A Calcined Mg/Al LDHs Strategy for High-Performance Steel Slag Cementitious Composites
Abstract
1. Introduction
2. Materials and Method
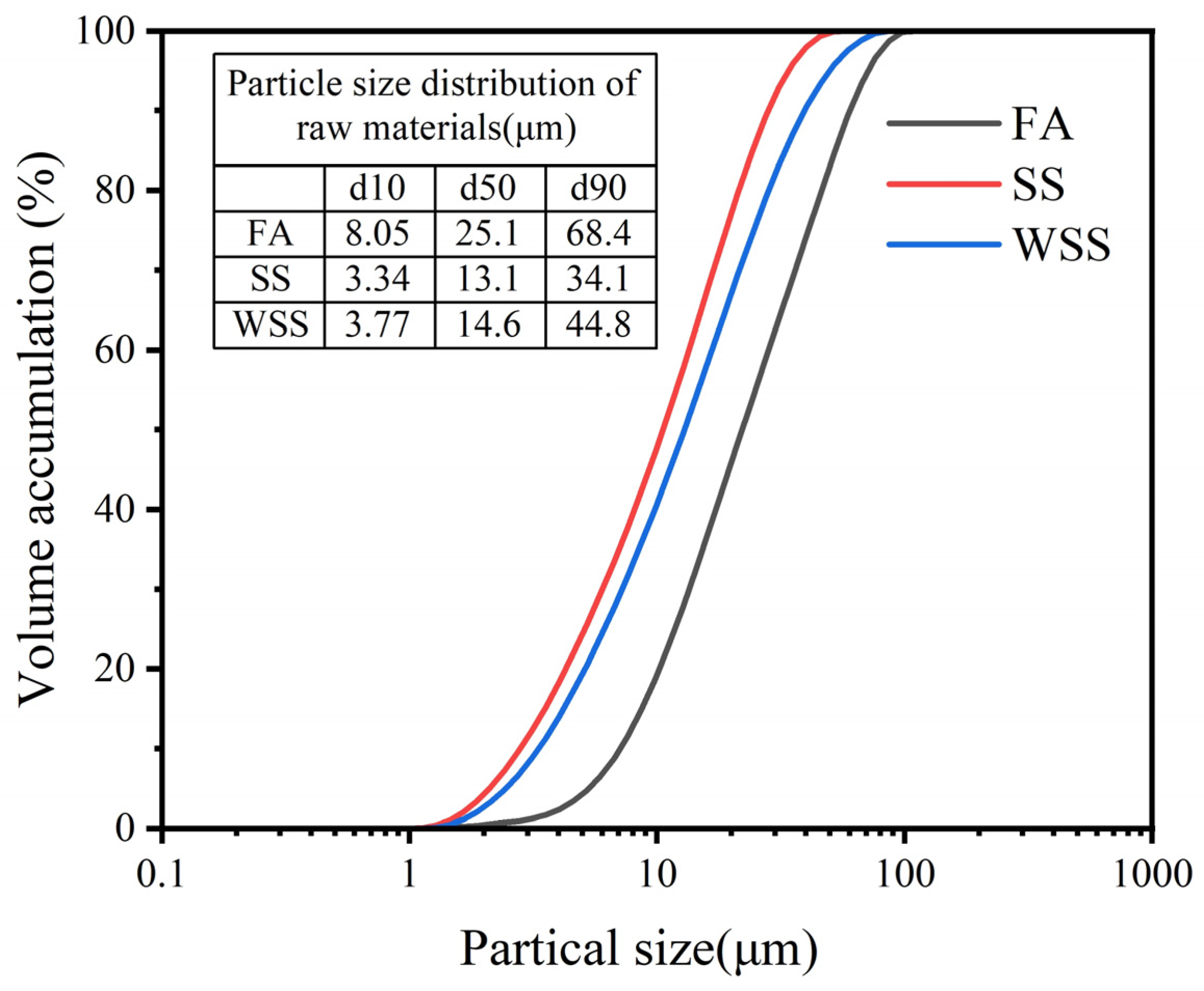
2.1. Raw Materials
2.2. Pre-Hydration for Steel Slag
2.3. Mix Composition and Sample Preparation
2.4. Characterization
3. Results
3.1. Chemical Structure and Composition of CLDH
3.2. Impacts of CLDH on Mechanical Properties
3.3. Impacts of CLDH on Hydration Kinetics
3.4. Impacts of CLDH on Mineral Compositions
3.5. Impacts of CLDH on Phase Content
3.6. Impacts of CLDH on Micromorphology and Chemical Composition
4. Discussion
5. Conclusions
- The hydration heat test results show that in the initial reaction stage, the hydration heat peak decreases due to the dilution effect of CLDH. As the hydration reaction proceeds, the cumulative heat release of the 1% CLDH sample exceeds that of the control sample when the reaction time approaches 3 d. Thus, CLDH exerts a certain promotional effect on the hydration reaction of the system.
