Abstract
Low-rank coal is difficult to upgrade using conventional flotation methods due to its high hydrophilic properties. Thus, it is necessary to explore new methods for upgrading and utilizing low-rank coal. In this investigation, a gaseous dodecane pretreatment method was used to enhance the flotation performance of low-rank coal. Pore distribution analysis, FTIR (Fourier Transform Infrared Spectroscopy), and contact angle measurements were used to study the surface properties of the coal sample. Size distribution and float-sink test results indicated that the coal sample contained a lot of clean coal with low ash content, which could be used as a high quality raw material for making coal water slurry. FTIR, pore distribution analysis, and contact angle results showed that the coal was very hydrophilic due to the high concentration of -OH group and the large number of pores and cracks on the coal surface. The hydrophobicity of the coal sample was significantly improved by the gaseous dodecane pretreatment method. Clean coal with 67.2% combustible matter recovery and 10.5% ash content was obtained by gaseous dodecane pretreatment flotation method. Coal water slurry with 60% concentration was prepared using the flotation clean coal.
1. Introduction
Even though energy research regarding new energy sources and technologies is becoming more popular than those regarding fossil fuels nowadays, coal still accounts for more than 50% of China’s primary energy consumption [1]. Due to the special nature of China’s resource distribution, a sudden reduction in coal consumption will cause a series of social and economic problems.
It is well-known that low-rank coal (LRC) is often used as thermal coal. China has abundant low-rank coal resources, such as lignite (13%), long-flame coal (13%), non-caking coal (15%), which accounts for about 40% of discovered coal resources [2]. During the coal preparation process, coal slime with a yield of about 15% or even higher can be produced. If the coal slime is directly burned or mixed into other products as medium coal without being upgraded, it will cause waste of resources and environmental pollution. Sometimes the coal preparation plant has to spend money taking the slime away. However, the coal slime usually has a fine particle size, which may be used as a high quality raw material for making coal water slurry (CWS) after being upgraded. In recent years, the demand for coal to product CWS has increased rapidly [3,4,5,6]. Therefore, the use of LRC slime to make CWS can greatly improve the economic benefits of the enterprise and reduce environmental pollution.
Flotation is an effective way to upgrade coal slime [7,8,9,10,11,12]. However, due to the high oxygen-containing functional groups and pores on the surface of LRC, the surface is highly hydrophilic and difficult to upgrade using conventional flotation method [13,14,15,16]. Nowadays, LRC flotation is a worldwide problem. In the past few decades, scholars have conducted many studies on improving the flotation efficiency of LRC. Xia et al. used a mixture of candle soot and hydrocarbon oil as a novel flotation collector for low-rank coal and found that the flotation recovery of low-rank coals using the mixture of candle soot and dodecane were higher than that using solo dodecane as collector [17]. Atesok and Celik found that the flotation behavior of low-rank coal was obviously improved using dry grinding method with colloctor [18]. Ahmed and Drzymala also proposed that the adsorption amount of the collector on the hydrophilic coal surface increased when using dry grinding method [19]. The surface of the low-rank coal contains a large number of pores, and the water molecules will first enter the pores during the flotation process, which leads to the formation of a stable hydration membrane on the surface of the low-rank coal, which affects the adsorption of the collector [20,21,22]. Xia et al. found that using heavy oil in the slime grinding process can significantly improve the hydrophobicity of the low-rank coal surface [23,24]. They believed that the pre-adsorbed oil collector can improve the hydrophobicity of the coal surface, weaken the hydration film on the coal surface, and enhance the adhesion of bubbles to coal particles. However, the dosage of the collector used in the dry-grinding process was huge. Since the oil collector has poor dispersibility during the grinding process, and it is difficult to enter into the pores of the coal surface.
Other techniques such as pore compression pretreatment [25], microwave pretreatment [26], ultrasonic pretreatment [27], dry grinding pretreatment [18], low-temperature pyrolysis [28] have been used to enhance the flotation of low-rank coal.
Recently, gaseous collector was used in flotation of low-rank coal. Shen et al. has proved that the gaseous oil collector was more easily adsorbed at the solid-gas interface [29]. Meanwhile, gaseous oil molecule was small enough to enter into the pores on the coal surface, which can improve the hydrophobicity inside the pores. Therefore, the purpose of this paper was to study the enhancement of low-rank coal flotation by the gaseous dodecane pretreatment method. The enhancing mechanism of gaseous dodecane pretreatment on the flotation recovery of low-rank coal was discussed. In this paper, a new method for the effective flotation of low-rank coal was proposed. Meanwhile, the feasibility of using flotation clean coal to make coal water slurry was studied.
2. Materials and Methods
2.1. Materials
Low-rank coal (LRC) samples were obtained from Shanxi province of China. −0.5 mm size fraction was used in the experiment. Table 1 illustrated the proximate analysis results of LRC. Table 2 showed the particle size distribution of LRC. The float-sink test results of the sample are given in Table 3. As presented in Table 2 and Table 3, the coal sample was fine with high ash content. The float-sink test results indicated that the coal slime contained a lot of clean coal with low ash content, which can be used as a high-quality raw material for making CWS. The coal sample with the density fraction of −1.3 g/cm3 was used for the FTIR (Fourier Transform Infrared Spectroscopy) measurement and contact angle tests. Dodecane were used as collector and methyl isobutyl carbinol was used as frother. Methyl isobutyl carbinol and dodecane were of analytical grade and obtained from Aladdin Reagent.

Table 1.
Proximate analysis of low-rank coal (LRC) sample (air-dried).

Table 2.
Particle size distribution of LRC sample.

Table 3.
Float-sink test results of LRC sample.
2.2. FTIR Measurement and Pore Distribution Analysis
A Fourier Transform Infrared Spectroscopy (IRAffinity-1) from Shimadzu Corporation in Japan was used to study the functional group on the coal surface. The coal sample was pressed into a pellet with KBr. The concentration of KBr was 1%. The spectrum was obtained between 4000 and 600 cm−1 with 4 cm−1 resolution.
A specific pore analyzer from MicrotracBEL Inc. in Japan was used to study the pore structure, including the pore volume (PV), pore size distribution (PSD), and average pore diameter (APD) of the LRC sample. In each test, 0.2000 g LRC sample was firstly placed into a sample tube and degassed in the adsorption system. Then, measurements of the pore structure were performed with liquid nitrogen at 77 K. Finally, the PV, PSD, and APD data were obtained by the Barrett–Joyner–Halenda (BJH) model.
2.3. Contact Angle Measurements
The coal samples that have been pretreated with gaseous dodecane were firstly pressed into a pellet under 10 M pressure for 1 min using a FW-4A powder compressing machine from Jiayuan Corporation in China. Then, DSA 30 from KRUSS, Inc. in Germany was used to measure the contact angles of water on coal surface before and after the gaseous collector pretreatment by sessile drop method [30]. Each measurement was repeated at least three times and the contact angle data points presented in this paper were the average value of the measurements.
