Enhanced Performance of Oxidation of Rosalva (9-decen-1-ol) to Costenal (9-decenal) on Porous Silicon-Supported Silver Catalyst in a Microstructured Reactor
Abstract
:1. Introduction
2. Experimental
2.1. Preparation of Microstructured Reactors
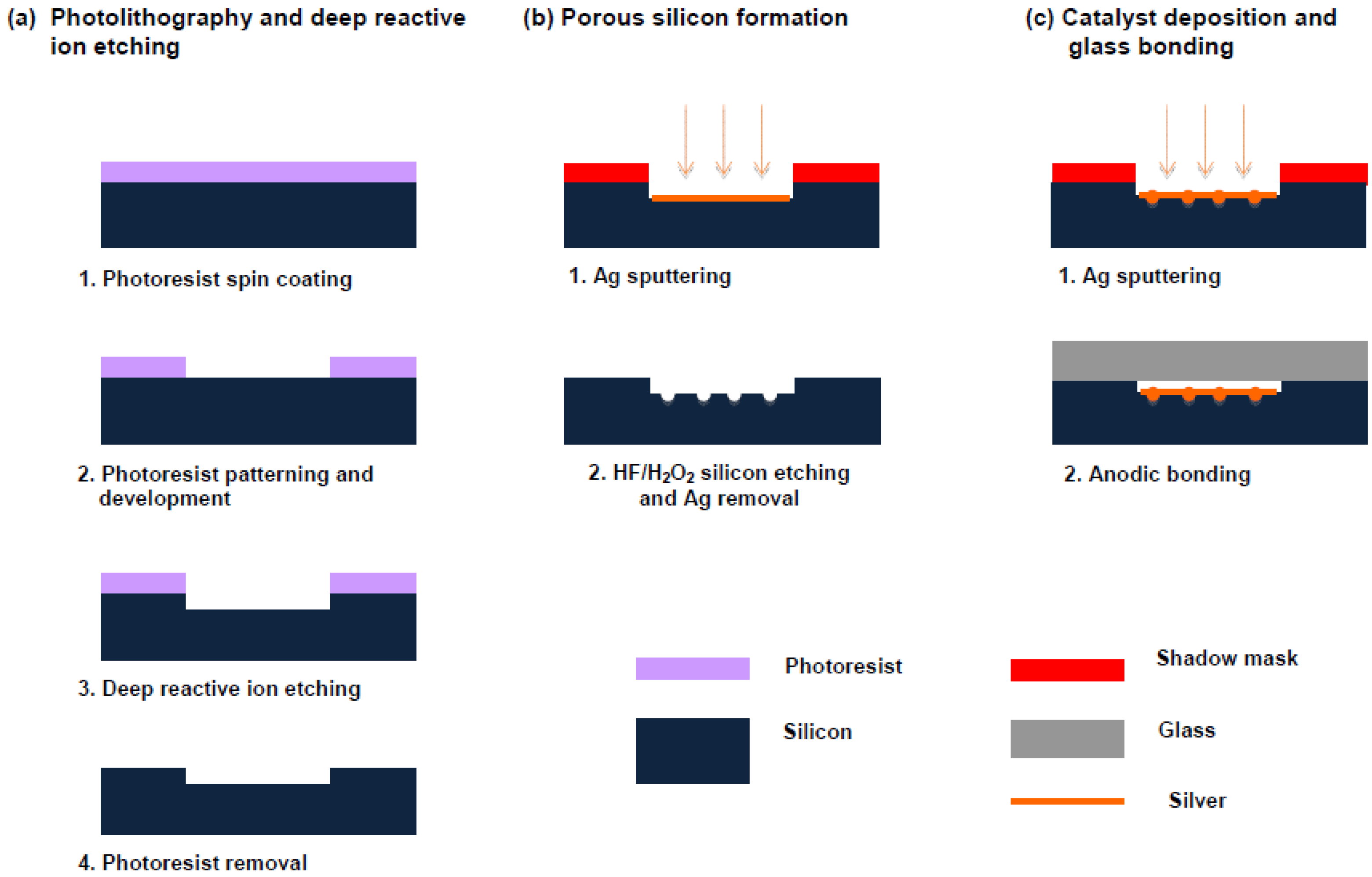

2.1.1. Porous Silicon Formation
2.1.2. Catalyst Deposition
2.1.3. Anodic Bonding
2.2. Characterization of Porous Silicon and Silver Catalyst
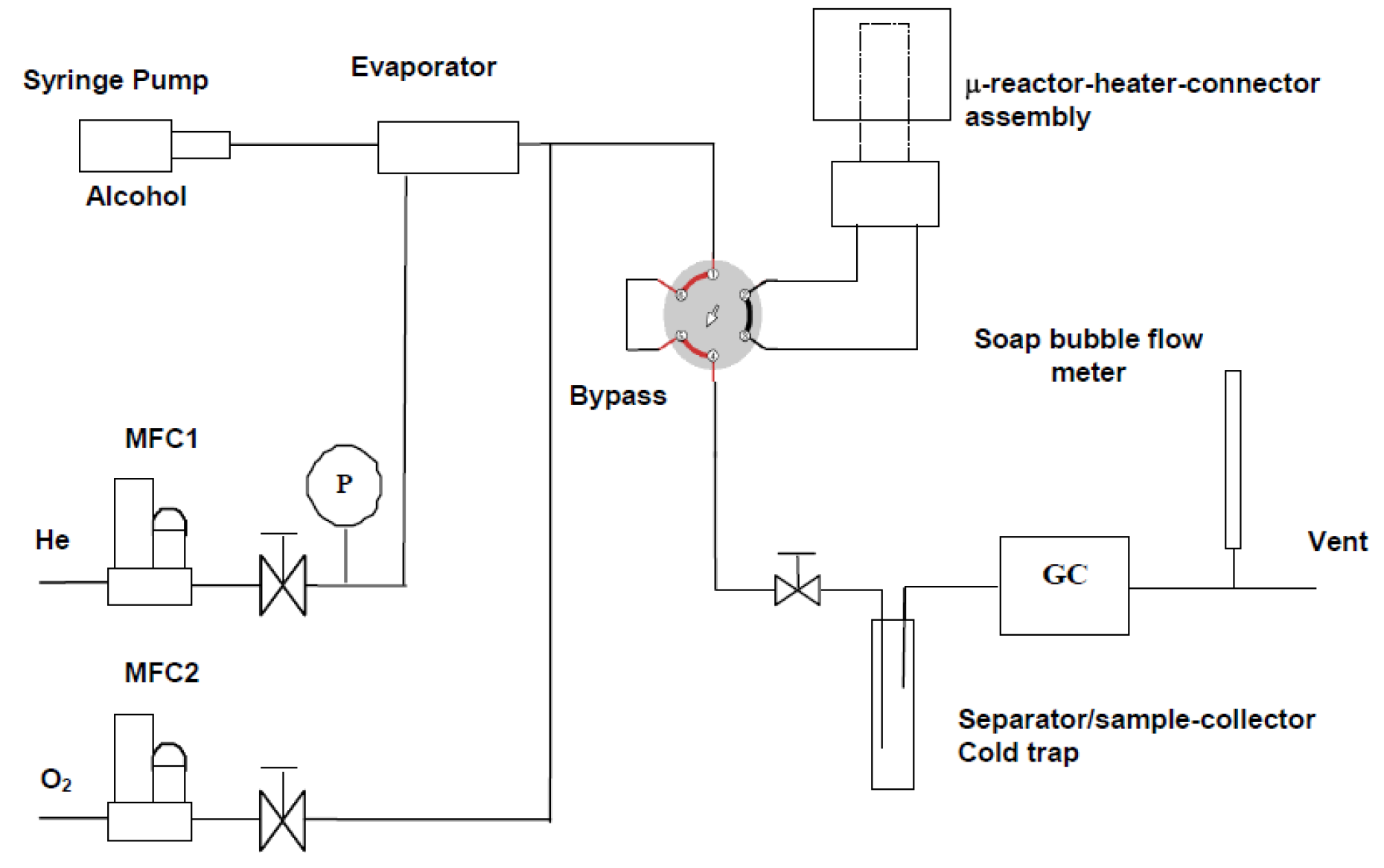
2.3. Experimental Set-Up and Procedure

3. Results and Discussion
3.1. Characterisation of Porous Silicon and Silver Catalyst



3.2. Rosalva Oxidation on Silver Catalyst
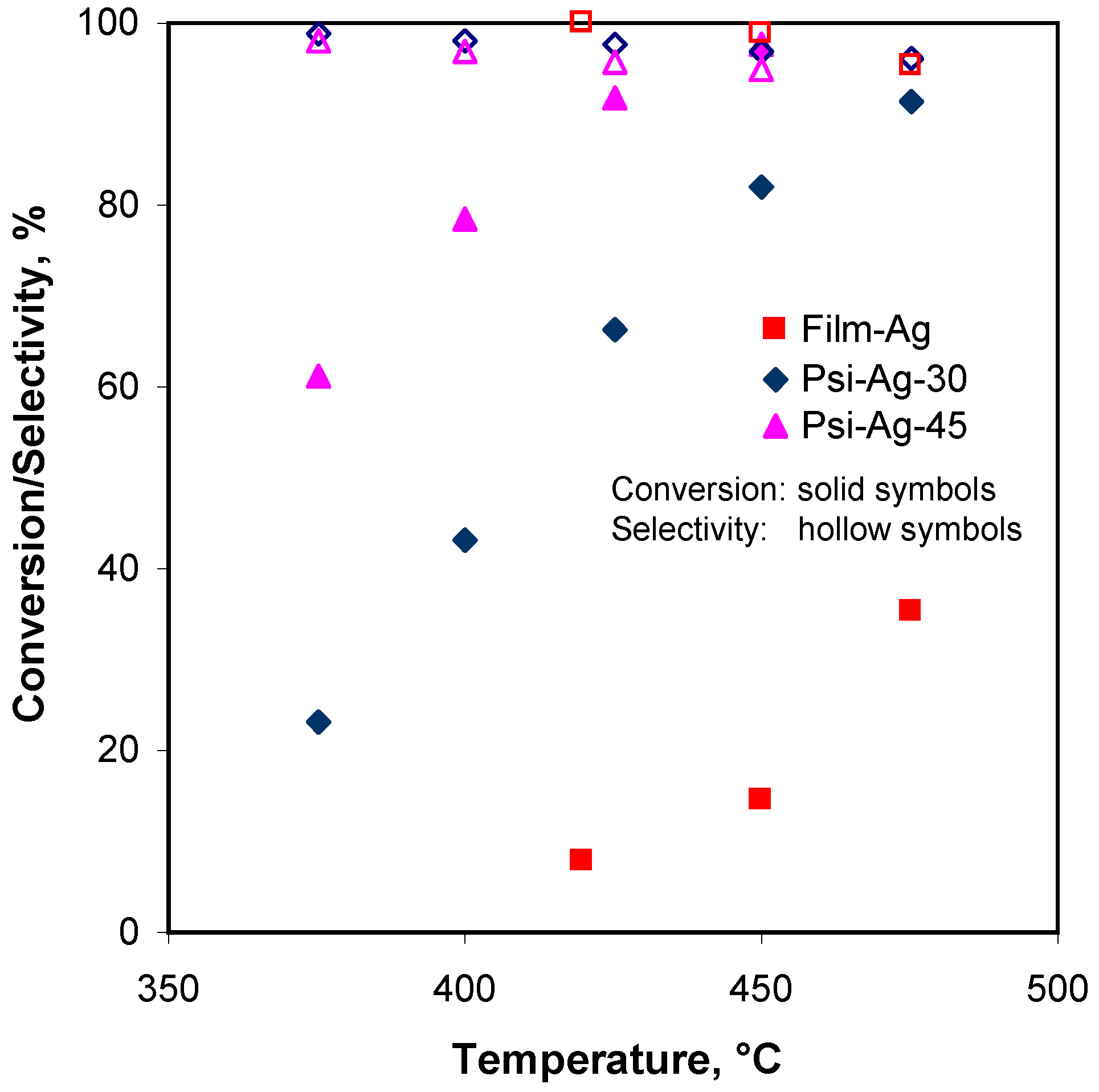
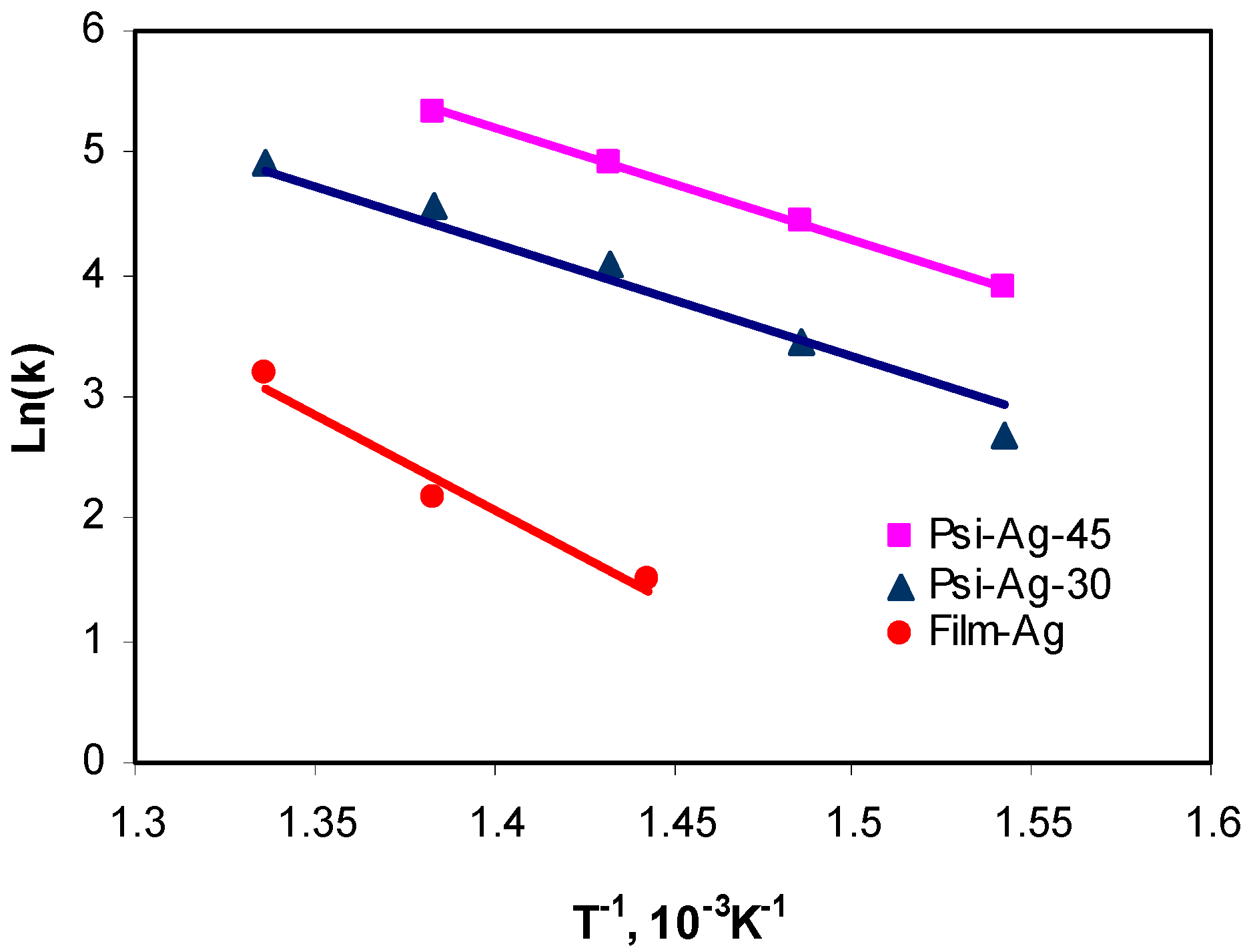
3.2.1. Effect of Reaction Temperature
| T, °C | Psi-Ag-30 | Film-Ag | ||||||
|---|---|---|---|---|---|---|---|---|
| X, % | Selectivity, % | X, % | Selectivity, % | |||||
| 9-decenal | 9-decenoic acid | CO2 | 9-decenal | 9-decenoic acid | CO2 | |||
| 375 | 23.3 | 98.7 | 1.3 | |||||
| 400 | 43.2 | 98.2 | 1.6 | 0.2 | ||||
| 425 | 66.3 | 97.6 | 1.7 | 0.7 | 7.7 | 100 | ||
| 450 | 81.8 | 96.9 | 1.2 | 1.9 | 14.4 | 98.9 | 1.1 | |
| 475 | 91.4 | 96.2 | 1.2 | 2.5 | 35.1 | 95.3 | 3.0 | 1.7 |
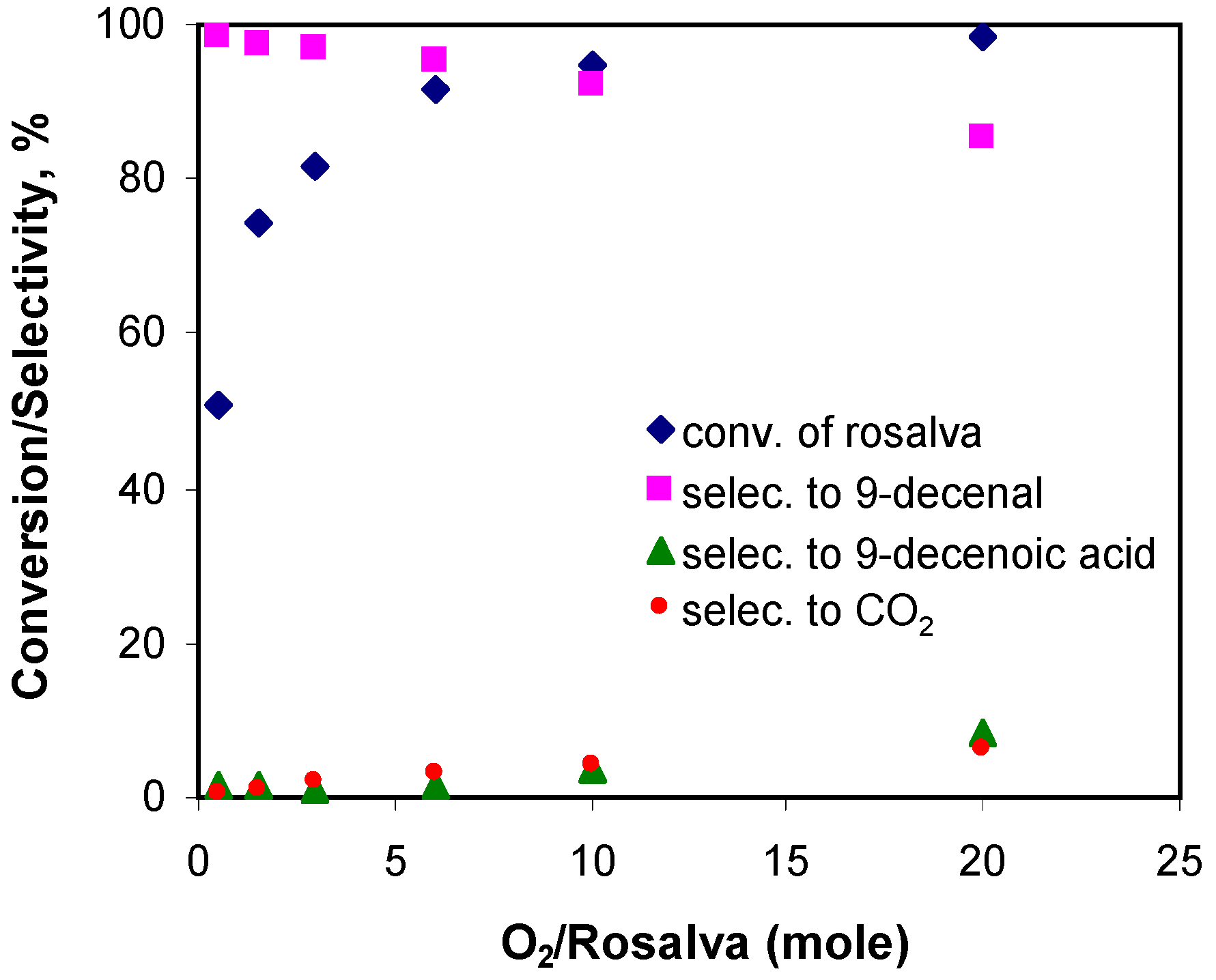
3.2.2. Effect of Inlet Oxygen Concentration
3.2.3. Effect of Inlet Rosalva Concentration

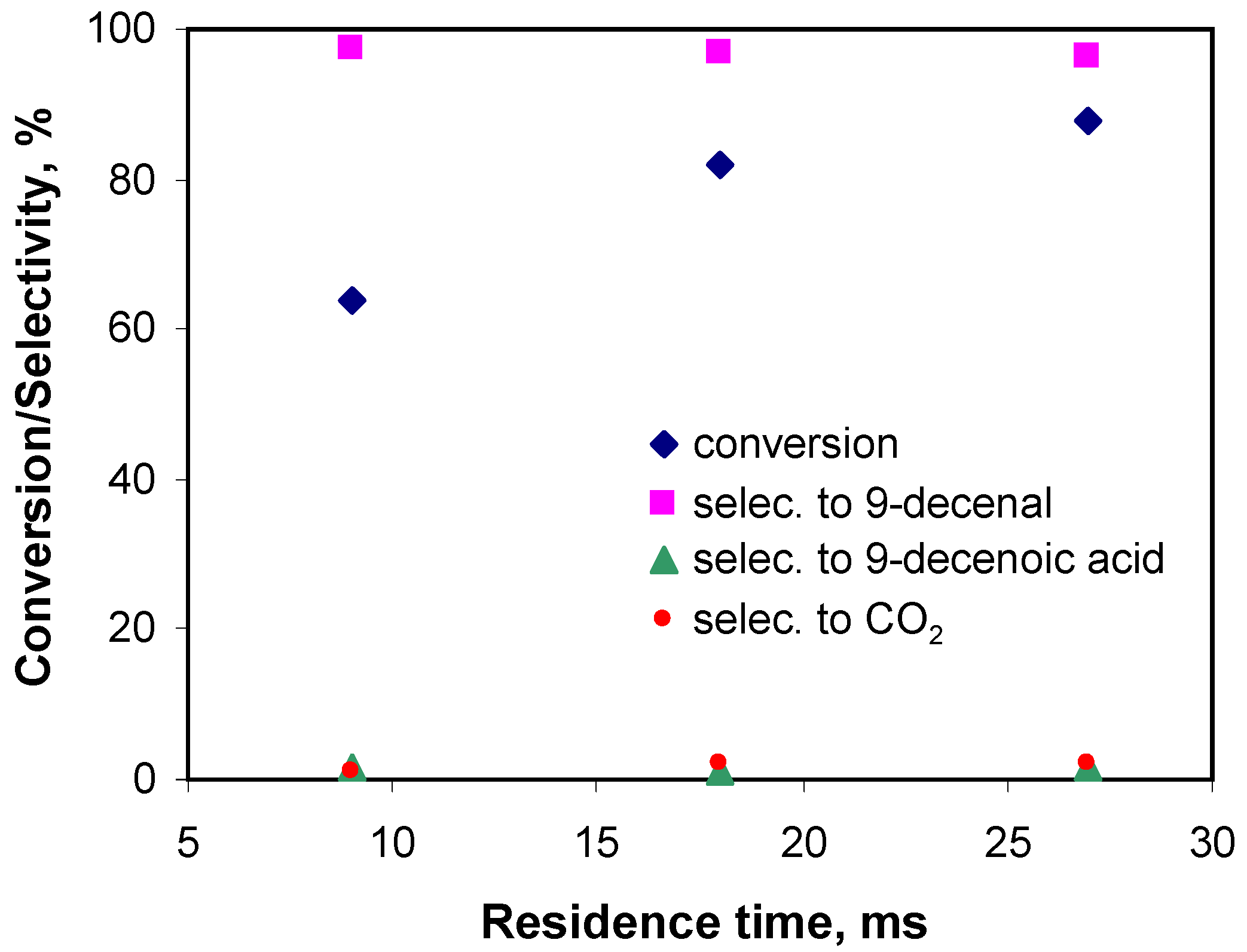
3.2.4. Effect of Residence Time
3.2.5. Effect of the Porous Silicon Layer Structure

