.1) |
| H [wt. %] | 10.4 (0.2) | 10.9 (0.2) | 11.2 (0.0) | 11.2 (0.1) |
| N [wt. %] | 5.0 (0.1) | 3.8 (0.0) | 2.8 (0.1) | 2.7 (0.0) |
| S [wt. %] | 0.7 (0.0) | 0.6 (0.1) | 0.8 (0.0) | 0.8 (0.0) |
| O a [wt. %] | 8.2 (0.4) | 9.2 (0.1) | 9.0 (0.4) | 9.4 (0.2) |
| H/Ceff b | 1.31 (0.03) | 1.41 (0.04) | 1.49 (0.01) | 1.48 (0.02) |
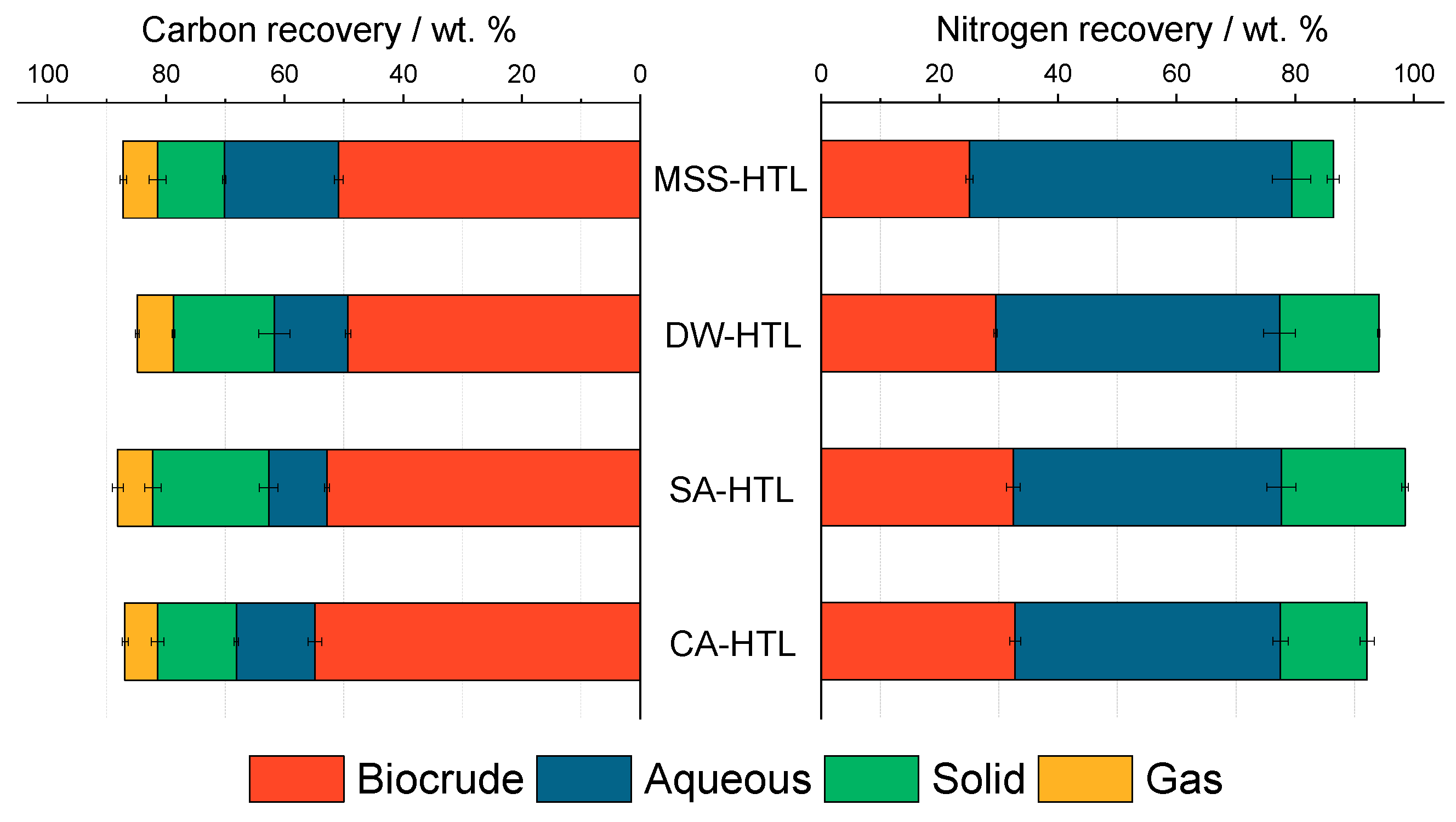
| C-recovery [wt. %] | 50.9 (0.8) | 49.3 (0.5) | 52.8 (0.5) | 54.9 (1.2) |
| N-recovery [wt. %] | 25.1 (0.6) | 29.5 (0.3) | 32.5 (1.2) | 32.7 (0.9) |
| Aqueous | ||||
| C-recovery [wt. %] | 19.3 (0.3) | 12.4 (2.6) | 10.0 (1.6) | 13.3 (0.4) |
| N-recovery [wt. %] | 54.3 (3.2) | 47.9 (2.7) | 45.2 (2.4) | 44.7 (1.3) |
| Solid | ||||
| C-recovery [wt. %] | 11.0 (1.2) | 17.0 (0.2) | 19.6 (1.6) | 13.3 (1.1) |
| N-recovery [wt. %] | 7.1 (1.0) | 16.7 (0.2) | 20.8 (0.6) | 14.6 (1.2) |
| Gas | ||||
| C-recovery [wt. %] | 5.3 (0.5) | 6.1 (0.3) | 6.0 (0.9) | 5.5 (0.5) |
| RT | Compound Name | MSS-HTL | DE-HTL | SA-HTL | CA-HTL | Class |
|---|---|---|---|---|---|---|
| 8.52 | Phenol | x | x | x | x | Phenol |
| 9.73 | 2-Cyclopenten-1-one, 2,3-dimethyl- | x | x | x | Ketone | |
| 10.32 | 2-Pyrrolidinone | x | x | N-heterocycle | ||
| 10.46 | p-Cresol | x | x | x | x | Phenol |
| 10.71 | 2,5-Pyrrolidinedione, 1-methyl- | x | x | N-heterocycle | ||
| 10.78 | Phenol, 2-methoxy- | x | x | Phenol | ||
| 10.78 | Phenol, 4-methoxy- | x | Phenol | |||
| 10.78 | 2-Cyclopenten-1-one, 3,4,5-trimethyl- | x | x | Ketone | ||
| 11.05 | 2-Pyrrolidinone, 1-propyl- | x | x | N-heterocycle | ||
| 12.35 | Phenol, 4-ethyl- | x | x | x | x | Phenol |
| 12.65 | 2-Piperidinone | x | N-heterocycle | |||
| 12.85 | Phenol, 2-methoxy-3-methyl- | x | Phenol | |||
| 14.91 | Indole | x | N-heterocycle | |||
| 15.78 | Piperidine, 1-pentyl- | x | x | x | N-heterocycle | |
| 16.29 | N-[2-Hydroxyethyl]succinimide | x | x | N-heterocycle | ||
| 16.66 | 1H-Indole, 3-methyl- | x | x | x | x | N-heterocycle |
| 17.96 | 1-Dodecanol | x | x | Aliphatic alcohol | ||
| 18.25 | 1-Pentadecene | x | x | Aliphatic alkene | ||