2.4. Gaseous Pretreatment Method
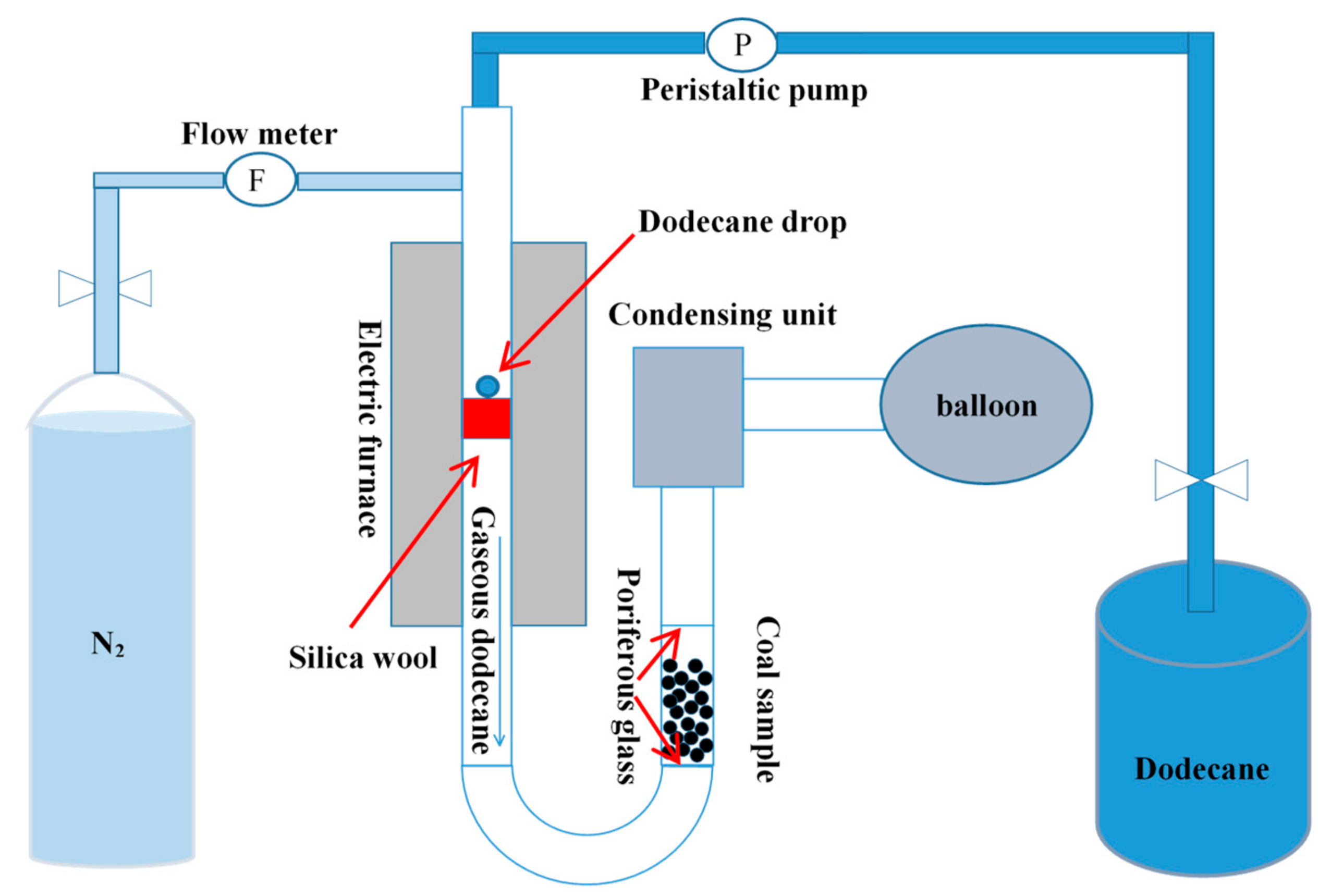
The gaseous pretreatment (GP) system was presented in Figure 1. First, the air in the system was blown out with high-purity nitrogen gas, and then the dodecane liquid was continuously drawn into the quartz tube by a peristaltic pump. In order to increase the heating time of dodecane, a silica wool was placed in the high temperature zone of the quartz tube. The coal sample was placed between two porous glasses. The gaseous dodecane passed through the coal sample from the bottom and was adsorbed by the coal sample. Unadsorbed gaseous dodecane and N2 were collected by a condensing unit and balloon. Six g original LRC sample or clean coal with −1.3 g/cm3 density fraction was used in each experiment. The original LRC sample was used as the feed of the flotation while the clean coal sample was used for FTIR measurements and contact angle tests. The feeding rate of dodecane was 0.015 mL/min. The dodecane dosages were 1 kg/t, 2 kg/t, 4 kg/t, and 6 kg/t. Throughout the test, high-purity nitrogen was continuously introduced into the system as a shielding gas. After the adsorption process, the weight of the coal sample, silica tube, and condensing unit were measured respectively to determine the adsorption amount of dodecane on the coal surface.
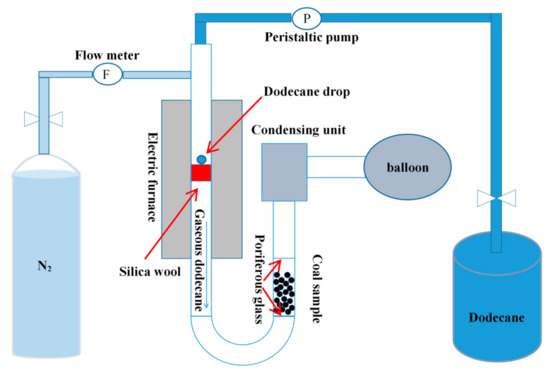
Figure 1.
Schematic of the gaseous pretreatment system.
Conventional pretreatment (CP) method was conducted to compare with the GP method. During the CP process, 6 g coal was firstly added into the flotation cell and prewetted by water for 1 min. Then dodecane was added and conditioned for 2 min.
2.5. Flotation Tests
All flotation tests were conducted in a 0.1 L micro-flotation cell with the impeller speed of 1400 r/min. The pulp concentration was 60 g/L. The pretreated coal was firstly added into the flotation cell with water and pre-wetted for 1 min. Then, frother was added and conditioned for another 0.5 min. At last, air flow was given. Finally, the concentrate and tailings were collected to measure the ash contents and yields. Since dodecane has been adsorbed on the coal surface during the pretreatment process, no collector was added in flotation process.
Conventional flotation was conducted to compare with the novel flotation process. The dosages of dodecane used in conventional flotation were the same with that used in the GP process. The frother dosage was 0.1 kg/t. The combustible matter recovery (CMR) of the concentrate was used to indicate the flotation behavior, which can be calculated from Equation (1).
where MF and MC represent the weight of the feed and clean coal, respectively, and AF and AC represent the ash contents of the feed and clean coal, respectively.