4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Sheldon, R.A.; van Bekkum, H. Fine Chemicals through Heterogeneous Catalysis; WILEY-VCH: Weinhein, Germany, 2001. [Google Scholar]
- Della Pina, C.; Falletta, E.; Prati, L.; Rossi, M. Selective oxidation using gold. Chem. Soc. Rev. 2008, 37, 2077–2095. [Google Scholar] [CrossRef]
- Mallat, T.; Baiker, A. Oxidation of alcohols with molecular oxygen on solid catalysts. Chem. Rev. 2004, 104, 3037–3058. [Google Scholar] [CrossRef]
- Biella, S.; Rossi, M. Gas phase oxidation of alcohols to aldehydes or ketones catalysed by supported gold. Chem. Commun. 2003, 9, 378–379. [Google Scholar] [CrossRef]
- Han, D.; Xu, T.; Su, J.; Xu, X.; Ding, Y. Gas-Phase selective oxidation of benzyl alcohol to benzaldehyde with molecular oxygen over unsupported nanoporous gold. ChemCatChem 2010, 2, 383–386. [Google Scholar] [CrossRef]
- Della Pina, C.; Falletta, E.; Rossi, M. Highly selective oxidation of benzyl alcohol to benzaldehyde catalyzed by bimetallic gold-copper catalyst. J. Catal. 2008, 260, 384–386. [Google Scholar] [CrossRef]
- Shen, J.; Shan, W.; Zhang, Y.; Du, J.; Xu, H.; Fan, K.; Shen, W.; Tang, Y. Gas-phase selective oxidation of alcohols: In situ electrolytic nano-silver/zeolite film/copper grid catalyst. J. Catal. 2006, 237, 94–101. [Google Scholar] [CrossRef]
- Yadav, G.D.; Mewada, R.K. Selectivity engineering in the synthesis of value added chemicals: Oxidation of 1-octanol to 1-octanal over nano-fibrous Ag-OMS-2 catalysts. Chem. Eng. Res. Des. 2012, 90, 86–97. [Google Scholar] [CrossRef]