- The compressive strength of steel slag cementitious materials containing CLDH is enhanced at all curing ages. Among them, the compressive strength of materials with 1% CLDH and 2% CLDH exhibits the most significant improvement: at 3 d of reaction, the compressive strength can increase by 21.5%, and at 28 d of reaction, it can still increase by 116.7%. However, when the CLDH dosage exceeds 2%, it will have a negative impact, especially on the compressive strength at 7and 28 d.
- SEM analysis shows that the addition of CLDH changes the morphology of the gel products. Due to the nucleation effect of CLDH, it promotes the formation of cluster gel products, which cover and combine with fly ash particles, thereby endowing the material with higher compressive strength. EDS elemental analysis reveals that the addition of CLDH changes the microstructure of C-(A)-S-H. When 1% CLDH is used, a higher Si/Ca ratio can be observed relative to the control group.
- The strengthening mechanism of CLDH on alkali-activated steel slag cementitious materials can be summarized into three pathways: (1) the micro-filling effect reduces pore defects; (2) CLDH does not significantly increase the amount of early hydration products (7 days), but optimizes the microstructure and elemental ratio of C-(A)-S-H gel; and (3) CLDH slightly promotes the hydration of steel slag after 7 days.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cembureau. The European Cement Association (Cembureau) 2020 Activity Report; Cembureau—The European Cement Association: Brussels, Belgium, 2021. [Google Scholar]
- IEA. Cement Technology Roadmap 2009—Carbon Emissions Reductions up to 2050; World Business Council on Sustainable Development (WBCSD): Geneva, Switzerland; International Energy Agency (IEA): Paris, France, 2009. [Google Scholar]
- Mikulčić, H.; Klemeš, J.J.; Vujanović, M.; Urbaniec, K.; Duić, N. Reducing greenhouse gasses emissions by fostering the deployment of alternative raw materials and energy sources in the cleaner cement manufacturing process. J. Clean. Prod. 2016, 136, 119–132. [Google Scholar] [CrossRef]
- Provis, J.L. Alkali-activated materials. Cem. Concr. Res. 2018, 114, 40–48. [Google Scholar] [CrossRef]
- Provis, J.L.; Palomo, A.; Shi, C. Advances in understanding alkali-activated materials. Cem. Concr. Res. 2015, 78, 110–125. [Google Scholar] [CrossRef]
- Li, L.; Xie, J.; Zhang, B.; Feng, Y.; Yang, J. A state-of-the-art review on the setting behaviours of ground granulated blast furnace slag- and metakaolin-based alkaliactivated materials. Constr. Build. Mater. 2023, 368, 130389. [Google Scholar] [CrossRef]
- Rashad, A.M.; Khafaga, S.A.; Gharieb, M. Valorization of fly ash as an additive for electric arc furnace slag geopolymer cement. Constr. Build. Mater. 2021, 294, 123570. [Google Scholar] [CrossRef]
- Guo, J.; Bao, Y.; Wang, M. Steel slag in China: Treatment, recycling, and management. Waste Manag. 2018, 78, 318–330. [Google Scholar] [CrossRef]
- Zhao, L.H.; Wei, W.; Bai, H.; Zhang, X.; Cang, D.Q. Synthesis of steel slag ceramics: Chemical composition and crystalline phases of raw materials. Int. J. Miner. Metall. Mater. 2015, 22, 325–333. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, M.; Mo, L.; Zhong, J.; Xu, M.; Deng, M. Probe into carbonation mechanism of steel slag via FIB-TEM: The roles of various mineral phases. Cem. Concr. Res. 2022, 162, 106991. [Google Scholar] [CrossRef]
- Hao, X.S.; Liu, X.M.; Zhang, Z.Q.; Zhang, W.; Lu, Y.; Wang, Y.G.; Yang, T.Y. Indepth insight into the cementitious synergistic effect of steel slag and red mud on the properties of composite cementitious materials. J. Build. Eng. 2022, 52, 104449. [Google Scholar] [CrossRef]
- Humbert, P.S.; Castro-Gomes, J. CO2 activated steel slag-based materials: A review. J. Clean. Prod. 2018, 208, 448–457. [Google Scholar] [CrossRef]
- Hou, J.W.; Lv, Y.; Liu, J.X.; Wu, Q.S. Expansibility of cement paste with tri-component f-CaO in steel slag. Mater. Struct. 2018, 51, 113. [Google Scholar] [CrossRef]
- Huo, B.B.; Li, B.L.; Huang, S.Y.; Chen, C.; Zhang, Y.M.; Banthia, N. Hydration and soundness properties of phosphoric acid modified steel slag powder. Construct. Build. Mater. 2020, 254, 119319. [Google Scholar] [CrossRef]