3. Results and Discussion
3.1. FTIR Results and Pore Structure Characteristics
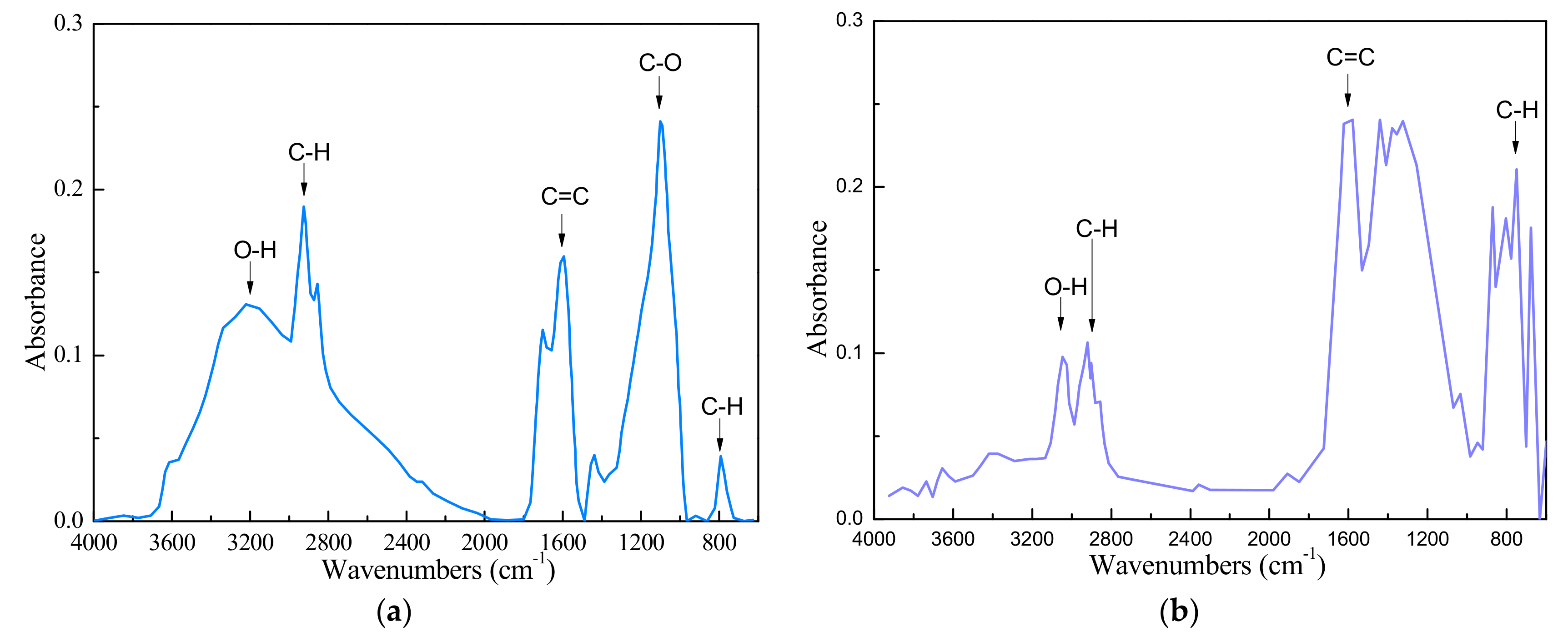
The FTIR results of the low-rank coal sample and anthracite were presented in Figure 2. It can be seen that the low-rank coal sample was characterized by a high concentration of -OH group as seen from the band in the 3200–3600 cm−1 region. The -OH group is the primary hydrophilic group in the coal. It indicated that the coal sample was very hydrophilic.
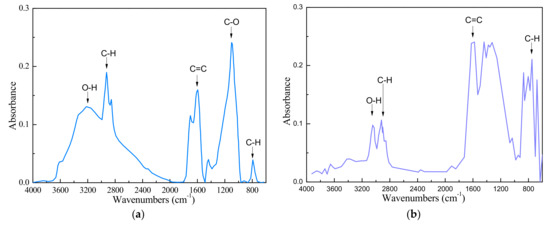
Figure 2.
FTIR(Fourier Transform Infrared Spectroscopy) result of LRC sample (a) and anthracite sample (b).
The characteristics of the pore structure of the coal samples were given in Table 4. A large number of pores and cracks existed on the coal surface, which could be filled by water molecules during the flotation process. Thus, a thick hydration shell may cover the coal surface and lead to poor floatability of the coal.

Table 4.
Characteristics of the pore structure of LRC sample.
3.2. Contact Angle Results
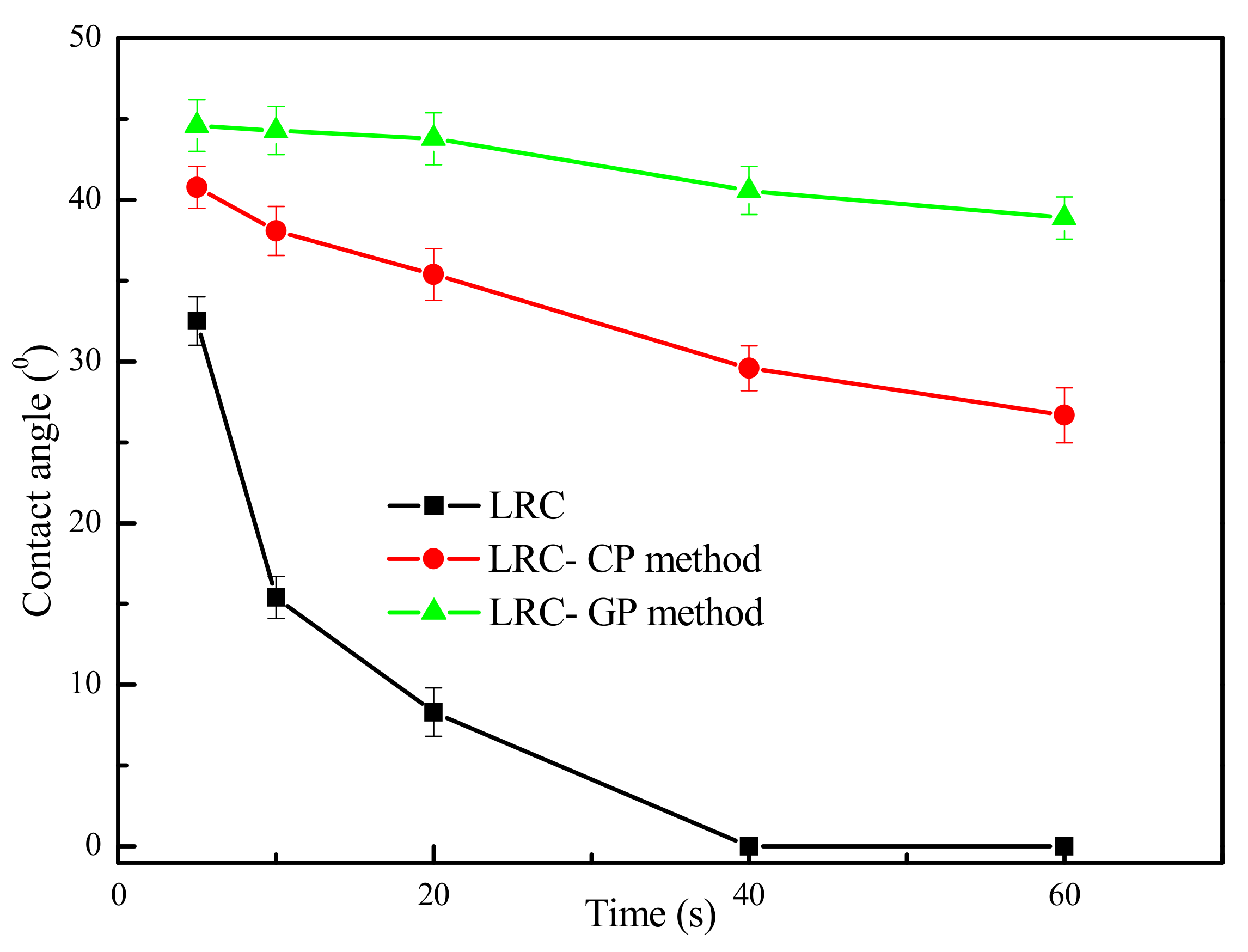
The contact angle results of water on LRC surface in the presence of the GP and CP method are presented in Figure 3. As shown in Figure 3, the contact angles of water on the LRC surface were very small and decreased rapidly with the increase in contact time. According to FTIR results and pore structure characteristics in Section 3.1, the surface of LRC was highly hydrophilic due to the high hydroxyl content. Thus, the water droplets can easily spread on the surface of the LRC. Furthermore, the water droplets may also penetrate into the pores and cracks of the coal surface.
