The Kinetics Investigation of CO2 Absorption into TEA and DEEA Amine Solutions Containing Carbonic Anhydrase
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
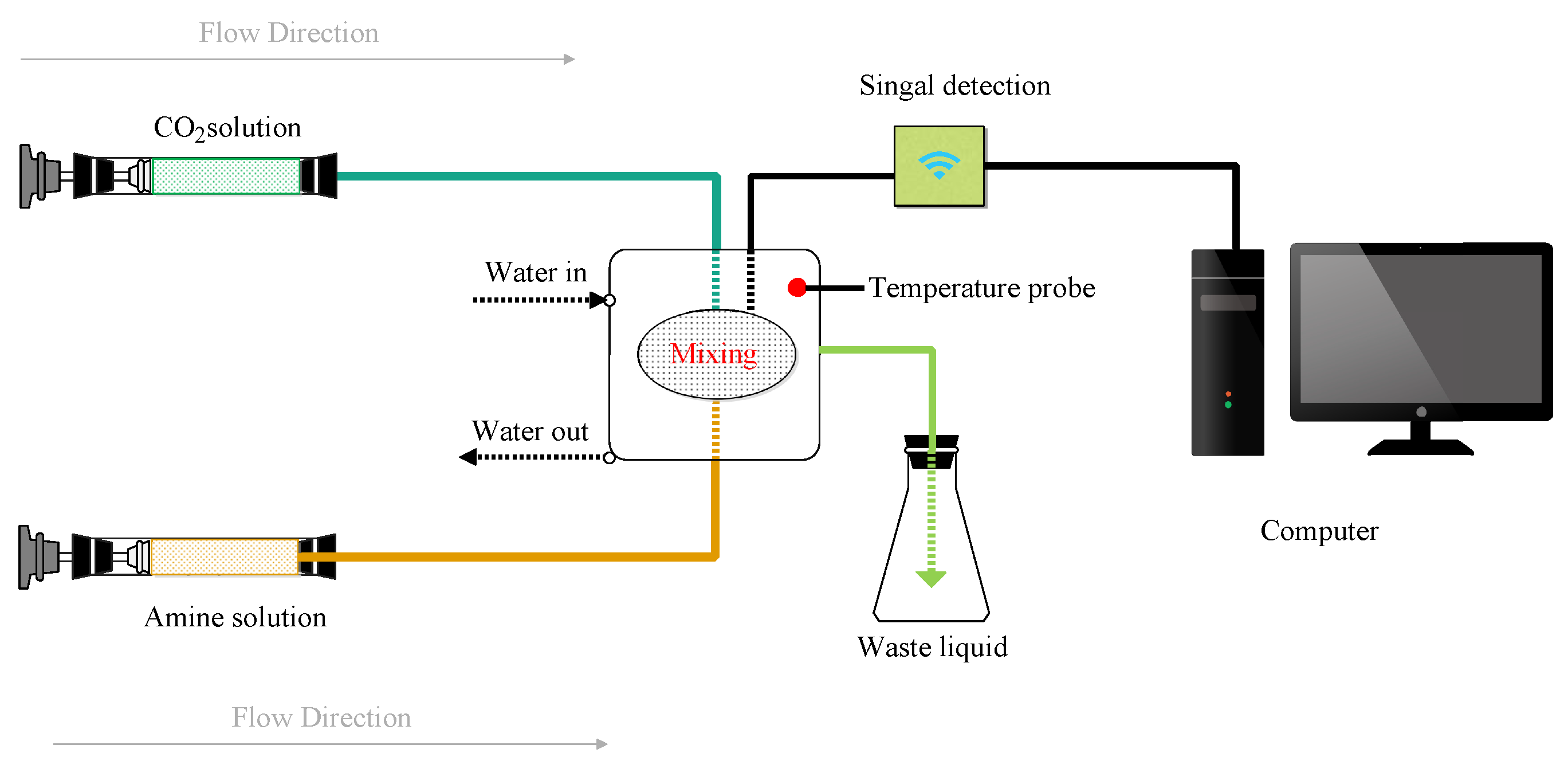
2.2. Experimental Procedure
2.3. Reaction Kinetics of CO2 Absorption
3. Results and Discussion
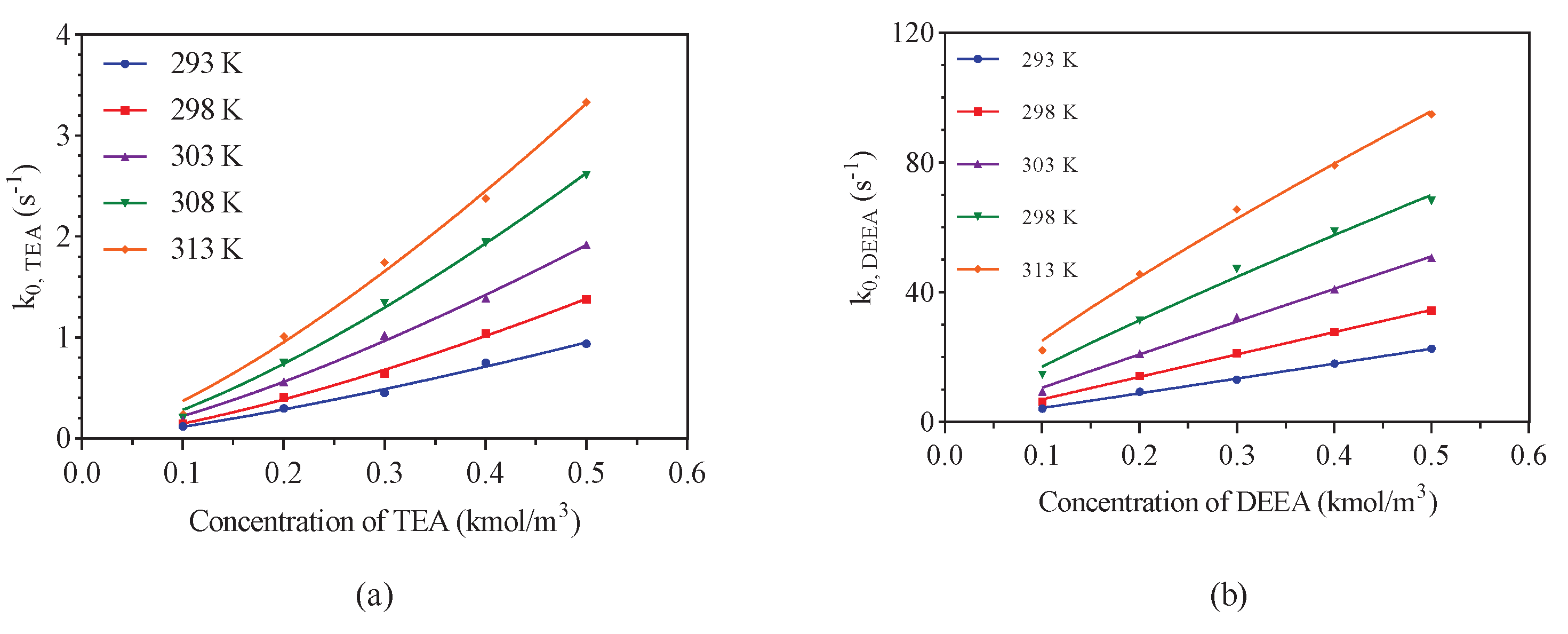
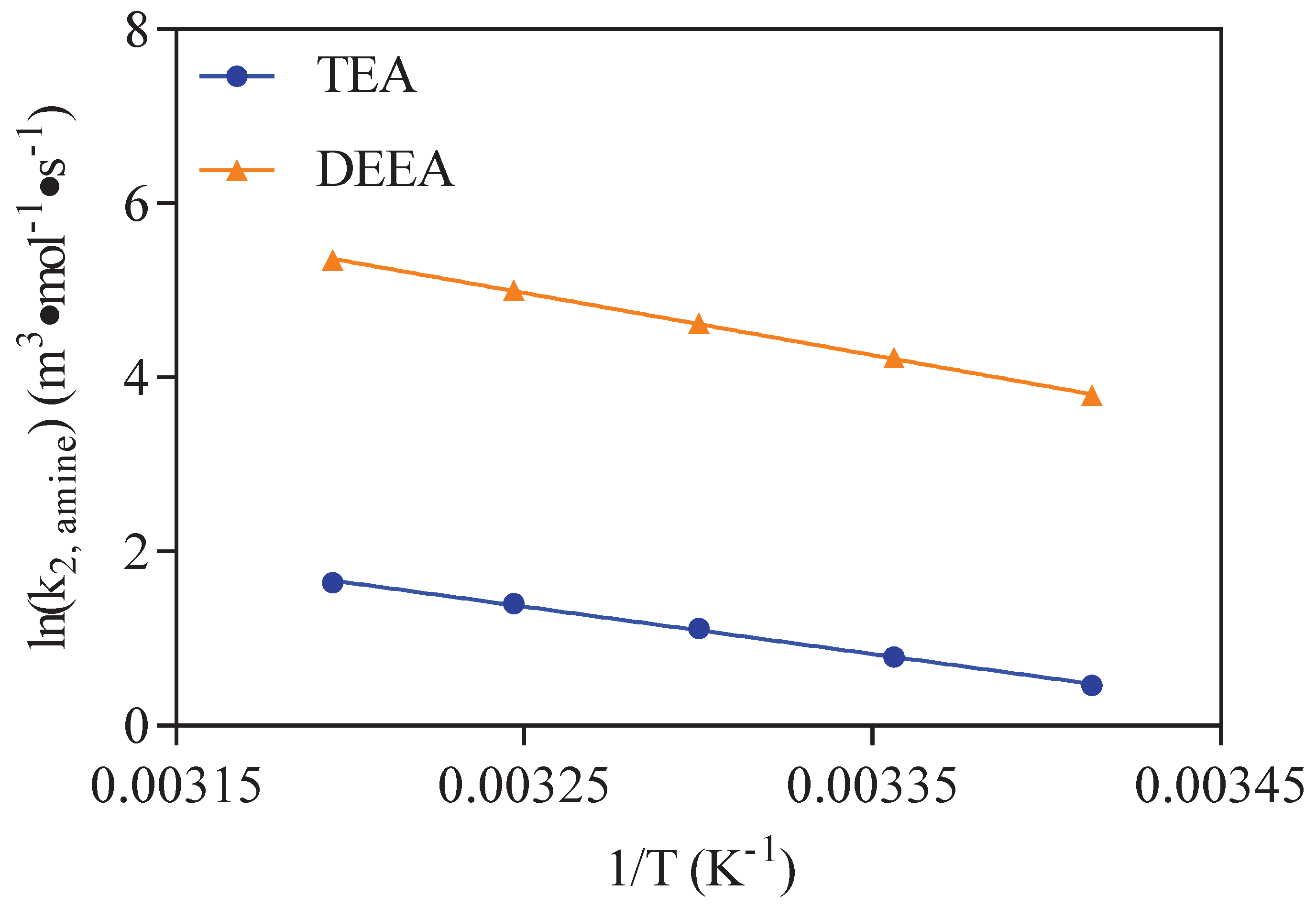
3.1. CO2-Amine-H2O System
3.2. CO2-Amine-H2O Containing CA
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TEA | Triethanolamine |
| DEEA | 2-(Diethylamino)ethanol |
| CA | Carbonic Anhydrase |
| HCA | Human Carbonic Anhydrase |
| CE | Catalyst Enhancement |
| Ea | activation energy (kJ/mol) |
| R | universal gas constant (0.008315 kJ/mol·K) |
| T | temperature (K) |
| concentration (kmol·m) | |
| first order reaction rate constant (s) | |
| second order reaction rate constant (m· mol ·s) | |
| first order reaction rate constant (s) without enzyme | |
| first order reaction rate constant (s) with enzyme | |
| reaction rate constant of CO2 reacting with hydroxyl ion in absence of enzyme | |
| reaction rate constant of CO2 reacting with hydroxyl ion in present of enzyme | |
| the total CO2 reaction rate (kmol·m·s) | |
| CE | catalyst enhancement |
| reaction rate constant of CO2 reacting with hydroxyl ion determined by Pinsent et al. | |
| parameter of Equation (14) | |