- Zhang, L.L.; Wang, Q.G.; Zheng, Y.C.; Cang, Z.Z.; Gisele, K.; Yu, C.S.; Cang, D.Q. Synergistic effect and mechanism of waste glass on the mechanical properties and autoclave stability of cementitious materials containing steel slag. Construct. Build. Mater. 2021, 311, 125295. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, Y.; He, B.; Jing, X.; Cang, D.; Zhang, L. Study on evolution of pores channel in carbonation steel slag samples with Fly Ash. Constr. Build. Mater. 2024, 411, 134471. [Google Scholar] [CrossRef]
- Papayianni, I.; Anastasiou, E. Effect of granulometry on cementitious properties of ladle furnace slag. Cem. Concr. Compos. 2012, 34, 400–407. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Jeon, D.; Yang, J.; Moon, J. Enhanced mechanical property of steel slag through glycine-assisted hydration and carbonation curing. Cem. Concr. Compos. 2024, 149, 105532. [Google Scholar] [CrossRef]
- Qu, J.; Zhang, Q.; Li, X.; He, X.; Song, S. Mechanochemical approaches to synthesize layered double hydroxides: A review. Appl. Clay Sci. 2016, 119, 185–192. [Google Scholar] [CrossRef]
- Chafiq, M.; Chaouiki, A.; Suhartono, T.; Hazmatulhaq, F.; Ko, Y.G. Interface engineering of LDH-based material as efficient anti-corrosive system via synergetic performance of host, interlayers, and morphological features of nature-mimic architectures. Chem. Eng. J. 2023, 462, 142239. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, J.; Zou, F.; Zhou, N.; Li, Y.; Lei, W. Ca-Al LDH hybrid self-healing microcapsules for corrosion protection. Chem. Eng. J. 2022, 447, 137125. [Google Scholar] [CrossRef]
- Duan, P.; Yan, C.; Zhou, W. Effects of calcined layered double hydroxides on carbonation of concrete containing fly ash. Construct. Build. Mater. 2018, 160, 725–732. [Google Scholar] [CrossRef]
- Jiang, H.; Tian, D.; Dong, M.; Lv, M.; Yang, X.; Lu, S. Effects of interlayer-modified layered double hydroxides with organic corrosion inhibiting ions on the propertiesof cement-based materials and reinforcement corrosion in chloride environment. Cem. Concr. Compos. 2024, 154, 105793. [Google Scholar] [CrossRef]
- Liu, J.; Gao, X.; Chen, T. Effect of SiO2-modified calcined layered double hydroxides on the properties of cement-based material: Crucial role of the phasetransformation induced by alkaline pore solution. Cem. Concr. Res. 2024, 178, 107465. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, F.; Long, Y.; Yu, L.; Yang, C.; Wen, M.; Yu, X.; Tian, Y.; Zhao, M. Improving the carbonation resistance of alkali-activated slag mortars by calcined Mg/Al layered double hydroxides. Appl. Clay Sci. 2022, 216, 106379. [Google Scholar] [CrossRef]
- Chen, M.; Liu, Q.; Zheng, H.; Yu, L.; Jiang, L.; Gu, Z.; Li, W. Understanding strengthening mechanisms of Ca-LDO on cementitious materials. Cem. Concr. Compos. 2024, 145, 105340. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, Z.; Zhu, H.; Duan, P.; Fang, Y.; Jiang, Z. Mechanisms of CLDH seeding on hydration kinetics of slag-based geopolymer: Towards aluminosilicate cement phase engineering. Compos. B Eng. 2024, 271, 111157. [Google Scholar] [CrossRef]
- Martins, A.C.; De Carvalho, J.M.; Costa, L.C.; Andrade, H.D.; de Melo, T.V.; Ribeiro, J.C.; Pedroti, L.G.; Peixoto, R.A. Steel slags in cement-based composites: Anultimate review on characterization, applications and performance. Constr. Build. Mater. 2021, 291, 123265. [Google Scholar] [CrossRef]
- Long, W.-J.; Xie, J.; Zhang, X.; Fang, Y.; Khayat, K.H. Hydration and microstructure of calcined hydrotalcite activated high-volume fly ash cementitious composite. Cem. Concr. Compos. 2021, 123, 104213. [Google Scholar] [CrossRef]
- He, B.; Zhu, X.; Lei, Y.; Jing, X.; Liu, Y.; Chen, Z.; Cang, D.; Birat, J.-P.; Tang, Z.; Zhang, L. Utilizing Alkaline Solid Waste for Low-Carbon Construction Material via In-Situ Calcium Phase Design. Nat. Commun. 2025, 16, 7275. [Google Scholar] [CrossRef] [PubMed]
- GB/T 17671-2021; Test method of cement mortar strength (ISO method). Standardization Administration of China, 2021.