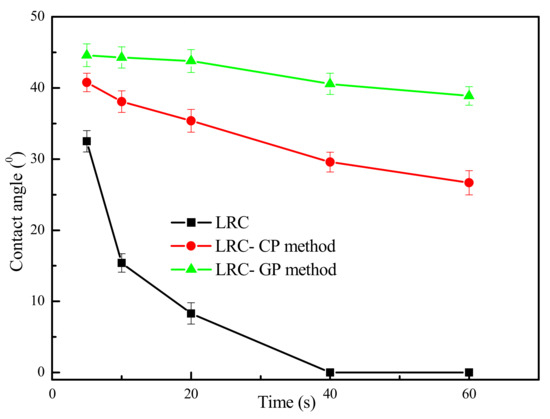
Figure 3.
Contact angle results of water on LRC surface in the presence of gaseous pretreatment (GP) and conventional pretreatment (CP) method (Dodecane dosage 6 kg/t) (The data points represent the mean values (n = 3) ± standard deviation.).
The contact angles of water on the surface of the coal obtained by the GP method were higher than that obtained by the CP method. Furthermore, the water droplets spread much slower on the gaseous pretreated coal surface. The contact angle results indicated that a more stable and hydrophobic coal surface was obtained by GP method, which could improve the flotation efficiency of LRC.
3.3. Dodecane Adsorption Results
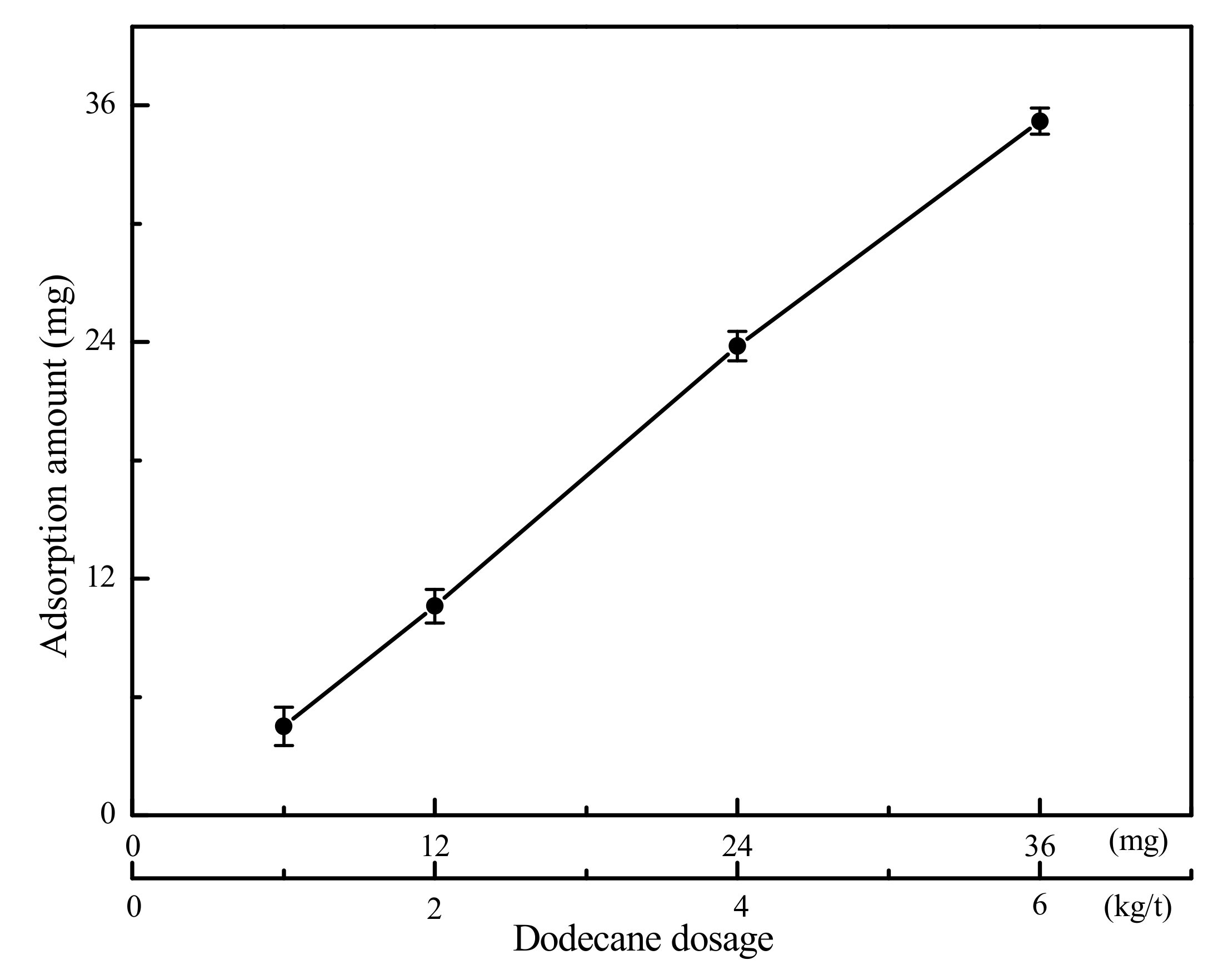
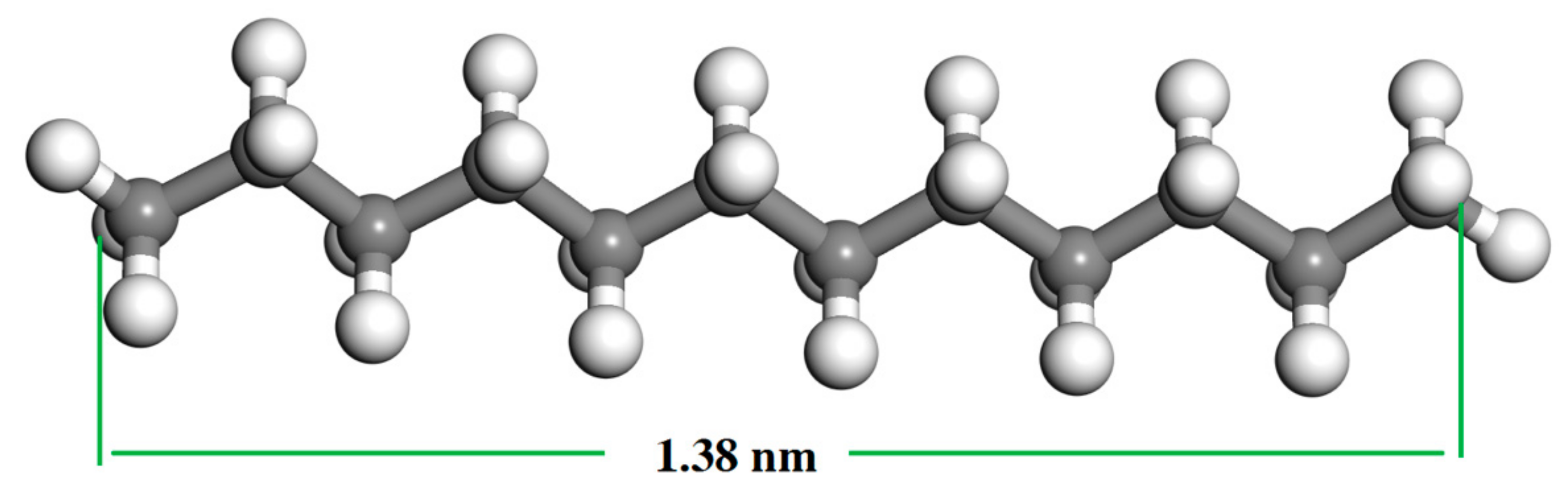
The adsorption results of gaseous dodecane on the LRC surface are presented in Figure 4. It can be seen from Figure 4 that the adsorption amount of dodecane on the LRC surface increased with the increase of dodecane dosage. Most of the dodecane was adsorbed by LRC except for some gaseous dodecane condensing on the surface of the quartz tube during the adsorption process. It indicated that LRC had a very strong adsorption capacity for gaseous dodecane. The gaseous dodecane was adsorbed on the surface and pores of the LRC mainly by van der Waals and Laplace forces. According to Section 3.1, the average pore diameter of the LRC sample was 8.658 nm. The length of a dodecane molecule is less