| parameter of Equation (14) |
Appendix A
Appendix A.1
| Amine Concentration (kmol/m) | Temperature (K) | ||||
|---|---|---|---|---|---|
| 293 | 298 | 303 | 308 | 313 | |
| TEA | |||||
| 0.1 | 0.12 | 0.15 | 0.18 | 0.20 | 0.24 |
| 0.2 | 0.30 | 0.41 | 0.56 | 0.75 | 1.01 |
| 0.3 | 0.45 | 0.64 | 1.02 | 1.34 | 1.74 |
| 0.4 | 0.75 | 1.04 | 1.39 | 1.94 | 2.38 |
| 0.5 | 0.94 | 1.38 | 1.92 | 2.61 | 3.33 |
| Reaction order | 1.30 | 1.39 | 1.34 | 1.38 | 1.36 |
| R value | 0.9926 | 0.9971 | 0.997 | 0.9974 | 0.9943 |
| DEEA | |||||
| 0.1 | 4.12 | 6.23 | 9.33 | 14.54 | 22.12 |
| 0.2 | 9.37 | 14.29 | 21.04 | 31.38 | 45.65 |
| 0.3 | 13.07 | 21.29 | 32.23 | 47.20 | 65.60 |
| 0.4 | 18.05 | 27.71 | 40.96 | 58.74 | 79.12 |
| 0.5 | 22.67 | 34.38 | 50.61 | 68.22 | 94.81 |
| Reaction order | 1.0 | 0.99 | 0.98 | 0.89 | 0.83 |
| R value | 0.9979 | 0.998 | 0.9968 | 0.9907 | 0.9939 |
| T (K) | (m·mol·s) | |
|---|---|---|
| TEA | DEEA | |
| 293 | 1.59 | 44.42 |
| 298 | 2.21 | 68.55 |
| 303 | 3.06 | 101.91 |
| 308 | 4,06 | 148.58 |
| 313 | 5.17 | 211.11 |
| CA Concentration (g/m) | Temperature (K) | |||||
|---|---|---|---|---|---|---|
| 293 | 298 | 303 | 308 | 313 | ||
| CA+TEA | ||||||
| 5 | 2.12 | 2.62 | 3.20 | 3.84 | 4.36 | |
| 15 | 9.04 | 10.20 | 12.42 | 14.71 | 17.94 | |
| 25 | 20.99 | 23.50 | 27.08 | 30.56 | 36.49 | |
| 35 | 25.07 | 29.28 | 33.26 | 39.75 | 47.16 | |