- Vagvolgyi, V.; Palmer, S.J.; Kristof, J.; Frost, R.L.; Horvath, E. Mechanism for hydrotalcite decomposition: A controlled rate thermal analysis study. J. Colloid Interface Sci. 2008, 318, 302–308. [Google Scholar] [CrossRef]
- Zheludkevich, M.L.; Poznyak, S.K.; Rodrigues, L.M.; Raps, D.; Hack, T.; Dick, L.F.; Nunes, T.; Ferreira, M.G.S. Active protection coatings with layered double hydroxide nanocontainers of corrosion inhibitor. Corros. Sci. 2010, 52, 602–611. [Google Scholar] [CrossRef]
- Tedim, J.; Kuznetsova, A.; Salak, A.N.; Montemor, F.; Snihirova, D.; Pilz, M.; Zheludkevich, M.L.; Ferreira, M.G.S. Zn-Al layered double hydroxides as chloride nanotraps in active protective coatings. Corros. Sci. 2012, 55, 1–4. [Google Scholar] [CrossRef]
- Xu, S.; Chen, Z.; Zhang, B.; Yu, J.; Zhang, F.; Evans, D.G. Facile preparation of pureCaAl-layered double hydroxides and their application as a hardening accelerator inconcrete. Chem. Eng. J. 2009, 155, 881–885. [Google Scholar] [CrossRef]
- Yang, Z.; Fischer, H.; Cerezo, J.; Mol, J.M.C.; Polder, R. Aminobenzoate modified MgAl hydrotalcites as a novel smart additive of reinforced concrete for anticorrosion applications. Construct. Build. Mater. 2013, 47, 1436–1443. [Google Scholar] [CrossRef]
- Chithiraputhiran, S.; Neithalath, N. Isothermal reaction kinetics and temperature dependence of alkali activation of slag, fly ash and their blends. Construct. Build. Mater. 2013, 45, 233–242. [Google Scholar]
- Bernal, S.A.; Provis, J.L.; Rose, V.; de Gutierrez, R.M. Evolution of binder structure in sodium silicate-activated slag-metakaolin blends. Cem. Concr. Compos. 2011, 33, 46–54. [Google Scholar] [CrossRef]
- Ke, X.; Bernal, S.A.; Provis, J.L. Controlling the reaction kinetics of sodium carbonate-activated slag cements using calcined layered double hydroxides. Cem. Concr. Res. 2016, 81, 24–37. [Google Scholar] [CrossRef]
- Zhang, S.; Li, Z.; Ghiassi, B.; Yin, S.; Ye, G. Fracture properties and microstructure formation of hardened alkali-activated slag/fly ash pastes. Cem. Concr. Res. 2021, 144, 106447. [Google Scholar] [CrossRef]
- Tsivilis, S.; Kakali, G.; Chaniotakis, E.; Souvaridou, A. A study on the hydration of portland limestone cement by means of TG. J. Therm. Anal. Calorim. 1998, 52, 863–870. [Google Scholar] [CrossRef]
- Thiery, M.; Villain, G.; Dangla, P.; Platret, G. Investigation of the carbonation front shape on cementitious materials: Effects of the chemical kinetics. Cem. Concr. Res. 2007, 37, 1047–1058. [Google Scholar] [CrossRef]
- Abdalqader, A.F.; Jin, F.; Al-Tabbaa, A. Characterisation of reactive magnesia and sodium carbonate-activated fly ash/slag paste blends. Constr. Build. Mater. 2015, 93, 506–513. [Google Scholar] [CrossRef]
- Gao, Y.; Zhu, X.; Corr, D.J.; Konsta-Gdoutos, M.S.; Shah, S.P. Characterization of the interfacial transition zone of CNF-Reinforced cementitious composites. Cem. Concr. Compos. 2019, 99, 130–139. [Google Scholar] [CrossRef]
- Shen, P.; Lu, L.; He, Y.; Wang, F.; Hu, S. The effect of curing regimes on the mechanical properties, nano-mechanical properties and microstructure of ultrahigh performance concrete. Cem. Concr. Res. 2019, 118, 1–13. [Google Scholar] [CrossRef]