- Yamamoto, R.; Sawayama, Y.S.; Shibahara, H.; Ichihashi, Y.; Nishiyama, S.; Tsuruya, S. Promoted partial oxidation activity of supported Ag catalysts in the gas-phase catalytic oxidation of benzyl alcohol. J. Catal. 2005, 234, 308–317. [Google Scholar] [CrossRef]
- Hoelderich, W.F.; Kollmer, F. Oxidation reactions in the synthesis of fine and intermediate chemicals using environmentally benign oxidants and the right reactor system. Pure Appl. Chem. 2000, 72, 1273–1287. [Google Scholar] [CrossRef]
- Hessel, V.; Hardt, S.; Löwe, H. Chemical Micro Process Engineering; Weiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Cao, E.; Gavriilidis, A.; Motherwell, W.B. Oxidative dehydrogenation of 3-methyl-2-buten-1-o1 in microreactors. Chem. Eng. Sci. 2004, 59, 4803–4808. [Google Scholar] [CrossRef]
- Haas-Santo, K.; Fichtner, M.; Schubert, K. Preparation of microstructure compatible porous supports by sol-gel synthesis for catalyst coatings. Appl. Catal. A 2001, 220, 79–92. [Google Scholar]
- Meille, V. Review on methods to deposit catalysts on structured surfaces. Appl. Catal. A 2006, 315, 1–17. [Google Scholar] [CrossRef]
- Losey, M.W.; Jackman, R.J.; Firebaugh, S.L.; Schmidt, M.A.; Jensen, K.F. Design and fabrication of microfluidic devices for multiphase mixing and reaction. J. Microelectromech. Syst. 2002, 11, 709–717. [Google Scholar] [CrossRef]
- Schilke, K.F.; Wilson, K.L.; Cantrell, T.; Corti, G.; McIlroy, D.N.; Kelly, C. A novel enzymatic microreactor with aspergillus oryzae β-galactosidase immobilized on silicon dioxide nanosprings. Biotechnol. Prog. 2010, 26, 1597–1605. [Google Scholar] [CrossRef]