| 50 | 38.55 | 44.38 | 50.45 | 59.28 | 70.57 | |
| CA+DEEA | ||||||
| 5 | 21.84 | 31.34 | 45.87 | 63.20 | 82.41 | |
| 15 | 24.97 | 31.51 | 43.34 | 58.98 | 79.34 | |
| 25 | 27.66 | 36.47 | 47.76 | 64.76 | 84.03 | |
| 35 | 29.84 | 37.16 | 49.06 | 65.36 | 83.58 | |
| 50 | 41.46 | 48.34 | 60.22 | 73.64 | 90.99 | |
| Amine Concentration (kmol/m) | Temperature (K) | |||||
|---|---|---|---|---|---|---|
| 293 | 298 | 303 | 308 | 313 | ||
| TEA+CA | ||||||
| 0.1 | 5.57 | 8.14 | 10.37 | 12.78 | 15.32 | |
| 0.2 | 7.54 | 9.17 | 11.09 | 13.25 | 16.80 | |
| 0.3 | 8.86 | 10.78 | 12.62 | 14.72 | 17.68 | |
| 0.4 | 10.99 | 12.92 | 15.47 | 17.41 | 20.31 | |
| 0.5 | 12.70 | 14.26 | 16.62 | 18.38 | 21.49 | |
| DEEA+CA | ||||||
| 0.1 | 7.66 | 10.44 | 13.88 | 18.38 | 26.84 | |
| 0.2 | 12.57 | 15.29 | 21.13 | 31.28 | 45.13 | |
| 0.3 | 16.59 | 20.62 | 29.68 | 41.09 | 56.92 | |
| 0.4 | 20.93 | 26.56 | 36.56 | 51.40 | 69.18 | |
| 0.5 | 24.97 | 31.51 | 43.34 | 58.98 | 79.34 | |
References
- Rochelle, G.T. Amine scrubbing for CO2 capture. Science 2009, 325, 1652–1654. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Rees, R.J.; Conway, W.; Puxty, G.; Yang, Q.; Winkler, D.A. Computational Modeling and Simulation of CO2 Capture by Aqueous Amines. Chem. Rev. 2017, 117, 9524–9593. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.H.; Sanpasertparnich, T.; Tontiwachwuthikul, P.P.; Gelowitz, D.; Idem, R. Part 1: Design, modeling and simulation of post-combustion CO2 capture systems using reactive solvents. Carbon Manag. 2011, 2, 265–288. [Google Scholar] [CrossRef]
- Supap, T.; Saiwan, C.; Idem, R.; Tontiwachwuthikul, P.P. Part 2: Solvent management: Solvent stability and amine degradation in CO2 capture processes. Carbon Manag. 2011, 2, 551–566. [Google Scholar] [CrossRef]
- Svendsen, H.F.; Hessen, E.T.; Mejdell, T. Carbon dioxide capture by absorption, challenges and possibilities. Chem. Eng. J. 2011, 171, 718–724. [Google Scholar] [CrossRef]
- Liu, B.; Luo, X.; Liang, Z.; Olson, W.; Liu, H.; Idem, R.; Tontiwachwuthikul, P. The development of kinetics model for CO2 absorption into tertiary amines containing carbonic anhydrase. Aiche J. 2017, 63, 4933–4943. [Google Scholar] [CrossRef]
- Lindskog, S.; Silverman, D.N. The Catalytic Mechanism of Mammalian Carbonic Anhydrases. EXS; Birkhäuser: Basel, Switzerland, 2000; Volume 14, p. 175. [Google Scholar] [CrossRef]
- Penders-van Elk, N.J.; Derks, P.W.; Fradette, S.; Versteeg, G.F. Kinetics of absorption of carbon dioxide in aqueous MDEA solutions with carbonic anhydrase at 298 K. Int. J. Greenh. Gas Control. 2012, 9, 385–392. [Google Scholar] [CrossRef]