than 1.38 nm (Figure 5) in atmosphere, which can directly enter into the pores on the coal surface, improving the hydrophobicity of the LRC. However, the diameters of dodecane drops produced by conventional flotation method are always larger than 3 μm.
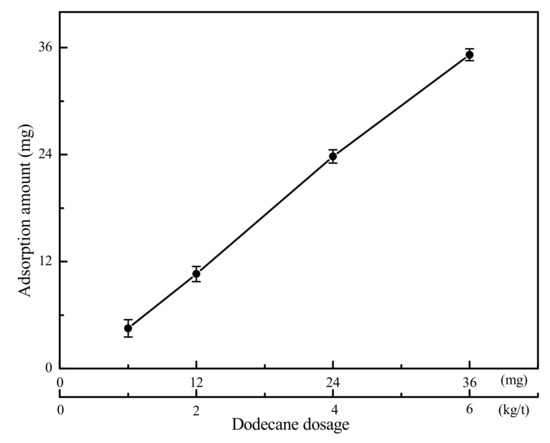
Figure 4.
Adsorption results of gaseous dodecane on LRC surface (The data points represent the mean values (n = 3) ± standard deviation.).
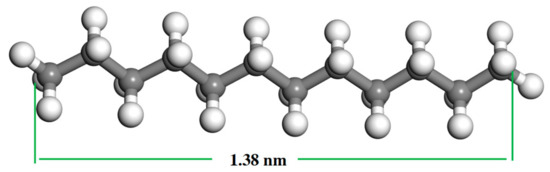
Figure 5.
Molecular structure of dodecane. Colored balls represent C in gray, H in white.
3.4. Flotation Results
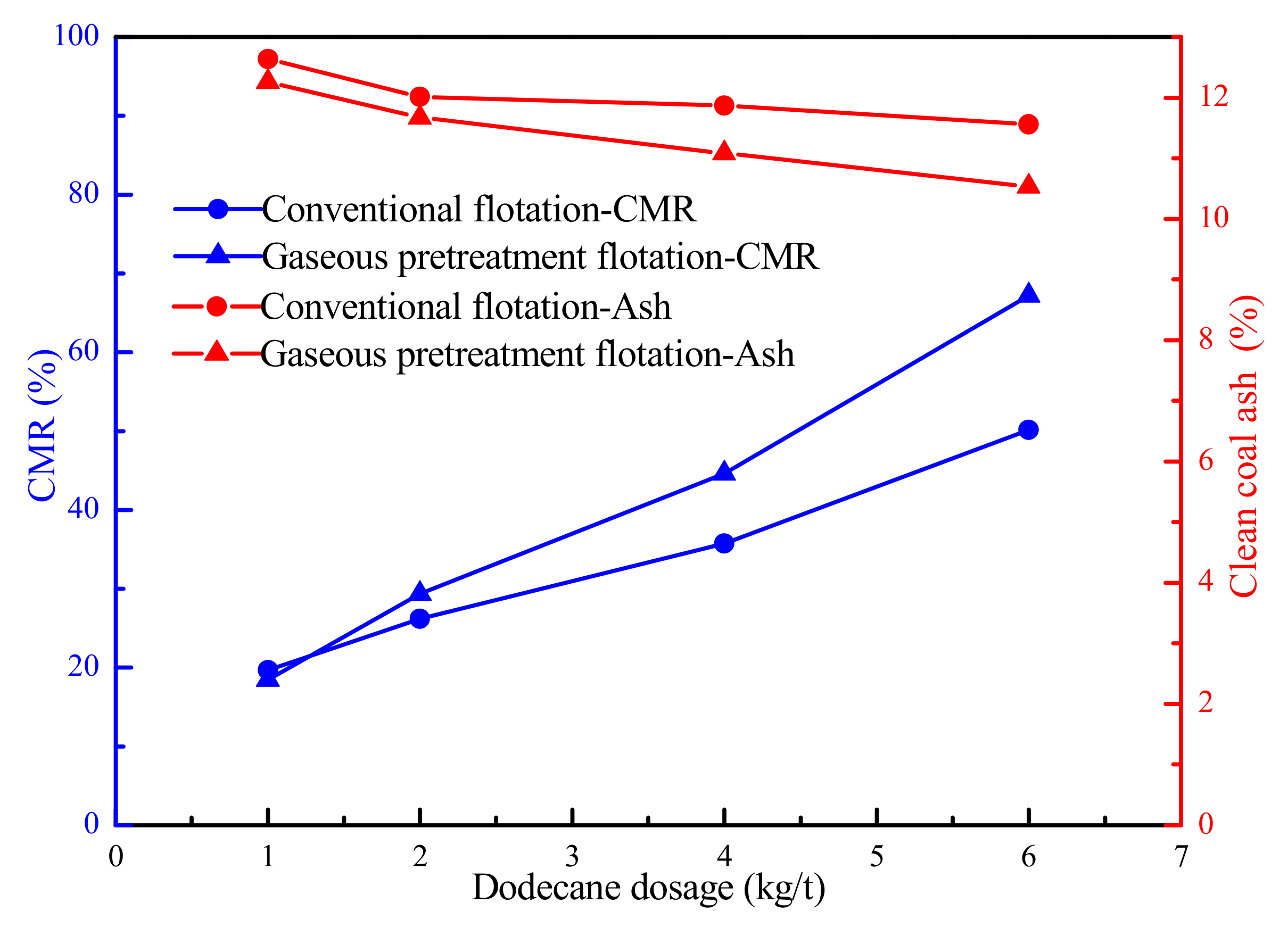
Figure 6 illustrated the flotation results of LRC in the presence of conventional flotation and the gaseous pretreatment flotation method. The combustible matter recovery (CMR) of the clean coal obtained by gaseous pretreatment flotation was much higher than that by conventional flotation method. However, the CMRs obtained by the two method were very close at low dodecane dosages. This was because some dodecane were condensed on the surface of the quartz tube, resulting in insufficient adsorption of the dodecane on the surface of the coal particles. It can also be seen that the ash contents of the clean coal obtained by gaseous pretreatment flotation were lower than that using conventional flotation method. It meant the pretreatment flotation method can promote the flotation recovery of the LRC and the flotation selectivity was acceptable.