- Huang, Z.; Geyer, N.; Werner, P.; de Boor, J.; Gösele, U. Metal-assisted chemical etching of silicon: A review. Adv. Mater. 2011, 23, 285–308. [Google Scholar] [CrossRef]
- Drott, J.; Lindström, K.; Rosengren, L.; Laurell, T. Porous silicon as the carrier matrix in microstructured enzyme reactors yielding high enzyme activities. J. Micromech. Microengineer. 1997, 7, 14–23. [Google Scholar] [CrossRef]
- Stewart, M.P.; Buriak, J.M. Chemical and biological applications of porous silicon technology. Adv. Mater. 2000, 12, 859–869. [Google Scholar] [CrossRef]
- Pichonat, T.; Gauthier-Manuel, B. A new process for the manufacturing of reproducible mesoporous silicon membranes. J. Membrane Sci. 2006, 280, 494–500. [Google Scholar]
- Clicq, D.; Tjerkstra, R.W.; Gardeniers, J.G.E.; van den Berg, A.; Baron, G.V.; Desmet, G. Porous silicon as a stationary phase for shear-driven chromatography. J. Chromatogr. A 2004, 1032, 185–191. [Google Scholar] [CrossRef]
- De Malsche, W.; Clicq, D.; Verdoold, V.; Gzil, P.; Desmet, G.; Gardeniers, H. Integration of porous layers in ordered pillar arrays for liquid chromatography. Lab Chip 2007, 7, 1705–1711. [Google Scholar] [CrossRef]
- Li, X.; Bonn, P.W. Metal-assisted chemical etching in HF/H2O2 produces porous silicon. Appl. Phys. Lett. 2000, 77, 2572–2574. [Google Scholar] [CrossRef]
- Chern, W.; Hsu, K.; Chun, I.; Azeredo, B.; Fang, N.; Ferreira, P.; Li, X. Non-lithographic patterning and metal-assisted chemical etching for manufacturing of tunable light-emitting silicon nanowire arrays. In Proceedings of Lasers and Electro-Optics/Quantum Electronics and Laser Science Conference: 2010 Laser Science to Photonic Applications, San Jose, CA, USA, 16–21 May 2010; IEEE: Piscataway, NJ, USA, 2010. [Google Scholar]