- Liu, H.; Liang, Z.; Sema, T.; Rongwong, W.; Li, C.; Na, Y.; Idem, R.; Tontiwachwuthikul, P.; Idem, R.; Tontiwachwuthikul, P. Kinetics of CO2 absorption into a novel 1-diethylamino-2-propanol solvent using stopped-flow technique. AIChE J. 2014, 60, 3502–3510. [Google Scholar] [CrossRef]
- Liu, B.; Luo, X.; Gao, H.; Idem, R.; Tontiwachwuthikul, P.; Olson, W.; Liang, Z. Reaction kinetics of the absorption of carbon dioxide (CO2) in aqueous solutions of sterically hindered secondary alkanolamines using the stopped-flow technique. Chem. Eng. Sci. 2017, 170, 16–25. [Google Scholar] [CrossRef]
- Ali, S.H.; Merchant, S.Q.; Fahim, M.A. Kinetic study of reactive absorption of some primary amines with carbon dioxide in ethanol solution. Sep. Purif. Technol. 2000, 18, 163–175. [Google Scholar] [CrossRef]
- Ali, S.H.; Merchant, S.Q.; Fahim, M.A. Reaction kinetics of some secondary alkanolamines with carbon dioxide in aqueous solutions by stopped flow technique. Sep. Purif. Technol. 2002, 27, 121–136. [Google Scholar] [CrossRef]
- Liu, H.; Sema, T.; Liang, Z.; Fu, K.; Idem, R.; Na, Y.; Tontiwachwuthikul, P. CO2 absorption kinetics of 4-diethylamine-2-butanol solvent using stopped-flow technique. Sep. Purif. Technol. 2014, 136, 81–87. [Google Scholar] [CrossRef]
- Donaldson, T.L.; Nguyen, Y.N. Carbon Dioxide Reaction Kinetics and Transport in Aqueous Amine Membranes. Ind. Eng. Chem. Fundam. 1980, 19, 260–266. [Google Scholar] [CrossRef]