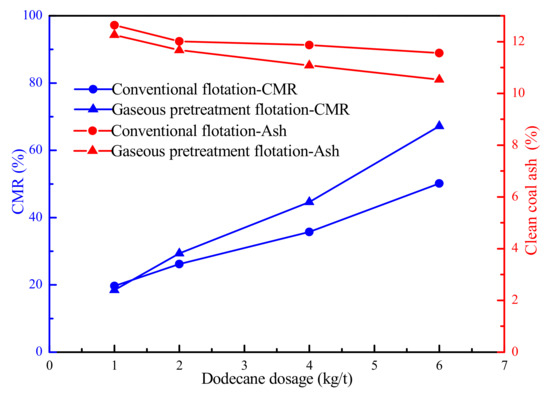
Figure 6.
Flotation results of LRC in the presence of conventional flotation and the gaseous pretreatment flotation method.
3.5. Probable Enhancing Mechanism
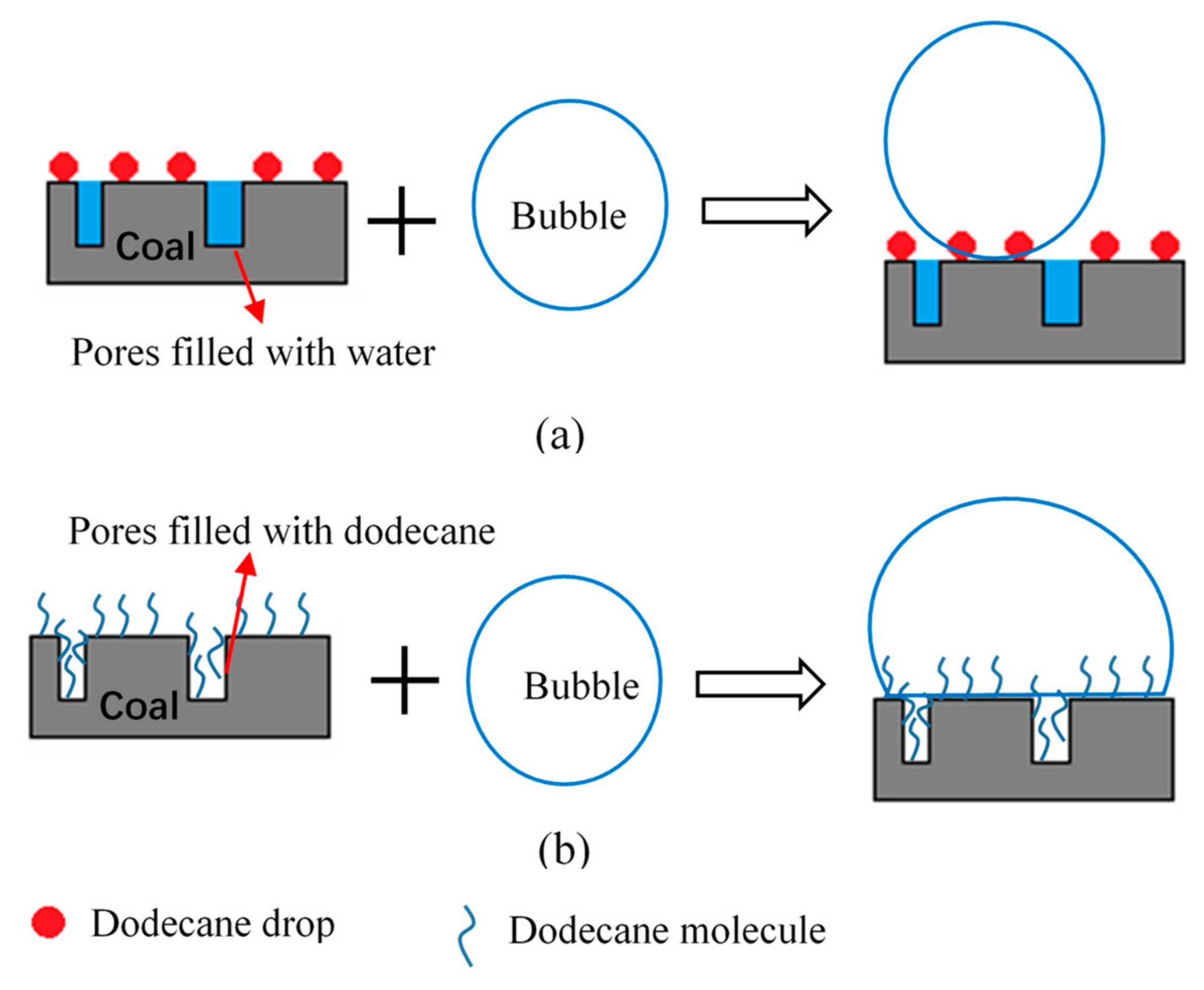
A probable enhancing mechanism of the LRC pretreatment with gaseous dodecane was proposed. Figure 7 shows the schematic representation of the enhancing process. Gaseous dodecane has better dispersibility and higher probability of adsorption on coal surfaces. Pores on coal surface are usually filled with water during the pre-wetting process in conventional flotation method. A thick hydration shell may cover the surface of low-rank coal. When coal particles collide with air bubbles, they cannot adhere to the surface of the bubble firmly as presented in Figure 7a. Furthermore, particles that have adhered to the surface of the bubble may also fall off. It can be seen from Figure 7b that the pores are firstly filled with dodecane when using pretreatment flotation method. The surface hydrophobicity of coal particles is further improved. Therefore, they can adhere to the surface of the bubble firmly when collide with air bubbles.
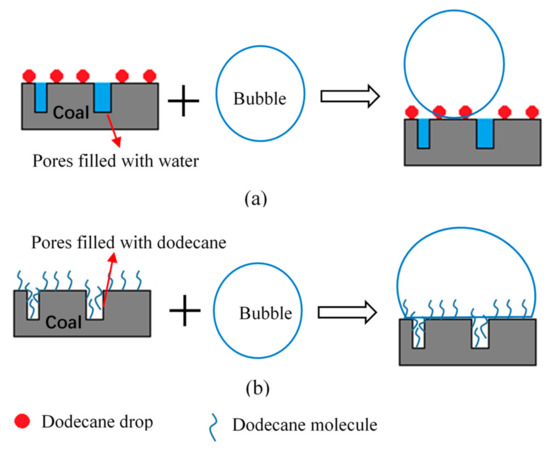
Figure 7.