- Banwell, M.G.; McRae, K.J. Chemoenzymatic synthesis of (+)-aspicilin from chlorobenzene. Org. Lett. 2000, 2, 3583–3586. [Google Scholar] [CrossRef]
- Bortolini, O.; Campestrini, S.; di Furia, F.; Modena, G.; Valle, G. Metal catalysis in oxidation by peroxides. Anionic molybdenum-picolinate N-oxido-peroxo complex: An effective oxidant of primary and secondary alcohols in nonpolar solvents. J. Org. Chem. 1987, 52, 5467–5469. [Google Scholar]
- McErlean, C.S.P.; Proisy, N.; Davis, C.J.; Boland, N.A.; Sharp, S.Y.; Boxall, K.; Slawin, A.M.Z.; Workman, P.; Moody, C.J. Synthetic ansamycins prepared by a ring-expanding Claisen rearrangement. Synthesis and biological evaluation of ring and conformational analogues of the Hsp90 molecular chaperone inhibitor geldanamycin. Org. Biomol. Chem. 2007, 5, 531–546. [Google Scholar] [CrossRef]
- Uyanik, M.; Akakura, M.; Ishihara, K. 2-iodoxybenzenesulfonic acid as an extremely active catalyst for the selective oxidation of alcohols to aldehydes, ketones, carboxylic acids, and enones with oxone. J. Amer. Chem. Soc. 2009, 131, 251–262. [Google Scholar] [CrossRef]
- Gallezot, P.; Ceroni, L.; Perrard, A. Oxidative dehydrogenation of rosalva to costenal on supported silver catalysts. J. Mol. Catal. A 1998, 129, L127–L130. [Google Scholar] [CrossRef]