- Versteeg, G.; van Swaaij, W. On the kinetics between CO2 and alkanolamines both in aqueous and non-aqueous solutions—I. Primary and secondary amines. Chem. Eng. Sci. 1988, 43, 573–585. [Google Scholar] [CrossRef] [Green Version]
- Versteeg, G.; van Swaaij, W. On the kinetics between CO2 and alkanolamines both in aqueous and non-aqueous solutions—II. Tertiary amines. Chem. Eng. Sci. 1988, 43, 587–591. [Google Scholar] [CrossRef] [Green Version]
- Carter, N.D.; Gros, S.J. The Carbonic Anhydrases: Cellular Physiology and Molecular Genetics. Plenum Press; Springer: Boston, MA, USA, 1991. [Google Scholar] [CrossRef]
- Lindskog, S. Structure and mechanism of carbonic anhydrase. Pharmacol. Ther. 1997, 74, 1–20. [Google Scholar] [CrossRef]
- Alper, E.; Deckwer, W.D. Kinetics of absorption of CO2 into buffer solutions containing carbonic anhydrase. Chem. Eng. Sci. 1980, 35, 549–557. [Google Scholar] [CrossRef]
- Penders-van Elk, N.J.; Fradette, S.; Versteeg, G.F. Effect of pKa on the kinetics of carbon dioxide absorption in aqueous alkanolamine solutions containing carbonic anhydrase at 298 K. Chem. Eng. J. 2015, 259, 682–691. [Google Scholar] [CrossRef]
- Wilk, A.; Wiecaw-Solny, L.; Krótki, A.; Piewak, D. Impact of the composition of absorption blend on the efficiency of CO2 removal. Chemik 2013, 67, 399–406. [Google Scholar]