Schematic representation of the enhancing mechanism of the gaseous dodecane pretreatment. (a) Conventional flotation and (b) gaseous pretreatment flotation.
3.6. Preparation of Coal Water Slurry (CWS)
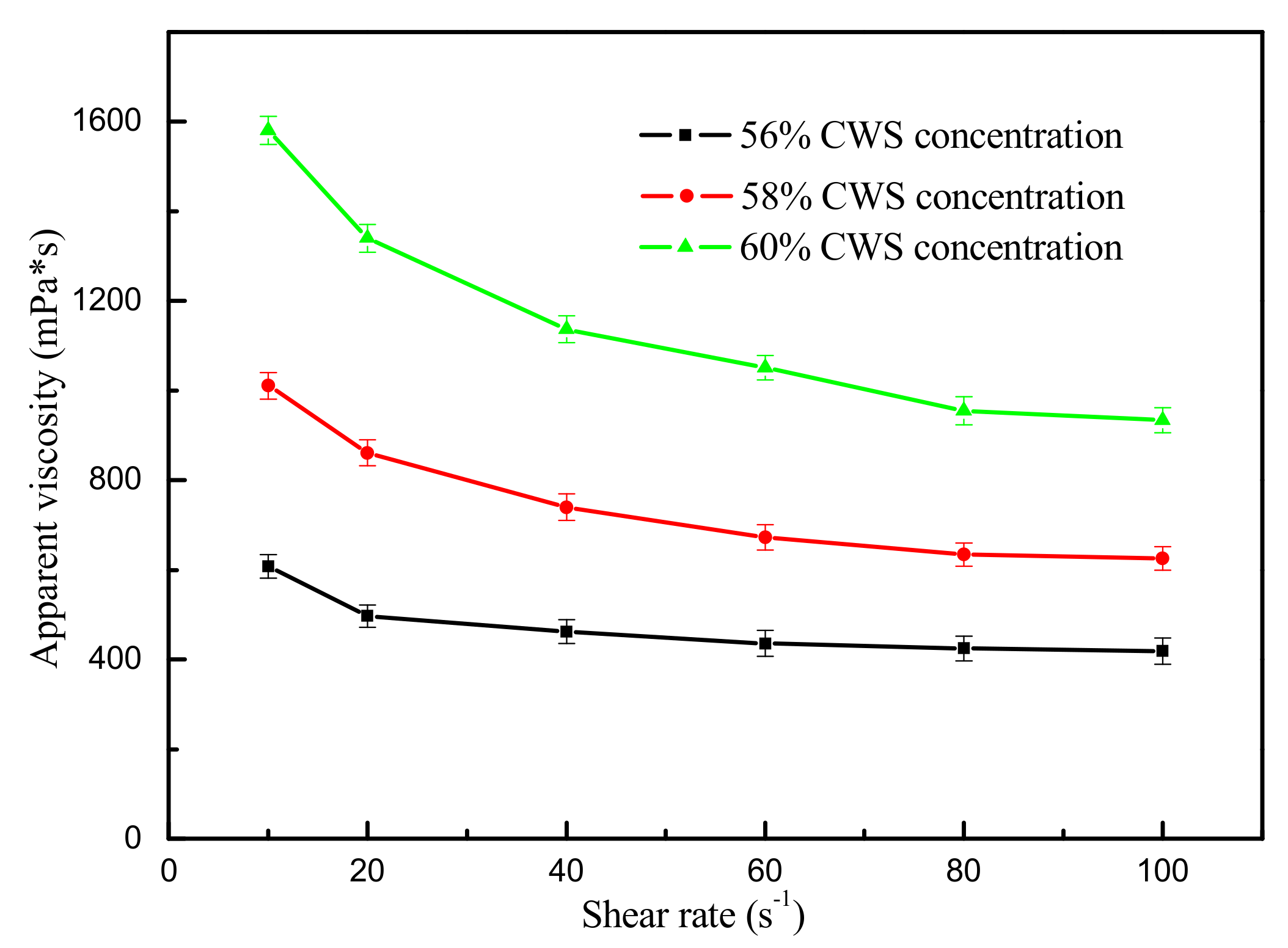
The size distribution of the flotation clean coal was presented in Table 5. It can be seen from Table 5 that the clean coal was mainly made up of fine coals, and the content of the −0.075 mm size fraction was above 70%, which met the requirements for coal particle size in the CWS production process. Humic acid was added as dispersant at 0.5%. Effect of shear rate on apparent viscosity of CWS in the presence of different CWS concentration is shown in Figure 8. The CWS with a concentration of 60% and an apparent viscosity of 950 mPa·s can be obtained using the flotation clean coal. The prepared CWS can be used as a high quality raw material for the Texaco gasifier.

Table 5.
Size distribution of the clean coal.
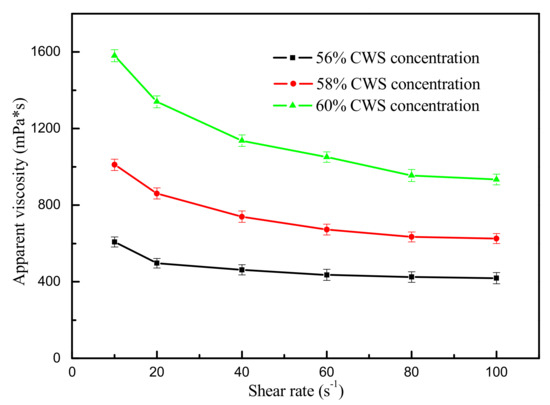
Figure 8.
Effect of shear rate on apparent viscosity of CWS in the presence of different concentrations (the data points represent the mean values (n = 3) ± standard deviation.).
4. Conclusions
In this investigation, gaseous dodecane pretreatment method was used to enhancing the flotation recovery of low-rank coal. The principal conclusions are:
- (1)
- Low-rank coal was very hydrophilic due to the high concentration of -OH group and the large number of pores and cracks of the coal.
- (2)
- The contact angles of water on the surface of the coal obtained by gaseous pretreatment method were higher than that obtained by conventional pretreatment method. And the water droplets spread much slower on the gaseous pretreated coal surface.
- (3)
- Low-rank coal had a very strong adsorption capacity for gaseous dodecane. The dodecane molecule can directly enter into the pores on the coal surface, improving the hydrophobicity of the LRC.
- (4)
- Clean coal with 67.2% CMR and 10.5% ash content was obtained using gaseous dodecane pretreatment flotation method. Coal water slurry with 60% concentration was made using the flotation clean coal.
Author Contributions
Conceptualization, L.S. and F.M.; Investigation, L.L. and C.X.; Methodology, F.M.; Validation, L.L.; Writing—original draft, L.S.; Writing—review & editing, C.X. and J.Z.
Funding
This research was funded by Natural Science Foundation of Anhui Province, grant number 1808085QE120, Key University Science Research Project of AnHui Province, grant number KJ2017A071, China Postdoctoral Science Foundation, grand number 2019M652163, Youth Fund of Anhui University of Science and Technology, grand number QN201608, National Natural Science Foundation of China, grant number 51874011, Excellent Youth Foundation of Anhui Scientific Committee, grand number 1808085J30.