- Cao, E.; Gavriilidis, A. Oxidative dehydrogenation of methanol in a microstructured reactor. Catal. Today 2005, 110, 154–163. [Google Scholar] [CrossRef]
- Tiggelaar, R.M.; Verdoold, V.; Eghbali, H.; Desmet, G.; Gardeniers, J.G.E. Characterization of porous silicon integrated in liquid chromatography chips. Lab Chip 2009, 9, 456–463. [Google Scholar] [CrossRef]
- Chaudhari, P. Hillock growth in thin films. J. Appl. Phys. 1974, 45, 4339–4346. [Google Scholar] [CrossRef]
- Sharma, S.K.; Spitz, J. Hillock growth and agglomeration in thin silver films. Thin Solid Films 1979, 61, L13–L15. [Google Scholar] [CrossRef]
- Sharma, S.K.; Spitz, J. Hillock formation, hole growth and agglomeration in thin silver films. Thin Solid Films 1980, 65, 339–350. [Google Scholar] [CrossRef]
- Waterhouse, G.I.N.; Bowmaker, G.A.; Metson, J.B. Oxygen chemisorption on an electrolytic silver catalyst: A combined TPD and Raman spectroscopic study. Appl. Surface Sci. 2003, 214, 36–51. [Google Scholar] [CrossRef]
- Waterhouse, G.I.N.; Bowmaker, G.A.; Metson, J.B. Influence of catalyst morphology on the performance of electrolytic silver catalysts for the partial oxidation of methanol to formaldehyde. Appl. Catal. A 2004, 266, 257–273. [Google Scholar] [CrossRef]
- Waterhouse, G.I.N.; Bowmaker, G.A.; Metson, J.B. Mechanism and active sites for the partial oxidation of methanol to formaldehyde over an electrolytic silver catalyst. Appl. Catal. A 2004, 265, 85–101. [Google Scholar] [CrossRef]
- Hayes, R.E.; Kolaczkowski, S.T. Introduction to Catalytic Combustion; Gordon & Breach Science Publishers: Amsterdam, The Netherlands, 1997. [Google Scholar]