- Pinsent, B.R.W.; Pearson, L.; Roughton, F.J.W. The kinetics of combination of carbon dioxide with hydroxide ions. Trans. Faraday Soc. 1956, 52, 1512–1520. [Google Scholar] [CrossRef]
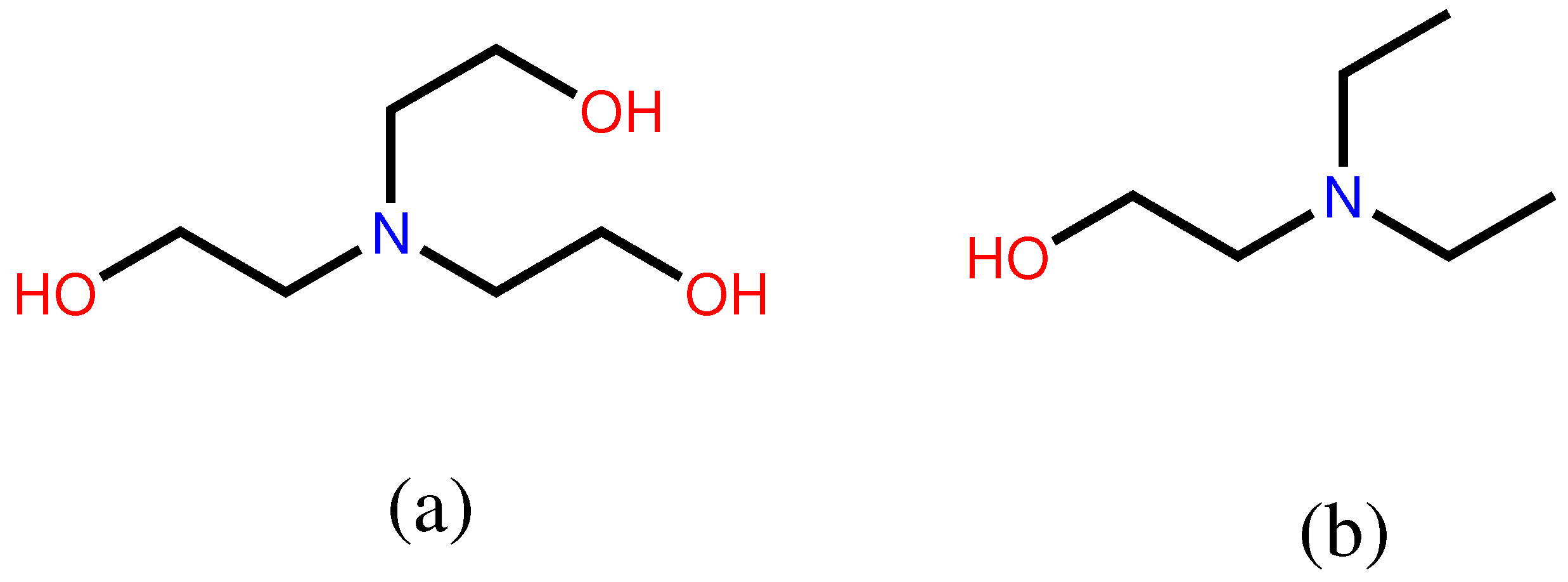





| Component | CAS | MW (g/mol) | Purity (%) | Supplier |
|---|---|---|---|---|
| TEA | 102-71-6 | 149.19 | 99% | aladdin |
| DEEA | 100-37-8 | 117.19 | 99% | aladdin |
| CA | 9001-03-0 | - | ⩾3000 units/mg | Sigma-Aldrich |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Cui, Z.; Tian, W. The Kinetics Investigation of CO2 Absorption into TEA and DEEA Amine Solutions Containing Carbonic Anhydrase. Processes 2021, 9, 2140. https://doi.org/10.3390/pr9122140
Liu B, Cui Z, Tian W. The Kinetics Investigation of CO2 Absorption into TEA and DEEA Amine Solutions Containing Carbonic Anhydrase. Processes. 2021; 9(12):2140. https://doi.org/10.3390/pr9122140
Chicago/Turabian StyleLiu, Bin, Zhe Cui, and Wende Tian. 2021. "The Kinetics Investigation of CO2 Absorption into TEA and DEEA Amine Solutions Containing Carbonic Anhydrase" Processes 9, no. 12: 2140. https://doi.org/10.3390/pr9122140
APA StyleLiu, B., Cui, Z., & Tian, W. (2021). The Kinetics Investigation of CO2 Absorption into TEA and DEEA Amine Solutions Containing Carbonic Anhydrase. Processes, 9(12), 2140. https://doi.org/10.3390/pr9122140