| 19.53 | 1-Tetradecanol | x | x | x | Aliphatic alcohol | |
| 22.01 | 1-Heptadecene | x | x | x | Aliphatic alkene | |
| 23.73 | Tetradecanoic acid | x | x | x | x | Aliphatic alcohol |
| 24.17 | 1-Hexadecanol | x | x | x | x | Aliphatic alcohol |
| 27.83 | Hexadecenoic acid | x | x | x | x | Fatty acid |
| 28.31 | Hecadecanoic acid | x | x | x | x | Fatty acid |
| 30.50 | Octadecen-1-ol | x | x | x | x | Aliphatic alcohol |
| 26.88 | 1-Octadecanol | x | x | x | x | Aliphatic alcohol |
| 32.39 | Octadecenoic acid | x | x | x | x | Fatty acid |
| 32.82 | Octadecanoic acid | x | x | x | x | Fatty acid |
| 28.12 | Hexadecanamide | x | x | x | x | Fatty acid amide |
| 28.51 | Hexadecanamide, N-methyl | x | x | x | x | Fatty acid amide |
| 29.03 | Hexadecanamide, N-ethyl | x | x | x | x | Fatty acid amide |
| 37.11 | Octadecenamide | x | x | x | x | Fatty acid amide |
| 37.92 | Octadecenamide, N-methyl | x | x | x | x | Fatty acid amide |
| 38.77 | Octadecenamide, N-ethyl | x | x | x | x | Fatty acid amide |
| 47.41 | Cholest-3-ene, (5.alpha.)- | x | x | x | x | Sterol |
| 48.19 | Cholest-2-ene, (5.alpha.)- | x | x | x | x | Sterol |
| 48.64 | Cholest-4-ene | x | x | x | x | Sterol |
| 49.05 | Cholest-5-ene | x | x | x | x | Sterol |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zimmermann, J.; Raffelt, K.; Dahmen, N. Sequential Hydrothermal Processing of Sewage Sludge to Produce Low Nitrogen Biocrude. Processes 2021, 9, 491. https://doi.org/10.3390/pr9030491
Zimmermann J, Raffelt K, Dahmen N. Sequential Hydrothermal Processing of Sewage Sludge to Produce Low Nitrogen Biocrude. Processes. 2021; 9(3):491. https://doi.org/10.3390/pr9030491
Chicago/Turabian StyleZimmermann, Joscha, Klaus Raffelt, and Nicolaus Dahmen. 2021. "Sequential Hydrothermal Processing of Sewage Sludge to Produce Low Nitrogen Biocrude" Processes 9, no. 3: 491. https://doi.org/10.3390/pr9030491
APA StyleZimmermann, J., Raffelt, K., & Dahmen, N. (2021). Sequential Hydrothermal Processing of Sewage Sludge to Produce Low Nitrogen Biocrude. Processes, 9(3), 491. https://doi.org/10.3390/pr9030491