- van Santen, P.A.; Neurock, M. Molecular Heterogeneous Catalysis-A Conceptual and Computational Approach; WILEY-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Che, M.; Bennett, C.O. The influence of particle size on the catalytic properties of supported metals. In Advances in Catalysis; Eley, H.P.D.D., Paul, B.W., Eds.; Academic Press: New York, NY, USA, 1989; Volume 36, pp. 55–172. [Google Scholar]
- Musselwhite, N.; Somorjai, G. Investigations of structure sensitivity in heterogeneous catalysis: from single crystals to monodisperse nanoparticles. Top. Catal. 2013, 56, 1277–1283. [Google Scholar] [CrossRef]
- Anderson, R.C.; Muller, R.S.; Tobias, C.W. Chemical surface modification of porous silicon. J. Electrochem. Soc. 1993, 140, 1393–1396. [Google Scholar] [CrossRef]
- Michael, J.S. Porous silicon in Practice: Preparation, Characterization and Applications, First ed.; Wiley-VCH: Weinheim, Germany, 2012. [Google Scholar]
- Tsybulya, S.V.; Kryukova, G.N.; Goncharova, S.N.; Shmakov, A.N.; Balzhinimaev, B.S. Study of the real structure of silver supported catalysts of different dispersity. J. Catal. 1995, 154, 194–200. [Google Scholar] [CrossRef]
- Mastikhin, V.M.; Goncharova, S.N.; Tapilin, V.M.; Terskikh, V.V.; Balzhinimaev, B.S. Effect of particle size upon catalytic and electronic properties of supported Ag catalysts: Combined catalytic, 109Ag NMR and quantum chemistry studies. J. Mol. Catal. A 1995, 96, 175–179. [Google Scholar] [CrossRef]