- Huang, H.; Teng, L.; Gao, X.; Khayat, K.H.; Wang, F.; Liu, Z. Effect of carbon nanotube and graphite nanoplatelet on composition, structure, and nano-mechanical properties of C-S-H in UHPC. Cem. Concr. Res. 2022, 154, 106713. [Google Scholar] [CrossRef]
- Zhao, Y.; Zheng, Y.; Cui, K.; Shen, P.; Poon, C.S.; Moon, J.; Peng, G.; Guo, R.; Xia, D. An innovative ternary carbon-fixing cementitious system of cement-fly ash-carbonated steel slag. Cem. Concr. Compos. 2025, 160, 106042. [Google Scholar] [CrossRef]
- Kani, E.N.; Mehdizadeh, H. Investigating gel molecular structure and its relation with mechanical strength in geopolymer cement based on natural pozzolan using in situ ATR-FTIR spectroscopy. J. Mater. Civ. Eng. 2017, 29, 04017078. [Google Scholar] [CrossRef]
- Ye, H. Nanoscale attraction between calcium-aluminosilicate-hydrate and Mg-Al layered double hydroxides in alkali-activated slag. Mater. Charact. 2018, 140, 95–102. [Google Scholar] [CrossRef]
- Li, H.; Xiao, H.G.; Ou, J.P. A study on mechanical and pressure-sensitiveproperties of cement mortar with nanophase materials. Cem. Concr. Res. 2004, 34, 435–438. [Google Scholar]
- Li, H.; Xiao, H.G.; Yuan, J.; Ou, J. Microstructure of cement mortar with nanoparticles. Compos. B Eng. 2004, 35, 185–189. [Google Scholar] [CrossRef]
- Bahmani, H.; Mostafaei, H. Sustainable high-performance concrete: Harnessing recycled rubber and slag for strength and eco-friendliness. Sci. Rep. 2026, 16, 7376. [Google Scholar] [CrossRef]









| Oxide wt. (%) | SiO2 | Al2O3 | CaO | Fe2O3 | K2O | MgO | MnO | Others |
|---|---|---|---|---|---|---|---|---|
| Fly ash (FA) | 56.179 | 20.936 | 8.425 | 6.306 | 2.293 | 1.669 | 0.147 | 3.882 |
| Steel slag (SS) | 16.852 | 2.748 | 42.006 | 23.837 | 0.051 | 5.551 | 5.204 | 3.719 |
| Water steel slag (WSS) | 16.763 | 2.687 | 41.990 | 24.129 | 0.051 | 5.380 | 5.281 | 3.662 |
| Samples | WSS | FA | Sodium Hydroxide | Sodium Silicate | Ms | w/b | CLDH |
|---|---|---|---|---|---|---|---|
| CLDH (0 wt%) | 70 | 30 | 2.31 | 14.36 | 1.4 | 0.33 | 0 |
| CLDH (1 wt%) | 70 | 30 | 2.31 | 14.36 | 1.4 | 0.33 | 1 |
| CLDH (2 wt%) | 70 | 30 | 2.31 | 14.36 | 1.4 | 0.33 | 2 |
| CLDH (3 wt%) | 70 | 30 | 2.31 | 14.36 | 1.4 | 0.33 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cui, F.; Tang, Z.; He, B.; Jing, X.; Chen, Z.; Cang, D.; Yang, Z.; Zhang, L. A Calcined Mg/Al LDHs Strategy for High-Performance Steel Slag Cementitious Composites. Processes 2026, 14, 974. https://doi.org/10.3390/pr14060974
Cui F, Tang Z, He B, Jing X, Chen Z, Cang D, Yang Z, Zhang L. A Calcined Mg/Al LDHs Strategy for High-Performance Steel Slag Cementitious Composites. Processes. 2026; 14(6):974. https://doi.org/10.3390/pr14060974
Chicago/Turabian StyleCui, Fuxiang, Zian Tang, Bingyang He, Xiaohuan Jing, Zhaohou Chen, Daqiang Cang, Zhijie Yang, and Lingling Zhang. 2026. "A Calcined Mg/Al LDHs Strategy for High-Performance Steel Slag Cementitious Composites" Processes 14, no. 6: 974. https://doi.org/10.3390/pr14060974
APA StyleCui, F., Tang, Z., He, B., Jing, X., Chen, Z., Cang, D., Yang, Z., & Zhang, L. (2026). A Calcined Mg/Al LDHs Strategy for High-Performance Steel Slag Cementitious Composites. Processes, 14(6), 974. https://doi.org/10.3390/pr14060974