Acknowledgments
The authors would like to thank Zhang Mingxu at Anhui University of Science and Technology for his help with this paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Xia, W.; Niu, C.; Ren, C. Enhancement in floatability of sub-bituminous coal by low-temperature pyrolysis and its potential application in coal cleaning. J. Clean. Prod. 2017, 168, 1032–1038. [Google Scholar] [CrossRef]
- Gui, X.; Liu, J.; Cao, Y.; Miao, Z.; Li, S.; Xing, Y.; Wang, D. Coal Preparation Technology: Status and Development in China. Energy Environ. 2015, 26, 997–1013. [Google Scholar] [CrossRef]
- Nyashina, G.S.; Kuznetsov, G.V.; Strizhak, P.A. Energy efficiency and environmental aspects of the combustion of coal-water slurries with and without petrochemicals. J. Clean. Prod. 2018, 172, 1730–1738. [Google Scholar] [CrossRef]
- Wang, R.; Ma, Q.; Ye, X.; Li, C.; Zhao, Z. Preparing coal slurry from coking wastewater to achieve resource utilization: Slurrying mechanism of coking wastewater–coal slurry. Sci. Total Environ. 2019, 650, 1678–1687. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, C.; Wang, Z.; Zhao, H.; Liu, H. Preparation of high concentration coal water slurry of lignite based on surface modification using the second fluid and the second particle. Fuel 2019, 242, 788–793. [Google Scholar] [CrossRef]
- Vershinina, K.; Shabardin, D.; Strizhak, P. Burnout rates of fuel slurries containing petrochemicals, coals and coal processing waste. Powder Technol. 2019, 343, 204–214. [Google Scholar] [CrossRef]
- Shen, L.; Liu, L.; Zhu, J.; Qiao, E. Effect of Oleic Acid on Froth Properties and Reverse Flotation Performance of Thermal Coal. Trans. Indian Inst. Met. 2018, 71, 1841–1846. [Google Scholar] [CrossRef]
- Chang, Z.; Chen, X.; Peng, Y. The interaction between diesel and surfactant Triton X-100 and their adsorption on coal surfaces with different degrees of oxidation. Powder Technol. 2019, 342, 840–847. [Google Scholar] [CrossRef]
- Xia, Y.; Wang, L.; Zhang, R.; Yang, Z.; Xing, Y.; Gui, X.; Cao, Y.; Sun, W. Enhancement of flotation response of fine low-rank coal using positively charged microbubbles. Fuel 2019, 245, 505–513. [Google Scholar] [CrossRef]
- Zhen, K.; Zhang, H.; Li, C.; Li, X. Effect of oxidized diesel oil on the flotation response of the low-rank coal by plasma oxidation method. Fuel 2019, 245, 13–20. [Google Scholar] [CrossRef]
- Yang, L.; Zhu, Z.; Qi, X.; Yan, X.; Zhang, H. The Process of the Intensification of Coal Fly Ash Flotation Using a Stirred Tank. Minerals 2018, 8, 597. [Google Scholar] [CrossRef]
- Xu, M.; Xing, Y.; Cao, Y.; Gui, X. Effect of Dodecane and Oleic Acid on the Attachment between Oxidized Coal and Bubbles. Minerals 2018, 8, 29. [Google Scholar] [CrossRef]
- Xu, M.; Xing, Y.; Cao, Y.; Gui, X. Waste colza oil used as renewable collector for low rank coal flotation. Powder Technol. 2019, 344, 611–616. [Google Scholar] [CrossRef]
- Cai, Y.; Du, M.; Wang, S.; Liu, L. Flotation characteristics of oxidized coal slimes within low-rank metamorphic. Powder Technol. 2018, 340, 34–38. [Google Scholar] [CrossRef]
- Xia, W.; Yang, J. Changes in surface properties of anthracite coal before and after inside/outside weathering processes. Appl. Surf. Sci. 2014, 313, 320–324. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, W.; Xu, H.; Zhuo, Q.; Sun, X. Adsorption Behavior of Methyl Laurate and Dodecane on the Sub-Bituminous Coal Surface: Molecular Dynamics Simulation and Experimental Study. Minerals 2019, 9, 30. [Google Scholar] [CrossRef]
- Xia, W.; Li, Y.; Nguyen, A.V. Improving coal flotation using the mixture of candle soot and hydrocarbon oil as a novel flotation collector. J. Clean. Prod. 2018, 195, 1183–1189. [Google Scholar] [CrossRef]
- Atesok, G. A new flotation scheme for a difficult-to-float coal using pitch additive in dry grinding. Fuel 2002, 79, 1509–1513. [Google Scholar] [CrossRef]
- Ahmed, H.A.M.; Drzymala, J. Upgrading difficult-to-float coal using microemulsion. Min. Metall. Explor. 2018, 29, 88–96. [Google Scholar] [CrossRef]
- Petukhov, V.N.; Girevaya, K.Y.; Kubak, D.A.; Girevoy, T.A. Interaction of the coal surface with water. Coke Chem. 2013, 56, 292–298. [Google Scholar] [CrossRef]
- Xia, W.; Wang, Y. Role of prewetting/immersion time in the attachment time between air bubble and Taixi oxidized coal. Int. J. Miner. Process. 2017, 163, 9–13. [Google Scholar] [CrossRef]
- Xia, W.; Yang, J. Effect of pre-wetting time on oxidized coal flotation. Powder Technol. 2013, 250, 63–66. [Google Scholar] [CrossRef]
- Xia, W.; Yang, J.; Zhu, B. Flotation of oxidized coal dry-ground with collector. Powder Technol. 2012, 228, 324–326. [Google Scholar] [CrossRef]
- Xia, W.; Zhou, C.; Peng, Y. Enhancing flotation cleaning of intruded coal dry-ground with heavy oil. J. Clean. Prod. 2017, 161, 591–597. [Google Scholar] [CrossRef]
- Yang, Z.; Xia, Y.; Li, M.; Ma, Z.; Xing, Y.; Gui, X. Effects of pore compression pretreatment on the flotation of low-rank coal. Fuel 2019, 239, 63–69. [Google Scholar] [CrossRef]
- Xia, W.; Yang, J.; Liang, C. Effect of microwave pretreatment on oxidized coal flotation. Powder Technol. 2013, 233, 186–189. [Google Scholar] [CrossRef]
- Peng, Y.; Mao, Y.; Xia, W.; Li, Y. Ultrasonic flotation cleaning of high-ash lignite and its mechanism. Fuel 2018, 220, 558–566. [Google Scholar] [CrossRef]
- Zhen, K.; Zheng, C.; Li, C.; Zhang, H. Wettability and flotation modification of long flame coal with low-temperature pyrolysis. Fuel 2018, 227, 135–140. [Google Scholar] [CrossRef]
- Shen, L.; Min, F.; Liu, L.; Zhu, J.; Xue, C.; Cai, C.; Zhou, W.; Wang, C. Application of gaseous pyrolysis products of the waste cooking oil as coal flotation collector. Fuel 2019, 239, 446–451. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, G.; Huang, J.; Eksteen, J.; Liu, X.; Zhao, Z. Characterization of coal particles wettability in surfactant solution by using four laboratory static tests. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 567, 304–312. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).