- Zuburtikudis, I.; Saltsburg, H. Linear metal nanostructures and size effects of supported metal catalysts. Science 1992, 258, 1337–1339. [Google Scholar]
- Christopher, P.; Linic, S. Shape- and size-specific chemistry of Ag nanostructures in catalytic ethylene epoxidation. ChemCatChem 2010, 2, 78–83. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cao, E.; Zuburtikudis, I.; Al-Rifai, N.; Roydhouse, M.; Gavriilidis, A. Enhanced Performance of Oxidation of Rosalva (9-decen-1-ol) to Costenal (9-decenal) on Porous Silicon-Supported Silver Catalyst in a Microstructured Reactor. Processes 2014, 2, 141-157. https://doi.org/10.3390/pr2010141
Cao E, Zuburtikudis I, Al-Rifai N, Roydhouse M, Gavriilidis A. Enhanced Performance of Oxidation of Rosalva (9-decen-1-ol) to Costenal (9-decenal) on Porous Silicon-Supported Silver Catalyst in a Microstructured Reactor. Processes. 2014; 2(1):141-157. https://doi.org/10.3390/pr2010141
Chicago/Turabian StyleCao, Enhong, Ioannis Zuburtikudis, Noor Al-Rifai, Mark Roydhouse, and Asterios Gavriilidis. 2014. "Enhanced Performance of Oxidation of Rosalva (9-decen-1-ol) to Costenal (9-decenal) on Porous Silicon-Supported Silver Catalyst in a Microstructured Reactor" Processes 2, no. 1: 141-157. https://doi.org/10.3390/pr2010141
APA StyleCao, E., Zuburtikudis, I., Al-Rifai, N., Roydhouse, M., & Gavriilidis, A. (2014). Enhanced Performance of Oxidation of Rosalva (9-decen-1-ol) to Costenal (9-decenal) on Porous Silicon-Supported Silver Catalyst in a Microstructured Reactor. Processes, 2(1), 141-157. https://doi.org/10.3390/pr2010141




